Abstract
Objective
Acute Exacerbation Chronic Obstructive Pulmonary Disease (AECOPD) is associated with an acute worsening of respiratory symptoms that have effects on lung function, quality of life and health economic burden. In addition, the development of pulmonary infections is a common complication of Chronic Obstructive Pulmonary Disease (COPD). In the pathophysiology of AECOPD, interleukin (IL)-6 is a pleiotropic cytokine that can be produced by inflammatory and primary lung epithelial cells in response to a variety of different stimuli. We aim to investigate the correlation between serum cytokine levels and AECOPD with pulmonary infection.
Methods
37 AECOPD patients diagnosed with pulmonary infection and 33 patients diagnosed with AECOPD only were selected. All COPD patients were diagnosed according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria. Serum samples for C-reactive protein (CRP) and cytokines were obtained from the patients immediately after admission. Serum concentrations of cytokines were measured using a fluorescent bead immunoassay on a flow cytometer. Logistic regression was used to identify risk factors for AECOPD co-infection of the lungs.
Results
Serum characterization of our cohort showed patients with AECOPD and pulmonary infection had higher levels of IL-6 and IL-10 compared with the AECOPD group, and IL-6 was independently associated with AECOPD with pulmonary infection. ROC curve analysis showed that IL-6 was a useful predictor of the incidence of pulmonary infection in AECOPD patients.
Conclusions
Our findings highlight the role of IL-6 in the pathogenesis of AECOPD with pulmonary infection.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a chronic respiratory disease characterized by incompletely reversible and restricted airflow.1,2 Acute exacerbation of COPD (AECOPD) is associated with an acute worsening of respiratory symptoms that have effects on lung function, quality of life and health economic burden. 3 Infectious (bacteria or viruses) and environmental (air pollution and meteorological effect) are major causes of AECOPD. 4 In addition, development of pulmonary infections is also a prevalent complication of COPD. 5 Dysfunction of immune system and overproduction of cytokines due to infections, tobacco, inhalation of toxic particle and air pollution have been shown to play a principal role in the pathophysiology of COPD and AECOPD.6–8
Cytokines and chemokines are recognized as key actors in the response against viral and bacterial infections. 9 Interleukin (IL)-6 sputum levels are negatively associated with forced expiratory volume in the first second (FEV1) and forced vital capacity (FVC) after antibiotic treatment in bronchiectasis. IL-6 affects lung disease (specifically COPD), which may prove to be an important field for future research.2,10 In addition, Wei et al. found that AECOPD patients had higher levels of IL-2, IL-4, IL-10, IL-17, and IgE than those with stable COPD or controls. 8 However, the role of IL-6 in AECOPD with pulmonary infection remains to be elucidated.
Hence, the objective of this study was to further elucidate some biomarkers associated with clinical features in AECOPD patients with pulmonary infection. We sought to identify specific biomarkers related to pulmonary infections in AECOPD.
Materials and methods
Patients
A retrospective case-control trial was performed. From June 2018 to May 2020, A total of 70 patients (58 males and 12 females) were diagnosed with AECOPD, among these, 37 were diagnosed with a pulmonary infection and 33 were not. Our study was approved by the Medical Ethics Committee of the Zhejiang Xiaoshan Hospital (Approval NO.2018-006).
The inclusion criterion was a diagnosis of AECOPD and pulmonary infection. The diagnosis and severity of COPD were according to the criteria of the 2017 Global Initiative for Chronic Obstructive Lung Disease (GOLD). 11 AECOPD was defined as an acute event characterized by a worsening of cough and sputum symptoms, and resulting in the production of purulent sputum and aggravation of dyspnea. For patients with AECOPD associated pulmonary infection, imaging results showed new patchy infiltrates, lobar or segmental consolidation, ground-glass opacities, or interstitial changes, with or without pleural effusion. Exclusion criterion were a diagnosis of pulmonary tuberculosis, pulmonary edema and cancer, use of immunologic agents (e.g., glucocorticoid, cyclosporin, tacrolimus or mycophenolate mofetil) in the past 6 months, immune diseases (e.g., rheumatoid arthritis, systemic lupus erythematosus, ankylosing spondylitis, Crohn’s disease), and mental disorders. The AECOPD associated pulmonary infection group comprised 33 males and four females aged 49–98 years old, (79.11 ± 10.64) on average. According to the criteria of GOLD, there were 6 cases of GOLD grade 1, 11 cases of grade 2, 16 cases of grade 3 and 4 cases of grade 4. In AECOPD group, there were 25 males and eight females aged 62–90 years old, with a mean of (78.27 ± 8.06). There were 5 cases of GOLD grade 1, 9 cases of grade 2, 16 cases of grade 3 and 3 cases of grade 4. Patients were treated with the inhalation corticosteroid (budesonide, beclometasone dipropionate and fluticasone propionate) according to clinical symptoms.
Laboratory measurements
Fasting venous blood was collected and then centrifuged at 1000 g for 30 min to obtain serum samples. Serum samples (about 5 mL) for leukocytes, C-reactive protein (CRP) and cytokines were obtained from the patients immediately after their admission. Serum concentrations of cytokines (IL-2, IL-4, IL-6, IL-10, TNF-α and IFN-γ) were measured using a fluorescent bead immunoassay on a flow cytometer (BD bioscience, USA) according to the instructions. All samples were analyzed blinded in the laboratory.
Statistical analysis
SPSS software (SPSS Inc., Chicago, IL, USA) was used to analyze data. The results were presented as the mean (SD) for all variables with normal distribution and as median (range) when not normally distributed. Student’s t-tests for independent samples and the Mann-Whitney U test for non-normal distribution were used to analyze differences in measurements between AECOPD associated pulmonary infection group and AECOPD group. To identify factors in AECOPD with pulmonary infection patients, binary logistic regression and odds ratios (ORs) were applied and 95% confidence intervals (CI) were calculated. The predictive value of IL-6 and CRP for the incidence of pulmonary infection in AECOPD was evaluated by a receiver operating characteristic (ROC) curve. The area under the ROC curve (area under curve, AUC) was used to determine the overall accuracy of each predictor (AUC ≥0.5 was considered to have a diagnostic value, the larger the area, the larger the value). p < 0.05 was considered statistically significant in all these tests.
Results
Baseline clinical data
Comparison of baseline data between the two groups.

AECOPD, Acute exacerbation of chronic obstructive pulmonary disease; GOLD, Global initiative for chronic obstructive lung disease.
p-values comparing the AECOPD + pulmonary infection and AECOPD group.
Inflammatory markers levels were higher in AECOPD associated pulmonary infection group
Comparison of blood cell counts and CRP in the two groups.

AECOPD, Acute exacerbation of chronic obstructive pulmonary disease. WBC, White blood cell. CRP, C-reactive protein.
p-values comparing the AECOPD + pulmonary infection and AECOPD group, *p < .05 is considered statistical significant.
Comparison of cytokines in the two groups.

AECOPD, Acute exacerbation of chronic obstructive pulmonary disease; IL, interleukin; IFN-γ, interferon γ; TNF-α, tumor necrosis factor α.
p-values comparing the AECOPD + pulmonary infection and AECOPD group, *p < .05 is considered statistical significant.
IL-6 is independently associated with pulmonary infection in AECOPD patients
Multivariate logistic regression analysis of factors.
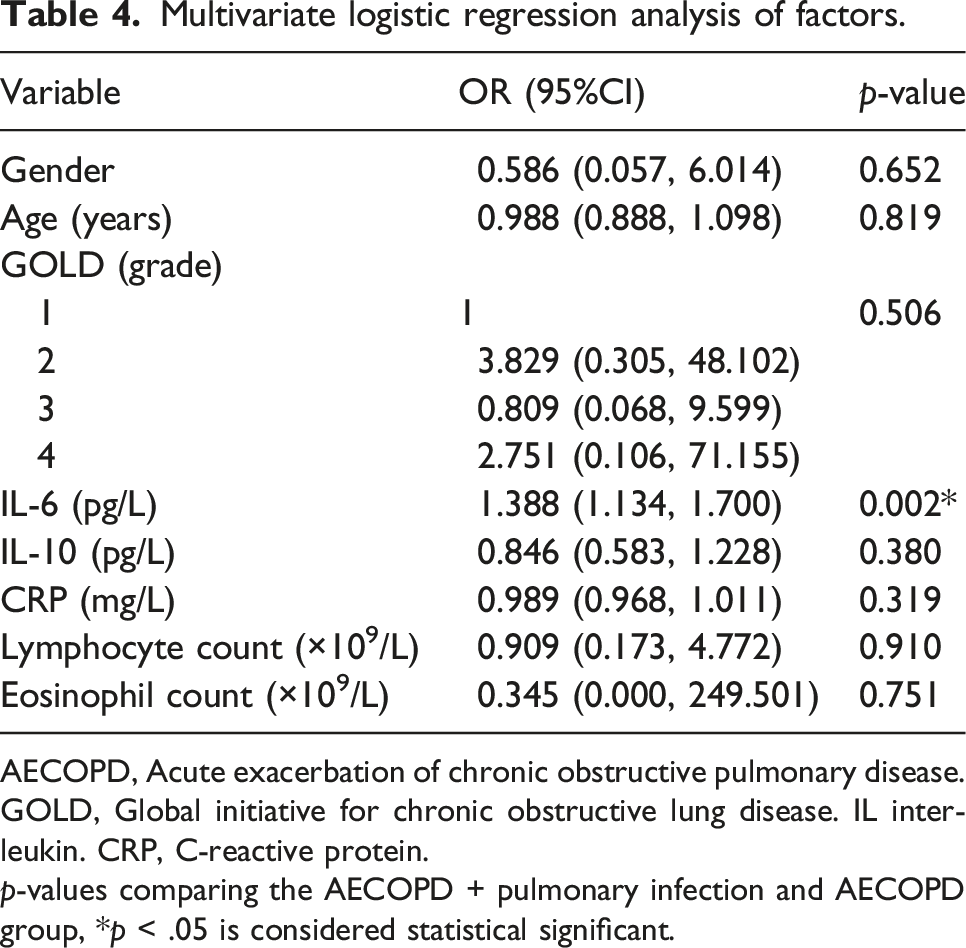
AECOPD, Acute exacerbation of chronic obstructive pulmonary disease. GOLD, Global initiative for chronic obstructive lung disease. IL interleukin. CRP, C-reactive protein.
p-values comparing the AECOPD + pulmonary infection and AECOPD group, *p < .05 is considered statistical significant.
Predictors of ROC curves for assessing the incidence of pulmonary infections in AECOPD
The results showed that CRP and IL-6 could be used to predict the incidence of pulmonary infection in AECOPD patients (p < .01). The AUC for IL-6 to predict the incidence of pulmonary infection in AECOPD was 0.934 with a standard error 0.029 (p < .001 compared with 0.5 which was set as a standard comparison for AUC). The AUC for CRP to predict the incidence of pulmonary infection in AECOPD was 0.781 with a standard error 0.058 (p < .001 compared with 0.5) (Figure 1). ROC curve analyze of IL-6 and CRP for predicting pulmonary infection in AECOPD patients.
Discussion
Chronic obstructive pulmonary disease (COPD) remains a global concern since its high prevalence, morbidity and mortality pose a challenge to health-care systems.3,12 According to the World Health Organization (WHO), COPD, which results in more than 120,000 deaths each year, is the third leading cause of global mortality, and AECOPD is the major cause of significantly increased mortality. COPD has now been recognized as a complex multicomponent disease characterized by chronic systemic inflammation, often coexisting with comorbidities. Pulmonary infection is one of the most common comorbidities associated with COPD. 12
Cytokines play a critical role in the initiation and propagation of inflammation in COPD. The pathophysiology of AECOPD is associated with increased numbers of goblet cells, mucus gland hyperplasia, fibrosis and pulmonary emphysema. 13 However, little information is available on how pulmonary infection affects the host immune responses in patients with AECOPD. Therefore, it is important to screen the biomarkers in AECOPD patients with pulmonary infections. In this study, we evaluated the association between pulmonary infection and the levels of 6 cytokines in 70 patients hospitalized with AECOPD.
C-reactive protein is increased in all components of the chronic systemic inflammatory syndrome and may serve as a precursor biomarker for all chronic diseases. 14 Thomas et al. reported elevated that CRP is associated with a 2- to 4-fold increased risk of pneumonia. 15 COPD patients have increased numbers of T and B lymphocytes, which are further increased during acute exacerbations or bacterial or viral infections. 16 In our study, the CRP concentrations and lymphocyte counts in AECOPD associated pulmonary infection group were statistically significantly increased compared to AECOPD group, suggesting that pulmonary infection increased inflammatory expression in AECOPD patients.
IL-10, a classical stimulator of B lymphocytes, suppresses the production of TNF-α, IL-2, and IFN-γ by Th1 cells through inhibiting the expression of nuclear factor-κB, a key inflammatory transcription factor. 17 Wei et al. reported that IL-10 levels were significantly higher in the AECOPD group than in stable COPD group. 8 Our study found that the levels of cytokine concentrations of IL-10 were significantly elevated in AECOPD associated pulmonary infection group compared to the AECOPD group.
IL-6 is a pleiotropic cytokine that can be produced by both inflammatory and primary lung epithelial cells in response to a variety of stimuli. 18 A previous study showed that 70% of patients with COPD experienced some components of systemic inflammation, while 16% had persistent inflammation by measuring C-reactive protein, IL-6, CXCL8, fibrinogen, TNF-α, and leukocytes. 19 One study found that in patients with COPD exacerbations, viral infection was directly associated with higher systemic levels of IL-6 and TNF-α, which are also known to contribute to inflammation-mediated tissue damage. 20 We demonstrated that plasma IL-6 at baseline was associated with pulmonary infection in AECOPD and this association was independent of gender, age, IL-10, CRP, lymphocyte count and eosinophil count, which were also taken into account in the model.
Our study has three limitations. First, the sample size was small and sample size calculation analysis was not performed. The other is the absence of cytokine profiles in respiratory samples, which may better mirror the pathogenic events taking place in the lungs of these patients. Finally, heart failure was not included in the exclusion criteria in our study. Therefore, further studies with larger sample size and respiratory samples are required to confirm our results.
Conclusions
In this study, we investigated the correlation between cytokine serum levels and AECOPD with pulmonary infection. Both IL-6 and IL-10 were elevated in patients with AECOPD with pulmonary infection compared with the AECOPD group, and IL-6 was independently associated with AECOPD with pulmonary infection. This suggests that IL-6 may play a role in the pathogenesis of AECOPD with pulmonary infection.
Footnotes
Acknowledgements
Thanks to all data collectors for their willingness during data collection.
Author contributions
Lei-lei Tang and Ling-di Zhang conceived the study, performed the majority of experiments, and writing; Xuan Mei and Jia-wen Yu participated in the collection of the human samples and made critical revisions; Guo-jun Jiang made critical revisions and provided approval of the final version of the manuscript to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medical Health Science and Technology Funding of Hangzhou (Grant No. 20190551), Science and Technology Plan Project of Hangzhou (Grant No. 20201231Y118) and Major Science and Technology Plan Project of Xiaoshan (Grant No. 2020310), The Natural Science Foundation of Zhejiang (Grants No. LY18H310009, 2018KY653, 2020377787).
Ethics approval
Ethical approval for this study was obtained from *Ethics Committee of Zhejiang Xiaoshan Hospital, Hangzhou, Zhejiang, China (APPROVAL NUMBER/ID 2018-006).
Informed consent
Written informed consent was obtained from all subjects before the study.
Consent to participate
Patients who participated in this research, signed the informed consent and had complete clinical data. Signed written informed consents were obtained from the patients and/or guardians.
