Abstract
The Omnipod→, a tubeless automated insulin delivery system, presents itself as a compelling innovation in diabetes management. Initially developed to overcome the limitations of conventional insulin pumps, the closed-loop system of the Omnipod→ offers more autonomy and freedom of choice to diabetes patients. This article addresses its clinical performance, real-life outcomes, and psychosocial impact. The current clinical trials and observational evidence suggest remarkable improvement in glycaemic control and patient satisfaction, particularly among Type 1 diabetes patients. Skin irritation, financial burden, and environmental considerations remain obstacles to large-scale use. Nevertheless, Omnipod→ is a vital step towards the next generation of diabetes technology, and its implications globally, particularly in Malaysia, continue to expand with rising diabetes incidence.
Keywords
Introduction: A Growing Need for Insulin Innovations
The rising prevalence of Type 2 Diabetes Mellitus (T2DM) affects approximately one in every five people in Malaysia, 1 thus, highlighting an increasing demand for more effective insulin delivery systems. The diverse pathophysiology of T2DM precludes all cases of T2DM from needing insulin therapy, leaving those with deficient insulin production with exclusive insulin demand. This contrasts with the cases driven by insulin resistance, where treatment options such as glucagon-like peptide-1 (GLP-1) agonists may suffice. 2 The Omnipod→, developed by Insulet Corporation (Massachusetts, United States), offers a modern, automated solution exclusively for patients requiring insulin.
Historical Context and Technological Advancements
Insulin therapy has undergone immense technological advancements since the insulin discovery in 1921 by Frederick Banting over the years. Increased innovation in insulin research has been driven by the need for less invasive, user-friendly alternatives, from the first syringe-based techniques to insulin pens. With the release of the first commercial insulin pump in 1979, closed-loop systems incorporating continuous glucose monitoring (CGM) technologies were made possible. 3 These systems, often referred to as automated insulin delivery (AID) or artificial pancreas systems, aim to mimic the body’s physiological insulin regulation. Originally conceptualised in the 1960s by Arnold Kadish, 4 the artificial pancreas gained traction with the development of Pfeiffer’s Biostator in the 1980s. 5 Recent initiatives like the OpenAPS initiative and the Juvenile Diabetes Research Foundation International (JDRF) Artificial Pancreas Project have made significant progress in the field. 6 Although these technologies were initially created for individuals with Type 1 Diabetes Mellitus (T1DM), those with insulin-dependent T2DM can also benefit from them.
Design and Functionality of the Omnipod→
The Omnipod→ system is a prime example of the innovative fruition from years of diabetic interventional research. Conceived by John Brooks III and Duane Mason, the device was inspired by the inconvenience experienced by Brooks’ son, a T1DM patient, who used a traditional tubed insulin pump. They envisioned a discreet, tubeless insulin delivery solution and developed the Omnipod→ system, which comprises two primary components: a wearable Pod and a Personal Diabetes Manager (PDM). The Pod, capable of holding up to 200 units of insulin, adheres to the skin and delivers subcutaneous insulin via a soft cannula. It can be worn on various sites of the body, which include the upper arms, thighs, abdomen, or buttocks. The PDM, connected wirelessly via Bluetooth, monitors insulin delivery, reservoir levels, and other operational metrics. It provides real-time alerts to the user through auditory and vibratory signals. This design eliminates the need for tubing, enhances mobility, and supports precision in insulin administration. 7
Two main versions of the Omnipod→ are currently available: the Omnipod DASH and the Omnipod 5. While both systems operate similarly, the Omnipod 5 includes integrated CGM capabilities with the incorporation of remote monitoring by the Horizon app, which is particularly useful for the caregivers of diabetic patients. The DASH requires a separate CGM device and connects solely via Bluetooth. The selection of the versions is dependent on the user preference and clinical context, as there is no definitive superiority between the two versions.
Evidence from Clinical Trials
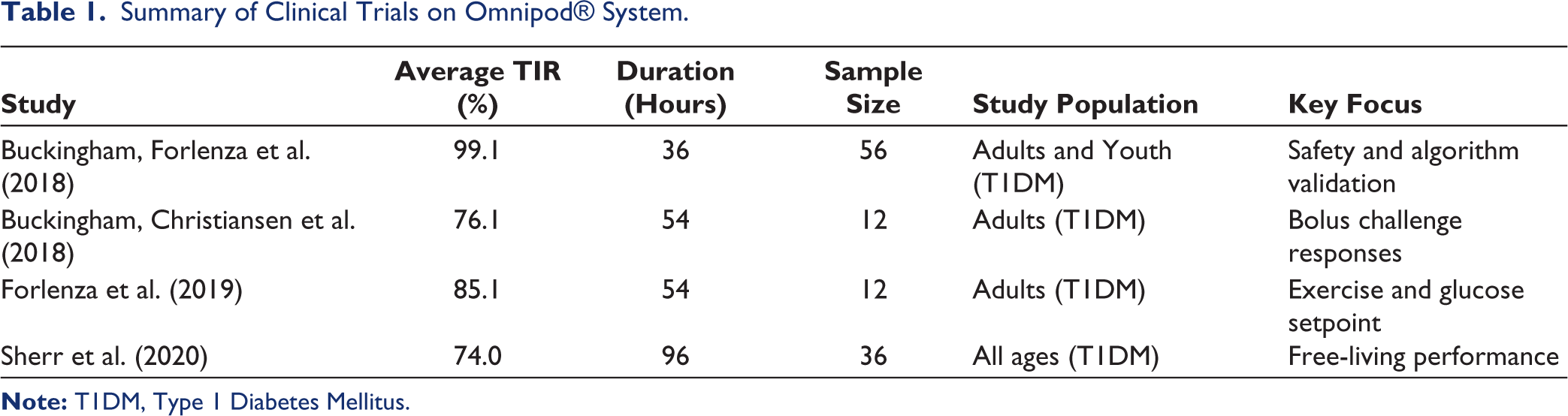
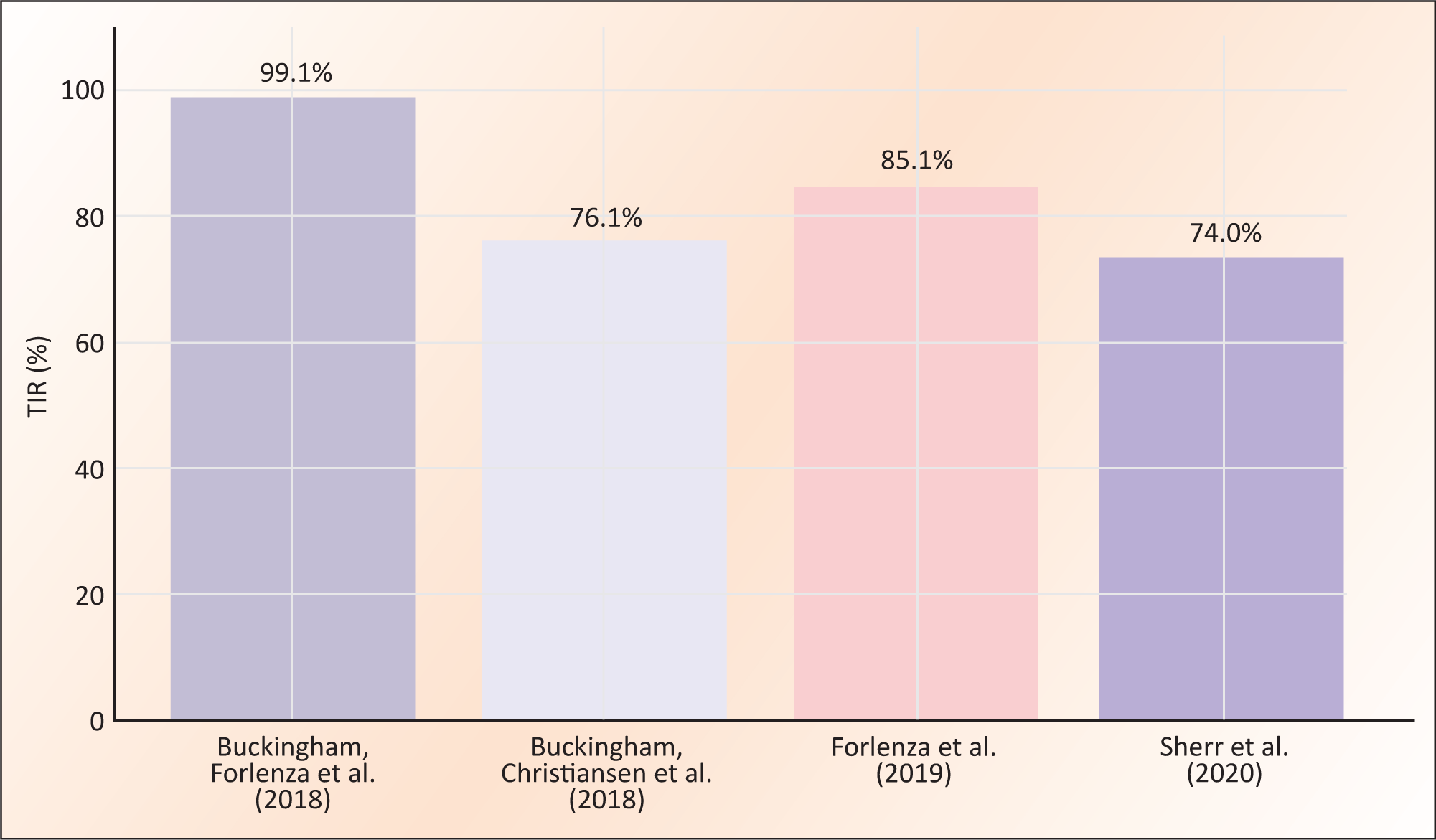
The clinical performance of the Omnipod→ system has been evaluated through a series of trials, mostly involving T1DM patients. Buckingham, Forlenza, et al. (2018) conducted a two-phase trial consisting of a 1-week outpatient period followed by a 36-hour inpatient phase. The trial demonstrated that the system’s model predictive control (MPC) algorithm, a process control method that satisfies a set of constraints, maintained glucose levels within the target range for 99.1% of the time during the inpatient phase. 8 Another study by Buckingham, Christiansen, et al. (2018) assessed the system’s response to various bolus scenarios, including overestimated, missed, and extended boluses. Despite the challenges, the Omnipod→ maintained a Time-in-Range (TIR) of 76.1% that exemplifies its adaptability. 9 A subsequent trial by Forlenza et al. (2019) introduced moderate intensity exercise and adjusted glucose setpoints to test Omnipod→ capability in the altered state of physiology. This trial achieved an 85.1% TIR, hence showcasing the system’s robustness under variable physiological conditions. 10 Sherr et al. (2020) furthered this research by evaluating the Omnipod→ over a 96-hour period in a hotel setting. The study encompassed both children and adult participants to provide a better scope of the study, which further demonstrated the device’s safety and reliability in a real-world, unsupervised environment. 11 Nevertheless, these studies often shared limitations, including small sample sizes, a narrow demographic focus limited to T1DM patients, and minimal representation of elderly users.
The aforementioned clinical findings are summarised in Table 1 to provide a comparative overview of study design, duration, sample sizes, and outcomes. This is the first review that encapsulates and tabulates all the current developments in insulin release of Omnipod systems. This table offers a concise view of the consistent effectiveness of the Omnipod→ across varied clinical environments. To visually highlight the clinical trial findings discussed, Figure 1 illustrates TIR performance across the four main Omnipod→ studies. The TIR metric, a key indicator of glycaemic control, reflects the percentage of time participants’ glucose levels remained between 70 and 180 mg/dL. This bar chart clearly demonstrates the high efficacy of the Omnipod→ system, especially in controlled inpatient settings, with notable variability under different real-world and test conditions.
Summary of Clinical Trials on Omnipod→ System.
Time-in-Range (TIR) Outcomes in Omnipod→ Clinical Trials.8–11
Trial Methodologies and Metrics
All the trials included in this review were standardised in the methodology. Each began with a 7-day outpatient phase where subjects adhered to their regular routines, and subsequently followed by an inpatient monitoring period. Primary outcomes examined included the percentage of time spent in hypoglycaemic (<70 mg/dL) and hyperglycaemic (≥250 mg/dL) states, while secondary metrics assessed mean glucose levels, standard deviation, and coefficient of variation. TIR, defined as the percentage of time glucose levels remain between 70 and 180 mg/dL, was a key metric in the study of the efficiency of insulin delivery. A TIR above 70% is considered a benchmark for effective glucose management, corresponding to approximately 17 hours per day within range. 12 All studies used various Dexcom CGM devices during monitoring.
Real-world Utilisation and Effectiveness
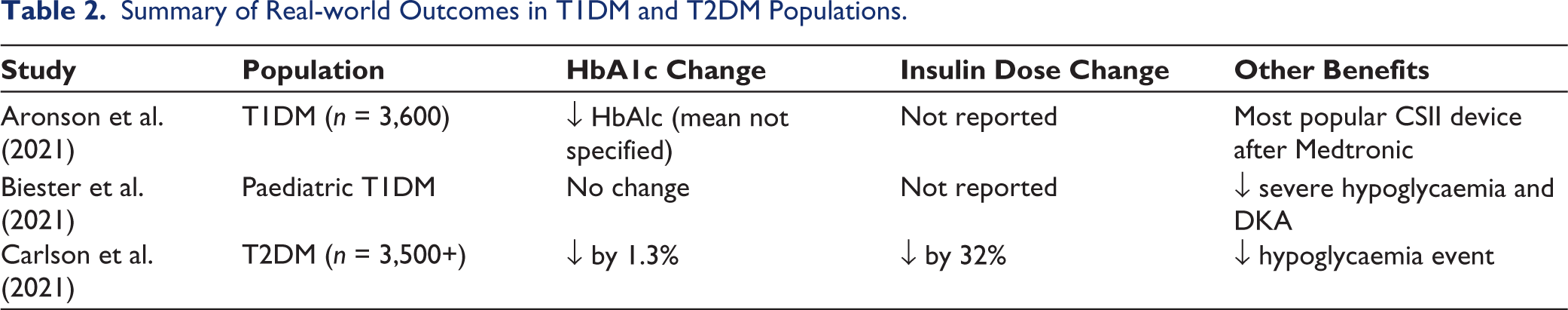
The compiled data from real-world implementation of the Omnipod→ system underscore its tangible impact beyond clinical trials. Aronson et al. (2021) reported that among 3,600 individuals from a Canadian registry, users of continuous subcutaneous insulin infusion (CSII) devices had significantly lower HbA1c levels, with Omnipod→ being the second most commonly used device. 13 Biester et al. (2021) analysed a cohort of paediatric and adolescent users over three years and oversaw a decline in acute complications, such as severe hypoglycaemia and diabetic ketoacidosis; nevertheless, HbA1c levels remained unchanged during the observations of the usage. 14 Bratke et al. (2021) in Norway reaffirmed the improved metabolic outcome, measured in HbA1c and the parameters such as the diabetic ketoacidosis incidence with Omnipod→ use in the paediatric populations, thus validated aforementioned results from clinical trial across age demography. 15
As the Omnipod system is not used solely on T1DM in the practical setting, Carlson et al. (2021) conducted a large-scale study for T2DM patients involving over 3,500 adults who used the Omnipod→ for 90 days. The results revealed an average 1.3% reduction in HbA1c and a 32% decrease in daily insulin dosage, irrespective of prior treatment regimen or age. This study also documented a reduction in severe hypoglycaemic episodes, underscoring the device’s efficacy in diverse populations. 16 Table 2 summarises the results of these real-world studies to emphasise both the clinical and economic value of the Omnipod→ for T1DM and T2DM patients.
Summary of Real-world Outcomes in T1DM and T2DM Populations.
Psychosocial Outcomes and Quality of Life
Psychosocial outcomes further affirm the Omnipod→’s value. A qualitative study by Stocco et al. (2024) in Australia explored the lived experiences of adult T1DM patients over a 12-week trial. Participants highlighted reduced anxiety about glycaemic control, greater lifestyle flexibility, and enhanced confidence in managing diabetes. The device’s discreet profile was praised for promoting normalcy and minimising stigma. Users appreciated the ability to engage in social and physical activities without the visibility and encumbrance of traditional pump tubing. However, concerns about cost and occasional device failure were noted. 17 Additional studies by Hood et al. (2023) and MacLeish et al. (2024) examined psychosocial outcomes among caregivers of children using the Omnipod 5, reporting similar improvements in emotional well-being and diabetes management confidence.18,19
Current Challenges and Limitations
Despite its advantages, several barriers hinder the broader adoption of Omnipod→ technology. Skin-related complications due to adhesives remain a recurring issue, especially among paediatric users. Berg et al. (2020) reported significantly higher treatment costs associated with managing skin problems, with paediatric patients incurring 56% more in related expenses compared to adults. Furthermore, allergic contact dermatitis has been documented in multiple case studies, implicating certain chemicals such as isobornyl acrylate, a known sensitiser used in the pod adhesives.20–22 These findings suggest a need for developing hypoallergenic adhesives and skin-friendly design improvements.
Financial burden is another critical concern. While the Omnipod→ offers considerable health and lifestyle benefits, the high cost of the device and its consumables can be prohibitive. 23 However, partial governmental subsidy can help alleviate financial issues with Omnipod use, one prime example being the Australian government subsidy for disposable pods in the Omnipod DASH→ via the National Diabetes Services Scheme (NDSS) found at community pharmacies. Such assistance was made available since December 2022 and was primarily indicated in T1DM cases. The middle- and low-income countries should follow suit with their own financial support schemas as the diabetic incidences are rising globally. Environmental sustainability also poses a challenge. A review by Avari et al. (2024) highlighted the significant labour and environmental impact involved in recycling Omnipod→ components, calling for innovations in biodegradable materials and simplified device design to reduce waste and cost. 24
Emergence of DIY Closed-loop Systems
Parallel to commercial advancements, a growing community of patients has begun exploring Do-It-Yourself (DIY) closed-loop systems. These solutions, often built using open-source software and mobile devices, allow users to enhance the functionality of their insulin delivery systems. Although not FDA-approved, these DIY systems are typically used by tech-savvy individuals willing to assume full responsibility for their insulin management, often possessing a deep understanding of the underlying technology and its inherent risks. 25 Their emergence, however, does bring attention to a gap in accessible, customisable solutions within the commercial landscape and points toward potential future developments in user-driven design.
In this context, understanding the characteristics of successful commercial systems is crucial. Notably, research evaluating the Omnipod→ automated insulin system in free-living conditions emphasises the significance of safety, performance, and adaptability in commercial systems. According to a study by Sherr et al. (2020), the Omnipod’s personalised MPC algorithm dramatically increased glycaemic control in children, adolescents, and adults without raising the risk of hypoglycaemia. These results highlight the importance of prioritising individual needs, safety, and practicality in user-centred AID, whether in regulated commercial offerings or DIY solutions. 11 The DIY community’s demand may continue to drive innovation in commercial systems, particularly for flexible, adaptable solutions that mirror the customisation that DIY users seek.
Conclusion and Perspectives for the Future
In conclusion, the Omnipod→ represents a revolution in diabetes technology. With proven clinical efficacy, favourable real-world outcomes, and strong psychosocial benefits, it transcends numerous limitations of the traditional insulin delivery systems. Despite the fact that it is now available only in some markets such as the United States, the United Kingdom, Germany, and the Netherlands, its broader use is encouraging, particularly in countries such as Malaysia. Given Malaysia’s vast diabetes load and the projected 49% of those undiagnosed, 26 the access to Omnipod→ would significantly improve management and quality of life.
It is highly suggested that the Malaysian decision-makers and healthcare stakeholders investigate making these technologies more available by way of subsidies, public-private partnerships, and awareness programs targeted at the most needy. In so doing, it could transform diabetes management in the nation for the better while encouraging innovation and growth in the local pharmaceutical and medical device sectors.
Footnotes
Acknowledgements
Not applicable.
Availability of Data and Materials
Not applicable.
Consent for Publication
All authors have reviewed and approved the final version of the manuscript and consent to its submission for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approval and Consent to Participate
This article is a perspective and does not report any original data involving human participants or animals. Therefore, ethical approval was not required. No patients or animals were involved in the writing of this manuscript.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
