Abstract
Objective
To examine the combination of heart rate recovery (HRR) and heart rate variability (HRV) in predicting atrial fibrillation (AF) progression.
Methods
Data from patients with a first detected episode of AF who underwent treadmill exercise testing and 24-h Holter electrocardiography were retrospectively analysed. Autonomic dysfunction was verified using HRR values. Sympathetic and parasympathetic modulation was analysed by HRV. AF progression was defined as transition from the first detected paroxysmal episode to persistent/permanent AF.
Results
Of 306 patients, mean LF/HF ratio and HRR did not differ significantly by AF progression regardless of age (< or ≥65 years). However, when the LF/HF ratio was divided into tertiles, in patients aged <65 years, the mid LF/HF (1.60–2.40) ratio was significantly associated with lower AF progression rates and longer maintenance of normal sinus rhythm. For patients aged <65 years, less metabolic equivalents were related to higher AF progression rates. For patients aged ≥65 years, a low HRR was associated with high AF progression rates.
Conclusion
In relatively younger age, high physical capacity and balanced autonomic nervous system regulation are important predictors of AF progression. Evaluation of autonomic function assessed by age could predict AF progression.
Keywords
Introduction
Atrial fibrillation (AF), the most common arrhythmia encountered in clinical practice, is a major threat to public health. 1 AF is classified into paroxysmal and persistent forms according to the pattern of termination, and is considered permanent when efforts to restore sinus rhythm fail. Most cases of paroxysmal AF gradually progress to chronic AF. 2 The exact pathophysiological processes leading to arrhythmia initiation are not yet fully understood, however, AF aetiology is multifactorial, and autonomic dysfunction may contribute to onset and maintenance of the condition. 3
Heart rate (HR) variability (HRV) analyses have revealed signs of abnormal autonomic modulation of the sinus node in patients with AF episodes during Holter monitoring.4–6 Chronotropic incompetence, which is an inadequate HR response during exercise, indicates an abnormality in autonomic tone and is associated with increased risk of AF.7,8 However, limited data exist on the association between autonomic dysfunction and AF progression. The aim of the present study was to examine whether the combination of HR recovery (HRR) and HRV data can predict AF progression.
Patients and methods
Study population
Data from consecutive patients with AF who underwent treadmill exercise testing (TMT) and 24-h Holter monitoring at Inha University College of Medicine and Inha University Hospital, Incheon, Republic of Korea, between 2007 and 2017, were analysed in this retrospective, observational study. Patients who had undergone TMT and 24-h Holter monitoring within 1 week after a surface electrocardiogram (ECG) revealing AF were included. The key exclusion criteria were as follows: (1) aged <20 years or >80 years; (2) history of medical treatment for AF before undergoing TMT and 24-h Holter ECG monitoring; (3) high-grade atrioventricular blocks; (4) severe hypertension (systolic blood pressure >200 mmHg, diastolic blood pressure >110 mmHg, or both); (5) uncontrolled symptomatic heart failure; (6) unstable angina not previously stabilized by medical therapy; and (7) inability to exercise given extreme obesity or other physical/mental impairment. Patients were divided into four groups according to AF progression and age (< or ≥65 years). AF was defined according to the 2016 European Society of Cardiology classification. 9 Paroxysmal AF was defined as self-terminating AF, in most cases within 48 h; persistent AF was defined as AF lasting >7 days, including episodes terminated by cardioversion, either with drugs or by direct current cardioversion, after ≥7 days; and permanent AF was defined as AF that is accepted by the patient (and physician). This study defined AF progression as a transition from the first detected paroxysmal episode to persistent or permanent AF over the follow-up period, detected using 12-lead ECG or Holter monitoring.10–15
Atrial fibrillation was diagnosed when standard 12-lead ECGs showed the typical AF pattern: absolutely irregular RR intervals and no discernible P waves. The first episode of AF was detected using a Marquette MUSE system (GE Medical Systems, Milwaukee, WI, USA) and the digital images were analysed using Adobe Acrobat X Pro software, version 10.0 (Adobe Systems, San Jose, CA, USA). Recurrent symptomatic episodes were pharmacologically managed by conventional antiarrhythmic drug therapy according to current AF management guidelines. Electrical cardioversion or catheter ablation was performed as necessary or in patients with persistent AF refractory to antiarrhythmic drug therapy.
Data regarding patient demographics, comorbidities, including CHA2DS2-VASc (congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, stroke or transient ischemic attack [TIA]-vascular disease, age 65–74 years, sex category) score, medication history, and echocardiography parameters (left atrial size and left ventricular ejection fraction), were collected and analysed.
The Institutional Review Board of Inha University Hospital, Incheon, Korea approved this study. Informed consent was waived because the research involved minimal risk to the subjects, the rights and welfare of the subjects are not adversely affected by the waiver, and the research could not be practicably conducted without the waiver. The reporting of this study conforms to STROBE guidelines. 16
Exercise stress testing
Data from patients with AF, who underwent TMT using a modified Bruce protocol according to the standard method, were analysed. The test was performed as recommended by established exercise testing guidelines. The target HR was defined as 85% of the age-predicted HR, assessed by the following equation: % predicted = (peak HR/ [220–age]) × 100%. The exercise protocol was symptom-limited, and patients were encouraged to discontinue exercise at peak symptoms rather than at achievement of age-related HR. The stress test was terminated if a decrease in blood pressure (≥20 mmHg), significant arrhythmia, or >2 mm ST depression from baseline were noted. Three predefined measurements were recorded: resting HR, peak HR, and recovery HR (defined as HR after 1 min of recovery). HRR was calculated as the reduction in HR from peak HR to recovery HR. Functional capacity was evaluated by assessment of metabolic equivalents (METs), where 1 MET = 3.5 ml/kg/min of oxygen consumption, based on the protocol and the total time elapsed in the final stage.
Analysis of Holter ECG recordings
The study population underwent 24-h Holter ECG monitoring (MARS & SEER Light Extend, version 8.0.2; GE Healthcare, Milwaukee, WI, USA) within 1 week of surface ECG monitoring. Holter ECGs were continuously recorded for 24 h during normal day and night activities. HRV was assessed through the following parameters of the time/frequency domain components from the 24-h Holter monitoring: standard deviation of normal sinus to normal sinus interval (SDNN), root mean square of successive RR interval differences (rMSSD), percentage of interval differences of successive normal sinus to normal sinus intervals >50 ms, low-frequency (LF) and high-frequency (HF) components, and the ratio of LF to HF (LF/HF) components. Frequency domain parameters were analysed by LF, HF, and LF/HF ratios, including night-time (00:00–06:00 h) and daytime (12:00–18:00 h) values. LF (0.04–0.15 Hz) and HF (0.15–0.04 Hz) power was calculated using the fast Fourier transform. Average values were recorded as hourly data. HRV measures were obtained only from noise-free normal sinus rhythm. Data acquisition, QRS detection and spectral analysis followed published guidelines. 17
Statistical methods
Categorical variables are presented as frequencies (n [%] prevalence), and were analysed using Pearson’s χ2-test or Fisher’s exact test. Continuous variables are presented as mean ± SD and were analysed using Student’s t-test or Wilcoxon rank–sum test. Independent predictors of AF progression were investigated by logistic regression using clinical factors, HRV parameters on 24-h Holter monitoring, and HRR parameters on TMT. Statistical comparisons were performed with IBM® SPSS® Statistics software for Windows, version 21.0 (IBM Corp., Armonk, NY, USA), and P values <0.05 were considered statistically significant.
Results
Patient clinical characteristics
A total of 306 consecutive patients with a first detected AF who underwent TMT and 24-h Holter monitoring were enrolled. Baseline demographic and clinical characteristics of patients categorized according to AF progression (with or without) and age (< or ≥65 years) are summarized in Table 1. The mean patient age was 55.4 ± 12.4 years, and 85 patients (27.8%) were women. During the mean follow-up period of 46.7 ± 40.8 months (range, 8‒251 months), 121 patients developed AF progression (82/228 patients [36%] aged <65 years and 39/78 patients [50%] aged ≥65 years, respectively).
Demographic and clinical characteristics in 306 patients with AF grouped according to age and AF progression.

Data presented as mean ± SD or n (%).
AF, atrial fibrillation; BMI, body mass index; CAD, coronary artery disease; CCB, calcium channel blocker; CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, stroke or transient ischemic attack (TIA)-vascular disease, age 65–74 years, sex category) score; CHF, congestive heart failure; CKD, chronic kidney disease; CVA, cerebrovascular accident; DM, diabetes mellitus; HTN, hypertension; LA, left atrium; LVEF, left ventricular ejection fraction; OAC, oral anticoagulant.
NS, no statistically significant between-group difference (P >0.05; Pearson’s χ2-test or Fisher’s exact test [categorical data]; Student’s t-test or Wilcoxon rank–sum test [continuous data]).
Fifty-nine patients (48.8%) with AF progression and 62 (33.5%) without AF progression received rhythm-control therapy (P = 0.008). In patients aged <65 years, those with AF progression were significantly older, had a higher body mass index (BMI), and tended to have a higher frequency of hypertension, heart failure, vascular disease and chronic kidney disease, and higher CHA2DS2-VASc scores (P < 0.05). In patients aged ≥65 years, baseline characteristics did not differ significantly between those with or without AF progression, except for weight and BMI (P < 0.05). A comparison of echocardiography findings revealed that patients with AF progression had significantly larger left atria and lower left ventricular ejection fraction versus those without AF progression, regardless of age (P < 0.05). At the time of AF diagnosis, there were no differences in most received medications. However, antiplatelet therapy and β-blockers were used more often in patients with AF progression versus patients without AF progression patients aged <65 years (P < 0.05).
HRR and METs during TMT
Differences in TMT results between patients with and without AF progression, categorized according to age, are shown in Table 2. Patients aged <65 years with AF progression had lower METs and HRs after 1 min of rest (P < 0.05). In patients aged ≥65 years, there were no significant differences in METs, but peak and recovery HRs were significantly higher in the AF progression group (P < 0.05). Division of METs into tertiles revealed different frequency patterns according to age category (Figure 1). The younger group (aged <65 years) included a higher frequency of patients with high METs than the older group (aged ≥65 years). In patients aged <65 years, the AF progression group had a significantly lower percentage of patients with high METs (P = 0.049). In those aged ≥65 years, the number of patients with low METs was relatively high, but there were no statistically significant differences between patients with or without AF progression.
Treadmill exercise test parameters in 306 patients with AF grouped according to age and AF progression.

Data presented as mean ± SD or n (%).
AF atrial fibrillation; HR, heart rate; HRR, heart rate recovery; METs, metabolic equivalents.
NS, no statistically significant between-group difference (P > 0.05; Pearson’s χ2-test or Fisher’s exact test [categorical data]; Student’s t-test or Wilcoxon rank–sum test [continuous data]).

Percentage of 306 patients with (+) or without (−) AF progression grouped according to METs by tertile, showing: (top) patients aged <65 years (n = 228); and (bottom) patients aged ≥65 years (n = 78). P = 0.049, AF progression (−) versus AF progression (+) in patients aged <65 years (Fisher’s exact test). AF, atrial fibrillation; METs, metabolic equivalents.
HRV during 24-h Holter monitoring
Differences in time/frequency domain HRV components between patients with or without AF progression categorized by age are summarised in Table 3. Among patients aged <65 years, the rMSSD was higher in the AF progression group than in patients without AF progression (P = 0.04). In patients aged ≥65 years, the SDNN was higher in the AF progression group versus patients without AF progression (P = 0.032). The mean LF/HF ratio did not differ significantly by AF progression regardless of age, but when classified into tertiles according to absolute values, differences in distribution were observed (Figure 2). In patients aged ≥65 years, the low LF/HF ratio showed the highest frequency, and there was no statistically significant difference in distribution between patients with or without AF progression. However, in patients aged <65 years, the distribution frequency of LF/HF ratios was different according to AF progression (P = 0.039). The AF progression free group showed the highest percentage of patients with mid LF/HF ratios. In patients with AF progression, the lowest proportion was seen in the mid LF/HF ratio, and a U-shaped distribution was observed (Figure 2).
24-h Holter monitoring parameters in 306 patients with AF grouped according to age and AF progression.

Data presented as mean ± SD or n (%).
AF, atrial fibrillation; HF, high frequency; LF, low frequency; pNN50, percentage of interval differences of successive normal sinus to normal sinus intervals greater than 50 ms; rMSSD, root mean square of successive RR interval differences; SDNN, SD of normal sinus to normal sinus interval.
NS, no statistically significant between-group difference (P > 0.05; Pearson’s χ2-test or Fisher’s exact test [categorical data]; Student’s t-test or Wilcoxon rank–sum test [continuous data]).

Percentage of 306 patients with (+) or without (−) AF progression grouped according to LF/HF ratios by tertile, showing: (top) patients aged <65 years (n = 228); and (bottom) patients aged ≥65 years (n = 78). P = 0.039, AF progression (−) versus AF progression (+) in patients aged <65 years (Pearson’s χ2-test). AF, atrial fibrillation; LF, low frequency; HF, high frequency.
Independent risk factors of AF progression
Factors affecting AF progression were analysed by univariate and multivariate regression (Table 4). In patients aged <65 years, the mid LF/HF ratio, METs, and age significantly correlated with AF progression (P < 0.05). In patients aged ≥65 years, only HRR was associated with AF progression (P = 0.032): HRR ≥40 bpm was associated with low AF progression rates. Further analyses revealed that in patients aged <65 years, the rate of maintenance of normal sinus rhythm was significantly higher in the mid-LF/HF ratio group than in the low and high LF/HF tertiles (P = 0.041; Figure 3). However, there were no statistically significant differences between the LF/HF ratio tertiles in patients aged ≥65 years (Figure 3). A comparison of METs revealed that sinus rhythm was better maintained in patients with high METs (>10) versus METs ≤10 in those aged <65 years (P = 0.021). There were no statistically significant differences in maintenance of sinus rhythm between different METs in patients aged ≥65 years (Figure 4).
Logistic regression analysis for clinical predictors of AF progression in 306 patients with AF grouped according to age.
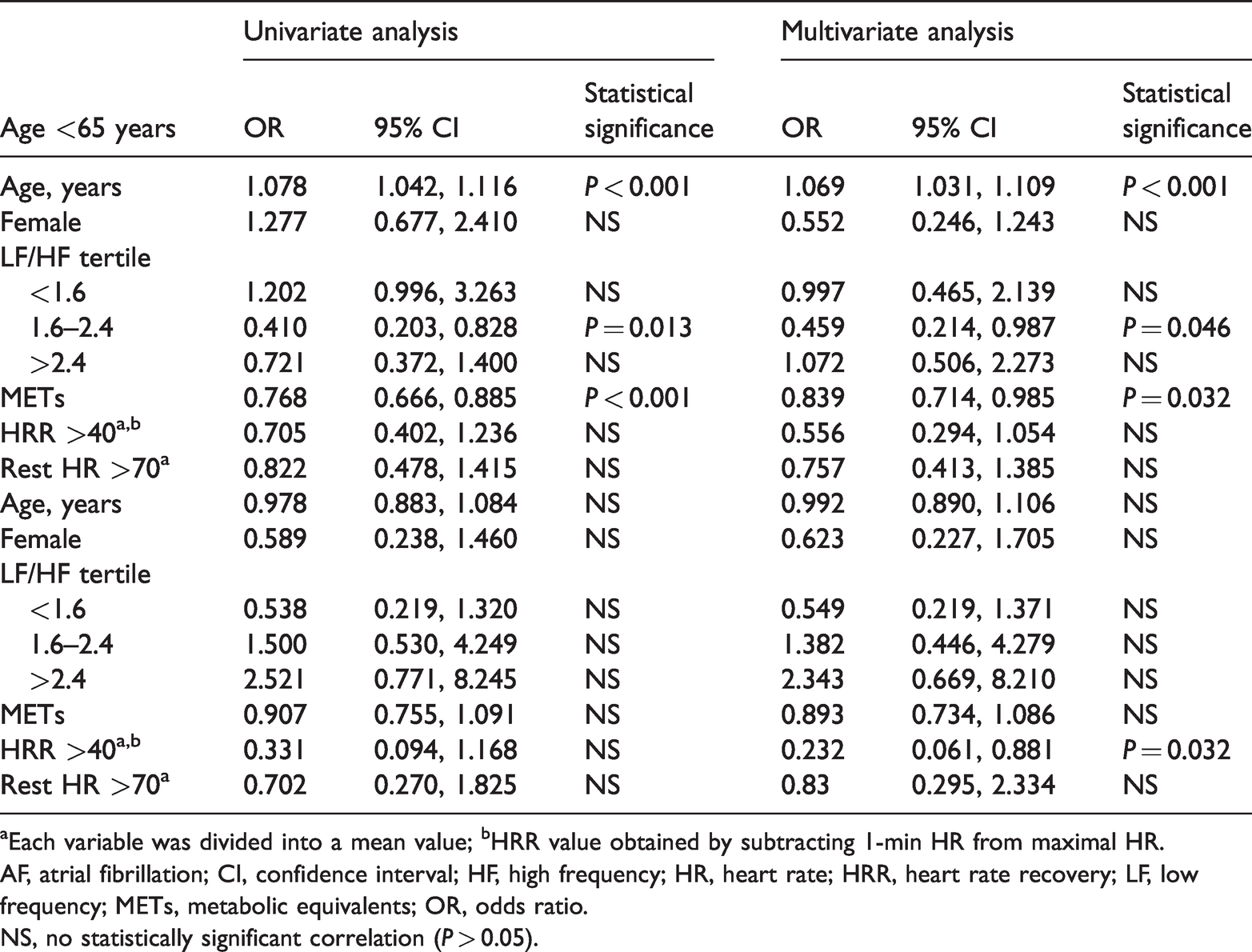
aEach variable was divided into a mean value; bHRR value obtained by subtracting 1-min HR from maximal HR.
AF, atrial fibrillation; CI, confidence interval; HF, high frequency; HR, heart rate; HRR, heart rate recovery; LF, low frequency; METs, metabolic equivalents; OR, odds ratio.
NS, no statistically significant correlation (P > 0.05).

Sinus rhythm maintenance in 306 patients with (+) or without (−) AF progression grouped according to LF/HF ratio, showing: (top) patients aged <65 years (n = 228); and (bottom) patients aged ≥65 years (n = 78). P = 0.041, LF/HF 1.60–2.40 versus other tertiles (Wilcoxon rank–sum test). AF, atrial fibrillation; LF, low frequency; HF, high frequency.

Sinus rhythm maintenance in 306 patients with (+) or without (−) AF progression grouped according to age and METs, showing: (top) patients aged <65 years (n = 228); and (bottom) patients aged ≥65 years (n = 78). P = 0.021, METs ≤10 versus METs >10 (Wilcoxon rank–sum test). AF, atrial fibrillation; METs, metabolic equivalents.
Discussion
The major findings of the present study are as follows: the mean LF/HF ratio and HRR did not differ significantly by AF progression regardless of age. When LF/HF ratio was divided into tertiles, in patients aged <65 years, the mid-range LF/HF ratio (1.60–2.40) was significantly associated with lower rates of AF progression and a longer maintenance of normal sinus rhythm. Below the age of 65 years, high METs and mid-range LF/HF ratios were related to lower rates of AF progression, while above the age of 65 years, a high HRR (>40 bpm) was associated with lower rates of AF progression.
Physical capacity and AF progression
A significant dose-response relationship has been shown between baseline cardiorespiratory fitness and a 20% reduction in the risk of AF recurrence for each MET increase in baseline cardiorespiratory fitness, 18 and physical activity is reported to be associated with a modestly reduced risk of AF in middle-aged women. 19 In addition, leisure-time exercise in older age is not shown to be associated with increased risk of AF. 20 In the current study, the relationship between METs and AF progression in patients aged <65 years may be related to the complex impact of physical activity on AF. Several small case–control studies have reported that long-term regular sports activity elevates the risk of AF in young and middle-aged men. Conversely, a prospective study of older men and women observed a reduced risk of AF with moderate-intensity physical activity, including leisure-time exercise and walking, but found no increase in risk with high-intensity physical activity.21–26 These findings imply that physical activity of different intensities may have age-dependent effects on the risk of later AF development. There are several possible mechanisms by which physical activity could influence the risk of AF. Exercise has positive effects on conventional cardiovascular risk factors, such as hypertension, diabetes, hypercholesterolemia, and obesity, and all of these effects also protect against AF. However, regular physical activity alters the balance of sympathetic and parasympathetic stimulation to the heart. Moreover, an increase in vagal tone can lead to a shorter atrial refractory period with greater dispersion and a higher risk of re-entrant rhythms, and an elevated vagal tone has been associated with AF onset. Moderate levels of physical activity seem to counterbalance the potentially adverse consequences of vigorous activity on atrial remodelling and electrophysiology. 19
Autonomic modulation and AF progression
When the association between resting HR and AF was examined in a restricted cubic spline model, an increased risk of AF was observed with decreasing HR. However, although not statistically significant, an upward trend was observed with increasing HR. 7 Lombardi et al. compared HRV before and after paroxysmal AF and observed a significant reduction in the LF/HF ratio. In most cases, predominant sympathetic and reduced vagal modulations were reflected by the presence of HR acceleration, predominance of the LF component, and an increased LF/HF ratio. Vagal predominance was detectable in about one third of the cases. 27 In the current study, patients aged <65 years showed different patterns of LF/HF ratios according to AF progression, and AF progression according to LF/HF ratio tertiles showed a U-shaped distribution, which supports the above results. HRV analysis can be performed on normal sinus rhythm. In patients without non-paroxysmal AF, measurement of HRV during sinus rhythm is recommended to evaluate autonomic function.
The pathophysiology of AF progression remains unclear, but the present results suggest that both sympathetic and parasympathetic dysregulation are important factors in AF progression. In patients aged ≥65 years, however, the LF/HF ratio was not associated with AF progression, but AF progression was associated with high peak and recovery HR. This may be interpreted as reduced regulation of HR due to parasympathetic disorder. Moreover, it was difficult to increase or decrease the HR during TMT. Therefore, this HR may not reflect the LF/HF ratio. In previous reports, age was the most important risk factor for AF.28,29 Imbalances in autonomic tone have been shown to facilitate the development of AF (particularly parasympathetic abnormalities), 30 and persistent AF (atrial sympathetic hyperinnervation) in patients with persistent AF. 31 Autonomic tone imbalance is also proposed to be an indicator of parasympathetic nervous system reactivation. Therefore, an abnormally slow rate of HRR after exercise may indicate a parasympathetic nervous system abnormality. 32 Increased age has been associated with increased mortality in response to exercise, partly due to its close association with chronotropic incompetence secondary to decreased physical fitness.33,34 Aging, similar to various cardiovascular diseases, increases sympathetic nervous system activity and decreases parasympathetic activity, which may contribute to age-related cardiac dysfunction and remodelling.35–38 This suggests that chronotropic incompetence is more important than METs in relatively older people.
Several studies have suggested the existence of a link between AF and the autonomic nervous system.4,39 Sympathetic and vagal mechanisms may facilitate arrhythmia initiation by reducing the atrial effective refractory period,40–42 and sympathetic activation may have an additional pro-arrhythmic role by increasing atrial automaticity. 41 The combined effects of sympathetic and parasympathetic stimulation are important factors in the initiation of AF. 43 For example, the inability to achieve an adequate HR response during exercise was shown to be independently associated with an increased risk of the development of AF. 7 In a study by Maddox et al., patients with an impaired HRR during exercise tolerance testing were found to be more likely to experience new-onset AF than patients without an impaired HRR. However, one of the study limitations was an inability to determine whether new-onset AF represented paroxysmal, persistent, or permanent AF. 44 The current study results suggest that autonomic dysfunction is associated with the progression of AF.
Study limitations
The results of the current study may be limited by several factors. First, the study was a relatively small retrospective analysis performed at a single centre. Larger prospective studies are required to evaluate the relationship between AF progression and HRR and HRV by age. Secondly, the study cohort was heterogeneous in terms of underlying heart disease and drug treatment. In particular, heart failure was not stratified or categorized by severity, and the AF substrate severity was not corrected.
Conclusion
The results of this study indicate that relatively younger age (<65 years), high physical capacity, and balanced autonomic nervous system regulation may be important predictors of AF progression. Higher HRR in elderly patients (≥65 years) was associated with lower rates of AF progression. Further larger size prospective studies are needed to confirm the present results.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by an Inha University Hospital Research Grant.
