Abstract
Introduction
Primary ciliary dyskinesia (PCD) is a genetic, congenital, and heterogenous disorder. Dyskinetic multiple motile cilia lining the airways cause impaired mucociliary clearance leading to recurrent and chronic infections of the upper and lower airways further resulting in destructive inflammation and bronchiectasis, and with time decline of lung function.1–3 The development of bronchiectasis, often seen already in childhood, provides an even more favorable environment for persistent infection of Gram-negative microorganisms such as Pseudomonas aeruginosa (PA), Achromobacter species (AS), Burkholderia species, (BS) and non-tuberculous mycobacteria (NTM) (Gram-positive) of the lungs. These infections cause a potentially further downward spiral with destruction and significantly increased treatment burden and reduced quality of life.4–7 Due to a general lack of evidence-based treatment in PCD, care is typically extrapolated from the care of cystic fibrosis (CF), which, only to a limited extend, resemble the lung conditions in PCD.8,9 Important differences exist, not only in the pathogenesis but also in the microbiological phenotypes, which emphasize the need for disease-specific treatments. 9,10 An important first step toward a more specified adaptation of PCD treatments is a solid foundation of knowledge on the conditions in PCD as well as how they differ from CF.
AS are Gram-negative, nonfermentive opportunistic pathogens. 11 The effect of AS infection on lung function in CF is considered both of significance and concern as chronic infection with AS in CF is associated with respiratory decline, increased frequency of exacerbations, and lung inflammation,12,13 as well as patient-to-patient transmission.14–16 In addition, AS is renowned for its resilience and intrinsic and acquired multidrug resistance traits that challenge the effectiveness of treatments. 17 The rarity of positive AS cultures from patients with PCD across PCD centers worldwide and the resulting lack of experience have so far left AS colonization in PCD an unexplored field with a concern that AS infection may play a significant but ignored role in PCD. 26 In this paper, we present a review of the prevalence and colonization of AS, including the distribution of early and persistent infections, as well as an overview of the associated antibiotic treatments, and the rate of successful eradication in a national cohort of patients with PCD.
Methods
This is a retrospective, single-center, nation-wide descriptive study of patients with PCD in Denmark. Historic sputum samples were reviewed for the previous 18 years and electronic health records reviewed for antibiotic treatments during infections with AS.
Data collection
Data was obtained from the Danish PCD registry 18 (patient demographics), MiBa (microbiology samples), and from reviews of individual health records (antibiotic treatments).
Patient demographics
The Danish PCD registry is associated with the Danish PCD center, a nationwide center facilitating the management, monitoring, and general care of pediatric and adult patients with PCD in Denmark. One hundred and forty-three patients with PCD are affiliated with the center and are intentionally scheduled for four annual visits to the outpatient clinic, including routine spirometry and sputum samples for microbiological examination.
Microbiology samples
All sputum samples analyzed for microorganisms are registered in an electronic microbiology database (MiBa), dating back to year 2002.
Antibiotic treatments
Systems used for electronic health records were replaced with a new national system in 2016 leading to limitations on how far back in time records were available, hence records were only reviewed from 2010 and onward. Records were reviewed manually, for prescribed antibiotics together with any deviations due to poor compliance or adverse effects during the course of infection. Due to the retrospective design and the challenge of designating antibiotics only targeted AS, the listed covers all antibiotics given during the period including antibiotics possibly aimed at other targets. The review and noting continued until AS had not been cultured for 1 year, as well as resumed if growth reoccurred.
Detection of AS
Achromobacter species is detected routinely in the clinical microbiology laboratory. All sputum samples and airway secretions are Gram-stained to ensure the origin from the lower airways and cultured on selective media. These media include a Sabouraud plate, a 7% NaCl plate, a B. cepacia plate containing colistin and gentamicin, a “blueplate” (modified Conradi Drigalski’s medium) selective for Gram-negative rods, and non-selective media including 5% Danish blood agar and chocolate agar (Statens Serum Institute, Copenhagen, Denmark). Before 2011, isolated bacteria were identified using biochemical profiling based on API 20NE (bioMérieux, France), and from 2011, MALDI-TOF MS (Bruker Daltronics, Germany) was used. 19
Treatment regimens for AS in Denmark
In Denmark, we have currently no specific guidelines for the treatment of AS infections in patients with PCD. In general, the choice of treatment follows the same guidelines as for patients with CF, with the infection treated according to the same standards as infections with PA. 20 At initial isolation, the first choice of treatment is 3 weeks of inhaled colistin as well as oral amoxicillin with clavulanic acid. 21 With continued growth, 14 days of intravenous antibiotic therapy either with piperacillin+tazobactam, meropenem or ceftazidime in combination intravenous tobramycin, oral trimethoprim/sulfamethoxazole, or chloramphenicol, or inhaled colistin, or ceftazidime is applied followed by 3 months of inhaled ceftazidime in combination with oral trimethoprim/sulfamethoxazole. However, the treatment is often hampered due to acquired resistance and adapted accordingly. One important exception is in vitro resistance to ceftazidime where we, based on primary antimicrobial susceptibility testing, continue inhalation treatments.
Inclusion and exclusion criteria
We included patients with a verified diagnosis of PCD according to the latest guidelines from the European Respiratory Society (ERS). 22 Patients with fewer than four microbiological samples or less than 1 year of continuous samples were excluded.
Infection state
Infections with AS were classified as persistent when more than three isolates of AS, at least 1 month apart, had been cultured within the preceding 12 months and otherwise classified as intermittent. Infections were regarded as eradicated when there had been no isolates of AS for 6 months, requiring a minimum of three samples with a minimum of 1 month between samples.
Lung function
Historical lung function data was used to assess lung function. The progression of lung function 2 years prior to infection in patients who became persistently infected was compared to the lung function during the first 2 years of infection. Further, subjects were matched 1:1 to age-matched (±2 years) controls without significant infection in order to compare the evolution of lung function to unaffected individuals.
Statistics
Discrete data is reported as counts and percentages while continuous data is reported as medians including interquartile and/or ranges. Fishers exact/Pearson’s Chi-squared test was used to calculate p-values for proportions, while the Wilcoxon signed rank test was used for continuous variables. Linear mixed effects models with random intercepts were applied to assess changes in lung function, with z-tests performed to compare slopes. p-values < 0.05 were considered significant. R version 3.6.3 was used for statistical analysis.
Results
Study population
The Danish PCD registry currently contains data of 148 patients with PCD. We successfully managed to link 140 [95%] of these patients with data from MiBa. We excluded four patients that did not meet the inclusion criteria (<4 samples and/or <12 months of data) and included 136 patients in the study (Figure 1). Microbiologic data was available for a median (range) of 14.6 (1–18.6) years with 72 (4–244) samples per patient, corresponding to 6 (1–25) samples per year. The median follow-up time for patients with positive AS was 17.5 (1–18.6) years (Table 1). Flow diagram of patient inclusion and allocation according to sample results. The Danish PCD registry contained information on 148 patients with PCD of which 12 had insufficient microbiology samples and were excluded, leading to a total of 136 patients included for further review. Patient demographics at first sample. an (%); Median (IQR). bN = 36 patients were too young to perform spirometry at their first sample.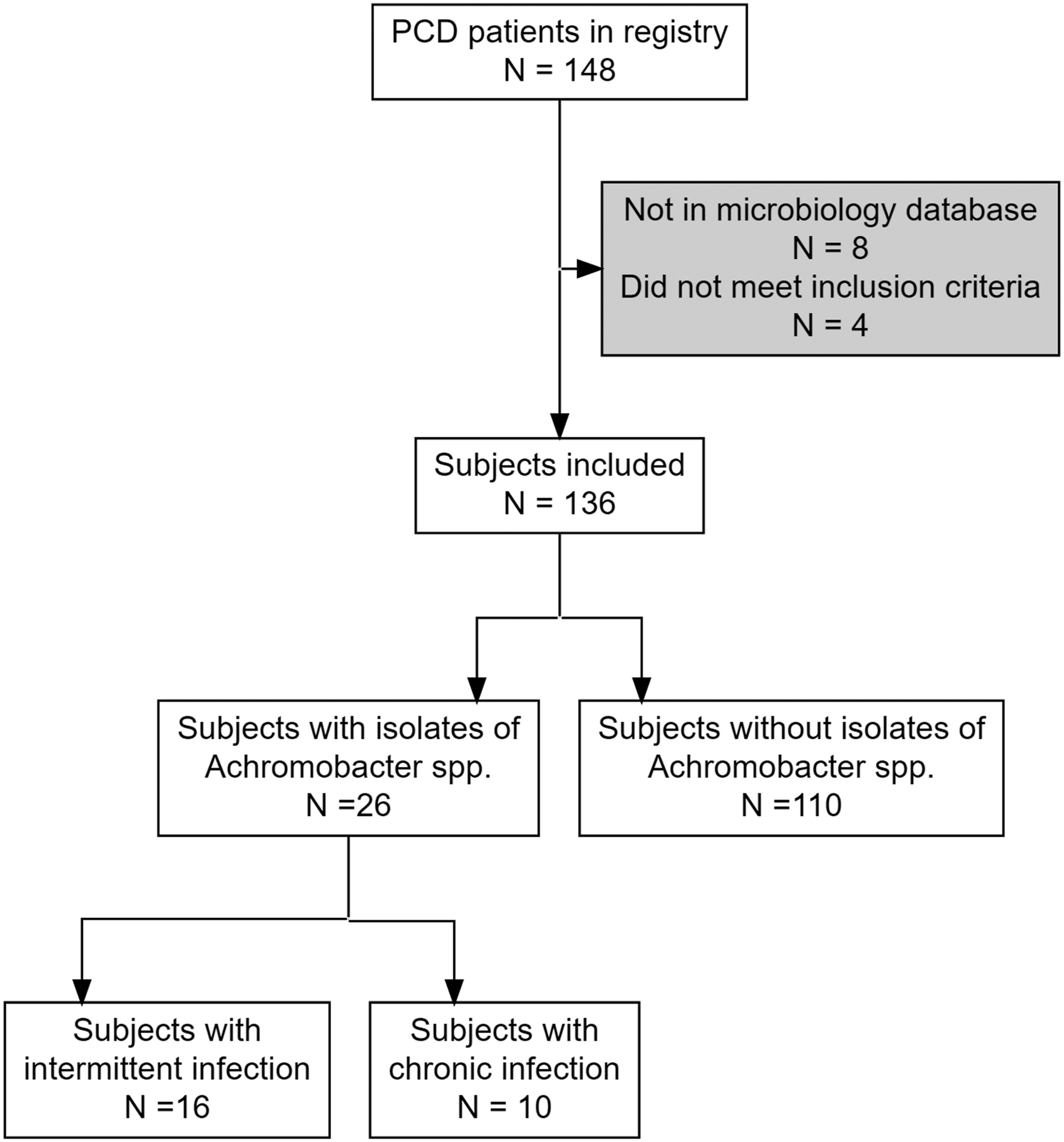

Occurrences of AS in patient samples
In total, 26 patients had cultures of AS in airway secretions. The annual prevalence (new and ongoing AS infections) ranged from 1.4 to 9.1% (median 4.6%), while the annual incidence of new infections ranged from 0 to 3% (median 1.2%) (Figure 2). The observed proportion of patients (%) with Achromobacter species infection reported annually. The proportion of patients who within a given year have had a positive culture with AS (prevalence) as well as the proportion of patients where the culture had not been preceded by positive samples in the previous year. Data are calculated on an annual basis, and the horizontal dotted lines indicate the average values.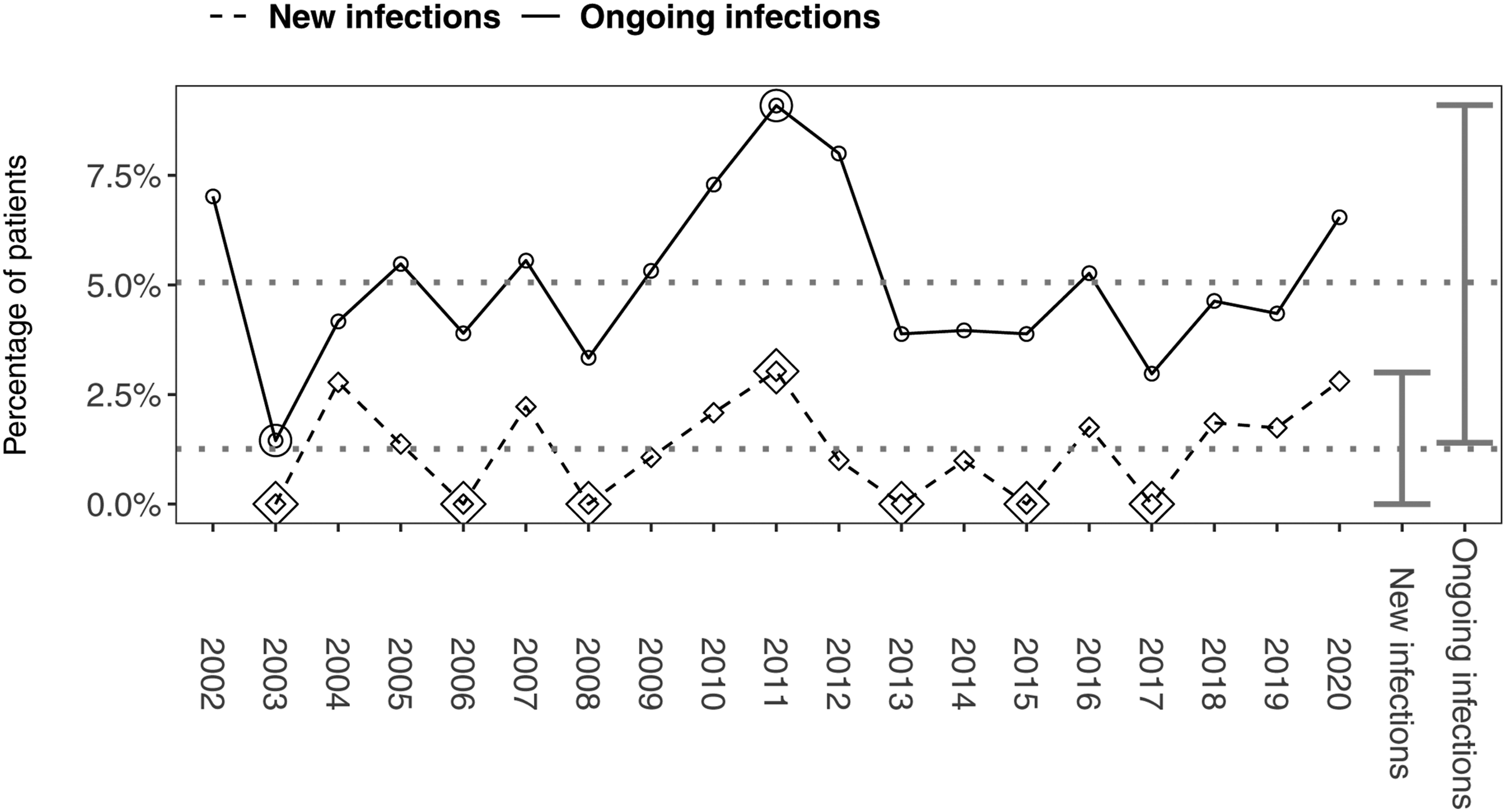
Characteristics of the AS infections
Infection characteristics.

aMedian (IQR); n (%).
bWilcoxon rank sum test; Fisher’s exact test; and Pearson’s Chi-squared test.
Association with lung function
The progression of FEV1 percent predicted in patients persistently infected was significantly different during infection (63.7 with an annual change of −2.2) when compared to the two preceding years (59.7 +1.2 per year), p = 0.017. The course prior to infection differed significantly to the course in unaffected controls (68.8–1.8 per year), p = 0.045, while the course during infection did not, p = 0.241 (Figure 3). Impact of persistent Achromobacter infection on lung function. Lung function in cases who became persistently infected with AS reported as FEV1 percent predicted. The rate of decline during the first 2 years of infection is compared to the decline during the 2 years prior to infection, as well as decline in 1:1 age-matched uninfected controls. The slope was significantly different between cases prior to and during infection (p = 0.017) as well as between cases prior to infection and controls (p = 0.045). The rate of decline during infection was not different from the rate of decline in uninfected controls (p = 0.241).
Antibiotics and eradication success
Antibiotic resistance
Resistance patterns during the first 2 years of persistent infection are presented in Figure 4. In general, low levels of resistance were seen for piperacillin/tazobactam, amoxicillin/clavulanate, meropenem, imipenem and colistin, while most strains were resistant to tobramycin and ciprofloxacin. Antibiotic resistance patterns in patients with persistent infection. Resistance patterns for patients who became persistently infected reported at approximately 12-month intervals for the first 2 years.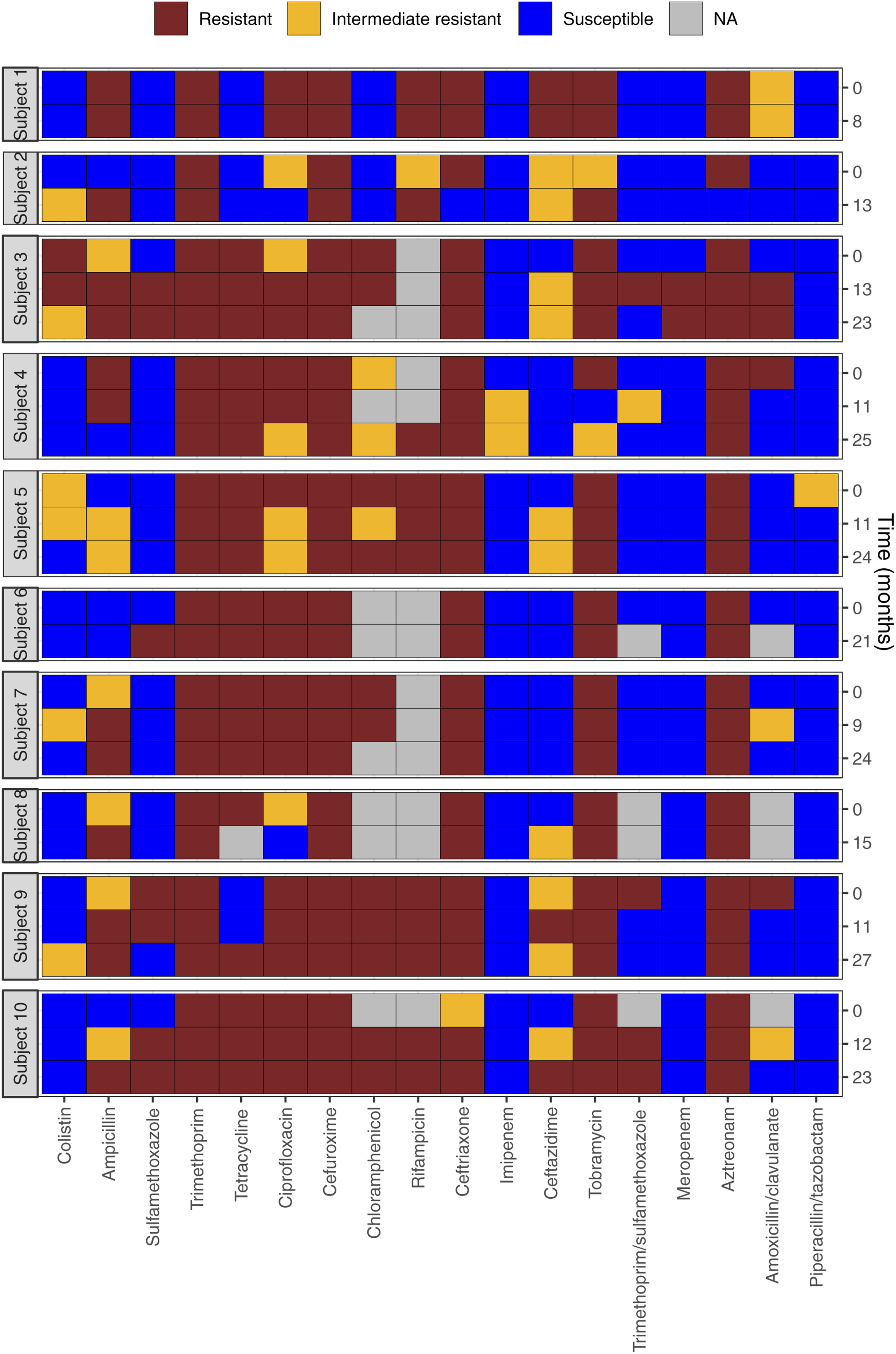
Eradication
Successful eradication of AS was achieved in 61.5% (16/26) patients. For patients with intermittent infection fulfilling the eradication criteria, 70% (7/10) only had AS cultured once (i.e., one positive sample), while the remaining three patients had a median (range) infection length of 0.8 (0.6–1.2) years. Eradication of persistent infections was achieved in 60% (6/10) of patients after a median (range) of 4.5 (0.7–7.8) years (at least three samples more than 1 month apart for six consecutive months without any isolates). The amount of prescribed antibiotics according to patient records (available from 2010 and onward) is visually illustrated as a timeline in Figure 5. Five patients had eradication prior to 2010 and hence these patients are missing.
Intravenous antibiotics
A total of 81% (17/21) patients received intravenous antibiotics, 1–10 times (median of 2) with a median (range) duration of 14 (5–41) days. Meropenem was the most frequently administered intravenous antibiotic used 64.1% (34/64) of times, followed by a combination of meropenem/tobramycin used 10.9% (7/64) of times.
Inhalation antibiotics
Nearly all patients (85.7%, 18/21) received inhalation antibiotics. The total quantity ranged from 1 to 19 (median of 4), with a median (range) duration of 79 (8–724) days for each treatment. Colistin was the most prevalent antibiotic used in inhalation therapy (68.5% of treatments 61/89), followed by tobramycin (16.9% of treatments, 15/89).
Oral antibiotics
Orally administered antibiotics were prescribed to 95.2% (20/21) patients a varying number of times ranging from 1 to 38 courses (median of 3). Treatment durations ranged from 5 to 947 days (median 27). Amoxicillin and clavulanic acid accounted for 51.7% (62/120) treatments and was the most frequently used oral antibiotic followed by azithromycin and ciprofloxacin, both accounting for 15.8% (19/120) of treatments. Treatment courses with antibiotics during and one year after Achromobacter species isolation. Overview of the amount, type, and length of antibiotic treatments given during and one year after the last sample with AS. The bigger dots indicate positive cultures of AS while the smaller dots (asterixes) indicate cultures without AS. As patient records are not available from before 2010, the period prior to this is not covered.
Discussion
Summary of main findings
This is the first study in which respiratory infections with AS in patients with PCD are directly compared with clinical and treatment data. Calculated annually, we found that an average of one percent of our patients contracted a respiratory infection with AS, and that an average of five percent of the patient cohort each year had had at least one positive sputum culture with AS. The chance that an initial positive culture of AS remained a stand-alone event even without treatment was quite high. This often gives rise to a dilemma also given the typically high rate of antibiotic resistance in AS, as to whether treatment should always be initiated at the first culture or whether one can wait for the next positive culture. Some studies have indicated that infection is commonly lost without specific eradication attempts. 23 However, we found that the chance of eradication in the early phase of positive cultures during the intermediate term is quite high. However, one-third remained intermittent with an unknown outcome, and another one-third developed persistent infection with a long duration (median of 6.6 years), and a high rate of antibiotic treatments that in two-thirds led to successful eradication of the infection. We addressed potential differences at first isolation between patients that managed to clear the infection and patients in whom the infection became chronic. We found no differences in age but observed a significantly lower FEV1 percent predicted at first isolation. However, this should be interpreted with caution due to the small number of patients included but is nevertheless consistent with what has previously been reported in patients with CF both concerning PA and AS. 24 We did not find a significant impact of AS on lung function when comparing the course of FEV1 percent predicted in patients with persistent infection to uninfected age-matched controls. However, the course of lung function prior to infection within the group of patients with persistent AS did differ to the course during infection (Figure 3), and our study does rise minor concerns toward low FEV1 and risk of contracting AS since significantly lower FEV1 percent-predicted at first isolation was associated with definite established or unambiguous development toward evolution of persistent infection.
In CF, AS is considered an opportunistic pathogen that causes healthcare-associated infections. In the US, 42% of Achromobacter-infected patients were infected with Achromobacter xylosoxidans while the rest were infected with other AS. 16 Since 1996 AS—in the CF population—has shown increasing prevalence from a few percent to approximately 5 percent in 2019, and virtually absent until school age showing increasing prevalence to between 5 and 10 percent in adolescence and beyond. 24
A previous study of the Danish PCD cohort found a median annual prevalence of A. xylosoxidans of 5.3%, 5 which is slightly different from the figures we present in this report. This may be explained by the fact that we did not limit the search exclusively to xylosoxidans but possibly also by a continuous review and revision of patient diagnoses according to updated guidelines and the greater availability of genetic analysis. 15
Perspectives, to treat or not to treat AS infection, and patient segregation
Our segregation and treatment strategy for PCD rely heavily on the way we manage AS infection in patients with CF. The start-up approach to CF is inspired by recommendations from Infection Prevention & Control (IP&C) guidelines in CF but also our own experiences and studies of the significance of AS infection for deteriorating development of CF lung disease.16,25 For patients with PCD, this implies a strict segregation from first culture of AS and initiation of the treatment regimen explained earlier. Furthermore, it is interesting that the impact of AS infection is considered rather controversial since there was no possibility of reaching a consensus on the management of AS infection in a recent IP&C PCD consensus document,
26
which probably reflects a large variation in experience with and opinion regarding AS infection among the PCD experts in the consensus panel
Treatment overview
According to the varying degree of infection persistence, we showed that the treatment intensity varied correspondingly. Many aspects of the treatment regimen are inspired by the evidence from antibiotic treatment of pulmonary PA infection in CF patients and our own experience with AS where it is highly recognized that AS has a natural innate resistance and, in most cases, even multi-drug-resistance as recognized already in the first isolate and with further development of increasing resistance. This is of great importance and influences our choice of prescribed antibiotics, which often include intravenous administration which is reflected in our findings of resistance patterns and the large armamentarium of antibiotics used in the treatment of this relatively small group of patients.
Strengths and the limitations of this study
Strengths
The strength of this study is the long observation period of 18 years (median 14.6 years) with a comprehensive thorough registration of all pathogenic bacteria, where in this study we have focused exclusively on AS occurrence, persistence, and treatment. The latter has consisted of a meticulous review of all patient records and notes regarding the detection of AS and antibiotic treatment. As all patients are associated with our center throughout life, it is possible to track all patients in terms of outcomes.
Limitations
A weakness of this study, though it is a built-in known premise, is the relatively small number of patients in the cohort and the small number of positive cultures for AS, which makes incidence and prevalence calculations relatively uncertain. Interestingly, however, we demonstrate a fairly stable prevalence comparable to what has been shown in CF. Age is severely left skewed (many young patients), but this is a common observation in all data set including registries as recently reported. 18 Further, information on treatments relies on patient records, that is, only our best guess of what really happened in terms of treatment duration and not everything is always reported. Changes in electronic health record systems and databases over the time-period of this study have also resulted in lack of data concerning treatment, which was only accessible from 2010, while microbiology could be tracked back to 2002. Finally, we often found contemporary overlap with other infections, which potentially could render treatment data as slightly non-specific particularly in cases where it was not explicitly stated which drugs were prescribed for which bacteria.
The implications for future research or clinical practice
Future studies of the prevalence, treatment, and potential influence of various bacteria on the development of PCD lung disease should be performed under controlled prospective circumstances. For this purpose, the new FOLLOW-PCD initiative where the clinical visits are recorded systematically for several parameters including microbiology and prescribed treatment is of great importance in combination with ongoing registration of clinical data in well-functioning registries.18,27,28 In this way, far more valid data from many PCD centers can contribute, among other things, to a greater understanding of the influence of AS and other.
In conclusion, Achromobacter species give rise to respiratory infections in patients with PCD with a prevalence comparable to what has been shown in patients with CF. Achromobacter can occur transiently, intermittently, or develop into a serious persistent lung infection associated with long-term antibiotic treatment. We recommend larger prospective multicenter studies to clarify the significance of both AS, but also other bacterial lung infections for the development of PCD lung disease.
Footnotes
Acknowledgments
The research pool from the University Hospital of Copenhagen (Rigshospitalet) sponsored a one-year grant and Ethris GmbH (Planegg, Germany) sponsored part time salaries for authors MGH and JKM. K.G.N. is a member of the European Reference Network of Rare Respiratory Disease (ERN-LUNG).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the This work was supported by the Children’s Lung Foundation (Denmark), BEAT-PCD European Respiratory Society, BEAT-PCD COST Action (BM1407), and the European Reference Network for Rare Respiratory Diseases (ERN- LUNG; project ID no. 739546).
