Abstract
Objective:
To evaluate the effectiveness and safety of a mobile application for carbohydrate counting and bolus calculation (CHOC-BC) in adults with type 1 diabetes mellitus (T1DM).
Research Design and Methods:
A 12-week randomized controlled trial was conducted at King Fahad Medical City, Riyadh, Saudi Arabia. Adults with T1DM on multiple daily insulin injections and using Libre 2 glucose monitors were randomized to either CHOC-BC or conventional treatment. The primary endpoint was time in range (TIR; 70–180 mg/dL).
Results:
A total of 127 participants (70 females) were included: 64 in the intervention group and 63 in the control group with a mean age of 26.56 ± 4.8 and 26.74 ± 6.52 years, respectively. After 3 months, the intervention group achieved better TIR than the control group (51.20% ± 11.61% vs. 46.17% ± 13.02%; mean difference [MD], 5.03; 95% confidence interval [CI], 0.70–9.36; P = 0.023). Application users showed a significant reduction in level 2 time above range (17.25% ± 11.61% vs. 24.10% ± 15.74%; MD, −6.85; 95% CI, −11.70 to −1.99; P = 0.006). No significant differences were observed in body weight or time below range.
Conclusions:
The CHOC-BC mobile application empowered users to achieve better glycemic control while maintaining a safe profile that avoids hypoglycemia and weight gain.
This is a visual representation of the abstract.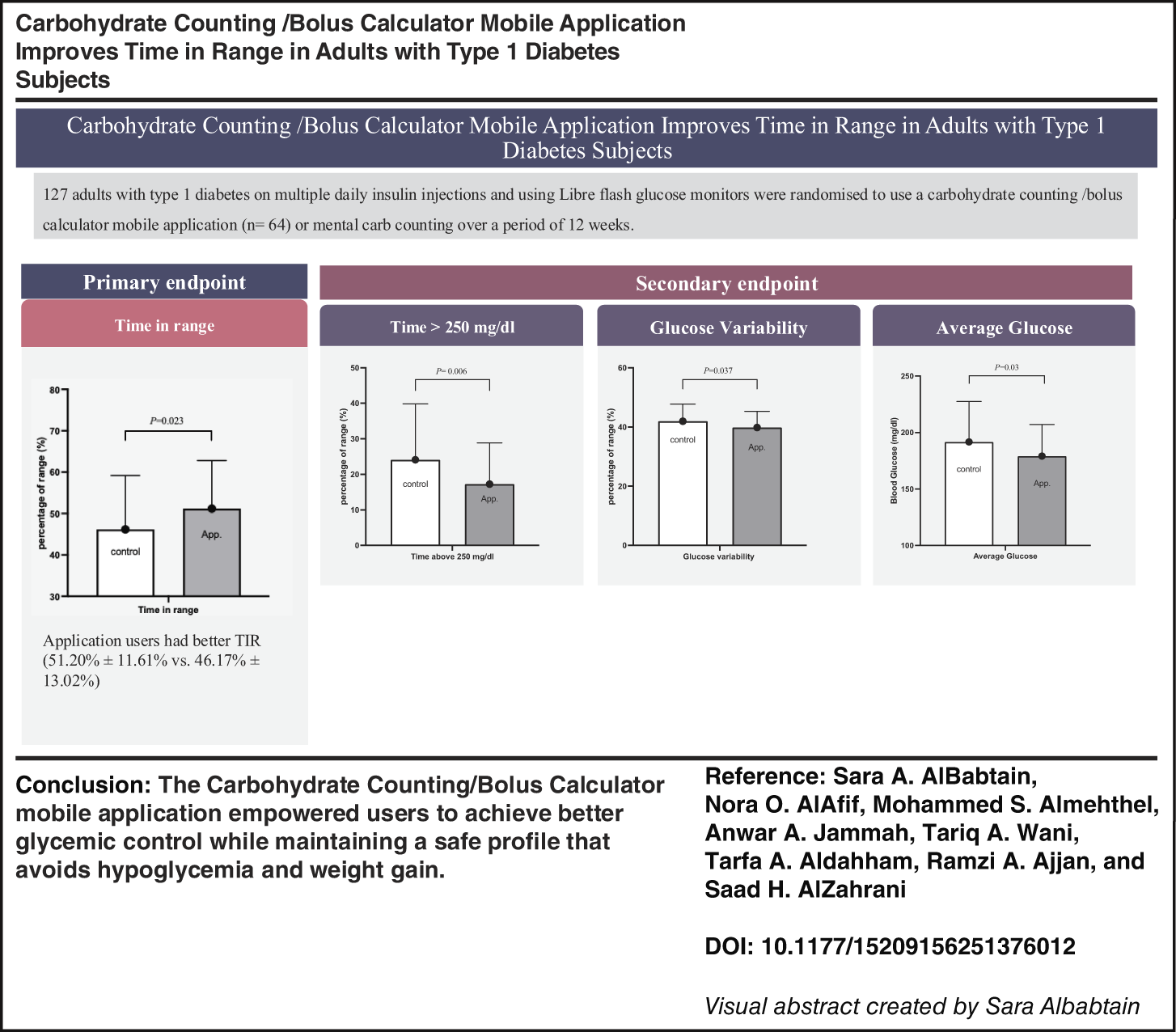
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
