Abstract
Being a versatile biopolymer, chitosan has been used to synthesize silver nanoparticles (SNPs) impregnated hybrid chitosan-nanosilver bionanocomposite (CSSNC) films
Introduction
There have been various diseases causing pathogens gradually devastating human health. The reason mainly due to the drug resistance in microorganisms as well as the uncontrolled antibiotics consumption lead to the impediment in disease management and hence requires effective and safe antimicrobial agents. According to the recent survey of the World Health Organization, the number of cancerous patients has been increased annually with 8.8 million deaths were recorded in 2015 and expected to be around 22 million by 2030.1,2 Chemotherapy is gradually replaced by natural product-based treatment which generally includes biopolymer-based materials owing to their magnificent antibacterial activity against pathogens without being imposing any adverse effect on the system. That’s why there seems a progress in utilization of biopolymers in biomedical sciences for renewable, sustainable, and environment benign substitute for petrochemicals.3,4
Chitosan (CS), a versatile biopolymer having similar structure like cellulose except the amino (–NH2) group present at C-2 position seems to be a suitable polysaccharide having almost all the required properties for its utilization in biomedical field.5,6 It has significant antimicrobial activity due to its polycationic nature which bind with negatively charged peptidoglycan bacterial cell wall and forestalls the transcription and lipid synthesis. Due to its beneficial and respectable antimicrobial activity against various pathogens and anticoagulating activity, 7 CS has found tremendous utilization in wound healing, 8 drug release, 9 tissue engineering, 10 sutures, medical textiles, 11 3D-bioprinting, etc.12,13
Besides, silver nanoparticles (SNPs) also exhibit excellent antibacterial activity against pathogens. Silver is toxic against microorganisms and so as for pathogenic bacteria present in human being. 14 Silver in nano dimension lead to the high surface to volume ratio and hence possess enhanced antimicrobial activity against various microorganisms. Moreover, the SNPs fabricated by green method make it suitable and less hazardous due to the active phytochemicals present in plants and its part.15,16
In this study, CS based in-situ and ex-situ silver nanocomposite has been successfully fabricated, characterized and examined for antibacterial activities against the most common Gram negative

Probable mechanism of interaction of CS and Ag NPs in ex-situ CSSNC.
Materials required
For this study, CS high MW, >75% degree of deacetylation was purchased from Himedia Chemicals, Mumbai, India. Analytical grade glacial acetic acid (99–100%) was purchased from Fischer Scientific, Mumbai. Silver nitrate was obtained from Merck, India. Chemicals including L-Ascorbic acid (PubChem CID: 54670067), Griess reagent, Trichloroacetic acid (TCA), Sodium nitroprusside, Ferric chloride, Hypochlorous acid, Dimethyl sulphoxide (DMSO), 2,2-diphenyl-1-picrylhydrazyl (DPPH) and MTT that is 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, all were procured from Sigma-Aldrich, Mumbai, India and used as obtained without further purification. Muller Hinton agar and Luria broth was purchased from HiMedia, Mumbai. HeLa cervical cancer cell line were acquired from National Cell Repository, NCCS, Pune, India. Glasswares used were dried in oven after being washed with chromic acid and double distilled water.
Experimental methods
Fabrication of CS based ex-situ nanocomposite
1% CS (w/v) solution was prepared by dissolving CS flakes in 1% (v/v) acetic acid solution and kept under stirring for 5 h, at 25°C. To this solution, SNPs synthesized from
Fabrication of CS based in-situ nanocomposite
CS 1% (w/v) was prepared as described above and 4 mM AgNO3 and
Characterization
The ex-situ and in-situ CSSNC films were characterized for maximum absorption intensity in the range of 200–600 nm under UV-Vis spectroscopy by using Shimadzu, 1800 spectrophotometer, Japan. Distilled water was taken as reference for the same. In order to determine the interaction of nanoparticles with the CS, FT-IR spectra were recorded by using the instrument, Bruker Tensor 37 spectrometer in the range of 500–4000 cm−1. HR-FESEM analysis of CS and CS based different nanocomposites films was carried out using ZEISS, Model V5.05 (Sigma) to confirm the presence of SNP’s. Wide angle X-Ray diffraction was observed to detect the crystalline phase of bionanocomposite films using Rigaku Ultima IV X-Ray Diffractometer furnished with graphite monochromator and Cu Kα radiation (λ = 1.5415 Å) functioned at 30 mA, 45 kV and 25°C. The moisture content was determined by measuring weight loss after drying the films in an air-circulating oven at 100 ± 5°C for 24 h. The moisture content was calculated as the percentage of water in the films using equation (1).

where, Ww is the weight of films initially at room temperature and Wd is the weight of films after drying. The surface properties of ex-situ and in-situ CSSNC films were also examined for hydrophilicity by means of static contact angle measurements using an optical contact angle analyzer, SEO Phoenix 150 at room temperature using sessile drop method. The thermal stability of the CSSNC films was measured by using the instrument TG/DTA6300 (SII EXSTAR6000).
Biological assays
Antibacterial screening
The antibacterial activity of synthesized nanocomposites was tested against two different bacterial strains i.e.
Determination of in vitro antioxidant activity
In vitro antioxidant activity of pure CS, plant extract bioreductant (EXT) and CSCNC were estimated by variety of free radical scavenging assays. Assay of standard antioxidants L-AA was performed along with all free radical scavenging assays.
DPPH assay
Radical scavenging activity of (CS, EXT and CSSNC) and standard antioxidant L-AA were assessed by using Blois method. 17 Forty microliter of working stock containing 10, 20 and 50 µg/mL of (CS, EXT and CSCNC) and standard antioxidant were taken in separate tubes. The volume of reaction mixture was adjusted to 2 mL by adding methanol. Thereafter 150 µL of 0.1 mM methanolic solution of DPPH was added to all tubes and vortex-mixed. The tubes were kept at 25°C for 20 min. The control was prepared without (CS, EXT and CSCNC) and a mixture of 40 µL of DMSO and 1.960 mL methanol was used for the baseline optical density correction. At 517 nm, the changes were observed for different compounds and were expressed in percent inhibition to obtain the radical scavenging activity from equation (2). Besides, the concentration of phytochemicals required to decrease 50% in optical density with reference to control referred as inhibition concentration (IC50) was also observed. 18

NO radical assay
Nitric oxide radical scavenging activity was measured by the Griess Illosvoy reaction. 19 NO production occurs on decomposition of sodium nitroprusside at physiological pH 7.2 in aqueous solution. Under aerobic conditions, NO reacts with oxygen produces stable products (nitrate and nitrite). Griess reagent is used to quantify these products. Nitric oxide scavengers compete with oxygen which leads to reduced nitrite ions production. For performing the assay, 1 mL of phosphate buffered saline (10 mM at pH 7.4) containing 10 mM sodium nitroprusside was added to 40 µl of DMSO containing 10–50 µg of (CS, EXT and CSCNC) and standard antioxidant was added separately and vortex mixed. This reaction mixture was incubated at 30°C for 2 h. After completion of incubation, 0.5 mL of (1% sulphanilamide and 0.1% N-(1-naphthyl)ethylenediamine dihydrochloride in 5% H3PO4) i.e. Griess reagent was added to each reaction mixture. Furthermore, a chromophore formed due to diazotization of the nitrite with sulphanilamide followed by coupling with naphthylethylenediamine dihydrochloride was read at 550 nm. Inhibition of nitrite formation by (CS, EXT and CSCNC) and the standard antioxidant L-ascorbate were calculated relative to the control. The formula used for calculating percent inhibition of nitric oxide radical generation is given in equation (3).
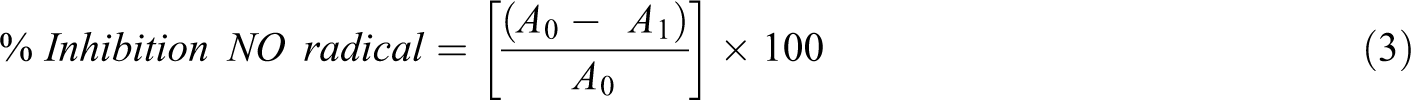
where A0 is the absorbance of control and A1 is the absorbance with (CS, EXT and CSCNC) and standard. IC50 value is the amount of sample (µg) required to scavenge 50% NO radical and was obtained from inhibition curve. 17
Total reduction capability
The reducing capacity of CS, EXT and CSCNC was determined by method as utilized by Aiyegoro
Cytotoxic studies
The MTT [3-(4,5-dimethyl-2-yl)-2,5-diphynyl tetrazolium bromide] assay was done to assess the cytotoxic effects of CS, EXT and CSCNC on HeLa cervical cancer cell line. Cells (1 × 104) per well were seeded overnight and 10 to 50 µg of standard antioxidant present in 40 µl of DMSO was supplemented in incubation media and incubated for 48 h. After that, 20 µL of MTT (5 mg/mL in PBS) was incorporated in the well plate and kept under 5% CO2 incubator at 37°C for 4 h. As a result, purple colored formazan (solubilized in DMSO (150 µL/well) at 570 nm) were obtained which clearly indicates the active mitochondrial dehydrogenase helps in reduction and thus allowing further studies for cell viability. 22
Results and discussion
Moisture content
Pure CS showed highest moisture content as compared to bionanocomposite films (Figure 2(a)). This might be due to interaction of water molecules with –OH and –NH2 groups of pure CS, but in case of bionanocomposite films of different compositions, there were less availability of free –OH and –NH2 functional groups due to the presence of Ag0 ions cross-linked with polymer matrix. There is decrease in percentage of moisture content with the increase in concentration of Ag0 ions in CS matrix (both ex-situ and in-situ) which is in good agreement with the literature. 23

(a) Moisture content % and (b) contact angle measurements of all the ex-situ and in-situ CSSNC.
Contact angle measurements
In order to determine the wettability of the CSSNC, contact angle measurements indicated the degree or extent of wetting when liquid and solid film surface interacted. Hydrophilicity of CSSNC having small contact angles attributed to have high wettability, while those possess large contact angles have low wettability. Figure 2(b) shows the average contact angle of the bionanocomposites, out of which CS9: AGE1 nanocomposite film had highest contact angle (>90°) while 1% CS film had the lowest (60°). This clearly indicates that SNPs embedded in CS matrix lead to decrease its hydrophilicity. 24
Thermogravimetric analysis
The TGA of pure CS and silver-loaded CS films have been reported as shown in Figures 3(a) and 4, respectively. Pure CS showed initial weight loss due to moisture and then decomposed at 280°C with 40% complete decomposition at 580°C, also reported by Thomas et al. 25 The CSSNC films, both ex-situ as well as in-situ showed decomposition of films into two steps (i) vaporization of moisture present in films from 75 to 80°C and (ii) the characteristic decomposition temperature from 190 to 260°C having broad peak (Figure 4). These decomposition temperature ranges were attributed to (i) evaporation water from all the films including pure CS whereas (ii) attributed to the degradation of CS polysaccharide chains and its deacetylated units. The pure CS possess low thermal stability as compared to its nanocomposites which showed their 40% weight loss at around 580°C due to the presence of SNPs in both the ex-situ and in-situ films. The results further extended by DTG as endothermic and exothermic peaks were observed.
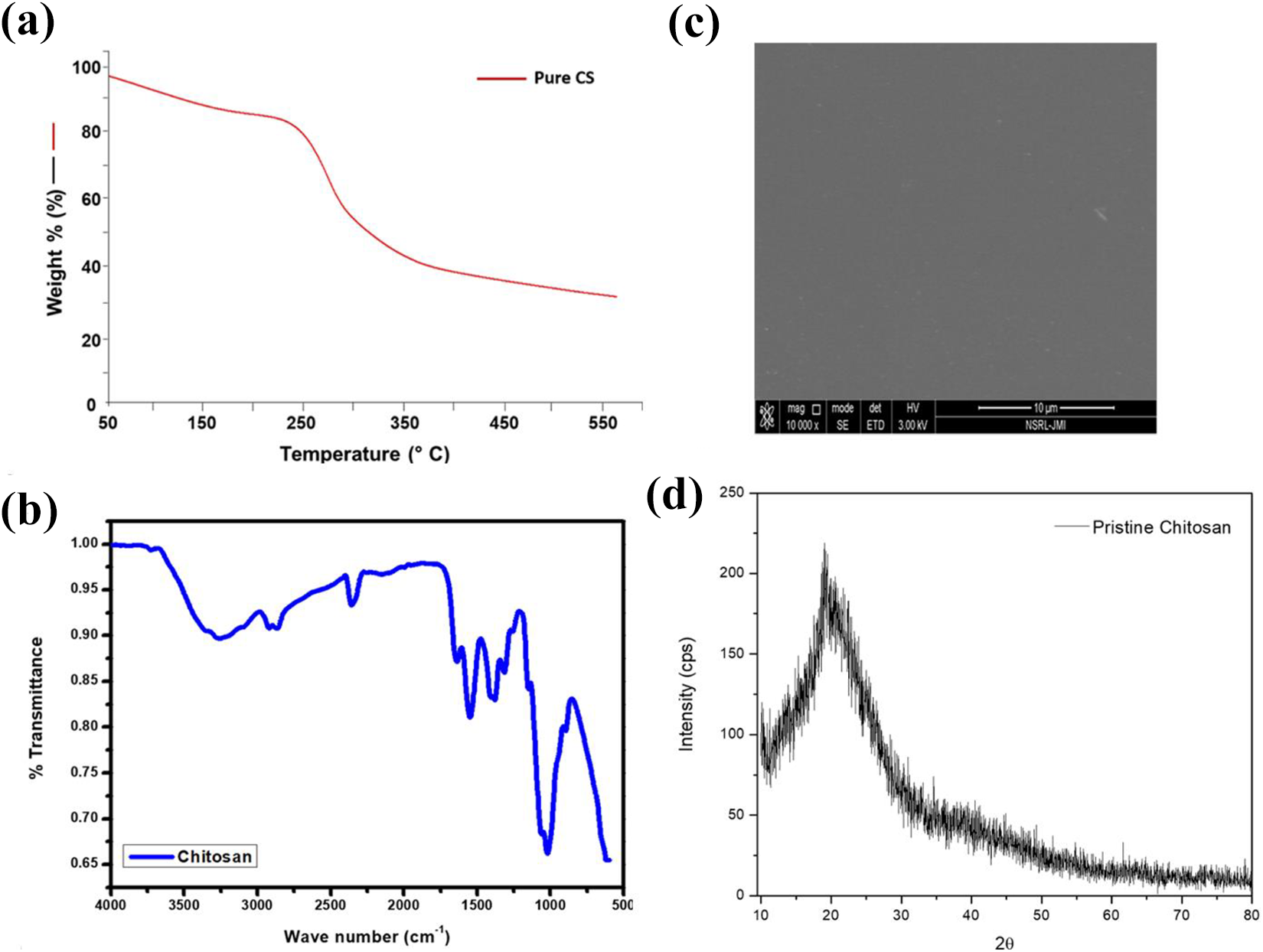
(a) TGA, (b) FT-IR spectra, (c) SEM image, and (d) XRD pattern of pure CS.
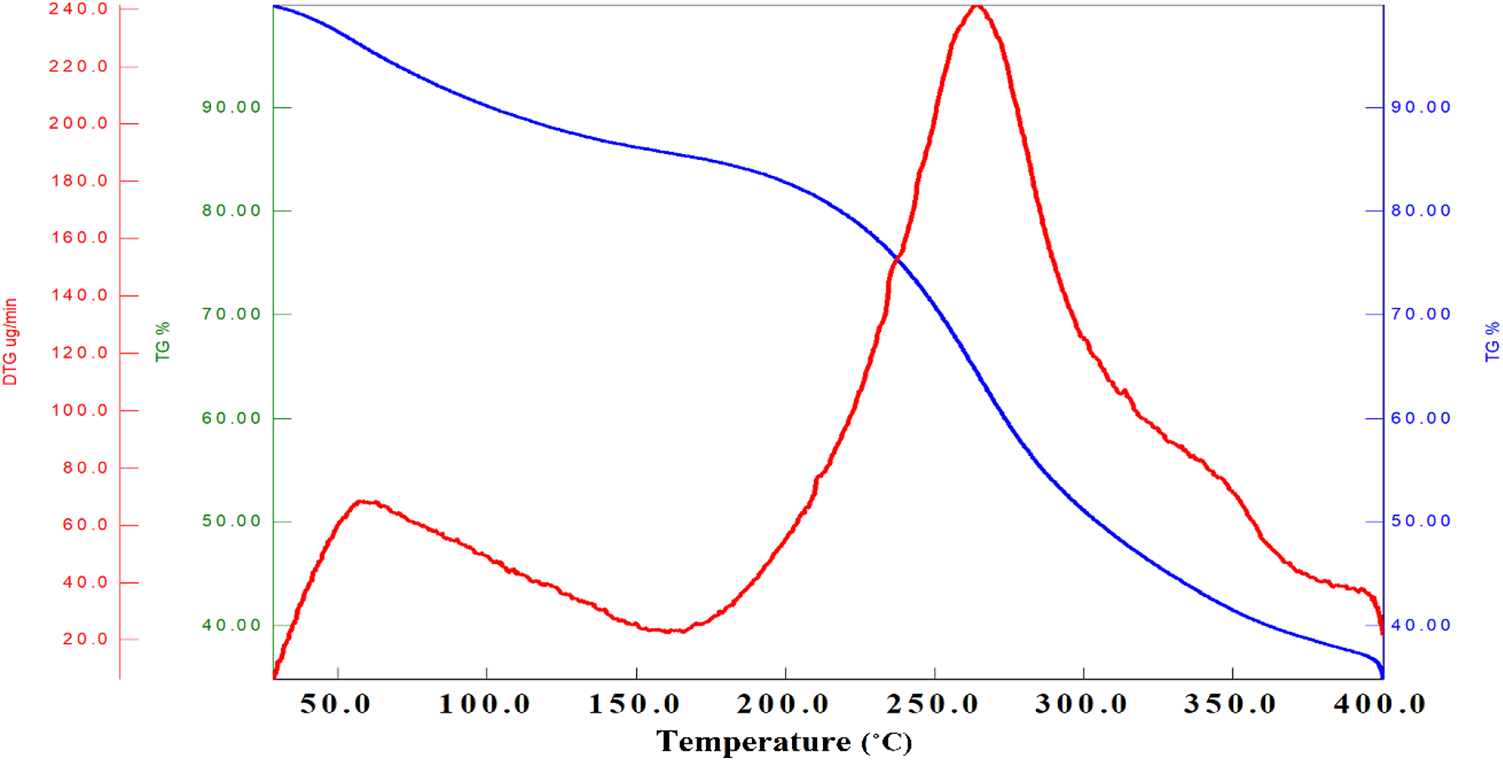
Thermal analysis of CSSNC.
UV-visible spectroscopy
The CSSNC films prepared by ex-situ and in-situ methods have shown the characteristic peak of SNPs at 440 nm and 480 nm, respectively (Figure 5). Since the color of pure CS film was slightly yellowish while CSSNC films appeared brownish, hence it was confirmed due to the Surface Plasmon Resonance (SPR band) of silver that SNPs formed successfully and thus impregnated within CSSNC films. This indicates that the nanoparticles prepared externally for ex-situ nanocomposite possessed high intensity which may be due the presence of larger number of SNPs formed as compared to in-situ.
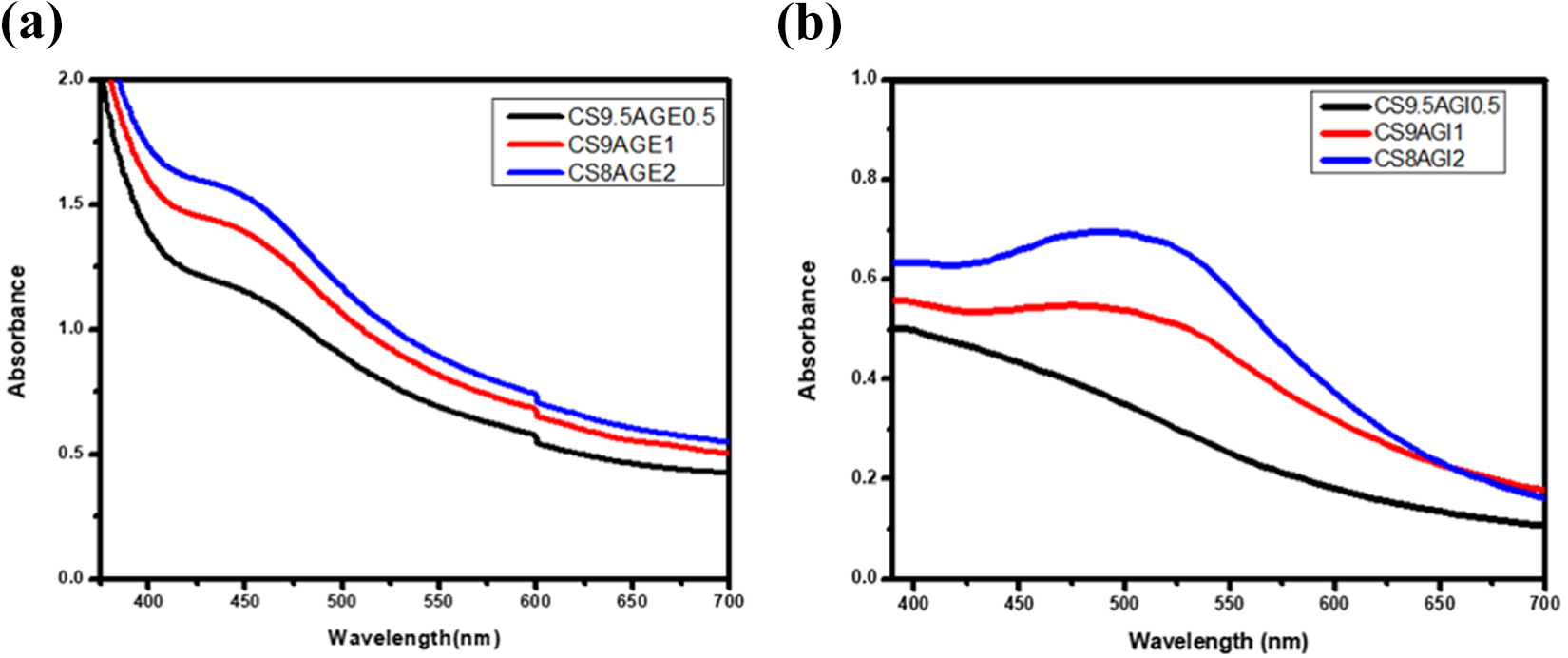
UV-visible spectra of (a) ex-situ and (b) in-situ CSSNC films.
FT-IR spectroscopy
The FT-IR spectrum of pure CS (Figure 3(b)) shows O–H and N–H stretching vibration ∼3000–3500 cm−1, C–H stretching at 2922 cm−1, C=O stretching (amide I) at 1640 cm−1, N–H bending (amide II) at 1549 cm−1, O–H bending at 1405 cm−1, C–N stretching (amide III) at 1322cm−1 and C–O–C stretching at 1026 cm−1. The FT-IR spectrum of ex-situ (Figure 6(a)) and in-situ (Figure 6(b)) CSSNC shows the shifting of N–H bending 1549 cm−1 to lower wave number 1539 cm−1 which confirmed the interaction of amino groups with SNPs. Furthermore, an increase in intensity of C–O stretching and decrease in intensity of O–H group has been observed indicating this interaction by weak intermolecular forces.1,26

FT-IR spectra of different composition of (a) ex-situ and (b) in-situ CSSNC films and XRD pattern of (c) ex-situ and (d) in-situ CSSNC films.
X-ray diffraction studies
The crystal structure of ex-situ and in-situ CSSNC was determined by XRD. Pristine CS shows its characteristic peak at 2θ = ∼20° (Figure 3(d))26,27 and the broadening of this peak may be due to its amorphous nature. Commercial SNPs shows XRD pattern at 2θ = 38.4°, 44.5°, 64.7°, and 77.6°, attributing respectively to the [111], [200], [220], and [311] planes of face-centered cubic (fcc) structure of silver. 28 Figure 6(c) and (d) shows that all the ex-situ and in-situ CSSNC exhibited same XRD diffraction pattern at ∼2θ = 7.7°, 37.3°, 43.7°, 63.6° and 76.9°. The diffraction peak obtained at 7.7° indicated the peak for CS and 37.3°, 43.7°, 63.6° and 76.9° were of fcc SNPs which attributed to planes (111), (200), (220) and (311) and are in good agreement with JCPDS file no. 89-3722 and available literature. 29
Surface morphology analysis
SEM was used to analyze the surface morphology of ex-situ and in-situ CSSNC films. The SEM image of pure CS showed a clear and smooth surface and even pattern indicating the solution was homogenized thoroughly (Figure 3(c)). SEM images of CSSNC film clearly indicates the spherical and uniform SNPs were embedded in the bionanocomposites of different composition as depicted in Figure 7. It shows that the SNPs in ex-situ films (Figure 7(a) to (c)) were well embedded in the CS matrix as compared to in-situ nanocomposite films (Figure 7(d) to (f)). These improved non-aggregated results revealed the compact distribution pattern of SNPs inside the CSSNC films of varying composition which better agreed with the result of Govindan et al. and Jung et al.26,30

SEM images of ex-situ (a to c) and in-situ (d to f) CSSNC films.
Antibacterial assay
On investigating the antibacterial activity of pure CS and CSSNC films against aforementioned bacterial strains, the ex-situ and in-situ CSSNC possess high zone of inhibition as compared to pure CS (Figures 8 and 9). The ex-situ CSSNC exhibited high antibacterial activity against

Zone of inhibition exhibited by ex-situ and in-situ CSSNC films.

Antibacterial study of (a and b) in-situ and (c and d) ex-situ CSSNC against
In vitro antioxidant activity
The ability to scavenge the free radicals generated in the system were explained in Figure 10(a) to (c) showing the antioxidant behavior of pure CS and ex-situ and in-situ CSSNC films. Since, the stability of ex-situ CSSNC were higher as compared to in-situ CSSNC and CS9AGE1 (CSBNP) was found to be the most stable as well as possess favorable physicochemical activities, therefore, it was optimized for further biological assays with pure CS and EXT.

Free radical scavenging activity of L-ascorbate, CS, EXT and CSBNP on (a) DPPH and (b) nitric oxide radicals (c) total reduction capability and for comparison L-ascorbate was considered as standard antioxidant and (d) anticancer activity of CS, EXT and CSSNC against HeLa cancer cell. Values are mean ± SD of three parallel measurements.
DPPH assay
This assay determines free radical scavenging activity of CS, EXT, CSBNP using DPPH in order to explore the efficacy of antioxidants present in aforesaid compounds so as to reduce purple DPPH radical to yellow DPPH in its non-radical form. This assay revealed a remarkable dose-dependent inhibition of DPPH radical upon addition of 10, 20 and 50 µg mL−1 concentration of each compound


Nitric oxide radical scavenging activity
Figure 10(b) shows that CS, EXT, and CSBNP exhibited moderate dose-dependent nitric oxide radical scavenging activity with concentrations 10, 20 and 50 µg mL−1 with an IC50 value of 48.26 ± 2.48, 38.16 ± 2.10 and 18.26 ± 1.98 µg mL−1 respectively, as compared to 51.43 ± 2.63 µg mL−1 for L-ascorbate (reference compound), (
Total reduction capability
In this study, the reducing capacity of CS, EXT and CSBNP were expressed as absorbance values increased from 0.731 ± 0.167, 0.65 ± 0.12 and 0.22 ± 0.11 at 10 µg mL−1 to 3.21 ± 0.123, 2.51 ± 0.103 and 1.41 ± 0.133, respectively at 40 µg mL−1 (Figure 10(c)). Among all the desired compounds, L-ascorbate showed highest reductive capacity at concentration range from 10 to 40 µg mL−1, with increased absorbance from 0.964 ± 0.167 to 3.42 ± 0.167 as the compounds shattered off on further rise above 40 µg mL−1 concentration. The order of reduction capability of CS, EXT, CSBNP and the standard L-ascorbate at 40 µg mL−1 were: L-ascorbate (3.42 ± 0.167) > CS (3.21 ± 0.123) > EXT (2.51 ± 0.103) and CSBNP (1.41 ± 0.133), (
Cytotoxic studies or anticancer activity
In order to assess the cell viability of CS, EXT and CSBNP compounds, MTT assay was performed with HeLa cell lines by treating with different concentration (10, 20 and 40 µg mL−1) of compounds (Figure 10(d)). This study revealed that the pure CS was not toxic at all the concentrations while the CSBNP exhibited low cell viability (22%) for cancerous HeLa cells with increasing concentration in dose-dependent manner especially, at highest concentration of 50 µg mL−1 after 24 h of treatment. As far as EXT was concerned, it decreased the percent cell viability for cancerous HeLa cells as compared to pure CS but less than the CSBNP which may be due to the active phytochemicals present in plant extract. Since, silver is toxic toward cancerous HeLa cells causing their death with low cell viability, hence when immobilized in biopolymeric matrix of CS to form bionanocomposite, it possesses significant anticancer activity. 31 The normal human peripheral blood mononuclear cells have shown about 98–100% cell viability against green synthesized SNPs already reported in previous work. 16
Conclusions
Apart from the toxicity of SNPs which limit its therapeutic applications, their potential to induce oxidation stress, cytotoxicity and genotoxicity indulge SNPs to be exploited in medical field as bionanocomposites being biosafety assessment. This study presented two different green approaches toward the synthesis of ex-situ and in-situ CSSNC (six different compositions) and were characterized for their confirmation of immobilization of SNPs inside the CS matrix. These CSSNCs were then examined under antibacterial assay against
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
