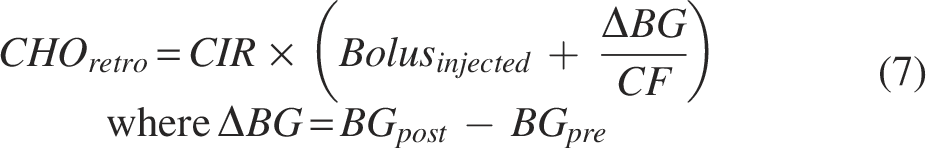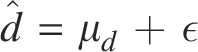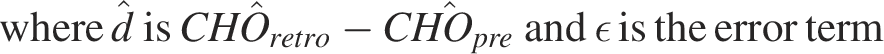Abstract
Introduction:
With the advent and increasing availability of continuous glucose monitoring (CGM), patients are now able to more easily and frequently determine their blood glucose levels than they would have using the finger prick blood test. Health care professionals can also review the patient’s prandial glucose profiles stored in the CGM system without intentional checking of glucose levels. However, given the large amount of data stored, methods for the utilization of such information need to be developed for better diabetes management.
Methods:
This study shows the retrospective estimation of carbohydrate amount using pre- and postprandial blood glucose levels via the following derived equation: (patient’s current carbohydrate-to-insulin ratio) × [injected bolus insulin + (postprandial blood glucose − preprandial blood glucose)/correction factor]. Accordingly, the retrospective estimates are systemically biased when the carbohydrate-to-insulin ratio (CIR) is not set appropriately. Insufficient adjustment of the correction factor (CF) also results in retrospective estimation bias for meals with large differences in pre- and postprandial blood glucose levels. We compared these retrospective estimates with patient-measured preprandial carbohydrate estimates to assess the bias. By using only meal data with small differences in pre- and postprandial blood glucose levels, the influence of CF on the bias was limited, and only the systemic bias caused by CIR was assessed. In the presence of a statistically significant systemic bias, the data suggest that the current CIR is set inappropriately.
Results:
We present the method by assessing CIR in a patient with newly diagnosed type 1 diabetes. CIR parameter assessments at 7 and 10 months from diagnosis are presented.
Conclusion:
A novel method for statistically assessing CIR was introduced. This method can support clinical decision-making when adjusting CIR for patients with insulin-dependent diabetes who engage in carbohydrate counting.
Keywords
Introduction
Diabetes mellitus is a globally prevalent chronic disease involving hyperglycemia, which leads to microvascular and macrovascular complications when uncontrolled. Type 1 diabetes mellitus (T1DM) is a type of diabetes characterized by progressive beta cell destruction typically caused by autoimmune response. Estimates show that around 8.4 million people had T1DM in 2021, with this number being expected to continuously increase. 1 In the Diabetes Control and Complications Trial (DCCT), intensive glycemic control designed to achieve near-normal glycemic control in T1DM patients prevented microvascular complications, including neuropathy, retinopathy, and nephropathy. 2 Intensive care also showed long-term protective effects against not only microvascular complications3–6 but also cardiovascular disease. 7 Various types of diabetes medications have been developed for glycemic control and diabetes complications; however, exogenous insulin replacement via injections or pumps remains the primary therapeutic approach for T1DM patients. 8
T1DM patients require insulin replacement therapy through multiple daily injections or insulin pumps, which consist of basal and bolus insulin. Although previous studies have reported on the theoretical dosage of basal and bolus insulin,9–11 adjustments are required for each patient. Given that patients have their unique characteristics and lifestyles, variations in insulin requirements within a day need to be considered. Meals have been among the various factors that considerably impact daily blood glucose variability. 12 To control prandial glucose fluctuations, appropriate dosage of bolus insulin is required, with its ideal dosage highly depending on the content of each meal.
To properly dose bolus insulin with flexible meal intake, insulin sensitivity parameters, such as carbohydrate-to-insulin ratio (CIR) and correction factor (CF), are needed for dose calculations. 13 CIR is a parameter that refers to an amount of carbohydrates covered or balanced by one unit of insulin. Meanwhile, CF is a parameter that indicates the extent to which blood glucose decreases by one unit of insulin. Therefore, the insulin dose that matches the estimated carbohydrate amount can be calculated by dividing the carbohydrate amount by CIR. Moreover, the insulin dose needed to correct preprandial blood glucose levels can be determined by dividing the preprandial blood glucose difference from the target level by CF. The bolus dose is the sum of the dose for carbohydrate and that for blood glucose correction. Administering the appropriate bolus dose for the meal keeps the corresponding postprandial glucose levels within its target range. To estimate the carbohydrate amount before each meal, the carbohydrate counting method has been used.
In the DCCT, carbohydrate counting had been used as one of the food planning methods in the intensive care group to meet their strict diabetes control. 2 The intensive care group achieved lower HbA1c levels than did the control group and experienced fewer incidences and progression of diabetes complications. Carbohydrate counting has three levels, with advanced carbohydrate counting (Level 3) providing information on how to match the bolus insulin dose with the carbohydrate amount in each meal with the use of CIR. 14 Recent studies have supported the efficacy and safety of advanced carbohydrate counting for T1DM treatment, 15 as well as its benefits in improving patient satisfaction and quality of life.16,17
With the continued advancements in technology, continuous glucose monitoring (CGM) has become widely used in clinical practice. 18 CGM provides a higher frequency of data, which can be understood as continuous data, than does finger prick blood glucose monitoring. Its usage has been proven beneficial in reducing hemoglobin A1c and the number of hypoglycemic episodes. 19 CGM has also been used to assess diabetes control. For example, time in range (TIR) is a metric that shows how long glucose values remain within the target range, with increased TIR having been associated with reduced microvascular complications. 20 To utilize CGM data for clinical decision-making, CGM data presentation needs to be standardized. In this regard, reporting of ambulatory glucose profile (AGP) with some CGM metrics, including TIR, is recommended in clinical settings.21–23 Aside from the standardized AGP report and CGM metrics, CGM data can also be interpreted in various ways. Objectives-based data interpretation can guide therapeutic modifications. 24 King also proposed a method for adjusting and titrating insulin dosage using CGM for pump-treated patients with T1DM. 25 The retrospective interpretation of blood glucose is key to adjusting the insulin dosage for each patient.
The current study presents a method for assessing CIR using multiple prandial glucose data extracted from CGM. In the Materials and Methods section, the formula to estimate carbohydrate content retrospectively was first derived. Retrospective estimates of carbohydrate amounts can be obtained by repeatedly applying the formula to each prandial glucose data. Second, the method to assess CIR by comparing these retrospectively estimated carbohydrate amounts with carbohydrate estimates measured by the patients was shown. In the Results section, these procedures are illustrated in a patient with T1DM.
Materials and Methods
Formula to retrospectively estimate carbohydrate content
Carbohydrate content was retrospectively estimated using the formula derived in this section.
Ideal bolus dosage
Injecting the ideal dosage of the bolus insulin can keep the postprandial blood glucose levels within target range. In contrast, injecting a suboptimal bolus insulin dosage for the meal can result in postprandial blood glucose levels out of the target range. Postprandial hyperglycemia indicates that more bolus insulin is needed, whereas postprandial hypoglycemia indicates that less bolus insulin is needed for the meal. CF, which is usually used to calculate insulin dosage to correct the current blood glucose level, is an insulin sensitivity indicator that was used in this study to estimate how much more or less bolus insulin would be needed to obtain the target postprandial blood glucose level. The additional or reduced amount of insulin required can be calculated as the difference between postprandial blood glucose levels and target glucose levels divided by the CF,
Components of bolus insulin
Based on the carbohydrate counting theory, the ideal bolus dose consists of a bolus for carbohydrate amount,
Equation (3) can be transformed into Equation (4).
By substituting Equation (1) into Equation (4), Equation (5) can be obtained. The carbohydrate component of bolus insulin can be obtained using Equation (5).
Retrospective estimation of carbohydrate amount
In the carbohydrate counting theory, the carbohydrate component of bolus insulin,
To summarize, Equations (5) and (6) can be used to derive the final Equation (7), which can be used to retrospectively estimate the carbohydrate amount,
Carbohydrate-to-insulin ratio assessment
Retrospective estimation and preprandial carbohydrate counting estimate the same carbohydrate amounts. As the retrospective carbohydrate estimation formula shows, retrospective estimates can be biased by inappropriate bolus insulin parameters, CF and CIR. In circumstances wherein bolus insulin parameters are set appropriately, retrospective carbohydrate estimates should be close to the corresponding patient-reported preprandial carbohydrate estimates,
To statistically quantify how
Accordingly, the confidence interval (CI) for
The CIR assessment method proposed is based on the CI for the systemic difference,
Patient eligible for μd CI-based CIR assessment
Some conditions need to be considered when applying the above method. The retrospective estimation formula considers only the balance between bolus insulin and carbohydrate load. Therefore, the effects from unadjusted basal insulin, endogenous insulin, hypoglycemia reaction, and residual bolus insulin need to be omitted from the data to which the method is applied. Therefore, eligible patients need to have moderately adjusted basal insulin settings and little preserved endogenous insulin secretion. Prandial data with insufficient time elapsed after another meal, another correction insulin, and hypoglycemia are excluded. Patients need to perform carbohydrate counting regularly and also need to record their logs to apply this method.
Patient data preparation and processing
In the following case presentation, CGM data were obtained through the manufacturer’s cloud system in comma-separated values format, from which meal timings, bolus insulin doses for each meal, and patient-recorded carbohydrate amounts were extracted. This study used 28-day sequential data. We then extracted 4-h prandial glucose values as prandial glucose trajectories. Each prandial glucose trajectory was categorized into breakfast, lunch, and dinner based on their start time, which spanned from 4:00 to 11:00, 11:00 to 17:00, and 17:00 to 24:00, respectively. How these prandial glucose data are included or excluded is illustrated in Figure 1. Only glucose data for the meals that have elapsed time of more than 4 h from another meal or another correction insulin injection and those more than 8 h from hypoglycemia are included. From the prandial glucose trajectories, glucose values after starting another meal, injecting another correction insulin, and postprandial hypoglycemia occurrence were excluded. To overview the prandial data, spaghetti plots describing prandial glucose trajectories for each meal and carbohydrate scatterplots comparing

Flowchart describing how prandial glucose values are included and excluded for the final CIR assessment. CF: correction factor,
Analysis of patient data was approved by the institutional review board. All data processing, visualization, and analysis were conducted in R version 4.5. 26
Results
We presented the CIR assessment method in a patient with T1DM who was engaged in carbohydrate counting. Seven months after having been diagnosed, the patient had a C-peptide of 0.23 ng/mL (plasma glucose 203 mg/dL) and HbA1c of 7.5%. The patient performed multiple daily injections of insulin. Long-acting insulin (glargine) was injected at 12 U once daily. The patient typically consumed 60, 60, and 70 g of carbohydrates for breakfast, lunch, and dinner. For these amounts of carbohydrates, the patient was instructed to use 11, 6, and 6 U of rapid-acting insulin (aspart), respectively, indicating that the CIRs were around 5.5:1, 10:1, and 11.7:1 for each meal timing. The typical total daily dose was 35 U, and CF was set at 50 mg/dL/U, which was derived from the 1700 rule.
9
A summary of the patient’s whole-day glucose levels is described in the AGP presented in Figure 2. To overview the extracted prandial glucose trajectories for each meal timing, spaghetti plots are shown (Fig. 3). How retrospective estimates,
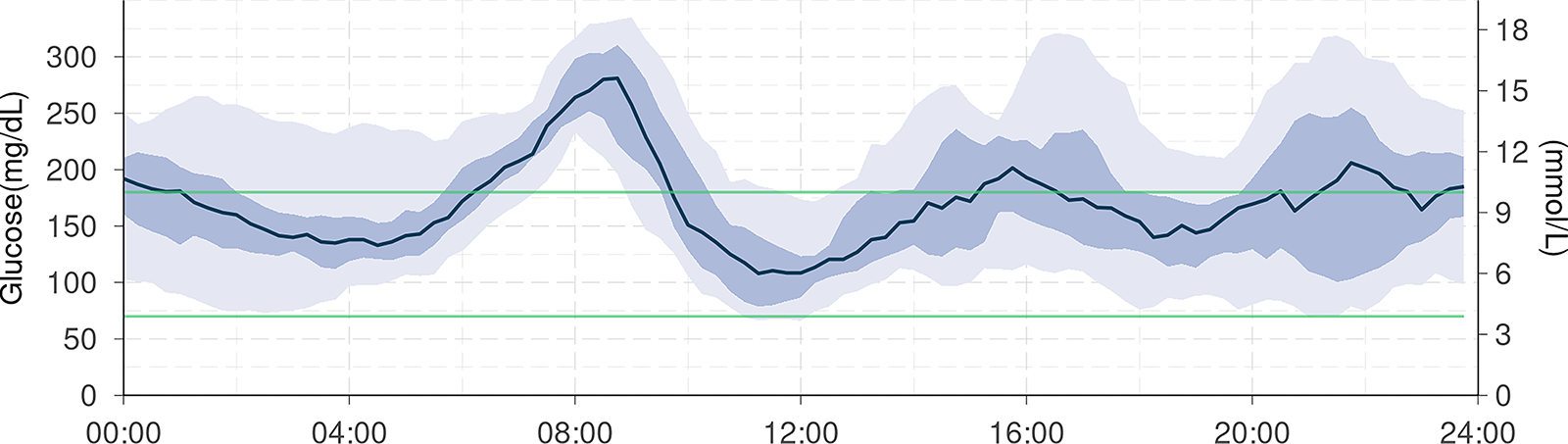
Ambulatory glucose profile for continuous glucose monitoring data at 7 months after diagnosis.

Spaghetti plots of prandial glucose trajectories for

Carbohydrate scatterplots comparing preprandial carbohydrate estimates with retrospective carbohydrate estimates for
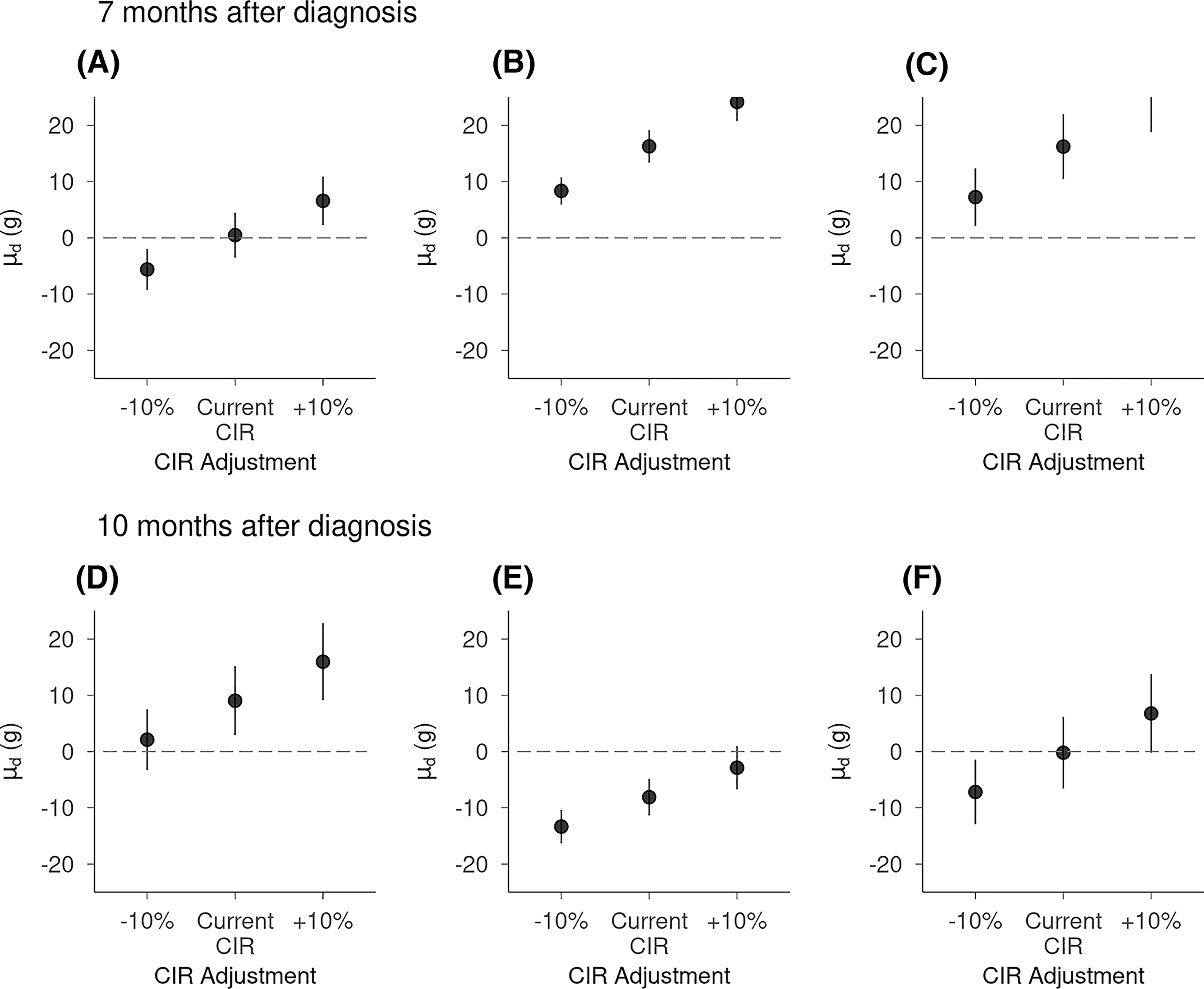
As mentioned earlier, to minimize the effect of CF in the assessment of CIR, prandial glucose trajectories with

Spaghetti plots and carbohydrate scatterplots for prandial data with small absolute values of

The same assessment was also conducted for data 10 months after the diagnosis. The patient’s C-peptide level decreased to 0.05 ng/mL (plasma glucose, 146 mg/dL), with an HbA1c of 6.4%. The patient continued with multiple daily injections. Detemir was injected twice daily at 8 and 7 U. The CIRs were 10:1, 8.6:1, and 10:1 for breakfast, lunch, and dinner, respectively. The typical total daily dose was around 35 U, and the CF was 50 mg/dL/U. The resulting plots are shown in Figure 7. The CIs for

Plots for data at 10 months after diagnosis.
CIR assessment
Based on the above results, the CIR assessment was conducted. When the CI for
Discussion
The current study presented a statistical method to assess CIR by comparing the retrospectively estimated carbohydrate with patient-measured preprandial carbohydrate. This method provides a logical framework to assess CIR for patients with insulin-dependent diabetes in clinical practice.
The novelty of this method lies not only in its clinical utility but also in its ability to provide statistical perspective for each patient’s daily data. The proposed method enables interpreting everyday prandial glucose profiles as repeated events of insulin and carbohydrate loading. By repeatedly measuring these events, prandial insulin-related parameters can be assessed more systematically than by just looking at a few arbitrary prandial glucose profiles.
Several key points should be noted to use this method in the clinical setting. First, this method does not directly provide information on the extent to which CIR should be adjusted. However, applying this method to the same data using a different hypothetical CIR value and assessing
Theoretical confidence intervals for
Second, even when the
Third, although the derived formula was based on a combination of widely adapted carbohydrate counting concepts, these concepts already simplify glucose metabolism, which is affected by various factors and should be complicated in reality. The process of adjusting the bolus insulin should account for each clinical situation. Special care should be taken when adjusting the CIR, especially in cases who frequently develop postprandial hypoglycemia. To easily determine the frequency of postprandial hypoglycemia, the number of postprandial hypoglycemia and its proportion in all included glucose trajectories are presented at the bottom of the carbohydrate scatterplots (Fig. 4 and Fig. 7D–F).
Fourth, this method cannot be applied to all patients with insulin-dependent diabetes. This method requires carbohydrate counting data and is not applicable to patients who do not regularly engage in carbohydrate counting. Such patients should not be forced to adopt this method and should instead be provided with alternative therapeutic approaches.
Fifth, CIR can change over time. Although this method can be applied repeatedly, it requires at least a few weeks to accumulate sufficient data. Complementary adjustment methods also need to be applied if the adjustments are required more frequently.
The following statistical points should also be considered. The current method eliminates glucose trajectories with the absolute value of
The current study assumed that the distribution of the differences in preprandial and postprandial estimates,
For better diabetes management, carbohydrate counting skills and CIR parameters need to be gradually improved and updated. Carbohydrate counting accuracy is one of the crucial factors in the presented method. Large errors in carbohydrate counting estimates produce large CIs for
To assess the carbohydrate counting accuracy, previous studies have used carbohydrate amounts defined by a team of experienced nutritionists as their true values. 27 In real-life settings, however, there are no ways to know the exact content of carbohydrate in each meal, which is an obstacle for patients improving carbohydrate counting skills. Once CIR is adjusted appropriately, retrospective estimation compared with preprandial estimation can tell how successful carbohydrate counting was for each meal, which is beneficial information to improve carbohydrate counting. The feedback through the retrospective estimation of carbohydrates can give patients opportunities to modify and improve their carbohydrate counting.
Even with innovative technologies, the benefits would be limited if patients do not have enough knowledge on diabetes. A previous study showed that in patients with T1DM, knowledge of diabetes and health literacy affected improvements in HbA1c among CGM users. 28 By visually sharing glucose profiles and logically assessing parameters, health care providers and patients can more easily share the disease status and their treatments, which should improve the understanding of their diseases.
Conclusions
This study presents a novel method to assess CIR for patients with insulin-dependent diabetes. This method also provides a statistical perspective of prandial CGM data. With the increasing availability of diabetes-related technology, methods that utilize new types of data need to be developed.
Footnotes
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
The authors declare that no funding was received for this research and/or publication of this article.
Authors’ Contribution
T.U.: Conceptualization, project administration, methodology, formal analysis, software, visualization, writing—original draft, and writing—review and editing. T.A., R.F., A.T., S.A., S.T., R.H., J.M., S.F., H.Y., and M.Y.: Validation. K.H.: Writing—review and editing.