Abstract
Background:
The benefits of Continuous Glucose Monitoring (CGM) on glycemic management have been demonstrated in numerous studies; however, widespread uptake remians limited. The aim of this study was to provide real-world evidence of patient attributes and clinical outcomes associated with CGM use across clinics in the U.S. based T1D Exchange Quality Improvement (T1DX-QI) Collaborative.
Method:
We examined electronic Health Record data from eight endocrinology clinics participating in the T1DX-QI Collaborative during the years 2017-2019.
Results:
Among 11,469 type 1 diabetes patients, 48% were CGM users. CGM use varied by race/ethnicity with Non-Hispanic Whites having higher rates of CGM use (50%) compared to Non-Hispanic Blacks (18%) or Hispanics (38%). Patients with private insurance were more likely to use CGM (57.2%) than those with public insurance (33.3%) including Medicaid or Medicare. CGM users had lower median HbA1c (7.7%) compared to nonusers (8.4%). Rates of diabetic ketoacidosis (DKA) and severe hypoglycemia were significantly higher in nonusers compared to CGM users.
Conclusion:
In this real-world study of patients in the T1DX-QI Collaborative, CGM users had better glycemic control and lower rates of DKA and severe hypoglycemia (SH) events, compared to nonusers; however, there were significant sociodemographic disparities in CGM use. Quality improvement and advocacy measures to promote widespread and equitable CGM uptake have the potential to improve clinical outcomes.
Keywords
Introduction
Intensive management of type 1 diabetes with the goal of maintaining tight glycemic control reduces the risk of long-term microvascular and macrovascular complications. 1 However, glycemic control remains suboptimal in the majority of patients with type 1 diabetes in the United States. The American Diabetes Association (ADA) guidelines recommend a hemoglobin A1c (HbA1c) goal of <7% for people with diabetes; however, in a 2018 study of the T1D Exchange Clinic Registry, only about 17% of children and adolescents <18 years old achieved HbA1c < 7.5 and 21% of adults achieved HbA1c <7.0%. 2
Advances in diabetes technology have aided clinicians and patients to monitor and manage glucose levels more readily and safely. Numerous studies have demonstrated the benefit of continuous glucose monitoring (CGM) in reducing HbA1c and incidences of diabetic ketoacidosis (DKA) and severe hypoglycemia (SH) in both adults and adolescents.3,4 In addition, studies have shown an improvement in quality of life with CGM use.1,3-5 Despite the existing clinical evidence and improved performance of CGM devices, widespread utilization and uptake of CGM has been limited, with only approximately 30% of patients in the T1D Exchange Clinic Registry using CGM in 2018. 2
The T1D Exchange Quality Improvement (T1DX-QI) Collaborative was established as a learning platform to evaluate the current state of care and engage diabetes clinics in collaborative quality improvement initiatives and data sharing. 6 The aim of this study was to provide real-world evidence of patient attributes and clinical outcomes associated with CGM device use in the T1DX-QI Collaborative.
Methods
The T1DX-QI Collaborative is a large cohort with over eighteen U.S. diabetes clinics engaged in data sharing and quality improvement to improve clinical outcomes. At the time of this IRB-approved analysis, electronic medical record data mapping was completed and available for 12,958 patients from 6 pediatric and two adult diabetes clinics. Data from all patients receiving care that the 8 clinics were included in the study. However, the 8 clinics was a convenience sample of the T1DX-QI clinics that have completed the EMR data transfer and quality assurance at the time of the analysis. This clinic sampling method could have led to selection bias, as there may be differences in centers included with respect to resource capacity, patient volume, and demography. Patients with one or more completed clinic encounters during the years 2017-2019 were included (N = 11,469). We examined the frequency of CGM use based on socio-demographic factors, as well as clinical outcomes including HbA1c, incidences of DKA and SH among CGM users versus nonusers. HbA1c levels were obtained as part of routine clinical care. DKA events were defined as a triad of hyperglycemia, that is, plasma glucose >250 mg/dL (>13.88 mmol/L), venous pH <7.3 and/or bicarbonate <15 mmol/L, with moderate or large ketone levels in urine or blood. SH was defined as an event that required assistance from another person due to altered consciousness.
Statistical Analysis
In this analysis, patients with type 1 diabetes were classified as CGM users if they indicated information on a device start date, or model/company of CGM within the past 1-year of their most recent diabetes clinic visit, and as CGM nonusers if this information was not recorded at their most recent clinic visit. Race/ethnicity was categorized as Non-Hispanic White, Non-Hispanic Black, Hispanic, Other, or Unknown. The Other category was composed of Asians, American Indian or Alaska Native, or Native Hawaiian. Insurance status was classified as public (including Medicare and Medicaid), private (including private and military insurance), or others (for patients whose insurance status was reported as other or unknown). Clinical outcomes considered in this study were HbA1c levels, DKA and SH. HbA1c was analyzed as a continuous variable, whereas DKA and SH were taken as categorical variables, with patients being classified as having had at least one event if it was recorded on EMR or reported by patients during clinic visits within the past year. Demographic data, CGM use (either real-time CGM or intermittent scan/flash device), insulin modality (injections or insulin pump), and the most recent HbA1c value were available from the most recent clinic visit. For additional confirmatory analysis, we also examined the distribution of mean A1c values within the last year of the most recent clinic visit. Differences in sociodemographic and clinical factors between CGM users and nonusers were examined using chi-square and
Results
Demographic and clinical characteristics for CGM users (N = 5483) and nonusers (N = 5986) are presented in Table 1. In this cohort of type 1 diabetes patients, 48% were CGM users: 60% used a real-time CGM, 2% used an intermittent scan/flash device and for 38% the device type was not specified. In this dataset, we had complete information on CGM use status, however, in the overall patient population insurance data were missing for about 10% (N = 1181) and race/ethnicity were missing for 6% (N = 711).
Participant Characteristics.
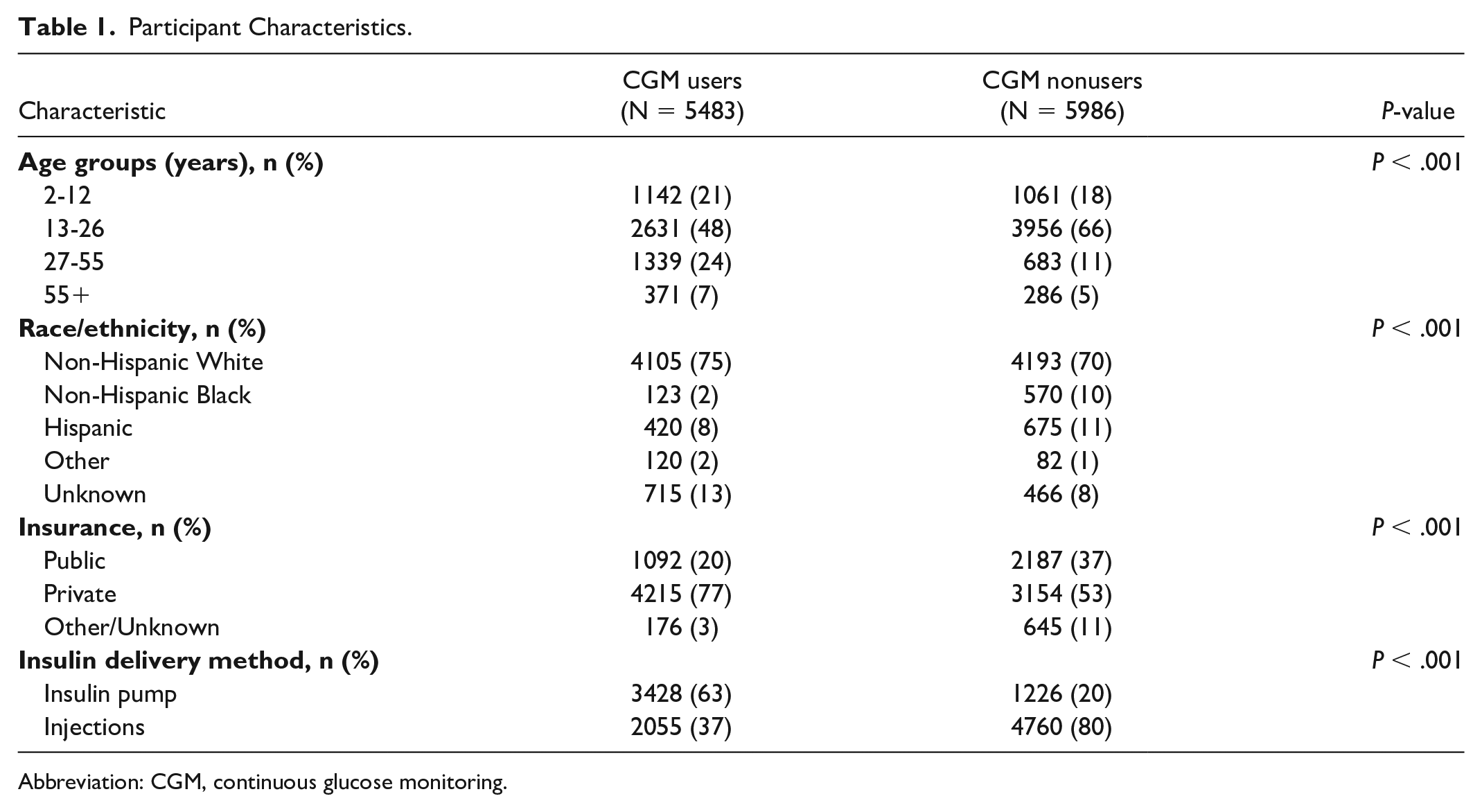
Abbreviation: CGM, continuous glucose monitoring.
In this real-world cohort, adult patients were more likely to be CGM users (66.2% of those aged 27-55 years, 56.5% aged 55 years and older) than pediatric patients (51.8% of patients aged 2-12 years, 39.9% aged 13-26 years) (
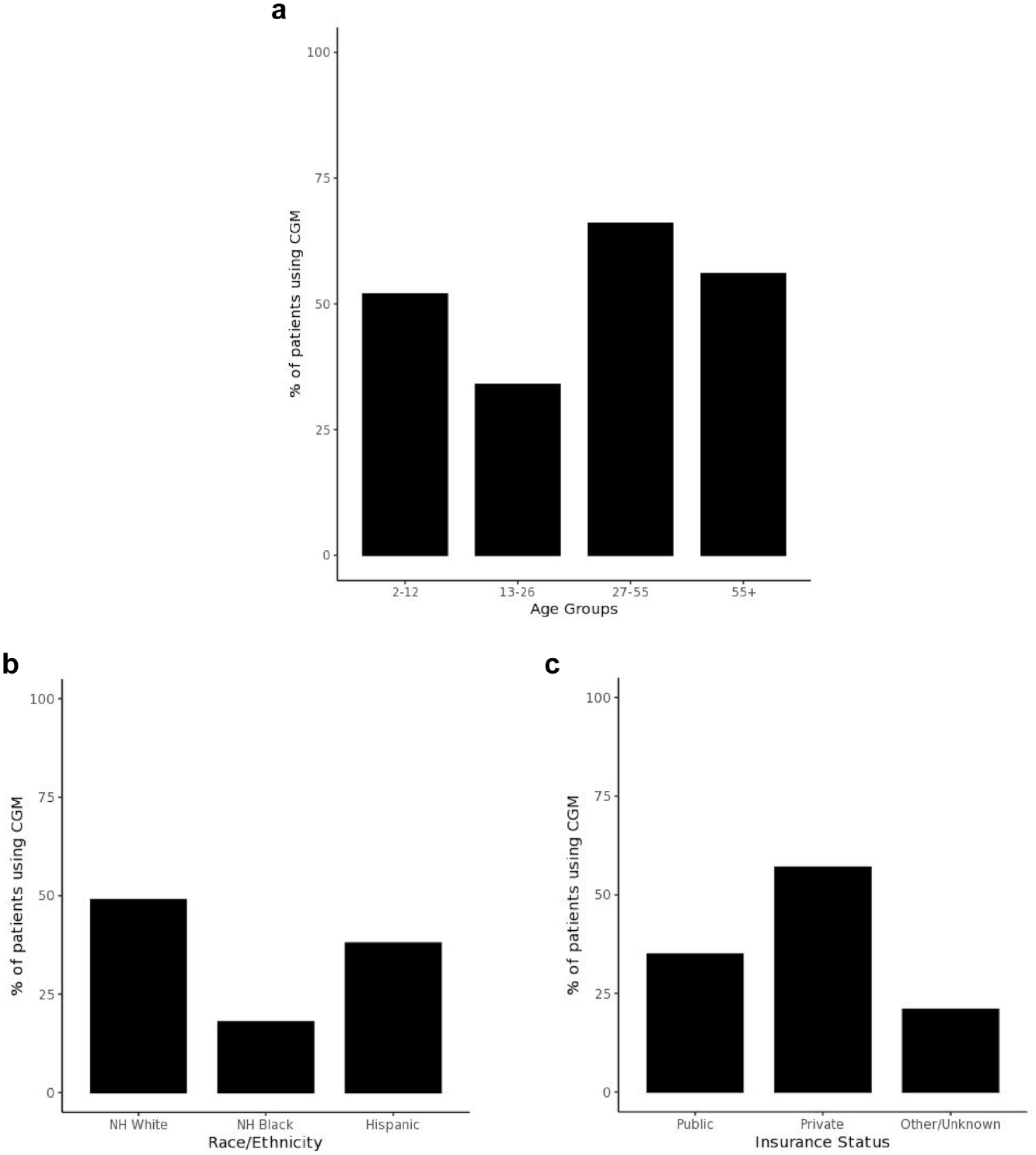
Patient attributes and CGM use. (a) CGM use by age in the T1DX-QI Collaborative. (b) CGM use by race/ethnicity in the T1DX-QI Collaborative. (c) CGM use by insurance status in the T1DX-QI Collaborative. Abbreviations: CGM, continuous glucose monitoring; T1DX-QI, T1D Exchange Quality Improvement.
CGM users in the T1DX-QI Collaborative had lower HbA1c compared to nonusers (median [interquartile range (IQR)]): 7.7% [6.9%,8.6%] vs. 8.4% [7.4%,9.5%];
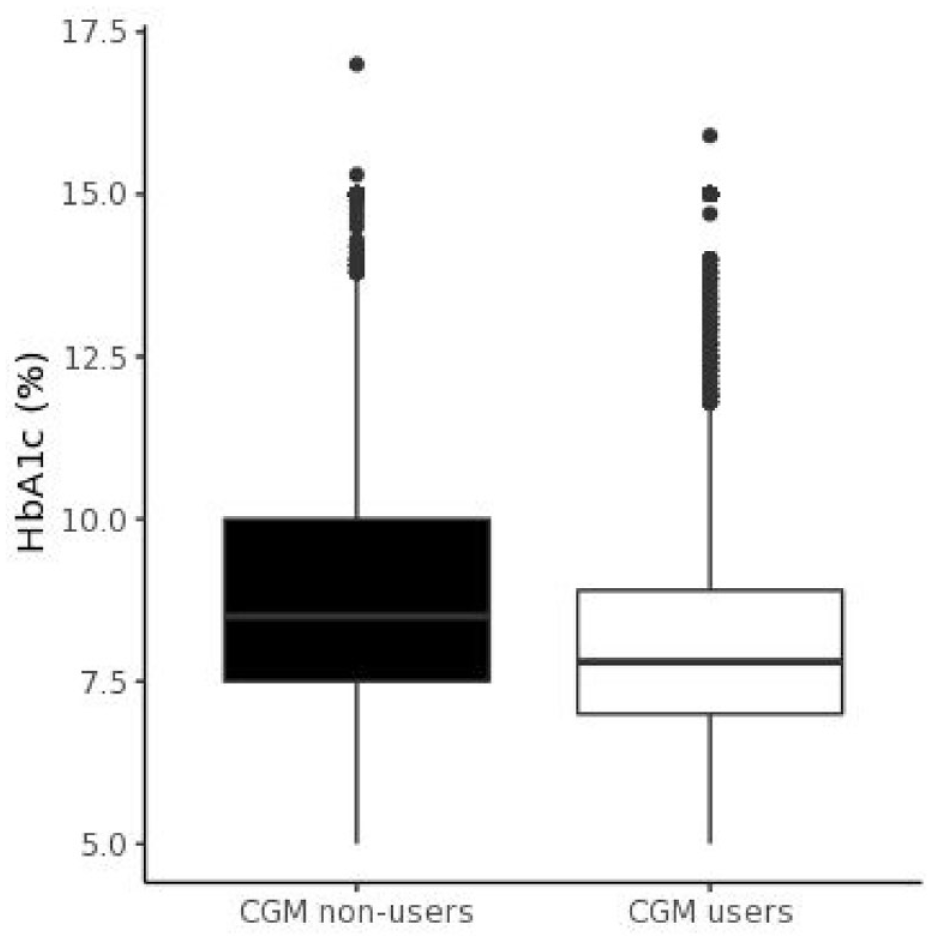
HbA1c levels by CGM nonusers versus CGM users in the T1D Exchange Quality Improvement Collaborative. (Median [IQR]: 8.4% [7.4%, 9.5%] for nonusers vs. 7.7% [6.9%, 8.6%] for CGM users;
CGM Device Use and Clinical Outcomes by Insurance Status and Race/Ethnicity of People With T1D.
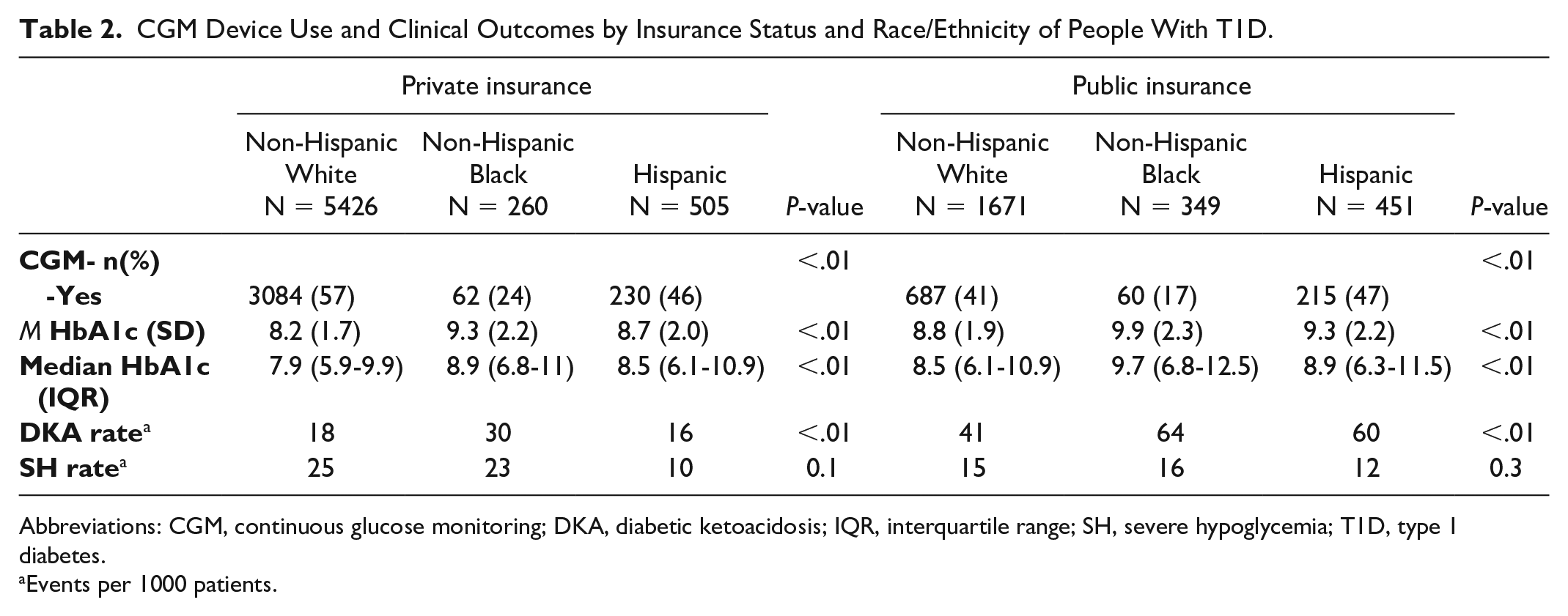
Abbreviations: CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; IQR, interquartile range; SH, severe hypoglycemia; T1D, type 1 diabetes.
Events per 1000 patients.
Discussion
This study highlights real-world evidence of patient attributes and clinical outcomes among CGM users in the T1DX-QI Collaborative, one of the largest type 1 diabetes cohorts available to-date. The findings from this study shed light on sociodemographic variability in CGM uptake with implications for targeted quality improvement interventions to promote more equitable device use in the type 1 diabetes population.
In this real-world study of patients in the T1DX-QI Collaborative, CGM users had better glycemic control and lower rates of DKA and SH events, compared to nonusers. These findings are in line with previous studies.3,4 The benefits of CGM were not available to all patients in the T1DX-QI Collaborative as rates varied across age groups, race/ethnicity and insurance type.
Interestingly, adults in the T1DX-QI Collaborative were more likely to use CGM than pediatric patients, which is discrepant from previous reports in the T1D Exchange Clinic Registry.2,7 The T1DX-QI Collaborative represents all patients in the clinic population, whereas the T1D Exchange Clinic Registry is only a subset of participants who were enrolled for research purposes. In this analysis of the T1DX-QI Collaborative, only 2 adult clinics were included, and diabetes technology use is a strong focus of their programs. Thus, the finding of higher CGM use in adults compared to youth with type 1 diabetes should be interpreted with caution given the limited sample of adult clinics. In the pediatric age group, younger children were more likely to use CGM than adolescents and young adults, which is consistent with previous studies2,8 and may be related to reluctance of teenagers and young adults to wear CGM.
The lower rates of CGM use in racial/ethnic minorities in the T1DX-QI Collaborative is concerning. Disparities in rates of device use have been reported in previous studies.9-11 Foster et al 2 demonstrated that African Americans have higher HbA1c than non-Hispanic whites or Hispanics across all age groups, even after adjusting for differences in socioeconomic status, so increasing CGM use is paramount for this group. Valenzuela et al 11 suggested that racial/ethnic disparities in device use may be perpetuated by subconscious racial stereotyping by providers assessing minority patients’ preparedness for the device.
Socioeconomic status and insurance coverage can impact diabetes device use (insulin pump and CGM) due to financial constraints of families.10,12 In this real-world analysis of U.S. clinics, patients with public insurance were less likely to use CGM than those with private insurance. The T1DX-QI Collaborative represents a geographically diverse collaborative with variability in Medicaid coverage of CGM across sites. The lack of Medicaid coverage for CGM in some states may have been a contributing factor in the disparity in CGM use by insurance type.
There were limitations in our study. First, this is a cross-sectional design, which does not allow determination of causality of CGM use with glycemic control or rates of DKA or SH. While DKA and SH were defined based on standard ADA clinical definitions, there may have been variation in how these events were captured across electronic medical record systems or self-report at clinic visits. Second, we employed convenience sampling to include 8 of the 18 T1Dx-QI member clinics in this study, as these sites were EMR mapped at the time of this analysis, while other centers were still undergoing the mapping process. This sampling method could have led to some selection bias, as there may be differences in centers included with respect to resource capacity, patient volume and demography. Third, data for race/ethnicity, insurance type, and CGM model were not available for all patients. Data on level of education and family income, important factors in diabetes outcomes, were not available for this analysis. Additionally, the disparities in CGM use may be influenced by unmeasured variables such as patient preference or provider biases in interpreting patients’ device preparedness. Lastly, this study analyzes data on type 1 diabetes (T1D) patients from 8 T1DX-QI member clinics that have completed the EMR data sharing and quality assurance process as of the time of analysis. While this includes the total population seeking care at each center, there may be some selection bias as people actively seeking care are likely to be overrepresented in this study population. The T1DX-QI cohort is a clinical and QI database of all patients seeking care at member clinics, which is notably different from the T1D Exchange Registry cohort, wherein, only data from select individuals who provided informed consent for research purposes were included in the data analyses. To that end, the distribution of certain key demographics in the T1DX-QI cohort, such as age, gender, and race/ethnicity, trends close to the those presented by other T1D U.S. population-based studies.13,14
This study reveals disparities in CGM uptake that could be addressed through quality improvement initiatives. In agreement with other studies demonstrating racial/ethnic disparities in adoption of diabetes technology, which have further been exacerbated during the COVID-19 pandemic,15,16 this study additionally demonstrates that despite adequate insurance coverage, inequities in diabetes device use by race/ethnicity and subsequent clinical outcomes still persist, alluding to other social and provider barriers which may play a role in prescription and adoption of CGM for diabetes management. To that end, the T1DX-QI Collaborative is engaged in applying quality improvement methods 17 to test and expand several interventions to increase CGM use17-19 and reduce inequities in device use. 20 Clinics are focused on patient education, clinician training, creating resources and navigating vendor and payor systems with patients and families. Essential to this quality improvement work has been co-production through partnership with patients/family experts to design the interventions as integral work of the T1DX-QI Collaborative. In an effort to promote widespread and equitable CGM use, clinics in partnership with patients and families have created a range of tools to be tested locally and spread as best practices across the collaborative.
Conclusion
In this real-world cohort of patients in the T1D Exchange QI Collaborative, we observed sociodemographic disparity in CGM use. Quality improvement and advocacy measures to promote widespread and equitable CGM uptake have the potential to improve glycemic control and reduce adverse events including DKA and SH.
Footnotes
Abbreviations
T1D, type 1 diabetes; CGM, continuous glucose monitoring.
Authors’ Note
Portions of this study were presented at the American Diabetes Association Scientific Sessions, June 2020 and 46th Annual ISPAD Conference, October 2020
Author Contributions
D. J. D., N. N., O. E. developed the concept for the manuscript. N. N. analyzed the data. D. J. D., C. X. wrote the manuscript. All authors reviewed, edited and approved the final version and take responsibility for contents of the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DJD has received independent consulting fees from Dexcom and Insulet. N. N., C. X., S. D. C., S. M., R. J. M., S. P., R. I., N. R., R. W., K. O., A. R., F. V., J. S., and O. E. have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Leona M. and Harry B. Helmsley Charitable Trust.
