Abstract
Chronic obstructive pulmonary disease (COPD) is a serious global health burden. Comprehensive management of COPD includes both pharmacologic and non-pharmacologic interventions aimed at improving disease-related functional capacity, health-related quality of life, and survival. The primary medications used for treatment of COPD are inhaled bronchodilator drugs which are delivered directly to the patient’s airways through a number of different mechanisms. Arformoterol, the (R,R) enantiomer of racemic formoterol, was the first long-acting beta agonist approved by the U.S. Food and Drug Administration (FDA) for nebulized delivery. We discuss the pharmacology, clinical efficacy, and safety of arformoterol, and provide recommendations for its use during longitudinal management of patients with COPD.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a serious global health burden, and the fourth leading cause of death overall [WHO, 2011]. In the United States, COPD is the third leading cause of death [Miniño et al. 2011] and is estimated to affect 5.1% of the total population [Akinbami and Liu, 2011]. Comprehensive management of COPD includes both pharmacologic and nonpharmacologic interventions aimed at improving disease-related functional capacity, health-related quality of life, and survival. The primary medications used for treatment of COPD are inhaled bronchodilator drugs which are delivered directly to the patient’s airways through a number of different mechanisms. For those patients who have difficulty with the coordinated breathing technique required to use newer devices, the use of nebulized delivery systems is recommended to ensure adequate medication delivery to the lower airways [Dolovich et al. 2005]. Although long-acting controller medications are recommended for the treatment of moderate severity COPD, until recently the only bronchodilators available in nebulized form were those having a relatively short duration of action. Arformoterol was the first long-acting beta agonist (LABA) approved by the US Food and Drug Administration (FDA) for nebulized delivery. In addition, the pharmacotherapy options for patients with COPD have expanded significantly in the past decade, and clinicians are gaining an increasing armamentarium of agents to apply to specific patient situations. The purpose of this review is to discuss the characteristics of arformoterol, and where it might best be applied during longitudinal management of patients with COPD.
Pharmacology and administration
Arformoterol is the (R,R) enantiomer of racemic formoterol. It has full agonist activity at the beta-2 adrenergic receptor on airway smooth muscle cells. With an onset of action around 7 minutes (peak at 1–3 hours), arformoterol acts similarly to inhaled rescue medications such as albuterol, but maintains activity for 12 hours or more. Studies of formoterol enantiomers have found that the (R,R) stereoisomer accounts for the drug’s bronchodilator activity, whereas the (S,S) stereoisomer may inhibit bronchodilation and cause other deleterious effects not mediated by beta-2 receptor binding [Handley et al. 2002]. (R,R) formoterol has been developed as the first approved maintenance bronchodilator intentionally composed of a single stereoisomer. Theoretically, this enhances the potency of the drug while reducing harmful side effects.
Arformoterol is packaged as an inhalation solution in 15 µg/2 ml unit-dose vials. Administration is by nebulization every 12 hours. This delivery method results in similar plasma concentrations of (R,R) fomoterol as the FDA-approved 12 µg dry-powder inhaler (DPI) dose of racemic formoterol [Kharidia et al. 2008]. The choice of nebulizer can significantly affect the delivered dose of arformoterol in vitro [Bauer et al. 2009], but whether choice of nebulizer affects clinical endpoints (in vivo) is unknown.
Efficacy
In prelicensing trials of arformoterol, the drug produced consistent improvements in lung function over placebo [Baumgartner et al. 2007; Hanrahan et al. 2008b] (see Figure 1). After 12 weeks of treatment, trough forced expiratory volume in 1 second (FEV1), FEV1 AUC0-12h, and peak FEV1 in the arformoterol 15 µg BID group were increased by 14.6%, 8.4%, and 22%, respectively. All increases were statistically significant when compared with placebo, and higher doses of arformoterol provided only small incremental increases. The increase in peak FEV1 met the suggested minimal clinically important difference of about 112 ml (the amount of change resulting in patient-perceived improvement) [Cazzola et al. 2008; Donohue, 2005]. Subgroup analysis indicated that the greatest increase in lung function was seen in those patients with baseline severe or very severe obstruction. One published study tested the single-day response to arformoterol 30 µg administered once daily compared with 15 µg twice daily and found no difference in FEV1 responses [Panettieri et al. 2009]. Licensing dose-ranging trials, however, found numerically higher increases in FEV1 responses with BID dosing, and FDA approval was sought for the 15 µg BID dosing schedule [US Food and Drug Administration Center for Drug Evaluation and Research, 2006].
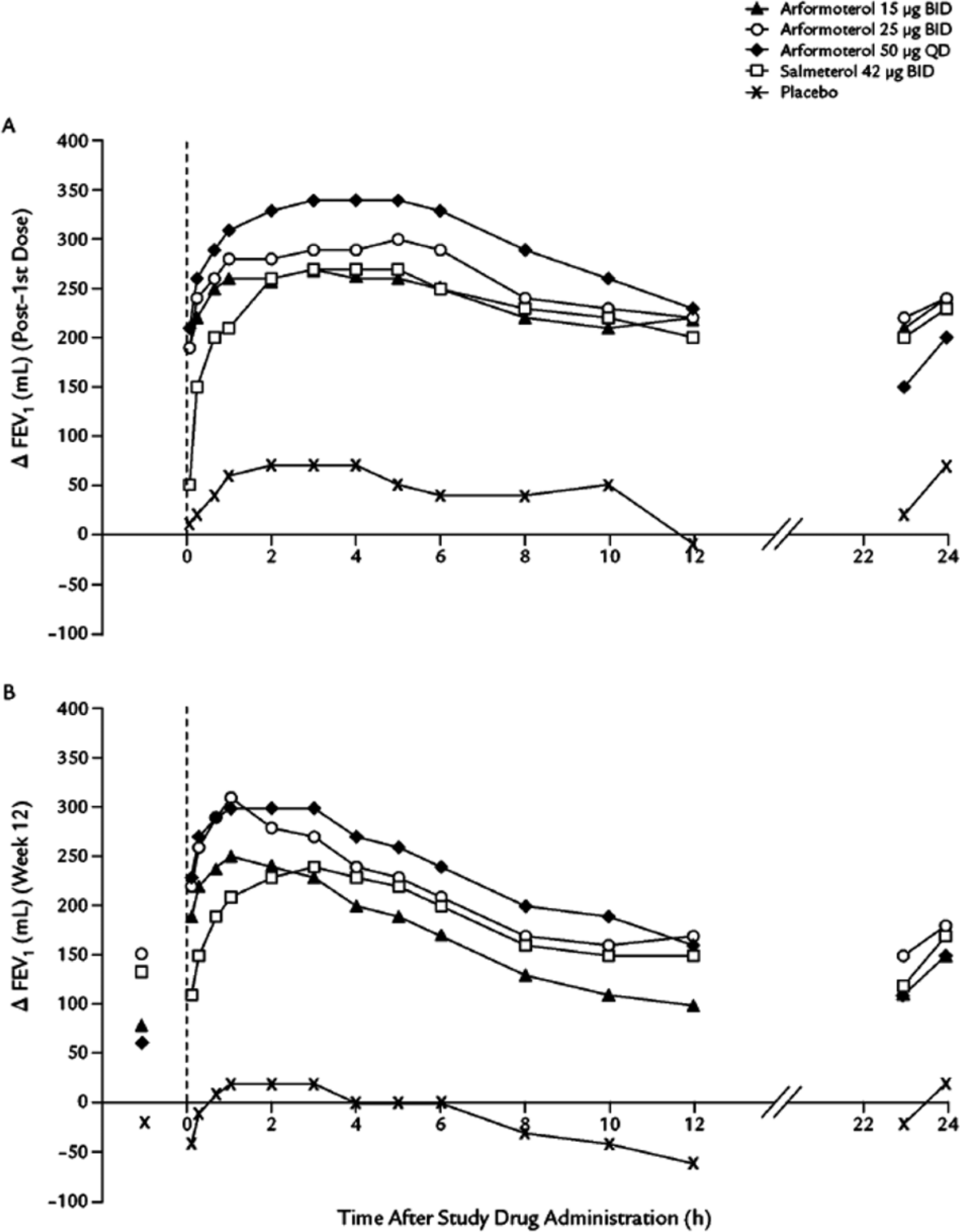
Changes From baseline in Forced expiratory volume in 1 second (FEV1) at weeks (A) 0 and (B) 12 in a 12-week trial in patients receiving arformoterol, salmeterol, or placebo for chronic obstructive pulmonary disease. Dashed line indicates time of study drug administration. Symbols prior to study drug administration at week 12 represent the mean change from baseline in visit predose FEV1 from baseline for each treatment group. (From Baumgartner et al. 2007, used by permission).
It is arguable that the clinical effects of arformoterol and its racemate should be the same, since the active molecule is the (R,R) form of the drug. Racemic formoterol is available in two formulations in the United States: a 12 µg DPI administered twice daily and a 20 µg/2 ml solution administered through nebulization twice daily. As noted previously, similar plasma levels of (R,R) formoterol are achieved with formoterol 12 µg DPI as with nebulized arformoterol 15 µg. Indeed, in a 6-month trial comparing formoterol DPI with nebulized arformoterol at 15 or 25 µg BID, improvements in lung function were similar between racemic formoterol and arformoterol 15 µg [Hanania et al. 2010]. To date, no studies have compared the nebulized form of arformoterol with nebulized racemic formoterol.
Salmeterol is another LABA approved for use as maintenance therapy in COPD. Statistically equivalent bronchodilation is seen between salmeterol and arformoterol, in both the pivotal 12-week clinical trials of arformoterol [Baumgartner et al. 2007; Hanrahan et al. 2008b], and a 12-month safety study of arformoterol 50 µg daily and salmeterol 42 µg BID [Donohue et al. 2008].
The use of combinations of long-acting bronchodilators may be considered for patients with persistent COPD symptoms or progressive airflow obstruction despite the use of a single maintenance therapy. Arformoterol has been studied in combination with tiotropium bromide, a once-daily long-acting muscarinic antagonist (LAMA) also approved for maintenance bronchodilator therapy in COPD [Tashkin et al. 2009]. In this 2-week study, each therapy alone produced similar levels of bronchodilation. Both spirometric measures and dyspnea index were further improved when the agents were combined (Figure 2).

Mean change in time-normalized FEV1AUC0-24 from study baseline at week 2 in a trial of arformoterol 15mcg, tiotropium 18mcg, and the combination. (Adapted from Tashkin et al. 2009).
Safety
Common to all LABA agents are potential side effects of stimulation at the beta-adrenergic receptor. These include hypokalemia, hyperglycemia, anxiety, nervousness, tremor, palpitations, and arrhythmias (particularly supraventricular tachycardias). In the prelicensing trials noted above [Baumgartner et al. 2007; Hanrahan et al. 2008b] there was a small increase in the incidence of nervousness, tremor, and insomnia as doses of arformoterol increased. There was a small numeric decrease in the number of COPD exacerbations with arformoterol treatment, and no difference in serious adverse events between placebo and arformoterol (Table 1). Effects on serum potassium and glucose concentrations were modest (greatest mean reduction in potassium of 0.19 mEq/l and greatest increase in glucose of 26 mg/dl). Specific data on cardiac arrhythmias during these studies was published separately [Hanrahan et al. 2008a]. The presence of arrhythmias was measured by 24-hour holter monitors at baseline, after the first study dose, and at weeks 6 and 12. Although there was a high rate of baseline atrial tachycardia (42%), treatment-emergent arrhythmias were not significantly increased by arformoterol treatment, and serious cardiovascular events did not differ between placebo and treatment groups. Fewer patients in the treatment groups used rescue albuterol inhalation. Additional studies of arformoterol safety are available since its approval. A single-day study in adults of arformoterol 15 µg BID compared with arformoterol 30 µg BID noted no serious adverse events [Panettieri et al. 2009]. A single-day study in pediatric subjects of consecutive doses of arformoterol 7.5 and 15 µg was performed over concern that arformoterol might be used in an off-label manner for asthma exacerbation. The medication was well tolerated, with no clinically important changes in heart rate, blood pressure, or serum glucose levels [Hinkle et al. 2011]. There was a substantial decline in serum potassium levels at 2 and 6 hours postdosing in the 0.5 mEq/l range.
Summary of safety events in two identically designed randomized trials comparing 12 weeks’ treatment with arformoterol 15mcg BID, arformoterol 25mcg BID, or arformoterol 50mcg QD with placebo. (Adapted from Hanrahan et al. 2008).

Recommendations for use
It is suspected that many patients taking inhaled corticosteroids (ICSs) do not need them [Barnes, 2010]. Recent FDA warnings regarding the safety of LABA agents in asthmatics may have inadvertently affected physician prescribing practices with regard to these agents in COPD. While there is no evidence of harm from treatment with LABA in COPD, there is evidence that the use of ICSs contributes to increased incidence of pneumonia [Drummond and Dasenbrook, 2008; Singh et al. 2009]. With this in mind, expanding the commercially available options for ICS-free long-acting bronchodilator therapy is appropriate.
Consistent with GOLD recommendations [Global Initiative for Chronic Obstructive Lung Disease (GOLD), 2011] and ACP/ACCP/ATS/ERS clinical practice guidelines [Qaseem et al. 2011] for the management of COPD, a long-acting maintenance bronchodilator should be used in many patients with moderate severity COPD or worse. ICSs need not be added unless the patient manifests frequent (two in the last year) exacerbations of their disease [Hurst et al. 2010]. When selecting a long-acting agent for a patient who has previously used only short-acting inhalers, both drug and device selection is important. Currently available data suggests that, at least for spirometric and dyspnea indices, the choice of LABA or LAMA is equivalent. There is data that tiotropium delays the time to first exacerbation of COPD when compared with salmeterol [Vogelmeier et al. 2011], but similar data in comparison to arformoterol (or racemic formoterol) is lacking.
Nebulized arformoterol solution can be combined with ipratropium bromide, acetylcysteine, and budesonide without changes in the amount of active component of these drugs in the admixture [Bonasia et al. 2007], although this study does not provide evidence of clinical efficacy or safety of this practice. More importantly, for patients who cannot coordinate inhalation technique for use with pressurized metered-dose inhalers (MDIs) or DPIs, who cannot manipulate those devices, or cannot generate sufficient inhalation velocity for adequate drug delivery, a nebulized delivery system may be superior [Dolovich et al. 2005]. Some patients will also express a preference for nebulized medication either due to perceived efficacy or relative cost. Arformoterol presents an appropriate choice for these patients. No studies are available to test whether it is superior to the nebulized form of racemic formoterol, although both of these medications appear equally effective in nondirect comparison [Gross et al. 2008; Hanrahan et al. 2008b].
Conclusion/summary
Arformoterol represents a unique agent in the clinician’s armamentarium. As one of two nebulized LABA agents, its single-stereoisomer pharmacology may provide effective bronchodilator activity with minimal side effects. However, clinical studies directly comparing arformoterol with nebulized formoterol are lacking. While in vitro and pharmacologic studies suggest that the (R,R) enantiomer of formoterol contains most of the beneficial effects of this medication, to suggest any clinical superiority of arformoterol over racemic formoterol at this time would be premature.
When a long-acting bronchodilator is needed for patients with moderate-severity COPD or worse, arformoterol is among the available agents that can be recommended without reservation. It provides the ability to prescribe a nebulized maintenance therapy with the convenience of twice-daily dosing. When patient characteristics include very severe airflow obstruction, inability to cooperate with DPI or MDI technique, or preference for nebulized drug delivery, arformoterol may be especially appropriate.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
James F. Donohue is an ad hoc advisor to Sunovion.
