Abstract
Due to their high capacity, nonwoven textiles have been used for a long time as potential filters to remove harmful air pollutants. In this study, needle-punch polypropylene layers were treated with powder activated carbon and nanosilver particle through pad drying and spray techniques, respectively. The morphology of treated layers was observed by a scanning electron microscope and laboratory apparatus were used to evaluate effects of microbe elimination from samples of passing polluted air. The gas adsorption efficiency of fabricated filter was measured by an air pollution device. Results showed that samples treated with activated carbon absorbed 38%, 100% and 100% of CO, NO and NO2 gases, respectively, compared with pristine samples. Staphylococcus aureus and Escherichia coli were completely removed from passing air by nanosilver-coated layer.
Introduction
According to United States Environmental Protection Agency (USEPA), air pollution is among the major environmental risks. It has caused the burden of chronic disease and premature death; such as cardiovascular and respiratory diseases [1]. Applying filters is one of the (very) practical ways to reduce such risks. Among conventional filters, textile filters have been developed and, in this field, nonwoven (NW) fabrics show extensive application because of their potential properties such as high porosity and absorbency, flexibility, and acceptable durability [2]. NW fabrics are generally sheets or webs, which lack a definite structure. The filtration process in NW fabrics is brought about by the interlocking nature of one fiber with the other [3].
Polypropylene fibers are one of the most important fibers used in this field due to their low cost and potential physical properties (light weight, resistance to moisture and chemicals). Polypropylene fibers are generally from isotactic type (most commercial tacticity) and considering their resistance to chemicals, melt spinning process is preferred for spinning. The fiber’s properties can also be enhanced by melt mixing with particulates and fibrous materials, as well as by melt blending with other polymers [4–8].
A great deal of effort has been put in to increase the textile filtration efficiency and establish new properties in order to meet upcoming necessities of modern societies. The most practical achievement is to take advantage of other material properties such as the gas absorption capacity of activated carbon. The combination of NW filters and activated carbon can effectively remove many gaseous pollutants, such as NO2 and CO, from the air and provide relatively acceptable air quality [9–12]. One disadvantage of such modified filters is the accumulation of pathogenic microbes inside the filter [13–16]. Furthermore, as they are used in the moist relatively warm environment, filter substrates possess great potential to stimulate microbial growth. On the other hand, the spread of chronic diseases such as asthma and influenza through the air can provide enough motivation to develop a new generation of filters with antimicrobial activity [17–21].
Some metals have oligodynamic effect (toxic effect on living cells, spores, molds and viruses), like mercury, silver, aluminum, lead and gold. Silver is considered as a more acceptable antibacterial agent in comparison with other antibacterial agents due to its economical aspects, efficiency and it has also been proved that its harmful effects on the human body are less [22–24]. As the size of silver particles decreases down to the nanoscale, their specific surface area and hence antibacterial efficacy increase considerably [25]. Acceptable activity of nanosilver (NS) particles against a wide range of microbes can be considered as one of the most potential antimicrobial property of NS to solve the aforementioned problems. Many studies have been conducted in this field. Filter layers have been achieved by doping fibers with such nanoparticles and plates, or textile fabric surfaces—whether woven or NW—coated with NS particles [26–32].
However, few studies have considered a filter with both absorption ability of harmful gases and pathogenic microbes at the same time. The present study aimed to produce a filter with these two properties. To do this, polypropylene NW layers were coated and treated with activated carbon and NS particles. The efficiency of fabricated filters was statically and dynamically tested against Staphylococcus aureus and Escherichia coli as the representatives for gram-positive and gram-negative bacteria, as well as against NO, NO2 and CO gases as the most conventional gaseous pollutants.
Experimental
Determination of minimum inhibitory concentration
Minimum inhibitory concentration (MIC) determination using the broth dilution method was followed to evaluate the antimicrobial effects of colloid NS [33]. NS (solution in water Cat. # PL-Ag-S10-10 mg, 10 nm diameter, Plasma Chem., GmbH, Germany) was freshly prepared before each experiment. A total of 1 mL of NS solution (0.1 mg/mL) was added to 1 mL of brain heart infusion broth (BHI broth; Merck, Darmstadt, Germany) and this mixture was serially diluted (two-fold) in the broth. S. aureus was precultured in BHI broth aerobically, 3–4 h prior to the test. Inoculum was adjusted to contain 108 colony forming units (CFU)/mL as measured by a spectrophotometer (OD600 0.7–0.8). Next, 0.1 mL aliquots of the inoculum were added to 0.9 mL of each of the NS broth serial dilutions. The test tubes were then incubated at 35–37°C in an aerobic atmosphere for 24 h. The MIC of NS was defined as the lowest concentration at which no visible bacterial growth could be detected. The test was performed three times. Similar results indicated the MIC of NS against S. aureus [33].
Impregnation of layers with powder activated carbon and colloidal NS
Samples with different concentration of active carbon and nanosilver particles.

Bacterial strains
The effects of the antimicrobial activity of samples were evaluated against S. aureus (ATCC 25923) and E. coli (ATCC 35218) in a ‘static state’ and against passing microbes in a ‘passing state.’ AATCC147-2004 standard test method was used for the static state [34]. S. aureus and E. coli were rehydrated by BHI broth and subcultured in nutrient agar (Merck, Darmstadt, Germany), then incubated at 37°C for 24 h. Colonies were harvested and suspended in BHI broth. The turbidity was adjusted to an equivalent of 0.5 McFarland turbidity standard using either bacteria suspended in BHI broth or just pure BHI broth (a turbidity of 0.5 McFarland units corresponds approximately to 1.5 × 108 CFU/mL). To achieve the lawn of bacterial growth, 0.1 mL of the suspension was inoculated by a sterile cotton-tipped swab onto the surface of a nutrient agar plate [35].
Antimicrobial activity of samples
Sterile samples were cut into 25 × 50 mm, placed directly on the inoculated plates, and incubated at 37°C for 24 h. The zone of inhibition around the specimen was determined with Equation (1) and the growth interruption was investigated.

An experimental setup was assembled for testing the antimicrobial activity of filters in the passing state (Figure 1). The air (7 cm3/min) was, respectively, pumped through the specimen and a high-efficiency particulate air (HEPA) filter (for collecting the microbes passed). The bacteria (0.5 McFarland E. coli and S. aureus) were injected through the inlet located before the sample injection point, hence, the growth of microbes on the samples and HEPA filters could be reported.
Set up for the evaluation of antimicrobial activity of specimens in air passing state: 1. compressor; 2. flow meter; 3. air inlet; 4. bacteria injection inlet; 5. specimen and its holder; 6. HEPA filter and its holder; 7. air outlet; 8. connecting tubes.
Air permeability test
Air permeability of a fabric is defined as the volume of air passing through a defined area under a constant pressure. Samples were tested by an air permeability tester (M021S, Shierly Co, UK). According to BS5636 (British standard method for determination of the permeability of fabrics to air), the volume of air passing through a sample can be determined through the area of 508 ± 1 mm2 at 50 Pa [36].
Pollution test
An experimental setup was designed according to Figure 2. Environmental air with specific pollutant was pumped up (25 L/min) by a fan into the main pump and the specimen. An environmental monitoring station (Babuc-A, LSI-LASTEM Co, Italy) was placed at the end of the pipe. Only the specimen coated with activated carbon (100 g/L) and NS (1000 ppm) were used for this test.
Schematic of pollution test set up.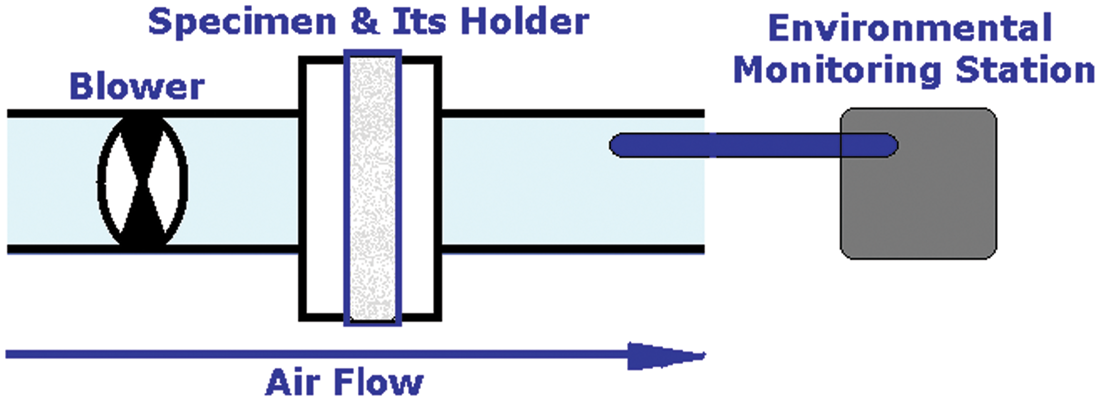
Morphological investigation
Samples coated with gold by a sputter coater (Fison Instruments, UK) and their morphological characteristics were investigated by a scanning electron microscope (SEM; LEO 440i, UK) at a voltage of 20 kV.
Results and discussion
Active carbon content of padded samples (wet pick up for all samples was the same 70%).

The MIC of NS was 62.5 ppm. As it is shown in Table 3 (also Figure 3), the raw layers coated with 1000, 500 and 125 ppm concentrations of NS particles showed antimicrobial properties. In contrast, such properties for layers coated with 250 ppm concentration of both activated carbon and NS particles were observed only in A100S1000 sample. This may be attributed to the absorption of NS particles by activated carbon, leading to the decreased antimicrobial activity.
Zone of prohibition for polypropylene samples sprayed with silver nano colloids with different concentration: (a) 1000 ppm, (b) 500 ppm, (c) 250 ppm, (d) 125 ppm, (e) 62.5 ppm. Inhibition zone of samples (wet pick up for all samples was the same 70%).

The antimicrobial activity test results in air passing state with Escherichia coli.

The antimicrobial activity test results in air passing state with Staphylococcus aureus microbe.

Air permeability test results of the various layers.

Each test repeated five times and the means reported.
The samples coated only with NS particles showed no reduction in the volume of pollutants passing through them (Tables 4 and 5). However, the samples treated with activated carbon caused a reduced volume of pollution for NO, CO and NO2 by about 100%, 38.7% and 100%, respectively (only A100S0 sample was tested; Table 2).
Figure 4(a) shows a micrograph of powder activated carbon. The distribution of powder activated carbon on the fiber surfaces was dispersed thoroughly (Figure 4(b)). NS particles were placed on both fiber surfaces and particles of activated carbon (Figure 4(c)). But, due to the pore size of activated carbon and the size of silver nanoparticles, some particles may enter into activated carbons, and thereby, the reduction of antimicrobial properties can be predicted. Figure 4(d) shows the bacterium E. coli bacterial trapped on the surface of treated sample. Figure 4(e) shows the growth of S. aureus on the surface of raw polypropylene layer.
SEM images: (a) active carbon powder; (b) layer coated with active carbon; (c) layer coated with active carbon and nano silver 500 ppm; (d) trapping of E. coli on layer coated with active carbon and nano silver 500 ppm; (e) the growth of S. aureus on layer coated with active carbon and nano silver 500 ppm.
Conclusions
It was shown that coating the polypropylene NW layer with NS particles could deactivate the microorganism like S. aureus and E. coli. On the other hand, layer coated with activated carbon absorbed pollutant gases like NO, CO and NO2 from air passing through the layer. The presence of activated carbon and silver particles on a surface such as the surface of polypropylene fabric together would reduce the antimicrobial activity of silver particles. However, no remarkable effect was observed on the absorption ability of activated carbon. It can be concluded that to possess two properties simultaneously, antimicrobial and pollutant absorption; activated carbon and silver particles should be placed on two different layers. The fabricated NW seems proper for air filters, especially for respiratory ones which need further studies.
Footnotes
Funding
The work is supported by Textile Research Center at South Tehran Branch, Islamic Azad University and also by Jihad research Center at Tehran University.
