Abstract
Background
Breathlessness is a common symptom related to a significant health burden. However, the association of breathlessness with clinical characteristics, especially objective pulmonary test results is scarce. We aimed to identify the characteristics independently associated with breathlessness in Australian adults.
Method
The analysis used data from BOLD Australia, a cross-sectional study that included randomly selected adults aged ≥40 years from six sites in Australia. Clinical characteristics and spirometry results were compared for breathlessness (modified Medical Research Council [mMRC] grade ≥2).
Results
Among all respondents (n = 3321), 252 participants (7.6%) reported breathlessness. The main univariate associations were obesity, chronic respiratory diseases, heart diseases and being Indigenous Australians (odds ratios [ORs] = 2.78, 5.20, 3.77 and 4.38, respectively). Participants with breathlessness had lower pre-and post-bronchodilator lung function than those without. Impaired spirometry results including FVC or FEV1 below 80% predicted, or FEV1/FVC < LLN were independently associated with breathlessness (adjusted ORs = 2.66, 2.94 and 2.34, respectively).
Conclusions
Breathlessness is common among Australian adults and is independently associated with obesity, chronic respiratory diseases, heart diseases, being Indigenous Australians, and impaired spirometry. Multi-disciplinary assessment and comprehensive investigation is needed in clinical practice to address the many factors associated with breathlessness in the population.
Keywords
Introduction
Breathlessness is one of the most common symptoms in the general population and is strongly associated with a significant burden of poor health, including lower quality of life, greater health service use, and premature death.1–3 The most common causes of breathlessness are cardiorespiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD) and chronic heart failure. 4 In addition to cardiorespiratory diseases, other factors contribute to breathlessness. The London Respiratory Network identified the Breathing SPACE (smoking, pulmonary disease, anxiety/psychosocial factors, cardiac disease, exercise level/unfitness) as important and often interacting risk factors causing breathlessness. 5
Self-reported breathlessness has often been assessed using the modified Medical Research Council (mMRC) dyspnoea scale. 6 A study of lung disease in Latin America, found that characteristics including sex, age, education, forced expiratory volume in 1 s (FEV1), comorbid diseases, wheeze, cough, phlegm and health-related quality of life had a significant impact on breathlessness defined by mMRC. 7 According to the Australian national breathlessness survey (NBS), the prevalence of clinically important breathlessness (defined by mMRC dyspnoea grade ≥2, hereafter termed “breathlessness”) in adults was 9.5%, while over half (50.8%) of those with breathlessness did not report a current diagnosis of respiratory or cardiac disease. 1 Since diagnoses known to cause breathlessness often co-exist in real-world clinical practice, recognising the multiple causes of breathlessness is important for future symptom and disease management.
The Australian national breathlessness survey was a self-reported web-based survey that was not able to test for the association of breathlessness with objective clinical test results. The authors pointed out this limitation and identified gaps in knowledge about alternative causes of breathlessness in Australia. 1 The Burden of Obstructive Lung Disease (BOLD) Australia study collected information about breathlessness using the mMRC and also performed spirometry. BOLD Australia provided an opportunity to more thoroughly understand the risk factors for breathlessness and the relationship between other clinical characteristics and breathlessness.
The aim of our study was to identify the clinical characteristics independently associated with breathlessness and to investigate the relationship between lung function and breathlessness in Australian adults aged 40 years and over.
Methods
Study population
BOLD Australia was a cross-sectional study conducted between 2006 and 2012 of individuals aged ≥40 years from six study sites across Australia, including Sydney, rural New South Wales (15 towns randomly selected around the population centres of Orange and Wagga Wagga), Melbourne, Tasmania (Hobart and Launceston), Busselton, and Broome in Western Australia. In Western Australia, participants were recruited from a household census data in Broome and local Aboriginal communities within the Kimberley region or were randomly recruited from the Busselton Health Study. The study design has been described elsewhere.8,9 As part of the international BOLD study, some results from the Sydney BOLD site have previously been published, together with data from centres in 14 other countries. 3 However, for this report, Australian data included both Sydney and five other Australian sites. Participants who were not contactable, institutionalised, or aged younger than 40 years were excluded; more detailed information on the sample selection was published previously.8,9 Exclusion criteria for the present analysis were inability to walk due to conditions other than breathlessness and missing data for mMRC.
Assessments and definitions
The survey instrument used was the BOLD core questionnaire which covered demographics, breathlessness, other respiratory symptoms, self-reported diagnosed diseases, medication use, and healthcare utilisation.8,10 The level of activity limitation due to breathlessness was measured using the mMRC dyspnoea scale, which comprises five grades. 11 As in previous studies, clinically important breathlessness (hereafter referred to as breathlessness) was defined as mMRC dyspnoea scale grade ≥2. 1 The BOLD questionnaire did not specifically collect information about anxiety or depression, participants only answered one mood-related question (‘Have you felt downhearted and depressed during the past 4 weeks). Participants who reported that they felt downhearted and depressed all of the time, most of the time, some of the time or a little of the time were defined as having any downhearted and depressed feelings during the past 4 weeks.
Spirometry was performed at the same visit as survey data were collected, and was measured using the EasyOne spirometer (ndd Medizintechnik, Zürich, Switzerland), before and 15 min after the administration of salbutamol 200 µg through metered dose inhaler and spacer. 10 Participants were told to withhold using any bronchodilator (BD) inhalers for 6–12 h before the test. 8 All spirograms were reviewed and quality graded by a senior respiratory scientist. 8 The highest recorded FEV1 and forced vital capacity (FVC) from acceptable trials 12 were collected. Persistent airflow limitation was identified from post-BD FEV1/FVC using two criteria: less than 0.70, as recommended by the Global Initiative for Obstructive Lung Disease (GOLD), 13 and less than the lower limit of normal (LLN) as recommended by the European Respiratory Society. 14 The Global Lung Initiative (GLI-2012) tool provided by the European Respiratory Society was used to calculate the LLN. 14 Participants with an increase in FEV1 (or FVC) of ≥200 mL and ≥12% from baseline were defined as having positive BD responses. 15
Minimal data were collected from those who did not complete the full protocol and included six key questions about smoking, respiratory disorders and additional comorbidities (Supplementary information S1).
Data analysis
Data were analysed using SAS version 9.4 (SAS Institute Inc., Cary, NC). Participant characteristics were tabulated by the presence of breathlessness (mMRC ≥2) and reported using counts (proportions) for categorical variables and means ± standard deviations (SD) for continuous variables. The associations between breathlessness and clinical characteristics were evaluated using the Chi-squared test for categorical variables, and the analysis of variance for continuous variables. Logistic regression models were used to estimate crude and adjusted odds ratios (ORs) with 95% confidence intervals (CIs).
The selection of potential confounders for adjustment in the analysis was based on the causal inference approach. The Directed Acyclic Graphs (DAGs), included in the supplement (Figures S1–3 in the Supplementary Information), identified age, sex, BMI, smoking and socioeconomic status (using Socio-Economic Indexes for Areas [SEIFA]) 16 as potential risk factors. They could open a back-door pathway association between exposure (characteristics) and outcome (breathlessness) and therefore should be adjusted for in the analyses. 17
To look for independent associations between the spirometry results and breathlessness, we added a second multivariate logistic regression model. Persistent airflow limitation (using FEV1/FVC <0.70 and FEV1/FVC < LLN), abnormal FVC (below 80% predicted), abnormal FEV1 (below 80% predicted) and BD responsiveness (an increase in FEV1 or FVC of ≥200 mL and ≥12% from baseline) were chosen, as they were deemed to be the most specific and most objective measures. Each factor was added into a separate model together with the background variables, age, sex, smoking and socioeconomic status. Other factors associated with breathlessness in univariate analysis, including being Indigenous Australians, obesity, occupational dust exposure, respiratory symptoms (cough on most days, phlegm on most days and wheeze), respiratory diseases and heart diseases, were also included in these models.
Results
Of the 10,760 eligible participants included in the BOLD Australia study,
8
3522 completed the BOLD questionnaires and performed acceptable spirometry tests. Of these participants, 201 were excluded as they did not complete the mMRC questionnaire and/or were not able to walk due to conditions other than breathlessness, leaving a sample of 3321. Of the 3321 participants, 252 participants (7.6%) reported breathlessness. Independent associations between four key spirometry endpoints and clinically important breathlessness. Adjusted for age, sex, smoking, socioeconomic status, being Indigenous Australians, obesity, occupational dust exposure, cough on most days, phlegm on most days, wheeze, respiratory diseases and heart diseases; Obesity = body mass index (BMI) > 30 kg/m2; BMI <30 is the reference value. Abbreviations: FEV1, forced expiratory volume for 1s; FVC: forced vital capacity; LLN, lower limit of normal; GLI: Global Lung Initiative; BD: bronchodilator.
Demographic characteristics of the BOLD Australia sample, by clinically important breathlessness.
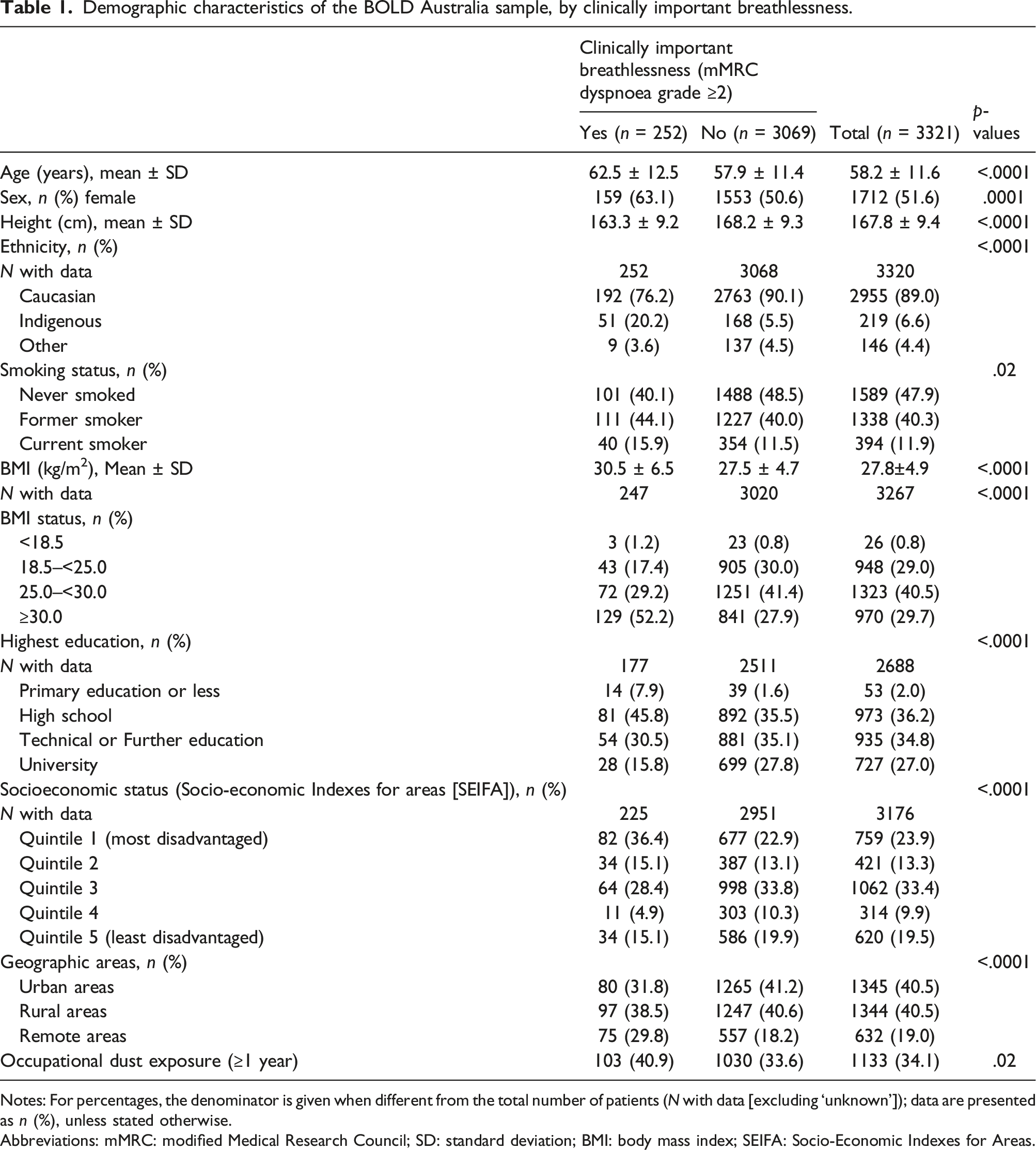
Notes: For percentages, the denominator is given when different from the total number of patients (N with data [excluding ‘unknown’]); data are presented as n (%), unless stated otherwise.
Abbreviations: mMRC: modified Medical Research Council; SD: standard deviation; BMI: body mass index; SEIFA: Socio-Economic Indexes for Areas.
Associations between clinical characteristics and clinically important breathlessness.

Notes: Data are presented as n (%), unless stated otherwise;
Abbreviations: mMRC: modified Medical Research Council; OR: odds ratio; CI: confidence intervals; SOB: shortness of breath; COPD: chronic obstructive pulmonary disease.
aAdjusted for age, sex, BMI status, smoking status and socioeconomic status.
bDo not include all observations due to missing.
In addition, a higher proportion of participants with breathlessness reported other chronic diseases including hypertension, diabetes, and stroke than participants without breathlessness (Table 2). Both any medication and respiratory medication use were reported more frequently by participants with breathlessness (Table 2). Many (61%) of participants with breathlessness reported feeling downhearted and depressed during the past 4 weeks, which was significantly higher than those without breathlessness.
Associations between lung function and clinically important breathlessness.
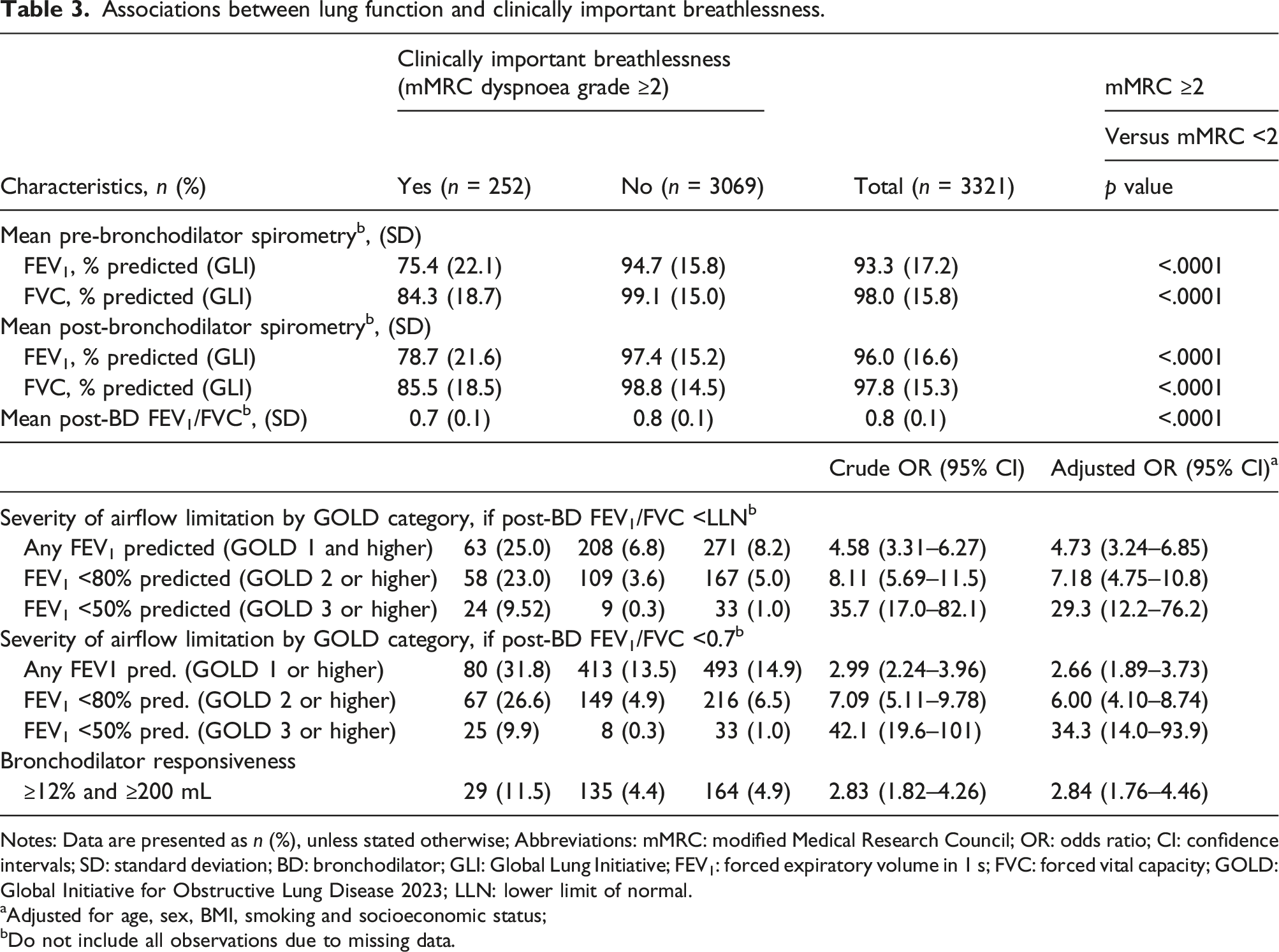
Notes: Data are presented as n (%), unless stated otherwise; Abbreviations: mMRC: modified Medical Research Council; OR: odds ratio; CI: confidence intervals; SD: standard deviation; BD: bronchodilator; GLI: Global Lung Initiative; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; GOLD: Global Initiative for Obstructive Lung Disease 2023; LLN: lower limit of normal.
aAdjusted for age, sex, BMI, smoking and socioeconomic status;
bDo not include all observations due to missing data.
The final model is shown in Figure 1. After adjusting for background variables (age, sex and socioeconomic status) and the other risk factors known to be associated with breathlessness, three spirometric parameters were independently associated with breathlessness. However, BD responsiveness was not significantly associated with breathlessness after taking into account respiratory symptoms and diagnoses. Other factors, including being Indigenous Australians, obesity, respiratory conditions, heart diseases, phlegm and wheeze (adjusted ORs = 3.46, 2.60, 2.96, 2.45, 1.82 and 2.00, respectively) were also independently associated with breathlessness (Table S1 in the Supporting Information). However, occupational dust exposure and cough were not associated with breathlessness after adjustment (Table S1).
Discussion
The principal findings of this population-based study are that: (1) the main risk factors among Australian adults aged ≥40 years reporting clinically important breathlessness, hereafter referred to as breathlessness, were obesity, chronic respiratory diseases, heart diseases and being Indigenous Australians; (2) participants with respiratory symptoms including phlegm and wheeze were more likely to report breathlessness; (3) Impaired spirometry results including reduced FVC, reduced FEV1 and airflow limitation was independently associated with reported breathlessness.
We found that 7.6% of the adults aged ≥40 had breathlessness. This is similar to the same age-group estimate from the Australian National Breathlessness Survey (NBS) (8.5%), 1 but higher than the 2.4% reported by another Australian survey in adult (≥18 years) members of an airline frequent flyer survey panel. 18 The NBS reported a similar proportion of breathlessness in both females and males aged ≥18 years 1 However our study found that breathlessness was more common in women, which was consistent with previous studies that included mild breathlessness (mMRC ≥1).3,19,20 The prevalence of current smokers and obesity among participants with breathlessness was higher than those without breathlessness, similar to the NBS. 1 Although occupational exposure was higher among participants with breathlessness than those without breathlessness, we found that occupational exposure was not independently associated with an increased prevalence of breathlessness, after adjusting for other factors.
In the present study, breathlessness was more common among participants with other respiratory symptoms, similar to a previous study. 7 In contrast to the previous finding, we found that cough was not independently associated with an increased prevalence of breathlessness, after adjusting for other factors. The NBS reported that breathlessness was more common in people with current respiratory or heart diagnoses than those without, 1 in line with our findings. We also found that diagnoses associated with breathlessness included hypertension, diabetes and stroke, consistent with a previous study. 3 Previous studies reported that breathlessness was more common in anxiety or depression,1,19 our findings also showed that participants with breathlessness were more likely to feel downhearted and depressed.
We observed participants with breathlessness had worse lung function than those without. Similar to our findings, a previous study reported a higher prevalence of breathlessness in people with lower absolute FEV1 or FVC. 21 Breathlessness was more common in participants with airflow limitation, and the odds of having breathlessness increased with the severity of airflow limitation, as observed previously. 22 However, 75% of participants with airflow limitation (defined by LLN) did not report breathlessness.
We observed high odds of clinically important breathlessness when post-BD FVC or FEV1 percent predicted was below 80%, or FEV1/FVC less than LLN, similar to the findings in the larger BOLD study. 3 However, our study did not confirm findings from a previous study that bronchodilator response for FVC was associated with breathlessness. 23 Efforts to achieve optimal lung function are likely to improve breathlessness and there are a range of medicines available to achieve this outcome. However, we know that breathlessness often persists despite optimal management of chronic respiratory disease. What is needed is earlier diagnosis and management of lung disease and also investigation of alternative non-pulmonary conditions that contribute to breathlessness.
We have previously reported from BOLD Australia that the prevalence of low FVC (<80% predicted) was substantially higher in Indigenous compared with non- Indigenous participants. 9 In the present multivariable analysis, we found that being Indigenous independently increased the odds of having breathlessness after adjusting for FVC and other factors. To our knowledge, this is the first study to report the independent association between Indigenous Australians and breathlessness. This finding could be further evidence of the health gap between Indigenous and non- Indigenous Australians. In Australia, interventions to improve pulmonary rehabilitation services in rural and remote areas are scarce and the sustainability of these interventions and their implementation in practice are not ensured. 24 It suggests that there may be a need for better access to pulmonary rehabilitation services in remote communities with Indigenous Australians.
A major strength of our study was that lung function was measured, enabling us to examine the relationship between spirometry results and breathlessness, which was not able to be examined in the NBS. 1 In addition, we used both fixed cut-off and LLN to define airflow limitation, allowing comparison with studies using either criterion. Other strengths included that data were from a large nationwide population sample 8 and the use of standardised measurements from the BOLD international protocol allowing comparisons between countries.8,10
The main limitations of the study included the cross-sectional design, which did not allow for the assessment of causality or long-term outcomes. The low overall response rate potentially introduced a degree of selection bias, with those included in the analysis being slightly younger and more likely to self-report a diagnosis of COPD compared with those who provided only minimal data. 8 Despite the large sample size, the number of participants at each mMRC grade was small, therefore we had limited ability to examine breathlessness at the more severe end of the mMRC scale.
Participants were not a truly random sample of the Australian population, since the six study sites were not randomly chosen but included geographic diversity. Broome and The Kimberley is a centre with a significant Aboriginal and Torres Strait Islander population and differs from the other Australian sites with a higher proportion of Indigenous Australians. Although post-hoc weights were used in previous work to adjust prevalence estimates to better reflect the Australian population, sample prevalence estimates were used in this analysis. 8 The single measure of spirometry was also a limitation as spirometry results can vary between tests and days so definitions that rely on thresholds can vary over different days. 25 Another limitation was that respiratory diagnoses were self-reported and may have been influenced by recall bias, however, these diagnoses were not the main focus of these analyses.
Our findings have significant implications for health service development in Australia, particularly in Indigenous communities. These findings indicate the importance of multiple risk factors independently associated with breathlessness including obesity, chronic respiratory diseases, heart diseases, being an Indigenous Australian, and impaired spirometry. Multi-disciplinary and multi-cause assessment can help to make more precise treatment decisions to guide clinical practice. Our findings will also continue to highlight breathlessness in Indigenous Australians, where better access to pulmonary rehabilitation services in remote communities with Indigenous Australians will help to improve symptoms such as breathlessness and chronic lung disease.
Conclusion
This study reports the main underlying risk factors for breathlessness and clinical characteristics of Australian adults over 40 years. The main risk factors among Australian adults reporting breathlessness, after adjustment for known risk factors for COPD (age, sex, smoking status and socioeconomic status), were obesity, chronic respiratory diseases, heart diseases, and being Indigenous Australians. Furthermore, we found a markedly higher prevalence of breathlessness amongst adults who had FVC or FEV1 less than 80% predicted, or FEV1/FVC less than LLN. We also found that Indigenous Australians were more likely to have breathlessness. Indigenous Australians may benefit from targeted interventions in the future. These findings build on earlier breathlessness research and demonstrate the relationship of spirometry measures after adjustment for known risk factors. They reiterate the importance of treatments to optimise lung function to minimise the negative consequences of breathlessness.
There is a need for further research linking risk factors, lung function, symptoms, and health outcomes such as quality of life and mortality, which could be valuable in reducing the national burden of breathlessness. Earlier diagnoses and treatment of diseases that contribute to breathlessness are needed to reduce the burden of breathlessness, particularly for Indigenous Australians.
Supplemental Material
Supplemental Material - Risk factors and clinical characteristics of breathlessness in Australian adults: Data from the BOLD Australia study
Supplemental Material for Risk factors and clinical characteristics of breathlessness in Australian adults: Data from the BOLD Australia study by Yijun Zhou, Maria R Ampon, Michael J Abramson, Alan L James, Graeme P Maguire, Richard Wood-Baker, David P Johns, Guy B Marks, Helen K Reddel and Brett G Toelle in Chronic Respiratory Disease.
Footnotes
Declaration of conflicting interests
MJA holds investigator initiated grants from Pfizer, Boehringer-Ingelheim, Sanofi and GSK. He has conducted an unrelated consultancy for Sanofi. He has also received a speaker’s fee from GSK. RWB reports cohort grants from the National Health & Medical Research Council. GBM has received funding for advisory boards with AstraZeneca. HKR holds investigator initiated grants from AstraZeneca, GlaxoSmithKline, Novartis, Perpetual Philanthropy. She has received consulting fees from AstraZeneca and GlaxoSmithKline and honoraria for advisory boards and independent medical education from AstraZeneca, GlaxoSmithKline, TEVA, Boehringer-Ingelheim, Sanofi, Getz and Chiesi. HKR holds non-funded leadership roles in the Global Initiative for Asthma (GINA) and National Asthma Council (NAC). All other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the BOLD study in Australia was funded by the National Health & Medical Research Council, Project Grant 457385. The BOLD study in Sydney was funded by grants from Air Liquide P/L, AstraZeneca P/L, Boehringer Ingelheim P/L, GlaxoSmithKline Australia P/L and Pfizer Australia P/L.
Ethical statement
Operations centre
Mrs Tessa E. Bird and Dr Wei Xuan (Woolcock Institute of Medical Research). Sydney: Professor Christine R. Jenkins, Mrs Tessa E. Bird, Dr Kate Hardaker and Dr Paola Espinel (Woolcock Institute of Medical Research). Busselton: the late Professor A. W. (Bill) Musk, Dr Michael L. Hunter, Ms Elspeth Inglis and Ms Peta Grayson (University of Western Australia). Kimberley: Professor David N. Atkinson, Mr Dave Reeve, Dr Nathania Cooksley, Dr Matthew Yap, Ms Mary Lane, Dr Wendy Cavilla and Ms Sally Young (University of Western Australia). Melbourne: Ms Angela Lewis, Ms Joan Raven, Ms Joan Green and Ms Marsha Ivey (Monash University). Tasmania: Professor E. Haydn Walters, Mrs Carol Phillips and Ms Loren Taylor (University of Tasmania). NSW Rural: Dr Phillipa J. Southwell, Dr Bruce J. Graham, Dr Brian Spurrell, Mrs Robyn Paton, Ms Melanie Heine, Ms Cassandra Eccleston and Dr Julie Cooke (Charles Sturt University).
Data Availability Statement
The data that support the findings of this study are available at reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
