Abstract
Background
The rationale for additional treatment with short-acting bronchodilators combined with long-acting bronchodilators for patients with chronic obstructive pulmonary disease (COPD) is not adequately studied.
Methods
We conducted a systematic review and meta-analysis to evaluate the efficacy and safety of a short-acting muscarinic antagonist (SAMA) therapy combined with a long-acting beta-2 agonist (LABA) in patients with stable COPD. Pulmonary function, dyspnea, health-related quality of life, exercise tolerance, physical activity, exacerbations of COPD, and adverse events during regular use were set as outcomes of interest.
Results
We included five controlled trials including two sets of publicly available online data without article publications for the meta-analysis. Additional use of SAMA plus LABA showed a significant improvement in the peak response in FEV1 (mean difference (MD) 98.70 mL, p < .00001), transitional dyspnea index score (MD .85, p = .02), and St George’s Respiratory Questionnaire score (MD -2.00, p = .008) compared to LABA treatment. There was no significant difference in the risk of exacerbation of COPD (p = .20) and only a slight trend of increased severe adverse events (OR: 2.16, p = .08) and cardiovascular events (OR: 2.38, p = .06).
Conclusion
Additional treatment with SAMA combined with LABA could be a feasible choice due to its efficacy and safety.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a leading cause of death worldwide despite its preventable and treatable nature. Persistent airflow limitations and dynamic hyperinflation due to small airway disease and emphysema are hallmarks of physiological changes in COPD and are important contributors to the development of respiratory symptoms such as dyspnea. 1 Physiological and symptomatic deterioration can contribute to exercise intolerance, physical inactivity, and exacerbations of COPD and may worsen the patient’s prognosis.2–4
Inhaled bronchodilators play central roles in the pharmacological treatment of stable COPD, aiming to improve symptoms, exercise intolerance, physical inactivity, and health-related quality of life (HRQOL) and reduce the frequency of exacerbations. In recent guidelines, long-acting bronchodilators (LABDs) are preferred for patients with persistent symptoms who need regular treatment.1,5 The use of short-acting bronchodilators (SABDs) can be considered an initial treatment choice for symptomatic patients to relieve breathlessness and exercise limitation. 6 Because of their rapid onset of action, SABDs could also be a choice for on-demand treatment of symptoms, regardless of the disease severity or the use of LABDs.1,5,7
Meanwhile, the rationale for additional treatment with SABDs is not adequately studied. There is not enough evidence for the precision use of short-acting bronchodilators as well. When considering additional treatment with SABDs on LABDs, combining bronchodilators of different classes such as muscarinic antagonists and beta2-agonist would be preferred in terms of efficacy and safety.8,9 Thus, combining short-acting beta2-agonist (SABA) with a long-acting muscarinic antagonist (LAMA) and a short-acting muscarinic antagonist (SAMA) with long-acting beta2-agonists (LABA) would be standard options. Although recent large-scale clinical trials have mainly focused on LABDs with/without ICSs, short-acting bronchodilators should also be refocused toward their more effective use. In the initial concept, we tried to evaluate the effectiveness and the risk of additional SABD in every combination of SABD(s) and LABD(s). However, in our preliminary literature search, there were not adequate reports/studies for systematic review and meta-analysis to discuss the proper use of the additional SABDs, and only additional treatment with ipratropium on salmeterol was evaluated in a previous meta-analysis. 10
Additional SAMA on LABA could be a choice of treatment in a certain situation such as the on-demand use in COPD patients with exertional dyspnea. Thus, in the present systematic review and meta-analysis, we evaluated the efficacy and safety of additional therapy of SAMA in addition to LABA treatment compared to LABA treatment alone in patients with stable COPD in terms of pulmonary function, dyspnea, HRQOL, physical activity, exercise capacity, and adverse effects.
Material and methods
Eligibility criteria and search
In the systematic review and meta-analysis, we followed the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 11
We screened interventional studies for eligibility in this systemic review, including clinical trials and observational studies, examining the effects or adverse events of additional treatment with SAMA combined with regular LABA treatment compared to LABA treatment among adult patients with stable COPD. No limitations of gender, ethnicity, or treatment duration were set.
We selected the outcomes of interest as (1) pulmonary function (peak response of the forced expiratory volume in 1 second (FEV1) from baseline by a single inhalation); (2) dyspnea (transitional dyspnea index (TDI) score); (3) HRQOL (change in St George’s Respiratory Questionnaire (SGRQ) score from baseline); (4) exercise tolerance (change in the 6-min walk distance (6MWD)); (5) physical activity (change in minutes of at least moderate activity defined as any physical activity >3 metabolic equivalents); 12 (6) exacerbations of COPD (the number of patients who experienced exacerbations during the study period); and 7–9) adverse events (serious adverse events, cardiovascular adverse events, and urinary difficulty). The outcomes were selected from the point of view of benefit and harm and also according to their clinical importance. The outcomes other than FEV1 were evaluated in the studies with a duration of 8 weeks or longer. Serious adverse events were defined as any adverse event that results in outcomes including death, a life-threatening adverse event, inpatient hospitalization or prolongation of existing hospitalization, a disability/incapacity, or a congenital anomaly in the offspring of a subject who received the drug or important medical events judged by investigators. We included all adverse events categorized as cardiovascular events and collected an adverse event related to the urinary frequency or difficulty.
We searched for eligible studies in the PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, and Ichushi (Database from Japan Medical Abstracts Society (URL; https://login.jamas.or.jp/[Japanese]) databases in July 2020, and updated the search in January 2023. using the search terms shown in the online supplement (Supplementary Table S1). We performed a literature search from the inception of the database to the present date (January 2023). We conducted manual searches of reference lists to identify additional reports. Reports published in English or Japanese were included in the analyses. We included studies with detailed data which were publicly available on the website even if the data was not published in an article form. This type of data was also included in a previous systematic review and meta-analysis. 10
Data collection process and risk of bias assessment
Two authors (K.T., Y.F.) screened the titles and abstracts of all studies identified by the search strategy to check their eligibility. Next, full-text assessments were performed to identify the studies for inclusion, and the data were extracted from the published articles and the data available on the website. The data extraction step was performed by two authors (K.T. and Y.F.). Two authors (K.T., Y.F.) evaluated the risk of bias according to the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions 6.1: 13 selection bias (random sequence generation and allocation concealment), performance bias (blinding of participants and personnel), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), reporting bias (selective reporting) and other bias, including a declaration of conflict of interest about the funding source. These steps were performed independently, and the results were reviewed by the six authors (K.T., S.S., Y.F., Y.Y., T.H., and S.M.) and the discrepancies were resolved by consensus in the meeting. We repeated this process for each data collection and assessment step. Finally, after the completion of these steps, all authors reviewed and approved the results of the screening of studies and assessment of the risk of bias.
Synthesis of results
In the meta-analyses, the inverse variance method for continuous data was used to compute the mean difference (MD) with 95% confidence intervals (CI), and the Mantel-Haenszel method for dichotomous data was used to compute the odds ratio (OR) with 95% CI using Review Manager Software version 5.4.1. 14 A random-effects model was used in the analyses. The degree of consistency among studies was assessed using the I2 value. Publication bias was examined by visual evaluation of funnel plots.
Results
Study selection and characteristics
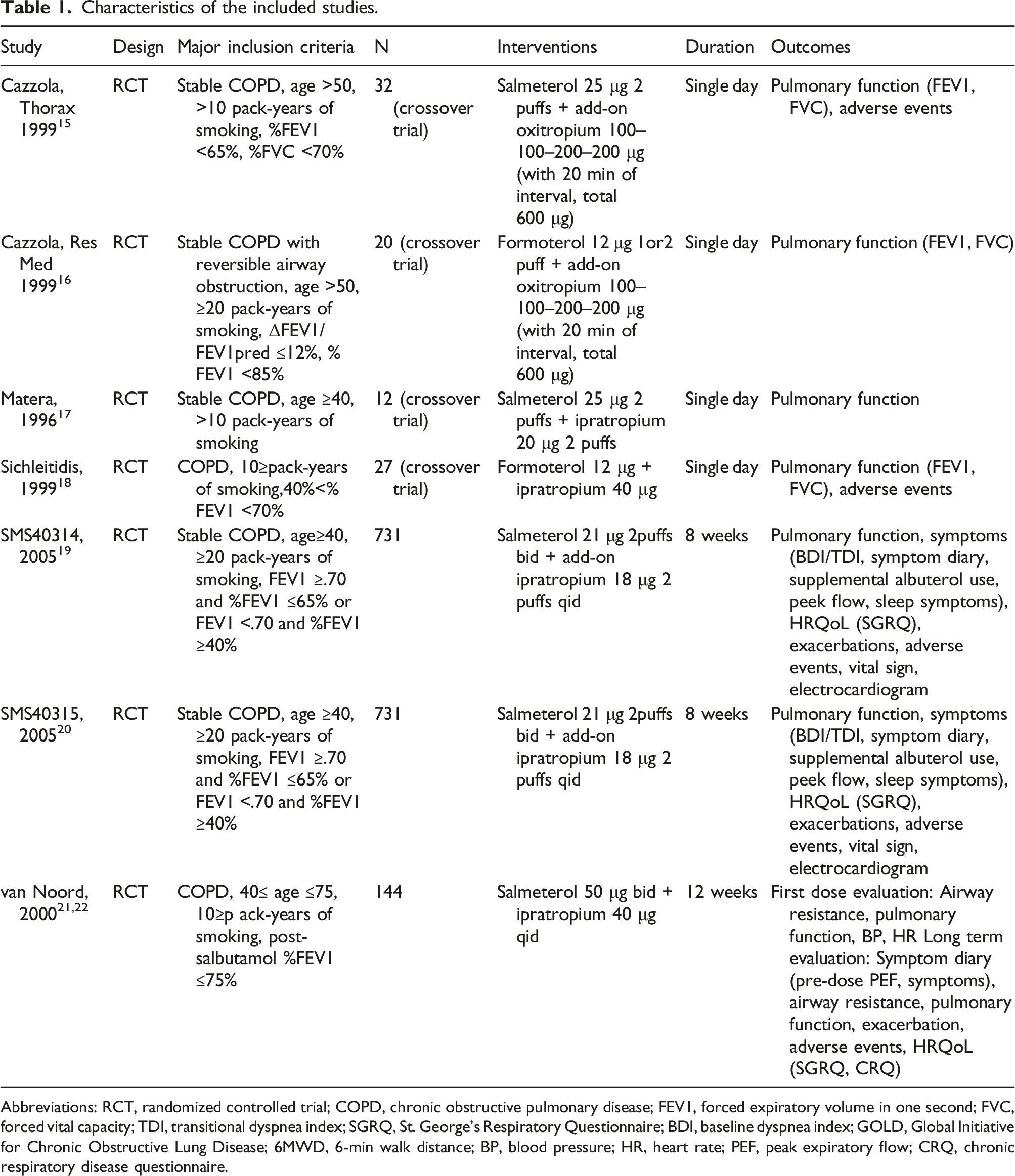
Using the search terms and manual searches, we identified and screened 1891 articles after excluding duplicates. Next, we assessed 55 full-text articles for eligibility and excluded 47 articles. Finally, we included seven randomized controlled trials (RCTs) (eight reports including two sets of unpublished data) in the qualitative analysis15–22 and included five RCTs in the meta-analysis (Figure 1). The characteristics of the included studies are summarized in Table 1. In the included studies with a longer study period of 8 weeks or longer, SAMA was inhaled regularly, four times a day. In the studies, the rescue use of SABA is allowed during the study period. There was no report aiming to evaluate the efficacy and safety of additional SAMA treatment with combination therapy of LAMA and LABA. All participants in the RCTs included in the systematic review were 40 years or older and had a smoking history of 10 pack-years or more. No patients with mild COPD were included in the RCTs. In the long-term studies, COPD patients with 40≤ Pre-bronchodilator %FEV1 ≤60%21,22 or FEV1 ≥.70 and 40≤% FEV1 ≤65%19,20 were included. Flow diagram of study selection. Characteristics of the included studies. Abbreviations: RCT, randomized controlled trial; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; TDI, transitional dyspnea index; SGRQ, St. George’s Respiratory Questionnaire; BDI, baseline dyspnea index; GOLD, Global Initiative for Chronic Obstructive Lung Disease; 6MWD, 6-min walk distance; BP, blood pressure; HR, heart rate; PEF, peak expiratory flow; CRQ, chronic respiratory disease questionnaire.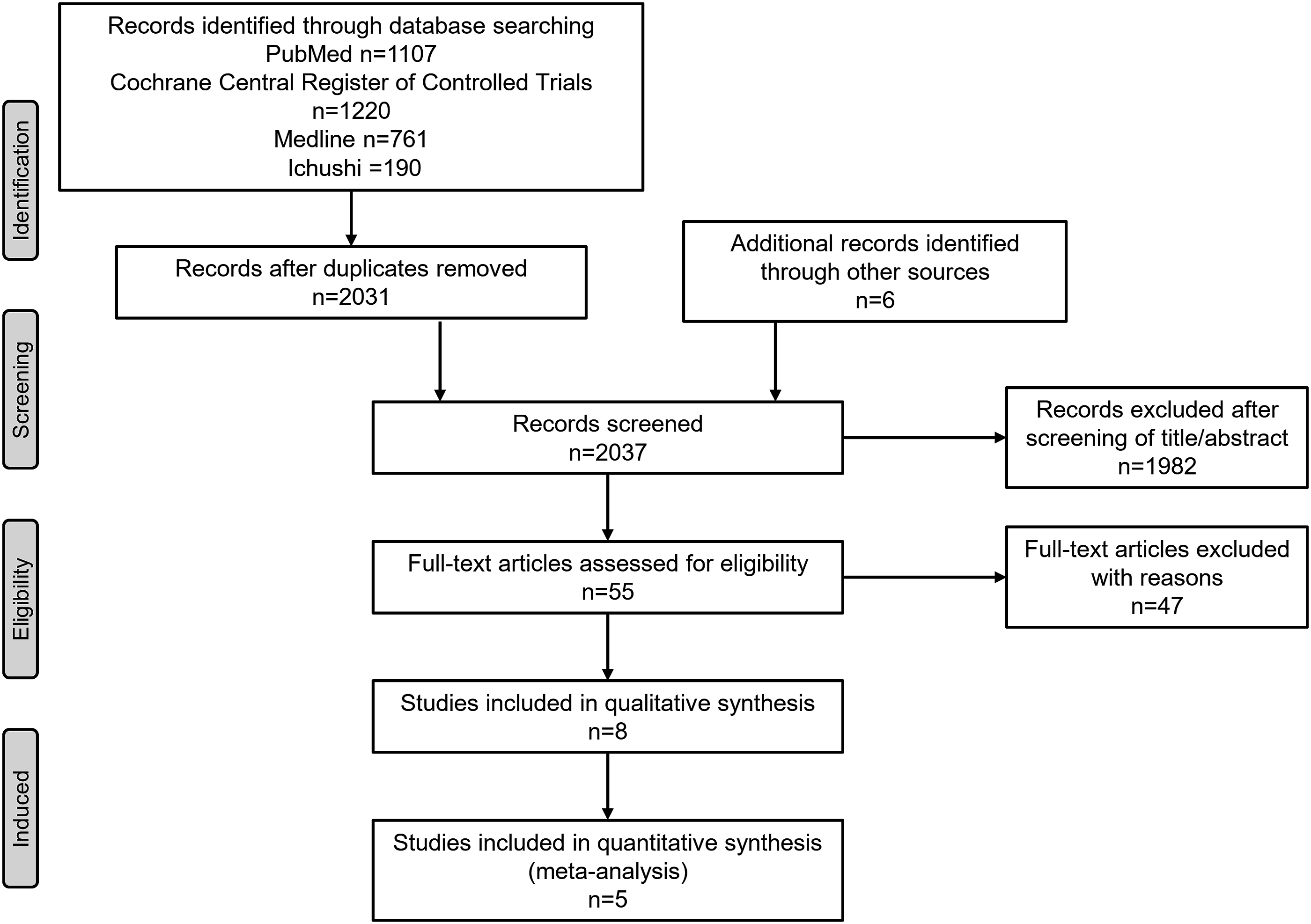
Risk of bias
The risk of bias in the included studies is provided in the supplemental figure (Supplementary Figure S1). Most RCTs included in the analysis had an unclear risk bias for allocation concealment (selection bias) and an insufficient declaration of the conflict of interest about the funding source. We judged that there was no study with a high risk of bias.
Outcome assessments
Pulmonary function: peak response in FEV1 from baseline by single inhalation
We included seven RCTs with an outcome related to FEV1.15–21 The peak response of FEV1 from baseline by a single inhalation was an outcome in three reports.18–20 We judged that there would be indirectness of the results because of the low number of included studies.
A meta-analysis was performed on these three studies (total 877 cases) (Figure 2(a)). Compared with placebo plus LABA treatment, additional treatment with SAMA plus LABA treatment demonstrated a significant improvement in FEV1 with MD 98.70 mL (95% CI: 63.13 to 134.26, p < .00001, I2 = 22%). It was equivalent to 100 mL of the minimal clinically important difference (MCID). The I2 statistic was 22%, showing mild inconsistency. It was judged that there was high certainty of evidence. An additional effect of short-acting muscarinic antagonists plus long-acting beta2 agonists on (a) pulmonary function (peak response in forced expiratory volume in 1 second (FEV1) from baseline by single inhalation), (b) dyspnea (transitional dyspnea index (TDI)), (c) health-related quality of life (change in the St. George Hospital Respiratory Questionnaire (SGRQ) score), (d) COPD exacerbations (the number of patients who experienced exacerbations during the study period), (e) serious adverse events, and (f) cardiovascular adverse events. Abbreviations: LABA, long-acting beta2-agonist; SAMA, short-acting muscarinic antagonist; CI, confidence interval.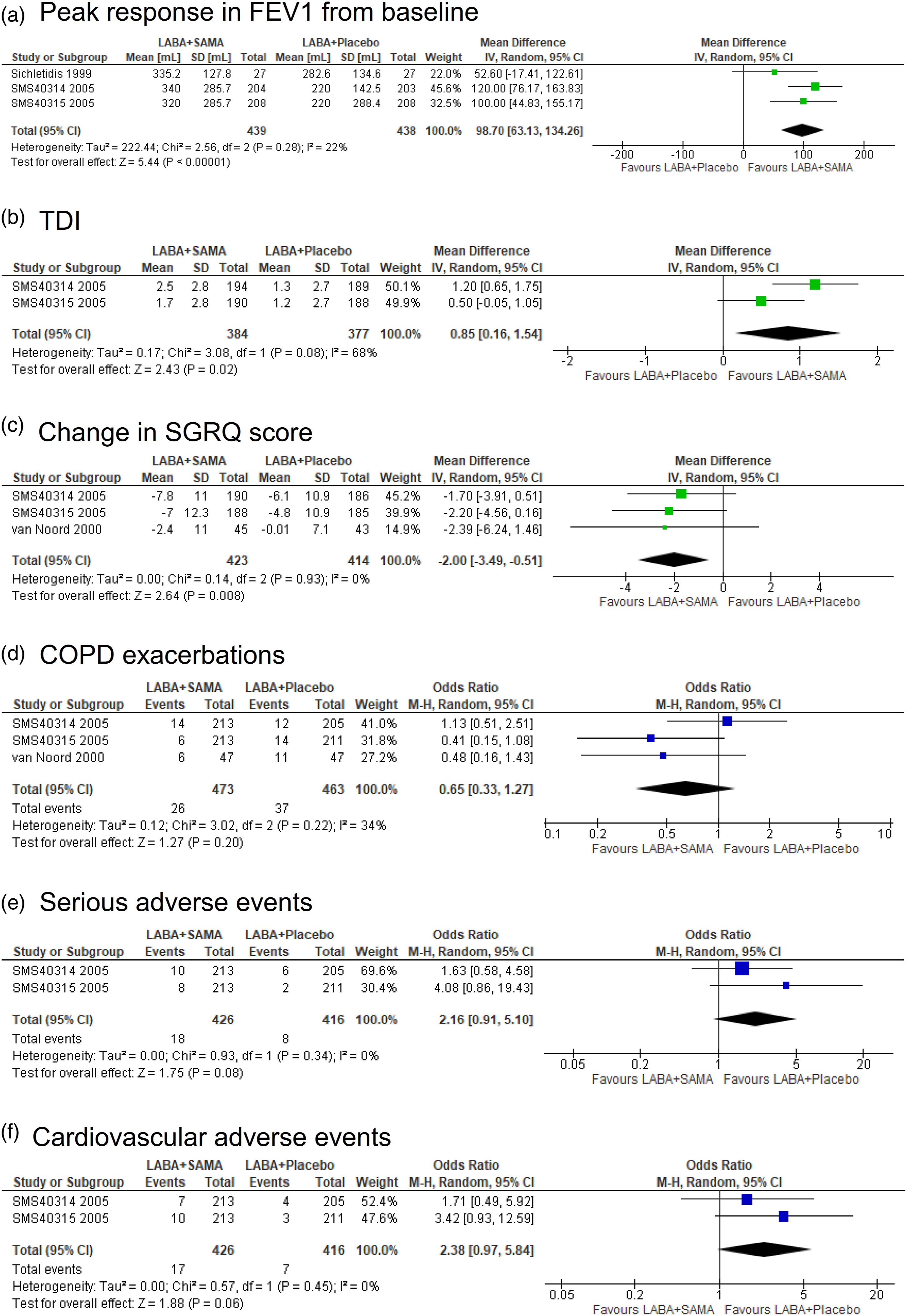
Dyspnea: TDI score
We included two RCTs,19,20 and a meta-analysis was performed on these studies (total 761 cases) (Figure 2(b)). Additional treatment with SAMA plus LABA treatment showed a significant improvement in the TDI score, with an MD of .85 (95% CI: .16 to 1.54, p = .02, I2 = 68%), compared to placebo plus LABA treatment, which did not reach the MCID (one point). The I2 statistic was 68%, showing moderate inconsistency. It was judged that there was a mild risk of bias and was moderate certainty of evidence.
HRQOL: change in the SGRQ score from baseline
We included three studies,19,20,22 and a meta-analysis was performed on these three studies (total 837 cases) (Figure 2(c)). Additional SAMA treatment plus LABA treatment demonstrated a significant improvement in SGRQ score with MD -2.00 (95% CI: −3.49 to −.51, p = .008, I2 = 0%) compared to placebo plus LABA treatment, which did not reach the MCID (four points). The I2 statistic was 0%, showing no inconsistency. It was judged that there was high certainty of evidence. Additional SAMA treatment plus LABA treatment significantly improved the activity domain of SGRQ score (MD: −2.33, 95% CI: −4.15 to −.50, p = .01, I2 = 0%) but only showed a trend of an increase in the impact domain (MD: −1.72, 95% CI: −3.49 to .06, p = .06, I2 = 0%) and the symptom domain (MD: −3.37, 95% CI: −7.42 to .67, p = .10, I2 = 65%).
Exercise tolerance: change in the 6MWD
There was no report evaluating this outcome.
Physical activity: change in minutes of at least moderate activity
There was no report evaluating this outcome.
Exacerbations of COPD
We included three RCTs19–21 and performed a meta-analysis (total 936 cases) (Figure 2(d)). There was no significant difference in the risk of exacerbation of COPD (odds ratio (OR): .65, 95% CI: .33–1.27, p = .20, I2 = 34%). The I2 statistic was 34%, which was judged to be moderately inconsistent. It was judged that there was moderate certainty of evidence according to the number of events and the confidence interval.
Adverse events during regular use: serious adverse events
We included three RCTs evaluating adverse events19–21 and performed a meta-analysis on the two RCTs in which serious adverse events were mentioned (total 842 cases) (Figure 2(e)).19,20 None of the serious adverse events during the double-blind treatment period in the two RCTs were considered to be related to the study medication by the reporting investigators. Compared to placebo plus LABA treatment, additional SAMA treatment plus LABA treatment showed a trend to increase severe adverse events (OR: 2.16, 95% CI: .91–5.10, p = .08, I2 = 0%). Although the I2 statistic was 0%, it was judged that there was mild inconsistency because of the small number of included studies and observed events. It was judged that there was moderate certainty of evidence.
Adverse events during regular use: cardiovascular adverse events
We included three RCTs evaluating adverse events19–21 and performed a meta-analysis on the two RCTs in which cardiovascular adverse events were mentioned (842 cases) (Figure 2(f)).19,20 The most common cardiovascular adverse event in the combination SAMA and LABA treatment was tachycardia, which was not frequently observed (4/426 cases, .94%). Compared to placebo plus LABA treatment, additional SAMA treatment plus LABA treatment showed a trend to increase cardiovascular adverse events (OR: 2.38, 95% CI: .97–5.84, p = .06, I2 = 0%). Although the I2 statistic was 0%, it was judged that there was mild inconsistency because of the small number of included studies and observed events. It was judged that there was moderate certainty of evidence.
Adverse events during regular use: urinary difficulty
Three RCTs evaluated adverse events.19–21 Adverse events in the urinary tract were mentioned in two RCTs,19,20 but we could not perform a meta-analysis because of the extremely low rates (1/842 cases). It was judged that there was low certainty of evidence.
Discussion
In the present systematic review and meta-analysis, a single dose of additional SAMA on a background of regular LABA treatment showed a significant improvement in pulmonary function (peak response in FEV1, MD 98.70 mL (95% CI: 63.13 to 134.26, p < .00001)) compared to LABA treatment alone, and the MD was equivalent to the MCID. Regular use of additional SAMA treatment plus LABA treatment significantly improved dyspnea (TDI score, MD .85 (95% CI:.16 to 1.54, p = .02)) and HRQOL (change in SGRQ score, MD −2.00 (95% CI: −3.49 to −.51, p = .008)), although the improvements did not achieve their MCID. There was no significant difference in the risk of exacerbation of COPD, and there was only a slight trend of an increase in severe adverse events or cardiovascular events. No meta-analysis could be performed for exercise tolerance, physical activity level, or the risk of urinary difficulty due to a lack of data.
In recent guidelines, combination therapy of LABA and LAMA was a mainstay of maintenance treatment in severe COPD patients, aiming to improve clinical outcomes such as pulmonary function, symptoms, and exercise tolerance.23–25 Short-acting bronchodilators are often added as rescue medication against a background of regular use of LABDs due to their rapid onset of action and further bronchodilation activity. Combination therapy of SABA and SAMA is reported to show a faster onset of action and greater peak bronchodilation compared to that of LAMA and LABA. 26 In the present meta-analysis, compared to placebo plus LABA treatment, additional therapy with SAMA plus LABA treatment significantly improved not only the peak response of FEV1 in a single inhalation but also TDI score and SGRQ score in regular use. The improvement did not reach their MCID but was equivalent to them for pulmonary function and dyspnea. And as reported in the previous meta-analysis, tiotropium shows higher trough FEV1 compared to ipratropium. 27 These findings would not recommend the regular use of SAMA on LABA instead of that of LAMA on LABA. However, the peak FEV1 on the first day of inhalation was reported to be similar between ipratropium and tiotropium. 28 When a rapid onset of action is expected, one treatment option would be to combine not LAMA but SAMA with regular LABA therapy, especially in patients with exertional dyspnea. This concept would support the application of short-acting bronchodilators for assistance in pulmonary rehabilitation and daily activities.29,30
We could not perform a meta-analysis on exercise tolerance under the combination therapy of LABA and SAMA. In the literature, it is shown that monotherapy of LABDs or SABDs improves exercise tolerance 31 and LAMA/LABA combination therapy improves exercise tolerance and physical activity compared to monotherapy. 32 In two RCTs, when SABA was added to LAMA treatment, home-based exercise training or daily activities showed significant improvement in the 6MWD33,34 and the additive effect of single inhalation of SABA not SAMA on LAMA on exercise capacity. 35 Additionally, we could not perform a meta-analysis on physical activity after combining SAMA with LABA treatment due to a lack of data. Two small studies, including one RCT, suggested promising effects of additional SABA therapy as an objective physical activity measure.29,34 In the present meta-analysis, the activity domain of SGRQ was significantly improved in patients with additional SAMA treatment. There is not adequate evidence to recommend the additional SABDs use on LABDs rather than LAMA/LABA for the improvement of physical activity and exercise tolerance. However, considering their rapid bronchodilation and symptom relief or the availability for on-demand/assisted use, SABDs use on LABDs could be a choice of treatment, especially in patients with marked exertional dyspnea. Future larger studies are warranted to evaluate the effect on exercise capacity and physical activity.
The additional SAMA on LABA showed no significant difference in the risk of exacerbation of COPD. Considering the frequency of COPD exacerbation or the longer trial periods of the recent clinical trial for LABDs,36,37 it would be difficult to expect a reduction of the exacerbation risk in 2 months. However, as the exacerbation risk is one of the important clinical outcomes in patients with COPD, we included the exacerbation risk in the outcome of the present analysis. Future studies would be required to evaluate the effectiveness of the reduction of the risk for exacerbation in longer periods.
Regarding adverse events, there were trends of increases in serious adverse events and cardiovascular events in combined SAMA with LABA treatment. However, none of the serious adverse events were judged unrelated to the study medication, and even the most frequent cardiovascular events, tachycardia, were seen in only a small proportion among SAMA + LABA combination treatment (.94%). In these studies, only one case of urinary difficulty was reported. It can be said that there is no remarkable risk for urinary difficulty with SAMA plus LABA treatment. Notably, one case–control study revealed that combination therapy of LAMA and SAMA significantly increased the risk of acute urinary difficulty compared to LAMA or SAMA monotherapy. 38 Therefore, some attention should be given to urinary problems during combination therapy with LAMA and SAMA.
There are some limitations in the generalizability of these findings because we could only perform a systematic review and meta-analysis of additional SAMA therapy plus LABA treatment due to a lack of data. First, a proper choice of additional short-acting bronchodilators based on the type of LABDs being used should be elucidated in terms of efficacy and safety. Combination therapy of muscarinic antagonists and beta2-agonists, rather than concomitant use of drugs in the same class, is considered to be the most effective. However, there is only one study that addressed the difference between additional treatment with SAMA plus tiotropium and that with SABA at the clinical dosage. 39 In that study, a single dosage of fenoterol provided greater additional bronchodilatation than that of ipratropium, and there was no significant difference in adverse events. Although these findings are consistent with the common understanding, there is not enough evidence regarding their long-term efficacy and safety. Second, the efficacy and safety of additional short-acting bronchodilator treatment plus combination therapy of LAMA and LABA should also be elucidated. Combination therapy with LAMA and LABA has an important position in recent COPD management, especially in patients with severe COPD, but it is not unusual for combination therapy to not achieve the expected symptom relief, and additional short-acting bronchodilators should be considered. Patients with severe COPD would be vulnerable because of their poor systemic condition, including multiple comorbidities. Special attention to adverse effects may be required in such patients. Finally, the validity of the on-demand use or the assisted use of short-acting bronchodilators needs further evaluation. In the present meta-analysis, except for pulmonary function, the efficacy and safety of the long-term regular use of additional SAMA treatment were evaluated. Some small studies have suggested that assistance with SABAs in addition to regular LABD treatment improves physical activity and HRQOL.29,30 SABA is routinely used for additional SABD therapy. However, SAMA also provides rapid bronchodilation or symptom relief. Since different classes of bronchodilators should be used in combination therapy, there would be more opportunities to utilize SAMA in clinical settings. Although the present analysis was not intended to compare additional SAMA and additional SABA or the effectiveness of on-demand reliever therapy with SAMA, SAMA instead of SABA could be a choice in certain situations and patients with exertional dyspnea. The effectiveness of on-demand use or the assisted use of short-acting bronchodilators due to their rapid onset of action should be confirmed in larger studies.
Conclusion
Additional therapy with SAMA plus background LABA treatment showed a significant improvement in pulmonary function, dyspnea symptoms, and HRQOL, but did not achieve their MCID. There was no significant difference in the risk of exacerbation of COPD. There was a minor trend of increased serious adverse events and cardiovascular events, but these adverse events may be tolerable. When a rapid onset of action is expected, one treatment option would be to combine SAMA with regular LABA therapy, especially in patients with exertional dyspnea.
Supplemental Material
Supplemental Material - The efficacy and safety of additional treatment with short-acting muscarinic antagonist combined with long-acting beta-2 agonist in stable patients with chronic obstructive pulmonary disease: A systematic review and meta-analysis
Supplemental Material for The efficacy and safety of additional treatment with short-acting muscarinic antagonist combined with long-acting beta-2 agonist in stable patients with chronic obstructive pulmonary disease: A systematic review and meta-analysis by Kazuya Tanimura, Susumu Sato, Yukio Fujita, Yoshifumi Yamamoto, Takashi Hajiro, Nobuyuki Horita, Tomotaka Kawayama, Shigeo Muro in Chronic Respiratory Disease
Footnotes
Author contributions
All authors were involved in the design of the systematic review and meta-analysis. K.T. collected, analyzed, and interpreted the data, and wrote the manuscript. S.S. analyzed and interpreted the data and edited the manuscript. Y.F. contributed to the collection, analysis, interpretation of the data. Y.Y. and T.H. contributed to the analysis and interpretation of the data. N.H. contributed to the analysis and interpretation of the data. T.K. contributed to the management of the project. S.M. analyzed and interpreted the data and assisted with the editing of the manuscript. S.M. takes responsibility for the integrity of the project as a whole, from its inception to the manuscript’s publication. All authors have approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.M. received honoraria from Boehringer Ingelheim Japan, AstraZeneca Japan, and Novartis Japan. T.K. received honoraria from AstraZeneca Japan, GlaxoSmithKline KK Japan, Novartis Pharmaceuticals Japan, Boehringer Ingelheim Japan, Sanofi Japan, and Kyorin Pharmaceutical Co. Ltd and research funding from Healios K.K. (Japan). S.S. received honoraria and research funding from Boehringer Ingelheim Japan.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the JSPS KAKENHI Grant Numbers JP20K08573 (S.M.) and JP22K20913 (K.T.). The present analysis was performed as a part of the project for the revision of the Japanese Respiratory Society Guidelines for the Management of Chronic Obstructive Pulmonary Disease. English editing fees are supported by the committee for the 6th edition of the Japanese Respiratory Society Guidelines for the Management of Chronic Obstructive Pulmonary Disease.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
