Abstract
Objective:
Multiple daily injections therapy in older adults with diabetes can negatively impact glycemic control and comorbidities. This issue may be overcome with advanced diabetes technology that reduces hypoglycemia and hyperglycemia. The present study evaluated real-world glycemic outcomes of a United States (US) cohort ≥65 years using the MiniMed™ 780G (MM780G) advanced hybrid closed-loop system.
Methods:
CareLink™ personal data as of December 18, 2024, for U.S. MM780G system users ≥65 years, were de-identified and analyzed. Metrics, including time in range (TIR 70–180 mg/dL), time in tight range (TITR 70–140 mg/dL), time below range 70 mg/dL (TBR70), and time above range 180 mg/dL and 250 mg/dL (TAR180 and TAR250, respectively), with and without recommended optimal settings (ROS, 100 mg/dL glucose target with 2 h active insulin time) were determined. Subanalyses based on age group (≥75 years) and type 1 diabetes (T1D) or type 2 diabetes (T2D) were, also, conducted.
Results:
The overall cohort (n = 8542) had a mean TIR, TITR, TBR70, TAR180, and TAR250 of 78.4%, 51.4%, 0.9%, 20.7%, and 3.6%, respectively, with a 6.8% glucose management indicator. For ROS users (n = 2753), TIR and TITR were higher (81.9% and 55.9%, respectively, P < 0.001), and TAR180 and TAR250 were lower (17.2% and 2.5%, respectively, P < 0.001). Data trended similarly among the population aged ≥75 years, and no differences were observed between T1D and T2D.
Conclusions:
In a real-world setting, a U.S. cohort aged ≥65 years using the MM780G system achieved consensus-recommended glycemic targets. Use of ROS enabled more users to achieve an even higher level of glycemic control.
Keywords
Introduction
According to the Centers for Disease Control and Prevention, an estimated 29.4 million people aged ≥18 years were diagnosed with diabetes (8.9% of the United States [US] population) in 2021. 1 Furthermore, the number of adults aged ≥65 years with diagnosed diabetes has increased to an estimated 13.8 million people (24.4% of the population aged ≥65 years). 1 A recent publication has estimated that there will be roughly 400,000, 450,000, and 530,000 people with type 1 diabetes (T1D) in the group aged ≥65 years by 2024, 2028, and 2033, respectively. 2 The increased incidence over an increasing life expectancy means higher diabetes prevalence in the Medicare population who may experience nondiabetes-related cognitive and physical impairment. Also, traditional insulin intensification therapy is known to cause side effects such as hypoglycemia that can lead to an increased rate of hospitalization for severe hypoglycemia. 3 Risk factors for hypoglycemia in this age group4–6 can be mitigated with proper education and carbohydrate counting and with advanced diabetes technologies (e.g., continuous subcutaneous insulin infusion [CSII] and continuous glucose monitoring [CGM]) that improve glycemic control.7,8 Despite this evidence, current coverage criteria of the U.S. Centers for Medicare & Medicaid Services (CMS) may impede access to CSII therapy for many Medicare beneficiaries with insulin-treated diabetes who would otherwise benefit from insulin pump use. 3 In addition, the American Diabetes Association (ADA) now recommends automated insulin delivery (AID) systems as the preferred method for improving glycemic outcomes in individuals with T1D and other forms of insulin-deficient diabetes (irrespective of C-peptide test results). 9
While glycated hemoglobin (HbA1c) is the most well-established diabetes outcome measure and is considered the key predictor for the development or progression of long-term complications in people with T1D and type 2 diabetes (T2D), it does not provide information about daily real-time glycemia or capture the impact that glucose excursions may have on quality of life. 10 Time in range (TIR) is a CGM-derived measure of the percentage of time a person spends in a target glucose range (70–180 mg/dL) as defined and recommended by the ADA 9 and international consensus. 11 Thus, the therapeutic HbA1c goal of <7% (53 mmol/mol) has been equated to a TIR of greater than 70%, whereas a TIR of at least 50% (an HbA1c corresponding to 8% [64 mmol/mol]) has been recommended for older or high-risk individuals. 11 TIR is recommended by medical societies as complementary to HbA1c10,12 and is a key metric of the quality of short-term glucose control and acute complications. 10
It is also important to consider that HbA1c cannot predict the short-term frequency and/or risk of experiencing hyperglycemia or hypoglycemia and that reducing the percentage of time spent below target glucose range (time below range [TBR]) defined as <70 mg/dL to <1% (versus <4%) is recommended for older/high-risk adults with diabetes. 13 Preventing excessive time above target glucose range (time above range [TAR]) defined as >250 mg/dL to <10% is also recommended for older/high-risk adults. 11 A recent study, HYPOAGE, in people with T2D older than 75 years showed that 30% of participants had a TAR250 higher than 10% and it was associated with higher mortality. 14 Safely reducing as much time as possible in hyperglycemia and hypoglycemia should, therefore, become a priority for managing T1D and T2D in adults aged ≥65 years.
For individuals with T1D or insulin-treated T2D, AID is a technologically advanced diabetes therapy, because it can automatically adjust (increase and suspend) insulin doses as a function of sensor glucose (SG) levels. The MiniMed™ 780G (MM780G) system is an advanced hybrid closed-loop (AHCL) AID system that received Conformité Européenne mark in June 2020 and was approved by the U.S. Food and Drug Administration in April 2023. The MM780G system includes an adaptive model-based (proportional integral derivative) controller that uses target glucose settings (100, 110, or 120 mg/dL and a temporary target of 150 mg/dL) and active insulin time (AIT) settings (2–8 h) to automatically deliver basal insulin and autocorrection insulin doses up to every 5 min, based on SG readings. System users estimate and announce the meal carbohydrate amount. To increase the effectiveness of the MM780G algorithm, the use of recommended optimal settings (ROS, a glucose target of 100 mg/dL for at least 95% of time and an AIT of 2 h for at least 95% of time) is suggested for adult populations.15–17
Several real-world data analyses and randomized clinical trials17,18 have shown that MM780G system users can meet or exceed all consensus-recommended glycemic targets, but no specific analysis has been conducted on a large population of older adults. The present real-world data analysis evaluates the impact of the MM780G system on glycemic outcomes in a U.S. population with T1D or T2D aged ≥65 years.
Methods
This retrospective study included de-identified MM780G system data from the U.S. CareLink™ personal platform, as of December 18, 2024. The evaluation included 8542 MM780G system users aged ≥65 years who self-reported T1D (n = 6249) or T2D (n = 1701) diagnosis and who consented to share their data. Data were analyzed by age group (≥65 and ≥75 years) per self-reported year of birth. HbA1c and adverse events were not available for analyses.
Statistical methods
To be included in the analysis, MM780G system users had at least 180 days of pump usage and at least 10 days of CGM data after initiating AHCL. Glycemic outcomes included the percentage of TIR (70–180 mg/dL), time in tight range (TITR, 70–140 mg/dL), TBR (<54 and <70 mg/dL), and TAR (>180 and >250 mg/dL) and the glucose management indicator (GMI). The percentage of users meeting: (1) TIR of >70%; (2) TBR70 of <4%; (3) TIR of >70% and TBR70 of <4%; (4) TIR of >50%; (5) TBR70 of <1%; and (6) TIR of >50% and TBR70 of <1% was determined. In addition, insulin delivery information, including total daily dose (TDD) of insulin, total bolus insulin, and autocorrection insulin in units and as percentages of TDD, was summarized. Data are presented by two age groups (≥65 years and ≥75 years) and by T1D and T2D; for an overall, ROS and non-recommended optimal settings (NROS) group. ROS users were designated as having a glucose target of 100 mg/dL for at least 95% of time and an AIT of 2 h for at least 95% of time. Means and standard deviations were used for continuous variables, and number of users and proportions were used for categorical variables. The comparisons between NROS and ROS users were performed using two sample t test, Wilcoxon signed-rank test, and Fisher exact test, as needed. All P-values reported are two-sided, and all analyses were conducted using SAS software, version 9.4 (SAS Institute, Inc., Cary, NC).
Results
Real-world data from 8542 U.S. users aged ≥65 years (mean age of 72 ± 11 years) were analyzed; and included 6249 with T1D, 1701 with T2D, and 592 who did not report diabetes type (3537 identified as “female”, 4623 as “male”, and 382 did not report demographic data). System settings and age assessment determined that 2753 (32.2%) used ROS and 2279 (26.7%) were aged ≥75 years (mean age of 79 ± 19 years).
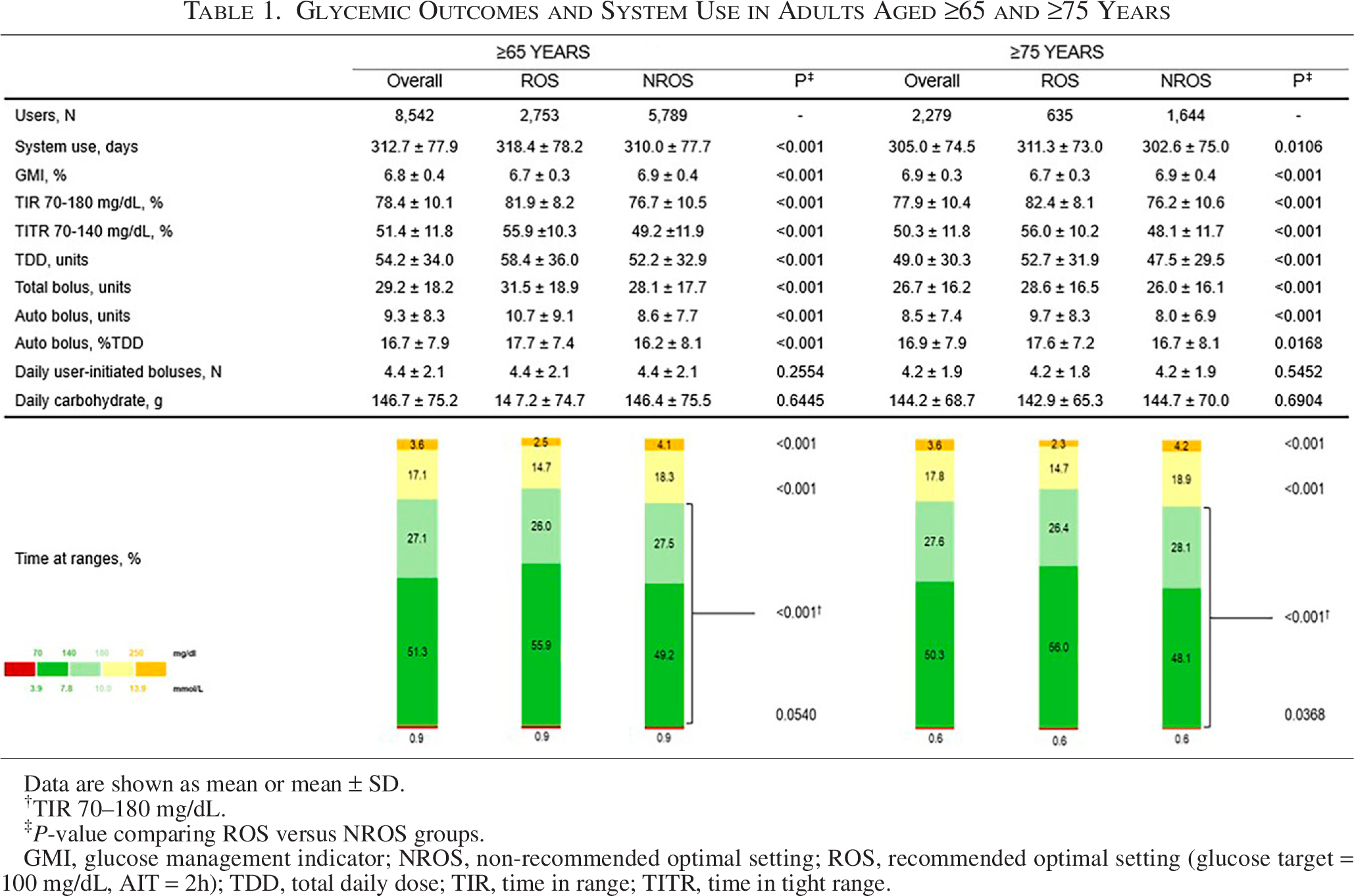
Table 1 shows that the overall group mean TIR was 78.4% and was higher for ROS users (81.9%, P < 0.001). Similarly, mean TITR was 51.4% in the overall population and 55.9% in the ROS group (P < 0.001). The ROS users versus NROS users had significantly improved TIR, TITR, and TAR250 (P < 0.001) with the same number of daily user-initiated boluses (Table 1). Similar results were achieved for users aged ≥75 years. In terms of glycemic targets for older/high-risk individuals, 99.2% of overall users achieved a TIR >50% (which comprised 100% of ROS users) and 80.2% achieved a TBR70 <1% (Fig. 1). Considering the targets recommended for younger/low-risk individuals, 78.9% of users achieved a TIR of >70% and 99.1% a TBR70 of <4%, which reached 92.6% and 99.5%, respectively, in the ROS users.
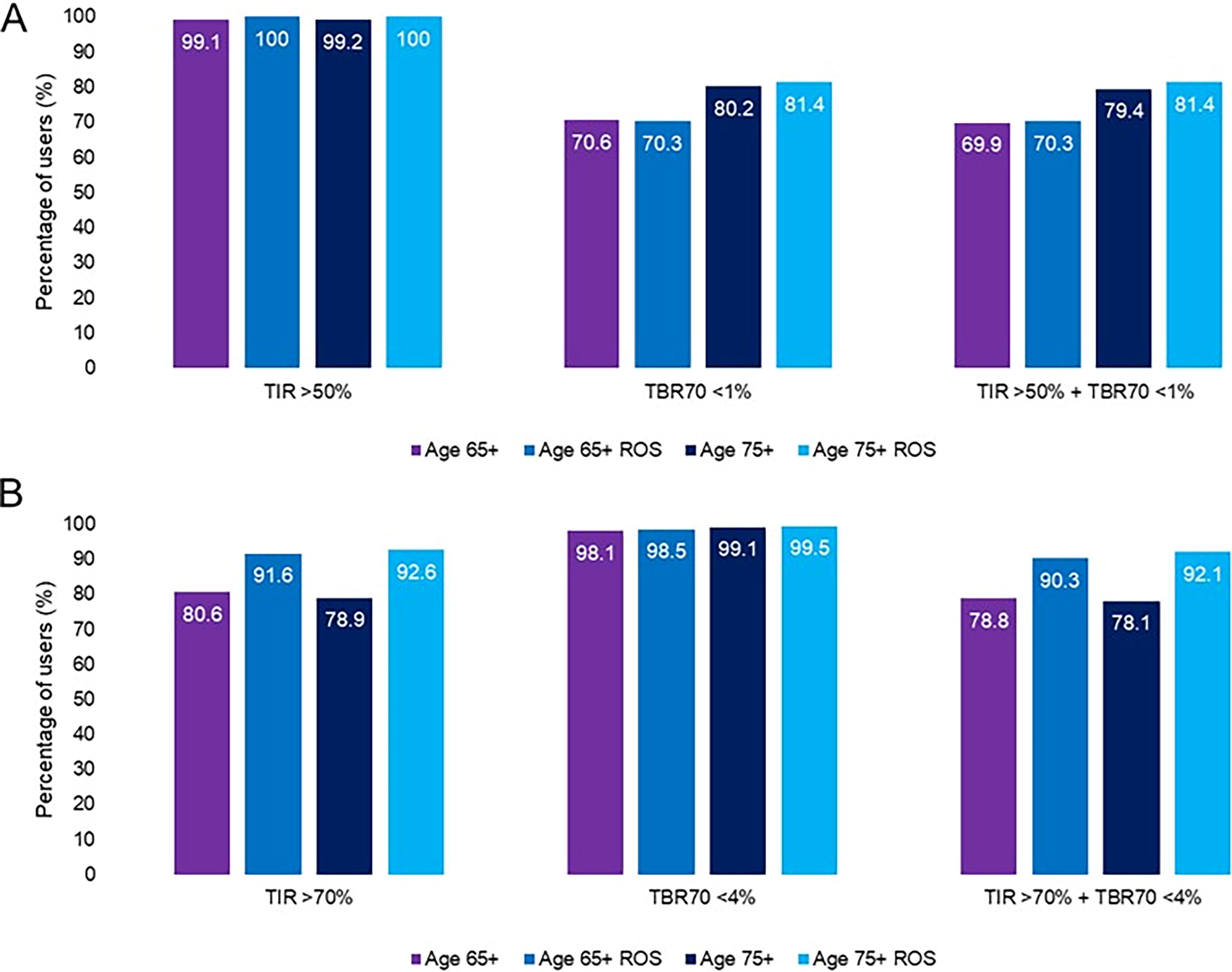
Achievement of consensus-recommended glycemic targets by age. Percentage of users by age group achieving
Glycemic Outcomes and System Use in Adults Aged ≥65 and ≥75 Years
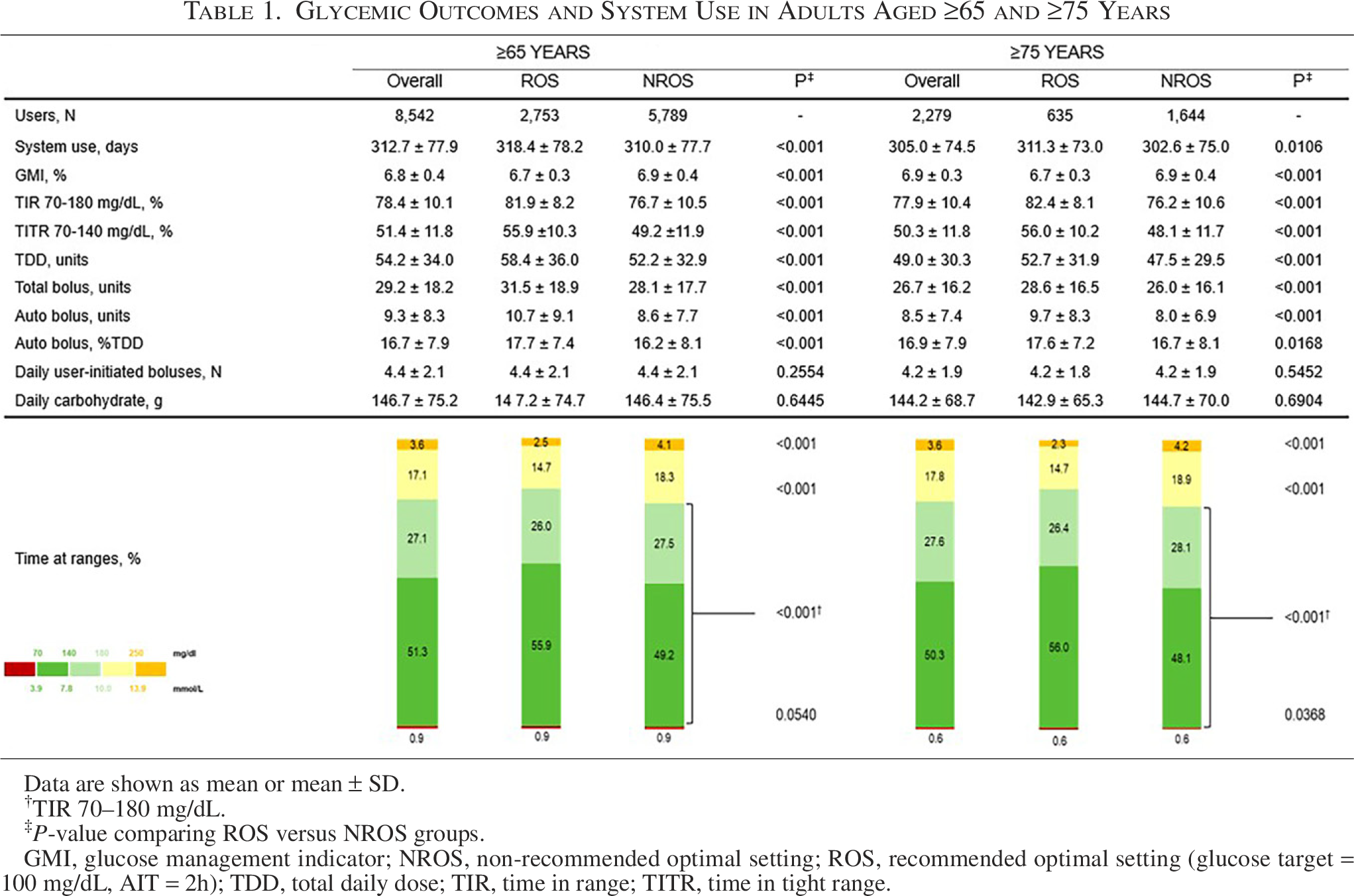
Data are shown as mean or mean ± SD.
TIR 70–180 mg/dL.
P-value comparing ROS versus NROS groups.
GMI, glucose management indicator; NROS, non-recommended optimal setting; ROS, recommended optimal setting (glucose target = 100 mg/dL, AIT = 2h); TDD, total daily dose; TIR, time in range; TITR, time in tight range.
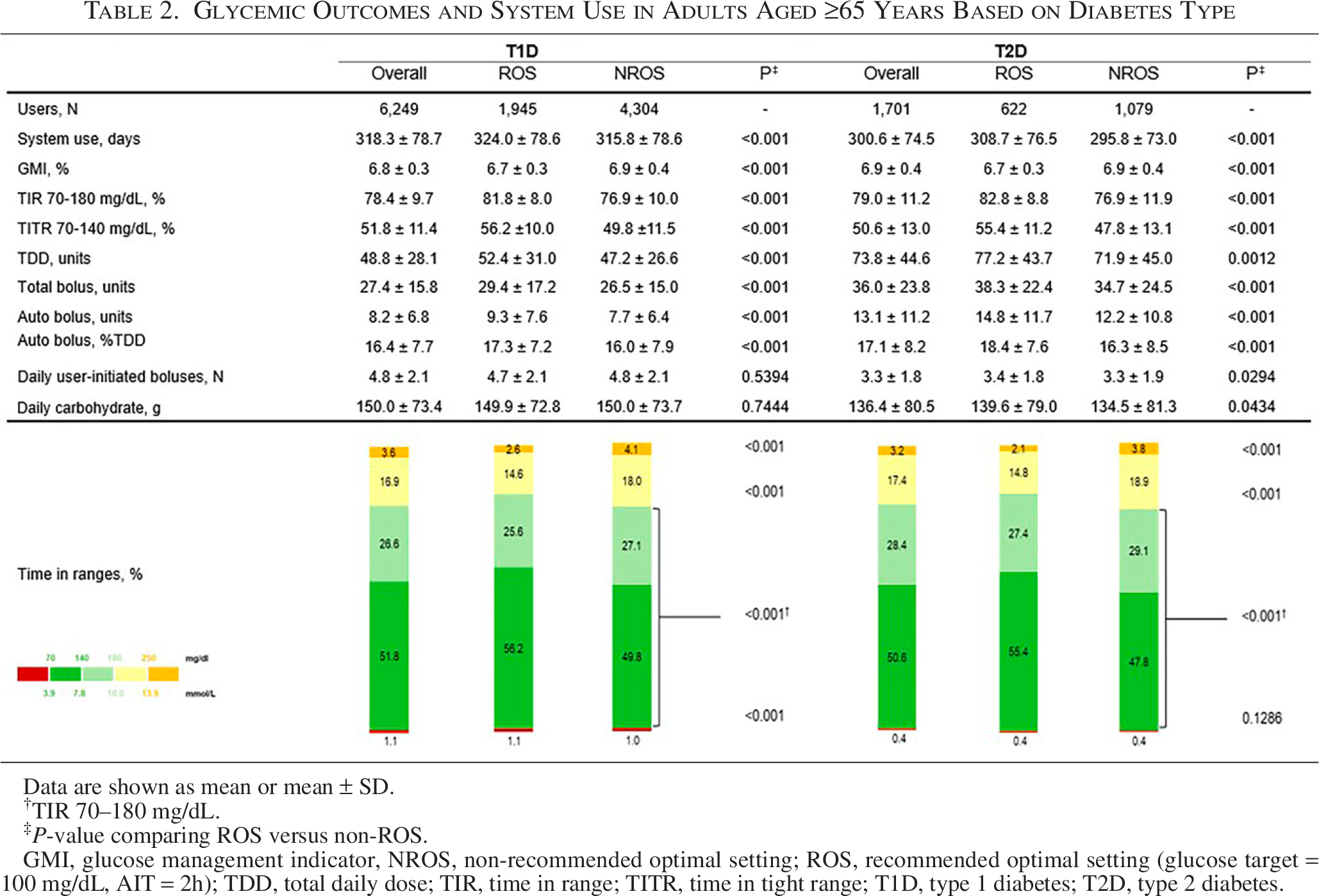
In MM780G system users with T1D and T2D, the mean TIR was 78.4% and 79%, respectively (81.8% and 82.8%, respectively, using ROS), the mean TBR70 was 1.0% and 0.4%, the mean TAR250 was 3.6% and 3.2%, and the TITR was 51.8% and 50.6%, respectively (Table 2). In addition, 98.5% of T2D users achieved a TIR of >50%, 91.4% a TBR70 of <1%, and 90.1% achieved both targets (Fig. 2).
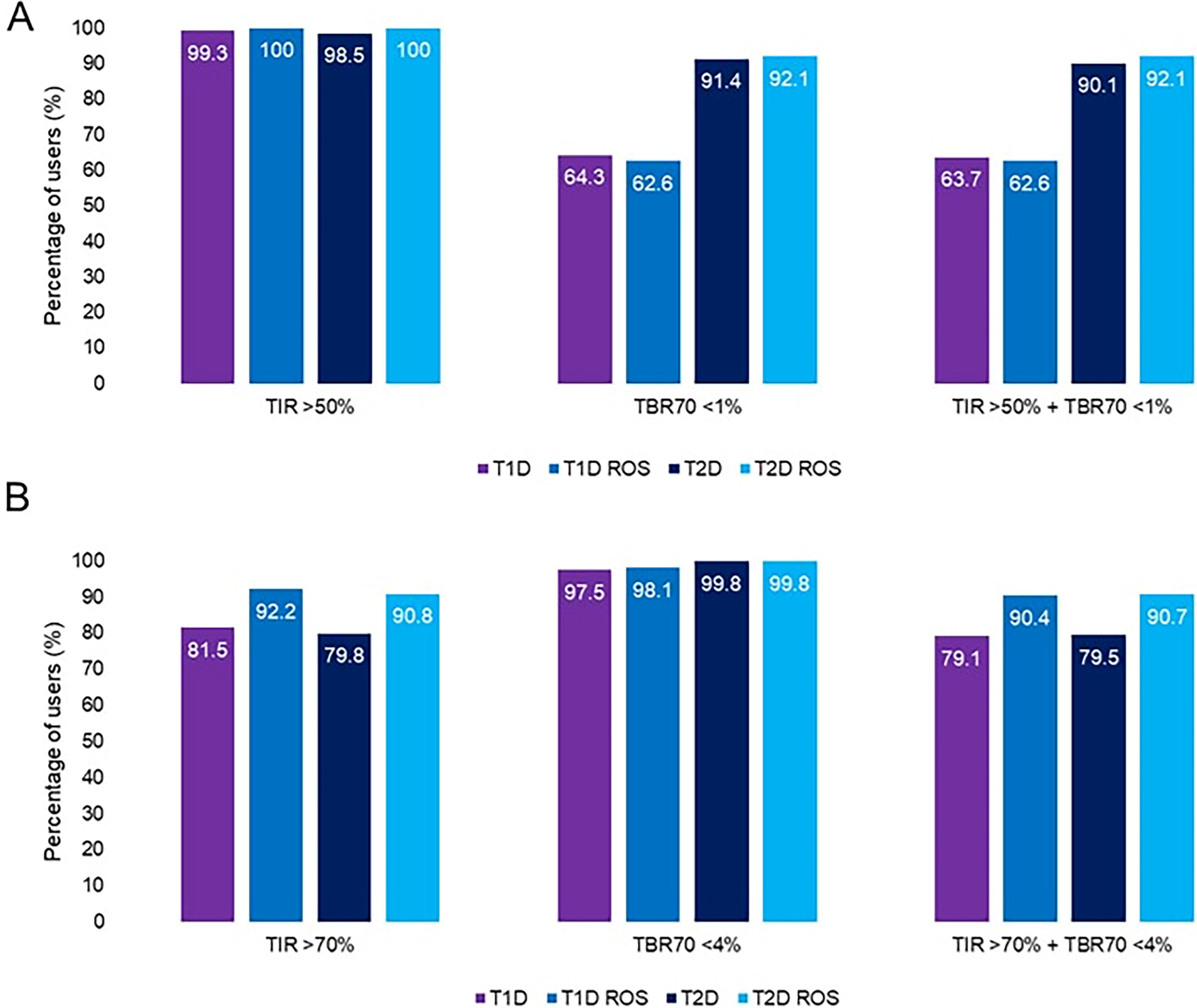
Achievement of consensus-recommended glycemic targets by diabetes type. Percentage of users by diabetes type achieving
Glycemic Outcomes and System Use in Adults Aged ≥65 Years Based on Diabetes Type
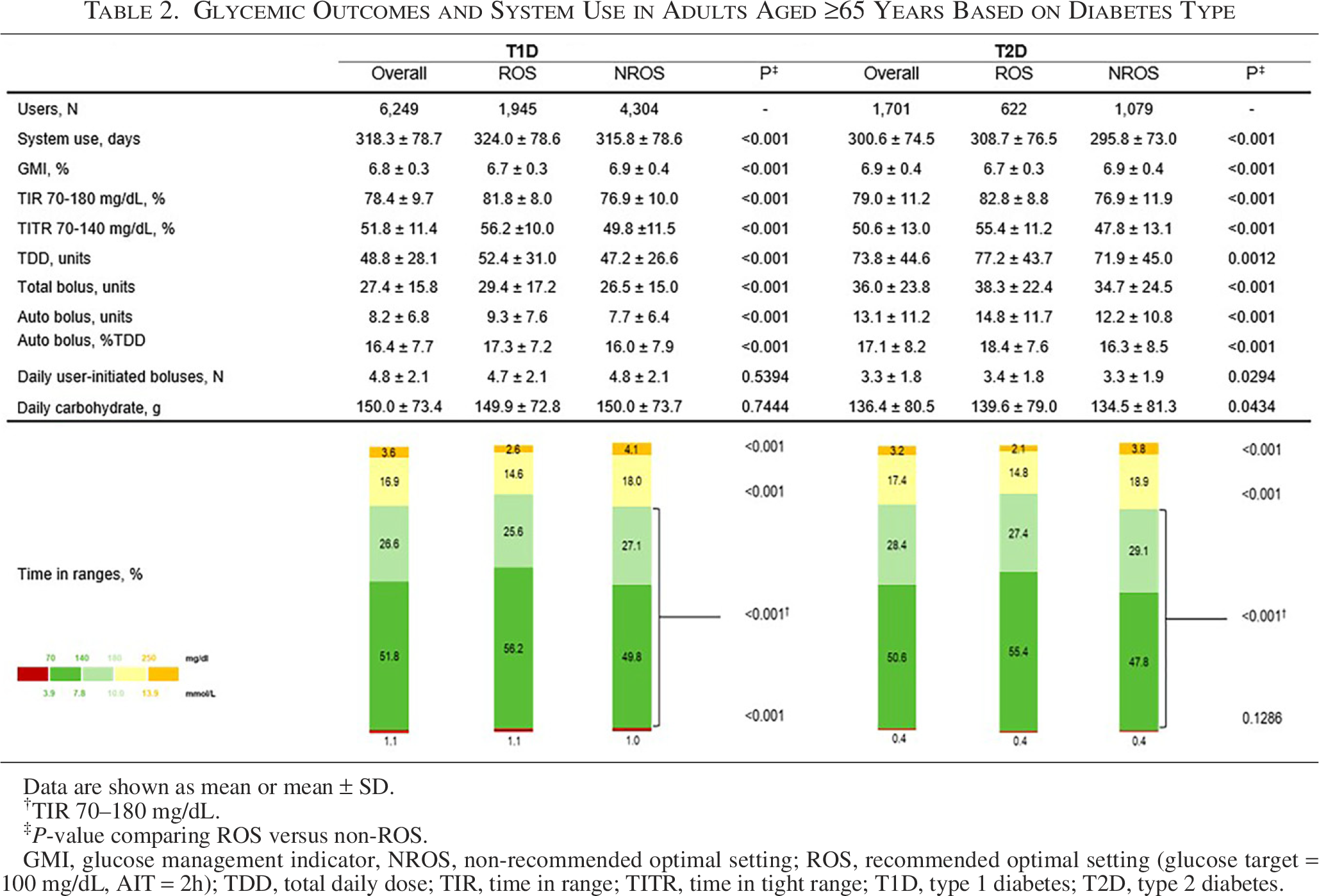
Data are shown as mean or mean ± SD.
TIR 70–180 mg/dL.
‡P-value comparing ROS versus non-ROS.
GMI, glucose management indicator, NROS, non-recommended optimal setting; ROS, recommended optimal setting (glucose target = 100 mg/dL, AIT = 2h); TDD, total daily dose; TIR, time in range; TITR, time in tight range; T1D, type 1 diabetes; T2D, type 2 diabetes.
Discussion
In the present study, real-world MM780G system users aged ≥65 years in the US with either T1D or T2D demonstrated CGM-derived glycemic control surpassing that recommended by international consensus for this age group. Regardless of diabetes type, use of the MM780G system with ROS allowed 100% to achieve the TIR of >50% target (91.6% achieved the TIR of >70%), with 70.3% meeting the composite TIR of >50% and TBR of <1% targets (90.3% achieved the composite TIR of >70% and TBR of <4%). This ROS-improved glycemia was similar to that observed in real-world MM780G system users with self-reported T1D16,17 and T2D 19 in the Europe, Middle East, and Africa (EMEA) region.
As noted above, the ROS are a glucose target of 100 mg/dL and an AIT of 2 h, both used for ≥95% of time. This recommendation is based on both clinical trial and real-world data demonstrating that these settings maximized TIR and TITR without increasing TBR.15–18 The reason why a greater percentage of users is not on these settings is complex, but may be predominantly a result of misunderstanding the meaning of AIT when used with the MM780G system compared with earlier AID systems from Medtronic. The MM780G system algorithm is markedly different from that of the MiniMed™ 670G and 770G systems where AIT in those systems represented an approximation of the physiological half-life of insulin. 15 In the MM780G system, it represents a “tuning knob” which controls how frequently the autocorrections are delivered and would be better termed “Algorithm Intervention Time.” The outcomes in the current study confirm that the TIR and TITR are improved by 5.2% and 6.7%, respectively, compared with those not using ROS in the cohort aged ≥65 years and 6.2% and 7.9%, respectively, in the cohort aged ≥75 years. The TBR value did not change and averaged <1% in both age cohorts, although the proportion was higher in users ≥75 years compared with those ≥65 years; this result may simply be explained by survivor or selection bias. Similar changes are seen in both age groups with T1D and T2D. More conservative settings can be used at MM780G system initiation if the user has a history of episodes of severe hypoglycemia or fear of hypoglycemia. However, close monitoring at 2- to 3-week intervals may permit the intensification of the settings if the hypoglycemia rate remains within target.
One large retrospective analysis has reported real-world CGM-derived glycemic outcomes of older adults using a different AID system. 20 Forlenza et al. evaluated 4243 Medicare recipients (aged 67.4 ± 10.9 years, n = 500 with T2D) who experienced significant improvement in outcomes, including TIR (from 64% pre-AID to 74% post-AID, P < 0.0001) during the AID use period, with 30% versus 19% (pre-AID) meeting specific composite targets as follows: GMI of <7%, TIR of >70%, and TBR of <1%, post-AID (P < 0.0001). A recent analysis of real-world MM780G system use in people with T1D ≥56 years showed that a TIR >70% was achieved by a majority of the 7415 users (79.2%), but that it reaches 93.7% with the use of ROS (n = 434/463), without increasing TBR, 21 which supports our study findings for older ROS users with T1D or T2D. This finding and the results from the present study show that glycemic benefits with the MM780G system do not deteriorate with increased age. The overall findings from the aforementioned real-world analyses strongly support the effectiveness and glycemia-improving benefits of AID therapy in older adults living with T1D or T2D.
The Global Burden of Disease and Risk Factors Study showed how the prevalence of T1D among people aged ≥65 years increased more than 25% between 1990 and 2019, concomitant with a decreased mortality at the global level. 22 Yang K, et al. highlighted how T1D is no longer a contributory factor in decreased life expectancy of older people with T1D mainly in regions with a high sociodemographic index, for example, North America, but that hyperglycemia remains a challenge. 22 The Centers for Disease Control and Prevention estimated that among 13.8 million people aged ≥65 years with diagnosed diabetes (24.4% of the population ≥65 years), 43% have an HbA1c >7% and 17% have an HbA1c >8%. 1 The T1D Exchange registry has also showed that the mean HbA1c of people ≥65 years of age living with T1D is 7.3% and it remains almost stable up to 80 years, although the lowest HbA1c values are reached by people using CSII + CGM. 23
An abundance of clinical and real-world evidence has demonstrated that TIR is a clinically meaningful and validated glycemic outcome metric. Numerous published studies have demonstrated a strong association between TIR and diabetes-related complications,24–27 including a virtual conversion of Diabetes Control and Complications Trial blood glucose measurements to CGM profiles. 28 In addition, Vigersky and McMahon reviewed 18 randomized controlled trials (RCTs) comprising 1179 individuals and concluded that a 10% change in TIR was approximately equal to a change in HbA1c of 0.8%, 29 whereas Beck and colleagues found that a 10% change in TIR was approximately equal to a 0.6% delta in HbA1c, in four RCTs. 24 Furthermore, a systematic review of AID system real-world evidence found that real-world retrospective analyses confirmed pivotal trial findings with larger and more diverse populations and follow-up periods of longer duration. 30
AID systems improve glycemic control by increasing TIR and reducing the time spent below and/or above range. 17 A recent systematic review evaluating the efficacy and safety of the MiniMed™ 670G, MM780G, Control‐IQ™, CamAPS FX, and Diabeloop Generation 1 systems demonstrated that MM780G achieved the highest TIR and lowest TAR250 and that all AID systems decreased TBR compared with standard therapy. 31 Real-world evidence from 13,202 system users in the EMEA region demonstrated that there are two predictors for reaching the highest TIR with the MM780G system. 32 The first one is age, where individuals >55 years reached a mean TIR of 76.8% in comparison with 72.3% achieved by the overall population (n = 101,629), and the second predictor is the use of ROS.19,33 Both findings were confirmed in our present analysis, which showed that ROS improved glycemic control without increased burden (i.e., more user-initiated insulin boluses).
Diabetes technology has been used mainly in T1D and is a recommended therapy in the majority of guidelines,9,33,34 but a growing body of evidence of AID use in T2D is emerging. In a recent meta-analysis conducted on seven RCTs, implementation of closed-loop AID in T2D as opposed to conventional therapy was demonstrated to improve glycemic control. AID systems significantly improved TIR by reducing time in hyperglycemia by 326 min (−23%) per 24 h and 184 min (−19%) during the daytime period compared with control. 35 Furthermore, the CLOSE AP+ trial evaluated the feasibility and safety of home health care-assisted AID in people with T2D (aged a mean of 69 years for both arms) unable to manage multiple daily injections (MDI) on their own. Results of the 12-week study revealed that TIR increased significantly by 27.4% and TBR decreased significantly by 27.7% in the AID group compared with those in the MDI group, with no severe adverse events. 36 Clinical trials of in-home AID system use, compared with basal or basal/bolus therapy (i.e., open loop or MDI), by adults with T2D have demonstrated safe and improved glycemic control,37,38 similar to that observed during AID system use in T1D. In the present real-world data analysis, no differences between T1D and T2D were observed, and a majority of both populations reached or exceeded consensus-recommended glycemic targets.
Despite the TIR association with HbA1c, TITR is emerging as a new metric because it represents the time in normal glucose range and, thus, may be more closely correlated with diabetes complications. To validate that TITR is a better option to assess glycemic variability, Shah et al. reported that healthy individuals spent 96% of the time in the glycemic range of 70–140 mg/dL 39 and that it was associated with incident retinopathy. 27 This was validated by Kovatchev et al. who determined that TITR had a higher hazard ratio for the risk of retinopathy and microalbuminuria than HbA1c or TIR. 28 Thus, TITR is emerging as an important marker for setting both T1D and T2D glycemic goals. 40 Although a target for percentage of TITR has not been established for older/high-risk adults yet, our data suggest that it may be 50% or as high as 55% since it is not associated with an increase in hypoglycemia.
The present analysis has several limitations, including the self-reported identification of diabetes type and age. Consequently, all user data available in the CareLink™ personal platform were considered. Potential selection bias may arise from the unknown duration of diabetes, the absence of individual medical histories, missing data on cognitive or physical impairment (e.g., in manual dexterity, vision, or hearing), caregiver involvement, and the use of glucose-lowering medications other than insulin, particularly for T2D. Addressing these limitations in future studies could enhance the accuracy and reliability of the findings. The major strengths of this work are that data are validated and come from a large number of system users aged ≥65 years and that there are no study-related biases that could overestimate the outcome, as the system is enabled by an automatic upload of data every 24 h. 41
Conclusions
In conclusion, our analysis of over 8000 MM780G system users aged ≥65 years in the US has demonstrated glycemic control reaching or exceeding consensus-recommended targets for all the CGM-derived parameters, which are consistent with those observed in younger adult and pediatric MM780G system users. The improved outcomes, with ROS use, could help U.S. adults aged ≥65 years with diabetes avoid acute and chronic complications. Our study provides strong reassurance in AID performance in this specific population. The rigorous results, inclusive of the ADA recommendation for AID systems as the preferred method for individuals with T1D (Level A evidence), 9 and other forms of insulin-deficient diabetes (irrespective of C-peptide test results) 9 compel reconsideration of CMS clinical coverage criteria for those who are Medicare eligible.
Authors’ Contributions
All authors interpreted and discussed the data, and reviewed the article. M.L. and A.A. analyzed the data. J.S. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The MiniMed™ 780G system includes technology developed by DreaMed Diabetes (Petah Tikva, Israel).
Footnotes
Author Disclosure Statement
All authors are employees of Medtronic Diabetes.
Funding Information
This work was funded by Medtronic Diabetes.
Prior Presentation
Portions of data within this work were presented at the 85th Scientific Sessions of the American Diabetes Association (June 20–23, 2025, Chicago, Illinois).