Abstract
Purpose
This study aimed to investigate the associations of the Geriatric Nutritional Risk Index (GNRI) and the simplified Geriatric Nutritional Risk Index (sGNRI) with Chronic Obstructive Pulmonary Disease (COPD).
Methods
We assessed 161 patients with COPD and 947 healthy controls. The sGNRI was derived using the formula: serum albumin (g/dL) + 0.1 × body mass index (BMI, kg/m2). Primary outcomes included the BODE (body mass index, airflow obstruction, dyspnea, exercise capacity) index and hospital length of stay (LOS). The secondary outcome was the prevalence of COPD.
Results
Malnourished patients (GNRI <95.40 or sGNRI <5.89) exhibited lower BMI and albumin levels, as well as worse pulmonary function (all
Conclusions
Compared with GNRI, sGNRI may serve as a more practical tool for identifying malnutrition-related risk among patients with COPD.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a primary cause of morbidity worldwide. 1 In 2019, the global prevalence of COPD was approximately 10.3%, affecting about 391.9 million people, with more than three-quarters of global COPD cases occurring in low- and middle-income countries. 2 Current evidence suggests that malnutrition is prevalent among people with COPD.3,4 A recent analysis demonstrated that nearly a quarter of COPD patients are in an impaired nutritional state, 5 which is associated with poor quality of life, worse physical performance, and an increased risk of exacerbation. 6 Additionally, the main contributors to malnutrition in COPD patients include insufficient dietary intake, increased resting energy expenditure, enhanced thermic response to nutrients, and humoral factors.6,7 Given these factors, there is a critical need for diagnostic tools that are both practical and suitable for accurately identifying malnutrition.
The Geriatric Nutritional Risk Index (GNRI), initially developed by Bouillanne et al., 8 has been extensively demonstrated in numerous studies to predict the prognosis of various diseases, such as pneumonia and cancer.9–12 Additionally, GNRI has been associated with the prevalence of depression, stroke, and peripheral artery disease.13–15 More recently, a study by Xuemin Chai et al. 16 found the GNRI to be a predictor of all-cause mortality in COPD patients. Nonetheless, the clinical utility of GNRI is limited by its computational complexity and the lack of straightforward interpretability.
The above-mentioned shortcomings highlight the need for more simplified assessment tools. Recent evidence suggests that the simplified Geriatric Nutritional Risk Index (sGNRI) calculation offers a more practical approach for malnutrition screening in maintenance hemodialysis patients. 17 However, its validity in COPD remains to be established.
To address this gap, this study analyzed the predictive utility of both GNRI and sGNRI in assessing the prevalence and severity of COPD, while comparing their diagnostic performance.
Materials and methods
Study design and participants
This was a retrospective case-control study. All data were obtained from 296 patients admitted for COPD at the Third Affiliated Hospital of Wenzhou Medical University from February 1, 2018, to July 5, 2022, and from 2196 healthy individuals admitted to the same hospital during the same period. For the COPD patients, the inclusion criteria were as follows: (1) age ≥40 years; (2) diagnosis of COPD confirmed by Forced Expiratory Volume in one second (FEV1)/Forced Vital Capacity (FVC) < 0.7; (3) availability of data on serum albumin, body mass index (BMI), BODE (BMI, airflow obstruction, dyspnea, exercise capacity) index, and hospital length of stay (LOS). Initially, 215 patients were included, and a total of 54 patients were excluded based on the following criteria: (1) patients with asthma; (2) hyponatremia (<135 mmol/L) and hypernatremia (>145 mmol/L) 18 ; (3) severe hepatic insufficiency, renal failure, or malignant tumors. 19 Consequently, the final analysis included 161 COPD patients. Separately, a total of 947 healthy people aged ≥40 years with available data on albumin and BMI were included.
Data collection
Demographic information, including age, sex, smoking status, and comorbidities, was obtained from patients’ electronic medical records. Blood biochemical parameters, such as albumin, were collected within 24 h of admission. The pulmonary function tests were performed by experienced clinicians using spirometry. In addition, the BODE index was assessed in COPD patients within 7 days of hospital admission. Prolonged LOS was defined as ≥ 11 days, according to the 75th percentile of LOS in our sample. 20
Primary outcomes and main exposure
The primary outcomes were the BODE index and LOS, which are comprehensive measures of COPD severity. The BODE index is a multidimensional grading system for COPD, 21 whereas LOS reflects the severity and resource utilization. 22 The secondary outcome was the prevalence of COPD. The sGNRI was calculated as follows: sGNRI = serum albumin (g/dL) + 0.1 × BMI (kg/m2). 17 The GNRI was calculated based on serum albumin levels (g/dL), present body weight (PBW, kg) and ideal body weight (IBW = height 2 (m2) × 22). See the following formula for details: GNRI = 14.89 × serum albumin +41.7 × PBW/IBW. 8 When the PBW exceeded the IBW, their ratio was considered to be 1.
Statistical analysis
Statistical analyses were performed via SPSS Statistics 27.0 software and R version 4.3.3. Skewed continuous variables were expressed as medians and interquartile range, while normally distributed continuous variables were expressed as mean ± standard deviation. Categorical variables were expressed as frequencies and percentages. Intergroup differences for skewed continuous variables were compared using the Mann-Whitney U-test, whereas normally distributed continuous variables employed independent samples t-tests. The Chi-square test was performed for categorical variables. Propensity score matching (1:1 match, caliper 0.02) was used to compare baseline characteristics, GNRI and sGNRI between the two groups (with and without COPD). COPD patients were divided into malnutrition-related risk group (GNRI <95.40 or sGNRI <5.89) and no malnutrition-related risk group (GNRI ≥95.40 or sGNRI ≥5.89), with the cut-off value for both GNRI and sGNRI determined by the maximum Youden index (sensitivity + specificity - 1). Logistic regression analysis was used to evaluate GNRI and sGNRI as independent predictors of the prevalence of COPD. Spearman correlation analysis was conducted to assess the correlation between nutritional assessment tools and primary outcomes. Univariate and multivariable logistic regression analyses and linear regression were used to screen for risk factors associated with BODE ≥5 and LOS. Model one included age and smoking status. In Model 2, FEV1/FVC and comorbidities were adjusted, including hypertension and heart disease. Restricted cubic spline (RCS) curves were further used to explore the exposure-response relationship. Additionally, we compared the accuracy of GNRI and sGNRI by analyzing receiver operating characteristic (ROC) curves, positive predictive value (PPV; the probability that subjects with a positive screening test truly have the condition), and negative predictive value (NPV; the probability that subjects with a negative screening test truly do not have the condition). Finally, subgroup analyses were conducted to explore the stability of the results.
Results
Participant characteristics
A comparison of the baseline characteristics of the COPD group and the non-COPD group is shown in Table S1. A total of 1108 participants were included in our study, with a mean age of 50.00 years and a female-to-male ratio of 1.21:1. The cohort comprised 161 participants with COPD and 947 without COPD. After propensity matching, the COPD group exhibited significantly lower values for albumin, GNRI, and sGNRI (all
Comparisons of baseline characteristics and outcomes between malnutrition-related risk and no malnutrition-related risk groups.

COPD: Chronic obstructive pulmonary disease; BMI: Body Mass Index; GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index; FVC: Forced Vital Capacity; FEV1: Forced Expiratory Volume in one second; FEV1/FVC: forced expiratory volume in one second to forced vital capacity ratio; BODE: BMI, airflow obstruction, dyspnea and exercise capacity; LOS: hospital length of stay.
Correlation of GNRI and sGNRI with COPD
Logistic regression analyses of GNRI and sGNRI for COPD and non-COPD groups.

COPD: Chronic obstructive pulmonary disease; GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index.
To further visualize these relationships, Spearman correlation analysis was conducted, revealing that while both GNRI and sGNRI were negatively associated with the BODE index, only sGNRI was negatively associated with LOS (Figure 1). Association of GNRI and sGNRI with BODE and LOS. Note: GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index; BODE: BMI, airflow obstruction, dyspnea and exercise capacity; LOS: hospital length of stay. *indicates 
Univariate logistic regression analyses were next conducted to identify the risk factors associated with BODE ≥5. The significant risk factors identified included: BMI (OR = 0.770, 95% CI: 0.682-0.870,
Multivariable logistic regression analyses and linear regression of GNRI and sGNRI for BODE ≥5 and LOS.

GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index; BODE: BMI, airflow obstruction, dyspnea and exercise capacity; LOS: hospital length of stay.
RCS curves were plotted and revealed negative linear correlations for both GNRI and sGNRI with BODE ≥5 (GNRI:
Accuracy of sGNRI compared with GNRI in predicting COPD
Comparison of predictive performance between GNRI and sGNRI for predicting BODE ≥5 and LOS ≥11day.
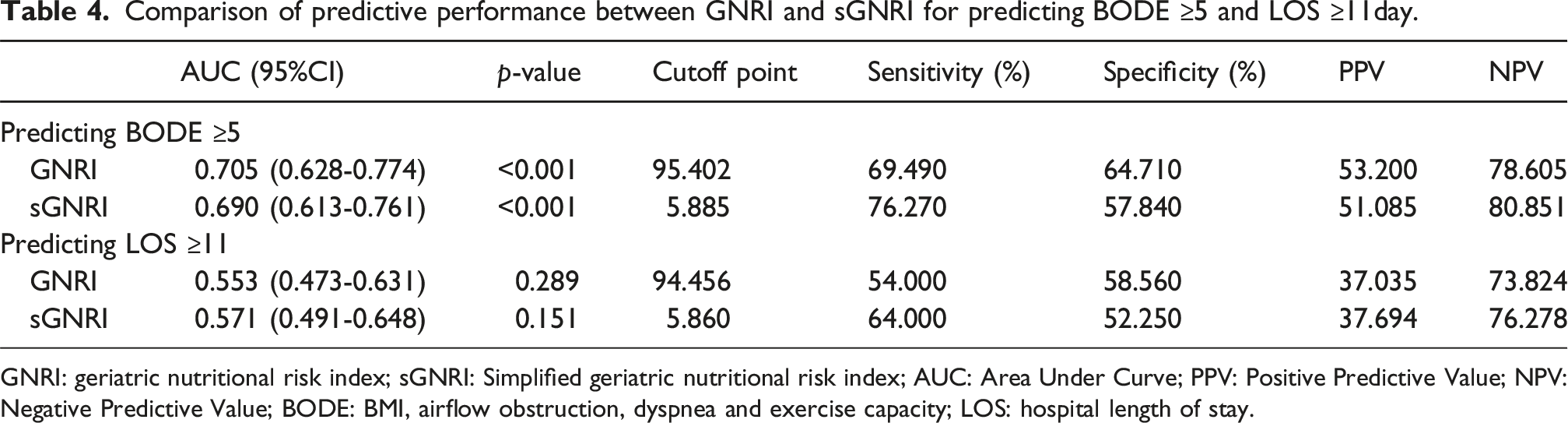
GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index; AUC: Area Under Curve; PPV: Positive Predictive Value; NPV: Negative Predictive Value; BODE: BMI, airflow obstruction, dyspnea and exercise capacity; LOS: hospital length of stay.
Predictive performance of GNRI and sGNRI in different subgroups
The association of GNRI and sGNRI with BODE ≥5 in subgroups.

GNRI: geriatric nutritional risk index; sGNRI: Simplified geriatric nutritional risk index; FEV1/FVC: forced expiratory volume in one second to forced vital capacity ratio; BODE: BMI, airflow obstruction, dyspnea and exercise capacity.
Discussion
To the best of our knowledge, this is the first study to investigate the feasibility of sGNRI in detecting the prevalence and severity of COPD. Our results demonstrated that both GNRI and sGNRI were negatively associated with the prevalence and severity of COPD. We also observed a significant interaction effect between hypertension and both GNRI and sGNRI. Current evidence suggests that individuals with hypertension are more prone to obesity, 23 which may account for the observed attenuation in the predictive capacity of GNRI and sGNRI in this population.
Compared with healthy individuals, patients with COPD are more susceptible to malnutrition, indicating a close association between these two conditions. In the present study, the severity of COPD was reflected using the BODE index and LOS. The BODE index is a straightforward grading system for COPD that incorporates one domain quantifying the degree of pulmonary impairment (FEV1), one capturing the patient’s perception of symptoms (the Modified Medical Research Council dyspnea scale), and two independent domains (the distance walked in 6 minutes and BMI). 21 The observed negative correlation between sGNRI and BODE suggests that malnutrition is associated with multiple domains of COPD severity. Similarly, sGNRI was inversely associated with LOS, indicating that malnutrition is linked to prolonged hospitalization, which could be explained by factors such as increased susceptibility to complications, reduced recovery capacity, or an increased treatment burden. 24
Existing tools for screening nutritional status have their limitations. The longitudinal measurement of body composition necessitates technologies like bio-electrical impedance (BIA) and dual energy X-ray absorptiometry (DXA), which remain inaccessible in many primary hospitals with limited medical resources.25,26 Other nutritional markers, such as Onodera’s prognostic nutritional index (OPNI), Nutritional Risk Screening 2002 (NRS 2002) and Nutric score, exhibit lower sensitivity compared to the GNRI in geriatric patients with respiratory failure in the intensive care unit (ICU). 27 The GNRI, first proposed by Bouillanne et al., 8 has recently been applied to various diseases, including acute ischemic stroke, heart failure, and non-small cell lung cancer.28–30 Importantly, we found that GNRI was a valid tool for predicting the prevalence and severity of COPD, consistent with the findings reported by Xuemin Chai et al. 16
Despite its advantages, the calculation of the GNRI can be relatively complex. To the best of our knowledge, the sGNRI represents a mathematical simplification of the GNRI, achieved by dividing the serum albumin coefficient and rounding the BMI coefficient. 17 Therefore, this simplified formula yields single-digit values, enhancing interpretability. In a study by Shunsuke Yamada et al. 17 sGNRI was identified as a promising surrogate marker of nutritional status and mortality in patients receiving maintenance hemodialysis. Notably, in the present study, we found a negative association of sGNRI with both the prevalence and the severity of COPD.
To further evaluate the predictive performance of sGNRI and GNRI, ROC analysis was conducted, revealing subtle differences between the two indices in predicting the severity of COPD. The sGNRI demonstrated higher sensitivity and NPV, making it more suitable for the rapid identification of potentially malnourished patients in primary care and community screening, thereby ensuring that high-risk populations are not overlooked. However, its lower specificity may lead to false positives and unnecessary referrals. Conversely, the GNRI’s superior specificity makes it more suitable for specialized hospitals or resource-rich environments, facilitating the confirmation of malnutrition and avoiding over-intervention. Therefore, the selection of screening tools should be determined through a comprehensive assessment of the clinical context, available resources, and specific screening objectives.
Given its accessibility, convenience and association with the BODE index and LOS, the sGNRI can serve as a valuable tool for predicting disease progression during both admission and outpatient visits. Therefore, sGNRI is conducive to initiating preventive measures before the patient deterioration, thereby improving patient outcomes and reducing healthcare costs associated with COPD exacerbations. In primary care settings, general practitioners (GPs), practice nurses and community-based allied health clinicians can integrate sGNRI into regular health assessments during regular check-ups. For instance, an sGNRI score below the threshold (e.g., <5.89) can trigger further nutritional assessment and intervention, such as nutritional supplement therapy. Such therapies include the intake of high-calorie foodstuffs, fruits, vegetables, and other essential nutrients. 6 This integration not only enhances the efficiency of nutritional risk identification but also contributes to the optimal allocation of healthcare resources by avoiding unnecessary complex assessments.
The sGNRI is composed of albumin and BMI, both of which are useful biomarkers for adult malnutrition. 31 In COPD, inadequate nutrition is a key factor driving the development of muscle weakness, exercise intolerance, and ultimately dyspnoea, 32 which may lead to a higher BODE score and prolonged hospitalization. Existing evidence indicates that hypoalbuminemia adversely affects prognosis and LOS of acute decompensated chronic obstructive pulmonary disease. 33 This is mechanistically supported by albumin’s role in antioxidant defense. 34 In addition, albumin is considered a negative acute-phase protein. 35 It is now understood that COPD is characterized by chronic inflammation. 36 Moreover, oxidative stress is a major driving mechanism in the pathogenesis of COPD. 37 A study revealed that COPD patients exhibit significantly lower serum albumin levels, indicating deficits in systemic anti-inflammatory and antioxidant defense mechanisms. 38 Additionally, a lower BMI level has been linked to prolonged LOS and exacerbation of COPD.39,40 The association can be explained by several factors, such as muscle loss and decreased lung function.41,42 Collectively, the robust association between sGNRI and COPD outcomes is supported by underlying biological rationale.
This study possesses several strengths that should be acknowledged. To our knowledge, this study is the first to suggest the feasibility of sGNRI for detecting the prevalence and severity of COPD. In addition, stable associations of sGNRI and GNRI with COPD were observed across most subgroups. Nevertheless, key limitations of this study should be acknowledged. First, given the single-center design of our study, the generalizability of our findings to other populations or settings may be limited. Multicenter cohorts are needed to further validate the conclusions of this study. Second, the sample size of COPD patients was limited, underscoring the importance of determining the effectiveness of sGNRI and GNRI in a larger COPD population. Third, given the retrospective nature of our study, more prospective longitudinal studies are warranted to clarify the clinical significance of the sGNRI and GNRI in COPD. Furthermore, the observed associations between both sGNRI and GNRI with COPD prevalence demonstrated limited predictive utility for incident COPD cases, warranting further investigation to elucidate these relationships. Moreover, the potential for residual confounding must be acknowledged as an inherent methodological limitation.
Conclusion
This study demonstrated the prognostic value of GNRI and sGNRI for clinical outcomes of COPD patients. However, given its simplified calculation and higher sensitivity, sGNRI may serve as a more practical tool for identifying malnutrition-related risk among COPD patients. Overall, these findings can help healthcare professionals identify individuals at risk of malnutrition, initiate timely preventive care and monitoring, and potentially mitigate adverse COPD-related outcomes.
Supplemental Material
Supplemental material - Associations of the geriatric nutritional risk index and simplified geriatric nutritional risk index with chronic obstructive pulmonary disease
Supplemental material for Associations of the geriatric nutritional risk index and simplified geriatric nutritional risk index with chronic obstructive pulmonary disease by Yiben Huang, Ruizi Xu, Xuanyang Chen, Siyao Chen, Zicong Dai, Xinran Li, Yiting Yu, Zihan Ye, Xianjing Chen, Chunyan Liu, Beibei Yu, Yage Xu, Xiaodiao Zhang in Chronic Respiratory Disease.
Footnotes
Acknowledgement
We would like to express our gratitude to all of the participants, as well as to the medical professionals from the Third Affiliated Hospital of Wenzhou Medical University.
Ethical considerations
The research protocols were approved by the Ethics Committees of the Third Affiliated Hospital of Wenzhou Medical University. The research adhered to the principles outlined in the Declaration of Helsinki.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Wenzhou Municipal Sci-Tech Bureau Program (No.Y20210842)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are not publicly available to ensure confidentiality of the patients but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
