Abstract
Chronotropic incompetence (CI; failure to reach the targeted heart rate (HR) on exercise) and a delayed HR recovery (HRR; ≤12 beats decline within the first minute after cessation) reflect autonomic dysfunction (AD) and predict adverse cardiac prognosis. As chronic obstructive pulmonary disease (COPD) is known to be associated with AD, we hypothesized that these patients may manifest these responses on exercise. The prevalence and predictors of these responses in COPD and their association with its severity have not been evaluated. Normoxemic, stable male patients with COPD (n = 39) and 11 healthy controls underwent lung function testing and incremental leg ergometry. HR responses were monitored during exercise and recovery to compute the HRR and CI. Of all the patients, 33 (84.6%) had at least one of the two exercise responses as abnormal, with the majority (23, 58.9%) having both an abnormal HRR and CI. The frequency of abnormal responses increased with increasing Global Initiative for Chronic Obstructive Lung Disease stage and body mass index, airflow obstruction, dyspnoea and exercise capacity index. After adjusting for smoking history and post-bronchodilator forced expiratory volume in 1 second, only a reduced diffusion capacity for carbon monoxide predicted abnormal HRR, though weakly. We concluded that abnormal HRR and CI are common in patients with COPD. These responses are observed with increasing frequency as the severity of disease increases.
Keywords
Introduction
Cardiovascular disease is a major cause of mortality in chronic obstructive pulmonary disease (COPD). 1 A probable cause of mortality is autonomic dysfunction (AD) that may predispose to arrhythmias. 2,3 Though there is a lack of direct evidence, AD predicts fatal cardiac events in diabetes mellitus, post-myocardial infarction state and heart failure 4 and may play a similar role in COPD.
The rate of recovery of heart rate (HR) within the first minute after cessation of exercise, termed ‘heart rate recovery’ (HRR), reflects parasympathetic reactivation and an abnormally delayed (≤12 beats) decline implies its dysfunction. 5,6 It has been shown to predict all-cause and cardiac mortality in population studies and in patients with cardiac disease. 6 –10 Though it has not been systematically investigated, a delayed HRR has been observed in studies on cardiac responses to exercise in COPD. 11,12 In a retrospective analysis, Lacasse et al. 13 found abnormal HRR to be associated with all-cause mortality in COPD. Chronotropic incompetence (CI), an attenuated HR response to exercise, is believed to represent an impaired sympathetic response and is another independent predictor of cardiac mortality. 14 –16 It is also associated with atherosclerosis 17 and diastolic heart failure. 18 In a recent retrospective study, it was observed that CI improved after lung volume reduction surgery in patients with COPD. 19 A prospective study to document and characterize CI has not been carried out in COPD.
Stewart et al. 2 demonstrated AD in patients with hypoxemic COPD, and later, we reported it even in normoxemic patients and in those with mild COPD. 3 As both an abnormal HRR and CI reflect AD, we hypothesized that an abnormal HRR and CI may occur in COPD. Being established as independent predictors of cardiac mortality, these markers of AD may find application as risk stratification tools in COPD and therefore need to be characterized. The prevalence and predictors of these markers in COPD and their association with severity of the disease have not been evaluated. With these objectives, the present study was carried out.
Materials and methods
The study was conducted in an outpatient setting at the Vallabhbhai Patel Chest Institute (New Delhi, India). It was approved by the Ethics Committee of Vallabhbhai Patel Chest Institute (MD Pulmonary Medicine project 3/2008). A total of 39 stable patients with a clinical diagnosis of COPD based on Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria 20 were included after a written informed consent.
Subjects
We intended to include patients with COPD due to their smoking habit. COPD in women in India is more often due to biomass fuel exposures than due to smoking and considering our hospital attendance patterns, it was felt that sufficient numbers of female smokers with COPD may not be found within the study period. Therefore, we selected only male patients with a history of smoking of ≥10 pack-years. Consecutive stable patients were screened for inclusion in the study. Patients with chronic respiratory failure (pulse oximetry saturation < 90% and partial pressure of oxygen (Pa
Initial assessment
After blood counts, routine biochemistry panel, plain chest radiograph (posteroanterior view) and a 12-lead standard electrocardiogram, the patients underwent lung function tests including spirometry and measurement of single-breath diffusion capacity for carbon monoxide (DLCO) and a 6-min walk test. Dyspnoea was graded according to the Modified Medical Research Council scale. The severity of COPD was staged according to the GOLD criteria. 20 Body mass index, airflow obstruction, dyspnoea and exercise capacity (BODE) index was calculated. 21 The BODE index scores each of these components in an ordinal manner and a total composite score is calculated. As it is not scored on a ratio/interval scale, we did not use it as a continuous variable but divided the patients into two categories, above and below the 50th percentile; these were labelled as ‘low’ and ‘high’ BODE index groups, respectively.
Inhaled bronchodilators were withdrawn 24 hours before investigations but corticosteroids were allowed. Spirometry was performed on a dry rolling seal spirometer (Benchmark lung function equipment, P.K. Morgan, Kent, UK) according to current recommendations. 22 DLCO was corrected for alveolar volume to obtain the Krogh’s constant (KCO). The 6-minute walk test was performed in accordance with the American Thoracic Society guidelines. 23 The 6-minute walk distance (6MWD) was used to calculate the volume of oxygen consumed and the exercise workload to be applied. The maximum HR (MHR) on ergometry was calculated (MHR = 220 − age). The target HR (THR) was set at 80% of MHR.
Leg ergometry
After 4 hours post-prandial and a 30-minute rest, a 3-minute stepwise test of increasing intensity of exercise was performed by leg ergometry on a manually braked cycle (Rehab Trainer 881E, Monark Exercise AB, Sweden). It was supervised by the same operator Vishal Bansal. The patients were instructed to pedal at frequencies of 50–55 cycles/min. In this range, the cycle delivered a constant workload. This frequency was maintained throughout the exercise and in the cooling phase. The predicted maximal oxygen consumption (V
HRR was calculated as the difference between the HR at peak exercise and at first minute into the cooling period. A cut-off point of ≤12 beats was taken as an abnormal HRR.
6
The utilization of HR reserve at peak exercise was expressed as the chronotropic response index (CRI)
15
and was calculated as follows:
Statistical analysis
Statistical analysis was carried out using SPSS 16.0 (SPSS Inc., Chicago, Illinois, USA) and Graph Pad Prism 4.01 (GraphPad Software, Inc., La Jolla, California, USA) softwares. The data were examined for normality of distribution by Shapiro–Wilk’s test and for homogeneity of variance by Levene’s test. Data on continuous variables are presented as mean ± SD. Student’s unpaired t test/Mann–Whitney’s U test was used for independent group comparisons. Comparison of data on continuous variables among multiple groups of patients was carried out by Kruskal–Wallis analysis of variance. Chi-square test was used to compare the association between abnormal HRR and CI. Correlation analysis was carried out to determine Pearson’s correlation coefficient. Multiple logistic regression was carried out to identify significant predictors of HRR/CI after adjusting for other variables. Abnormal/normal HRR and presence/absence of CI were the dependent binary variables and age, pack-years of smoking, post-bronchodilator forced expiratory volume in 1 second (FEV1) percentage predicted, percentage predicted DLCO and percentage predicted KCO were the predictor variables. A two-tailed p value of <0.05 was considered statistically significant.
Results
The clinical characteristics and baseline laboratory parameters are shown in Table 1. Patients with severe COPD had a lower body mass index as compared to those with mild COPD (p < 0.01), while age, smoking intensity and duration of disease were similar across categories by GOLD severity. There was a significant progressive deterioration of lung function parameters and BODE index with increasing severity.
Clinical characteristics and lung function across GOLD categories.
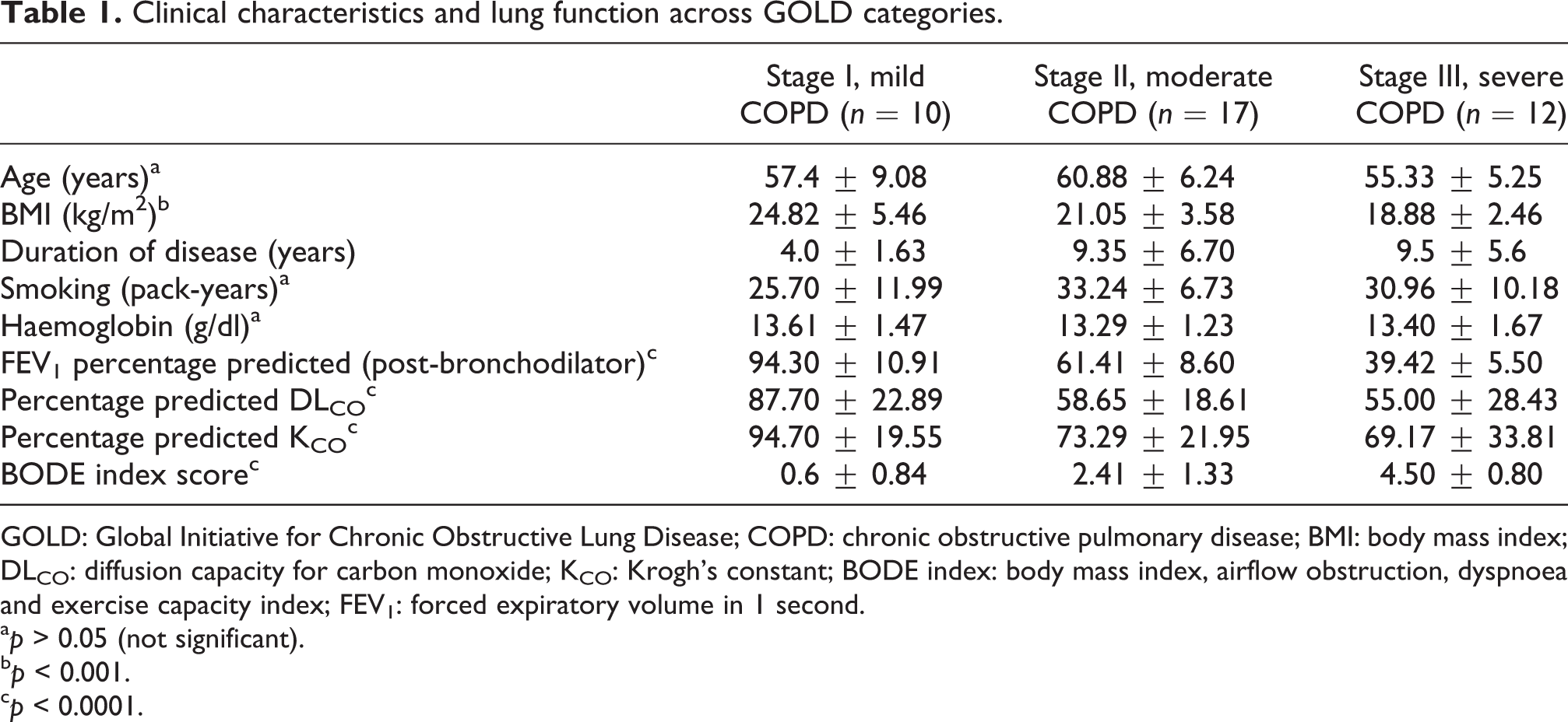
GOLD: Global Initiative for Chronic Obstructive Lung Disease; COPD: chronic obstructive pulmonary disease; BMI: body mass index; DLCO: diffusion capacity for carbon monoxide; KCO: Krogh’s constant; BODE index: body mass index, airflow obstruction, dyspnoea and exercise capacity index; FEV1: forced expiratory volume in 1 second.
a p > 0.05 (not significant).
b p < 0.001.
c p < 0.0001.
The duration of exercise (in seconds) was 444.7 ± 173.7 in patients and 637.7 ± 229.3 in controls. The HR response during exercise in patients and controls is shown in Table 2. The resting HR was not different (p > 0.05), but the latter achieved a higher peak HR (p < 0.001). The CRI and HRR were significantly reduced in patients (p < 0.01 and p < 0.0001, respectively). Only 13 patients could achieve the THR, 6 of the 10 mild and only 2 of the 12 severe COPD patients (p = 0.05). Majority of the patients (22, 56%) stopped exercise due to leg fatigue and only four stopped because of dyspnoea. All the normal subjects reached the THR.
HR response during exercise in patients and controls.

HR: heart rate; bpm: beats per minute.
a p > 0.05 (not significant).
b p < 0.001.
c p < 0.0001.
d p < 0.01.
Figure 1 shows the HRR and CRI across GOLD stages. The HRR in mild COPD was 17.8 ± 11.67 beats, 12.7 ± 8.86 beats in moderate and 3 ± 4.97 beats in severe COPD (p = 0.09). The CRI in these patients was 49.3 ± 15.89, 46.96 ± 13.45 and 37.3 ± 10.93 (p = 0.08), respectively. Comparison of patients with low and high BODE index scores (Figure 2) revealed that the latter had a lower HRR (15.42 ± 10.15 and 9.13 ± 5.45 beats, respectively, p < 0.05) and a lower CRI (49.19 ± 14.28 and 37.22 ± 10.08, respectively, p < 0.01). The HRR was significantly correlated with CRI (r = 0.54, p < 0.001).
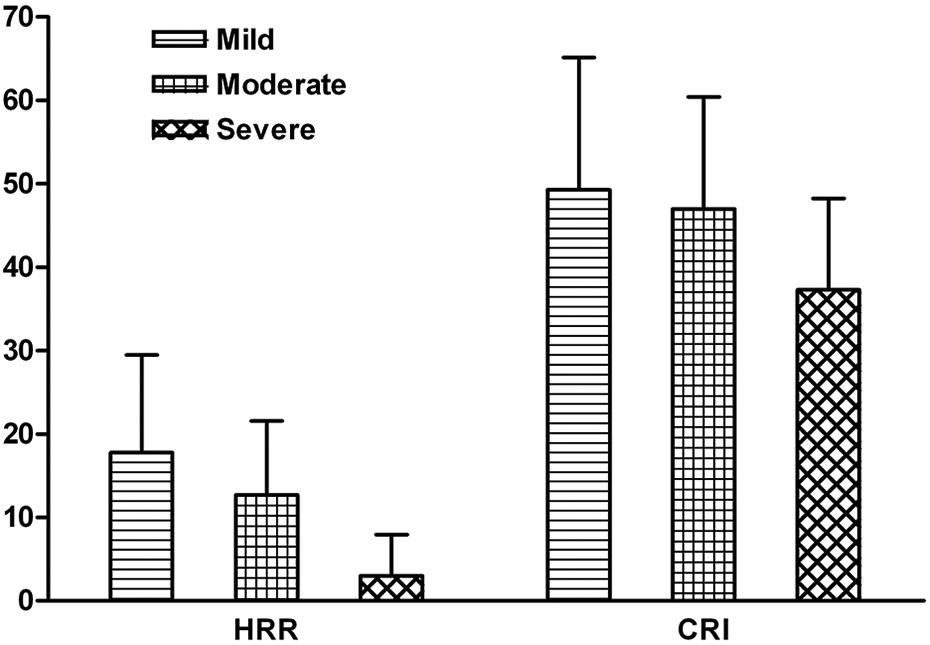
HRR and CRI according to GOLD categories of severity; p values of HRR = 0.09 and CRI = 0.08. HRR is expressed in beats in first minute, CRI is expressed in percentage. HRR: heart rate recovery; CRI: chronotropic response index; GOLD: Global Initiative for Chronic Obstructive Lung Disease.

HRR and CRI in patients with low and high BODE index scores (+ indicates p < 0.05, * indicates p < 0.01). HRR is expressed in beats in first minute, CRI is expressed in percentage. HRR: heart rate recovery; CRI: chronotropic response index; BODE: body mass index, airflow obstruction, dyspnoea and exercise capacity.
Of them, 29 (74.4%) patients but none of the controls had an abnormal HRR (p < 0.0001). CI was found in 27 (69.2%) patients but in only one control subject (p < 0.0001). Majority of the patients (33, 84.6%) had at least one of the autonomic responses as abnormal. Of the 39 patients, 23 (58.9%) had both abnormal HRR and CI, 6 (15.4%) had isolated abnormal HRR, and 4 (10.3%) had isolated CI. Only six patients (15.4%) had no abnormality in the two exercise responses.
Figures 3 and 4 show the proportions of subjects with abnormal HRR and CI across GOLD and BODE categories, respectively. The frequency of patients with abnormal HRR increased with increasing GOLD severity: 5 of 10 (50%) in mild, 13 of 17 (70%) in moderate and 11 of 12 (92%) in patients with severe COPD (0.05 > p < 0.1). While 4 of 10 (40%) in mild COPD had CI, the proportion of CI increased to 12 of 17 (70.5%) in moderate and to 11 of 12 (92%) in patients with severe COPD (p < 0.05). Four of the six subjects who had normal HRR and did not have CI belonged to the mild group. Abnormal HRR was observed in 16 of 24 (66.7%) patients in the low and 13 of 15 (86.7%) patients in the high BODE index groups (p > 0.05). CI was significantly more frequent in the high BODE index group (14 of 15 patients – 93.3%) compared to 13 of 24 (54%) patients in the low BODE index group (p < 0.01). All the six subjects who had normal HRR and no CI were in the low BODE index group.

Proportions of subjects with normal and abnormal HRR across GOLD severity and BODE index categories. HRR: heart rate recovery; BODE: body mass index, airflow obstruction, dyspnoea and exercise capacity; GOLD: Global Initiative for Chronic Obstructive Lung Disease.

Proportions of subjects in the absence and presence of CI across GOLD severity and BODE index categories. BODE: body mass index, airflow obstruction, dyspnoea and exercise capacity; CI: chronotropic incompetence; GOLD: Global Initiative for Chronic Obstructive Lung Disease.
Significant inverse correlations were observed between HRR and age (r = −0.41, p < 0.01) and pack-years of smoking (r = −0.35, <0.05), while positive correlations were found between HRR and percentage predicted DLCO (r = 0.37, p = 0.05) and percentage predicted KCO (r = 0.31, p < 0.05). Correlations were not significant between HRR and post-bronchodilator FEV1 percentage predicted (p > 0.05). CRI did not show significant correlations with any of the above variables (p > 0.05). On multiple logistic regression, KCO remained a significant, though weak predictor of abnormal HRR, after adjusting for age, smoking intensity and post-bronchodilator FEV1. It explained only 22% of the variability in HRR.
Comparison of clinical characteristics between patients with abnormal/normal HRR and present/absent CI is shown in Table 3. Patients with an abnormal HRR were older and had poorer lung function and lung diffusion parameters as compared to those with a normal HRR. On the other hand, patients with CI had higher BODE index scores (p < 0.05) as compared to those without CI.
Comparison of clinical characteristics between patients with abnormal/normal HRR and present/absent CI.

HRR: heart rate recovery; CI: chronotropic incompetence; DLCO: diffusion capacity for carbon monoxide; KCO: Krogh’s constant; BODE index: body mass index, airflow obstruction, dyspnoea, and exercise capacity index; FEV1: forced expiratory volume in 1 second
a p < 0.05.
b p > 0.05 (not significant).
c p < 0.01.
Discussion
We found that nearly 85% of normoxemic patients of COPD have either abnormal HRR or CI or both on exercise. These tended to occur together and were observed at all levels of severity, becoming more frequent with higher GOLD stages and significantly increasing with higher BODE index. Patients with abnormal HRR had greater airways obstruction and a lower diffusion capacity compared to those with a normal HRR. After adjusting for age, smoking history and airways obstruction, only a reduced diffusion capacity predicted an abnormal HRR, though weakly. No clinical or lung function parameter predictor of abnormal CI was identified.
The first minute rapid decline in HR following exercise-induced tachycardia is largely due to parasympathetic restoration and a slower decrease was first shown to mark an increased risk of mortality by Schwartz et al. 25 This was subsequently confirmed in other disease states independent of exercise conditions, peak and resting HR. 6 –10 Though not specifically investigated in COPD, an abnormal HRR has been noted in studies on cardiac responses to exercise in such patients. Chick et al. observed prolonged tachycardia and delayed recovery after exercise that was independent of FEV1. 11 In a retrospective analysis of data in a heterogeneous group of patients, including COPD, Seshadri et al. 12 observed that abnormal HRR was found with increasing frequency as FEV1 decreased. In another retrospective study, Lacasse et al. 13 found an association between abnormal HRR and increased risk of all-cause mortality among patients with an FEV1 <50% predicted. Ba et al. 26 followed the HR decay after maximal cycling exercise to study metabolic factors affecting the recovery in patients with COPD and identified greater lactic acid production and/or hypoxaemia as being associated with slower recovery. These factors determine the recovery later on after exercise. They did not study autonomic mechanisms of recovery that operate almost exclusively in the first minute. There are no previous reports of CI in COPD. Multiple definitions and lack of standardized criteria likely account for the wide range in reported prevalence of CI in different disease states. 27 The most often used criteria to diagnose CI is a failure to reach an arbitrary percentage (usually 80%) of the age-predicted maximal HR or a failure to attain 80% of the HR reserve from rest to peak exercise (CRI). 16,24,27 We defined CI by a strict criteria requiring both these conditions to be met.
Implications for clinical practice and future research
AD was first reported in advanced COPD with respiratory failure 2 and later abnormalities of HR variability were also documented in patients without hypoxemia. 28 Although Stewart et al. 2 found abnormalities largely in the parasympathetic limb, Stein et al. 28 observed a mixed sympathetic and parasympathetic modulation of HR in COPD. From our laboratory studies, we reported that both parasympathetic and sympathetic limbs of the cardiac autonomic control were affected even in patients without hypoxaemia and in mild COPD. 3 In the present study, the observation of an abnormal HRR suggests parasympathetic dysfunction, while the findings of CI raise the possibility of an impaired sympathetic response. The present study also confirms our earlier observations that AD develops early in COPD and becomes more frequent as the disease advances. This is likely to be a significant complication as cardiac events may be responsible for mortality even in mild COPD. 1,29
CI has been shown to be associated with carotid intima–media thickness 17 and diastolic dysfunction 18 in cardiac patients and may thus be a marker of cardiovascular involvement in COPD. An abnormal HRR and CI may therefore predict a bad cardiac prognosis in COPD as these do in other diseases. 6 –10,14 –16 This appears likely as both indices were associated with increasing BODE index, a well-recognized predictor of mortality in COPD, 21 and with increasing GOLD stage, a classification based on a reduced FEV1, that is a marker for cardiovascular mortality. 30 Therefore, abnormal HRR and CI may be useful tools for risk stratification in COPD. This needs to be explored in longitudinal studies.
Among the commonly measured clinical and physiological parameters, only a reduced diffusion capacity predicted an abnormal HRR, though weakly, and none predicted abnormal CI. It suggests that though AD is common in COPD, it probably develops independently of pulmonary involvement and may therefore be looked upon as an extrapulmonary manifestation.
Possible mechanisms for observed cardiac responses
The underlying mechanisms of impaired cardiac autonomic control during exercise are not known. Sustained tachypnoea on exercise in patients with COPD 11 may impair parasympathetic recovery, a response known to be enhanced by slower breathing. Reduced baroreflex sensitivity in COPD is associated with an impaired sympathetic response. 31 On the other hand, increased sympathetic activity has been shown at rest in COPD 32,33 that may counter parasympathetic restoration. However, we did not find any difference in the resting HR in patients and controls, and also between patients who had and did not have abnormal HRR and CI arguing against increased sympathetic activity at rest. Furthermore, Imai et al. 34 have also shown that excessive sympathetic nervous system activity does not play a dominant role in immediate HRR. An incomplete washout of anti-cholinergic drugs is unlikely to explain the delayed parasympathetic restoration after exercise as these drugs were stopped 24 hour earlier. Whether there is any subsensitivity of cardiac muscarinic receptors in COPD that may explain the abnormal HRR is not known. Furthermore, as anticholinergic drugs will delay HRR by their pharmacological action, the cardiac safety of these drugs may be questioned. Indeed, this has been a subject of several investigations though the results have been inconclusive. 35 Recently, Kawasaki et al. 24 suggested that a post-synaptic downregulation of β-adrenergic receptors in the sino-atrial node following sympathetic activation may be responsible for CI in cardiac patients. We speculate that similar to COPD, regular use of LABAs may produce a downregulation of β-receptors resulting in CI.
Strengths and limitations
Abnormal HRR and CI are well-known predictors of adverse cardiac prognosis. Although in recent years, there has been an increasing recognition of a significant cardiovascular risk in patients with COPD, the prevalence and predictors of these markers and their association with severity have not been evaluated in these patients. This is the first study to investigate these aspects of COPD. The study has identified markers that may be useful in risk stratification in COPD. Similar to their application in patients with coronary artery disease, abnormal HRR and CI may identify patients with COPD with increased risk of cardiac mortality. In view of lack of standard definitions and methods of computing CI, 27 we used a stricter definition as pointed out above to lend greater assurance to our observations.
The study is limited by being gender-specific; the reasons for non-inclusion of female subjects were explained above. Smoking itself may be a factor in causing AD. 36 Comparison of responses in patients with non-smoking COPD is therefore required to determine the contribution of COPD itself. This was not part of our study design. It may, however, be pointed out that all the patients in the present study were ex-smokers. The only way autonomic balance can be directly evaluated is by measuring the HR variability. If we had also measured HR variability during and after exercise, it would provide direct evidence of autonomic imbalance on exercise. Finally, the clinical implications are only hypothetical. The present study was cross-sectional in design and, therefore, outcome measures such as mortality or arrhythmias could not be built into the protocol. These need confirmation in a longitudinal study.
Factors such as exercise levels, medication, cardiovascular fitness, neuromuscular limitations and exercise modality may also modulate the chronotropic response and thus the observed CI may not necessarily represent sympathetic dysfunction. It is important to take into account the patient’s level of effort and reasons for terminating the exercise test before concluding that a patient has CI. 27 The respiratory exchange ratio is a more definitive and objective measure of physiological level of effort and may have provided a more definite confirmation of maximal exercise. Symptom-limited exercise also, however, ensures reasonably that maximal levels are reached. Thus, it is likely that inadequate metabolic activity was the cause of a reduced chronotropic response. Another important factor is the method of exercise. A treadmill exercise may allow achievement of higher HRs than a bicycle ergometry. 37 Several drugs such as beta agonists and theophyllines can confound the HR responses to exercise. However, inhaled beta agonists were withdrawn 24 hours before the test and none of the patients were on theophyllines or any other cardioactive drugs. Previous exercise habits and neuromuscular limitations may modulate the HR response to exercise. These were not objectively recorded. Ventilatory limitation may cause a premature stoppage of exercise and thus prevent the target HR from being achieved. It is now well established that dyspnoea on exercise in patients with COPD is related to dynamic hyperinflation and is the main reason for termination of exercise. 38 In most of our patients, dyspnoea was not the reason for stopping exercise. Thus, it can be inferred that exercise was not stopped due to ventilator limitations and lung mechanics alterations during exercise. In view of the above limitations, our observations on CI may be considered as preliminary and require confirmation.
Conclusions
To conclude, abnormal HRR and CI are observed during exercise in a majority of patients with COPD and tend to occur together. These occur even in early stages of the disease and increase in frequency with increasing GOLD-defined severity and the BODE index. Clinical and lung function parameters are poor predictors of these phenomena, with only a reduced diffusion capacity weakly predicting an abnormal HRR and no factor predicting CI. Therefore, their occurrence cannot be inferred and these need to be experimentally documented. As these indices of AD are established markers of poor cardiac prognosis, these measurements may find application as tools for risk stratification in COPD. The mechanisms underlying the pathogenesis of these abnormalities are not known and can only be speculated. The long-term significance of these abnormal responses needs to be evaluated in longitudinal studies.
Footnotes
Conflicting of Interests
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
