Abstract
Objective
Sodium-glucose cotransporter 2 (SGLT2) inhibitors have been studied for their effects on cardiovascular disease, cognitive impairment, dementia, Parkinson’s disease, and cerebrovascular disease, with evidence suggesting a reduced risk of mortality. This study aimed to examine the association between SGLT2 inhibitors and cerebrovascular event recurrence.
Methods
The cohort was extracted from the Korean National Health Insurance Service Database, including individuals aged 19 or older diagnosed with type 2 diabetes. SGLT2 inhibitor users were compared with metformin users using 1:4 propensity score matching.
Results
Among study subjects, 1563 (7.8%) experienced recurrent cerebrovascular events. SGLT2 inhibitor users showed lower recurrence of cerebral infarction compared to metformin users (adjusted hazard ratio=0.84, p=0.010). Among the types of cerebrovascular diseases, ischemic stroke was associated with a significantly lower risk of recurrence in SGLT2 inhibitor users than metformin users (aHR=0.70, p=0.020). In the SGLT2 inhibitor group, concurrent use of DPP4 inhibitors was associated with a reduced risk of cerebrovascular event recurrence (aHR=0.69, p=0.011).
Conclusion
SGLT2 inhibitor use was associated with a significantly lower risk of recurrent cerebral infarction compared to metformin, suggesting a potential role in the secondary prevention of cerebrovascular events in patients with type 2 diabetes.
Key messages
• SGLT2 inhibitors have been associated with a reduced risk of cardiovascular mortality. • This study found that SGLT2 inhibitors were significantly associated with a lower risk of recurrent cerebral infarctions compared to metformin in patients with type 2 diabetes. • According to our findings, SGLT2 inhibitors may have protective effects in addition to primary prevention, potentially providing further benefits to secondary prevention of cerebrovascular diseases. • The concurrent use of DPP4 inhibitors was associated with a reduced risk of recurrence of cerebrovascular events in the SGLT2 inhibitor group.
Introduction
Patients with type 2 diabetes have nearly double the risk of stroke compared to those without diabetes, and the condition is estimated to contribute to over 8% of first ischemic strokes. 1 Consequently, stroke prevention in this population is of paramount importance. The potential of glucose-lowering medications to prevent stroke remains unclear, with some agents, such as sulfonylureas and rosiglitazone, potentially increasing the risk of stroke and cardiovascular disease.2,3 The cardiovascular impact of dipeptidyl peptidase-4 (DPP-4) inhibitors varies, with saxagliptin associated with an increased risk of heart failure, whereas sitagliptin and linagliptin demonstrate a more neutral cardiovascular profile.4,5 In contrast, certain antidiabetic medications, including metformin, pioglitazone, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and glucagon-like peptide-1 (GLP-1) receptor agonists, have demonstrated cardiovascular benefits, offering not only improved glucose control but also a reduced risk of stroke.6,7
Since its 1998 UKPDS-based recommendation as first-line therapy, metformin has remained the standard of care for T2DM management. 8 Following the 2008 FDA mandate for cardiovascular safety, subsequent cardiovascular outcome trials (CVOTs) for newer classes, such as SGLT2 inhibitors, have consistently demonstrated cardiovascular protection. Notably, these benefits persist regardless of baseline or concomitant metformin use.9–13
SGLT2 inhibitors target proteins in the renal proximal tubules to reduce glucose reabsorption and promote urinary excretion. Beyond their glucose-lowering effects, SGLT2 inhibitors have demonstrated significant cardiovascular benefits, including reduced vascular inflammation, oxidative stress, and endothelial dysfunction. These mechanisms contribute to their anti-atherosclerotic effects, ultimately reducing cardiovascular risk in patients with T2DM. 14 All FDA-approved SGLT2 inhibitors are indicated for improving glycemic control in adults with T2DM, with additional indications for reducing cardiovascular death and hospitalization in heart failure and for slowing kidney disease progression. 15 Previous studies primarily evaluated SGLT2 inhibitors in patients concurrently using metformin with composite outcome of interest including myocardial infarction, coronary syndromes, stroke, heart failure, and cardiovascular death. Although current guidelines recommend SGLT2 inhibitors for patients with established atherosclerotic cardiovascular disease, evidence specifically addressing recurrent cerebrovascular events remains limited. 16 The objective of this study is to investigate the association between SGLT2 inhibitor use and the recurrence of cerebrovascular disease in patients with T2DM, comparing its effects to metformin.
Methods
Data sources and study population
This population based cohort study was conducted using Korean National Health Insurance Service Cohort Database. The database includes individual beneficiary and healthcare service information including diagnosis, procedures, and prescriptions of nearly 97% of Korean population. Diagnoses have been coded using the 10th revision of the International Classification of Diseases (ICD-10) and medications have been coded using the national procedure-coding system and the Anatomically Therapeutic Chemical (ATC) classification system. 17 (Supplementary Tables 1 and 2).
Patients were eligible for inclusion if they were aged 19 years or older, diagnosed with type 2 diabetes (ICD-10 E11-E14), and had been prescribed antidiabetic medications. Patients were excluded if they had type 1 diabetes (E10). Cerebrovascular events were defined by one or more claims of stroke, cerebral infarction or other cerebrovascular disease and related syndromes.
The study design was an active-comparator design, with metformin as the comparator. Metformin had typically been used as first line agent with positive results on cerebrovascular outcomes, although recent CVOTs trials might be interpreted as a limited benefit.18,19 The study period for this analysis started in 2013 when SGLT2 inhibitors became available.
This research was approved by the Chungbuk University Institutional Review Board (IRB) (CBNU-202302-HR-0023). The data in this study was used only for research purposes and coded to prevent personal identification.
Study designs
The index date was defined as the date of initial prescription of SGLT2 inhibitor or metformin. Patients who used antidiabetic medications more than once were selected. Patients were followed from the index date until the end of data collection or the outcome event or death. In case of death, the follow-up was censored. This study included first-ever event patients with cerebrovascular disease with the minimum period of 21 days between the start of antidiabetic medication and the outcome. 20
The primary outcome was the recurrence of cerebrovascular event (two or more episodes) identified using the codes of treatment and procedure related to cerebrovascular disease such as catheter, computed tomography (CT), magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). Those medical service codes were extracted from the health insurance activity classification fee code at the Health Insurance Review & Assessment Service. As a surrogate variable for the severity of cerebrovascular disease, the use of a ventilator, including intubation (which indicates severe cases), was used. 21
Statistical analysis
The cohort groups were matched by propensity score at a ratio of 1:4 with age, sex, index year, Charlson comorbidity index (CCI), and duration of antidiabetic medication use (greedy mode (k=4), and caliper at 0.2). Among these characteristics, CCI two years before the index date and co-medications and antidiabetic medications 120 days before the cohort entry were considered. 22 The independent t-test and chi-square test were used. For the study outcomes, the incidence rate was calculated. Cox’s proportional hazard regression analysis was used to estimate hazard ratios. Cramer’s V was calculated to assess the effect size between categorical variables and the association was considered relatively strong with a Cramer’s V>0.40. 23 Statistical significance was considered with a p-value<0.05. Statistical analysis was performed using SAS Enterprise Guide 8.3.
Results
From the database (n=1,137,861), 332,905 patients were selected who were diagnosed with T2DM, and to identify newly diagnosed diabetes patients, those with a claim history of diabetes diagnosis or diabetes medication from 2002 to 2003 were excluded. Patients who were diagnosed gestational diabetes (ICD-10 O24) or type 1 diabetes (E10) (n=31,418), under 19 years of age (n=6,801), using both SGLT2 inhibitor and metformin (n=4,968), and having overlapped administration period (n=151) were excluded. Since the SGLT2 inhibitors were approved after 2013 by Korea Ministry of Food and Drug Safety, subjects before 2013 were excluded (n=22,446). There were 1,322 SGLT2 inhibitors users and 5,278 metformin users after 1:4 propensity score matching (Figure 1). After matching, all baseline characteristics in the cohorts were well balanced, with standardized mean differences <0.10. Flowchart of patient selection.
Baseline characteristics.

Co-medications used among study subjects.

The total number of patients who experienced at least one cerebrovascular event after the index date was 1,608, with 1,258 in the metformin group and 350 in the SGLT2 group. The recurrence of cerebrovascular event occurred in 1,240 (23.5%) patients in metformin group and 300 (22.7%) patients in SGLT2 inhibitor group (p-value=0.010). Severe cerebrovascular events occurred in 29 patients (1.7%), including 23 in the metformin group and 5 in the SGLT2 inhibitor group.
Multivariable analysis was performed after checking the covariates to include variables that would not confound the other variables’ relationship with the outcome (Cramer’s V < 0.4). There were noticeable relationship between asthma and chronic lung disease (Cramer’s V=0.575), hypertension and angiotensin II receptor blocker (ARB) (Cramer’s V=0.658), and potassium sparing or loop diuretics and acid-suppressing agents other than proton pump inhibitors and histamine-2 receptor blockers (Cramer’s V=0.409). Variables with the lowest partial correlation with the dependent variable were initially considered for removal; as a result, asthma, ARB, potassium sparing or loop diuretics were excluded.
Multivariable analysis by types of cerebrovascular disease between SGLT2 Inhibitors and metformin.

a*Adjusted for gender, age, severity of cerebrovascular disease, concurrent medication use, comorbidities, and co-medication.
The hazard ratio was calculated using the metformin group as the reference group.
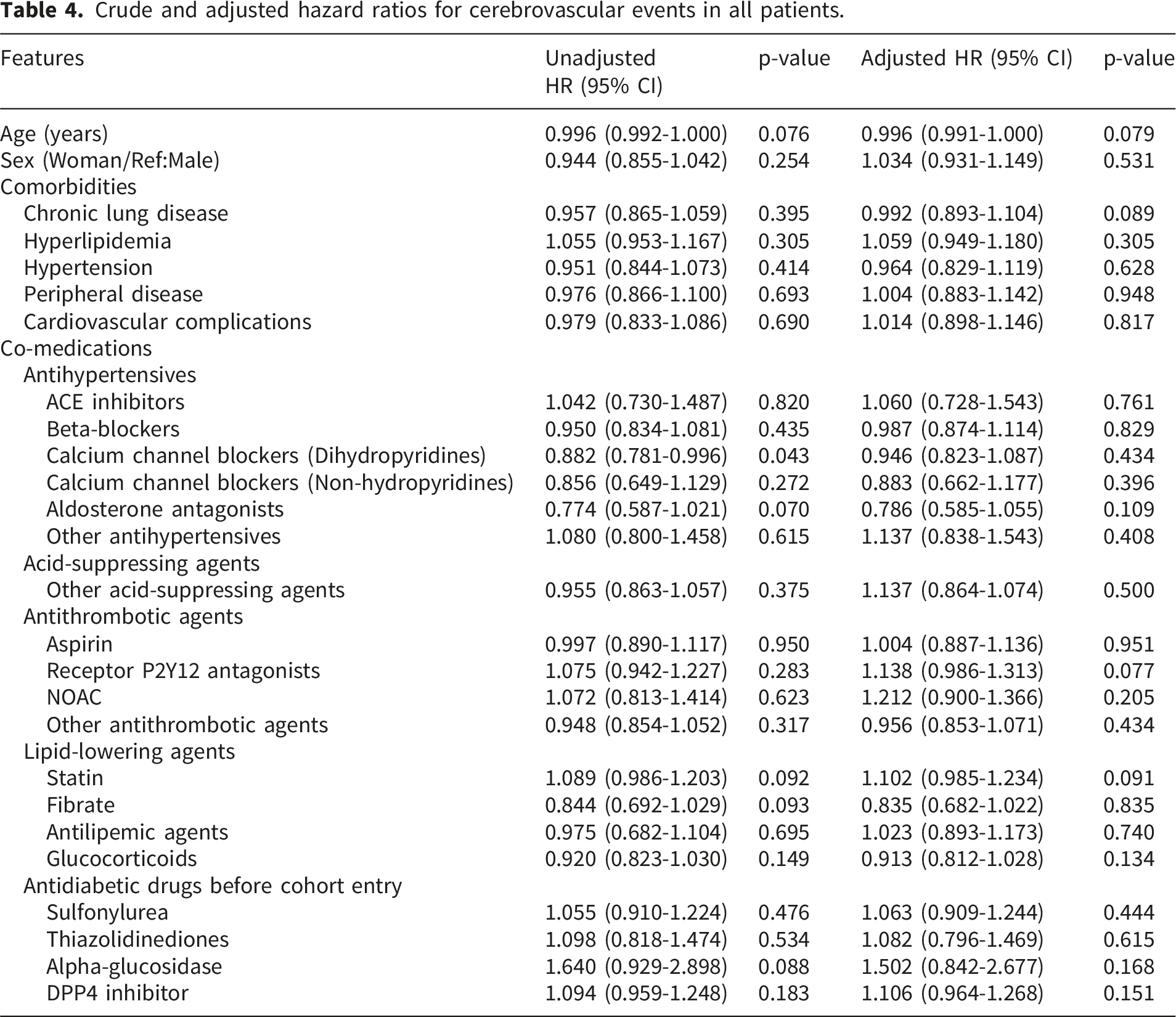
Crude and adjusted hazard ratios for cerebrovascular events in all patients.
Discussion
Our study investigated the impact of SGLT2 inhibitors compared to metformin on the recurrence of cerebrovascular events in patients with T2DM. After rigorous selection and propensity score matching, our findings suggest that SGLT2 inhibitor use is associated with a significantly lower risk of recurrent cerebrovascular events compared to metformin. This result highlights the potential cerebrovascular benefits of SGLT2 inhibitors among T2DM with established cerebrovascular disease.
Our study supports previous studies indicating the cerebrovascular protective effects of SGLT2 inhibitors.24–26 Proposed mechanisms include improvements in endothelial function, reductions in arterial stiffness and inflammation, as well as favorable effects on blood pressure and body weight, all of which contribute to cerebrovascular risk reduction.27–30 These pleiotropic effects support the role of SGLT2 inhibitors in cardiovascular protection, particularly in the secondary prevention of cerebrovascular disease.
Experimental studies further suggest potential atheroprotective and neuroprotective mechanisms of SGLT2 inhibitors, including reduced atherosclerotic lesion formation, attenuation of platelet–leukocyte aggregation, and induction of ischemic tolerance in cerebral microvascular cells independent of glucose lowering.31,32 Together, these findings indicate that SGLT2 inhibitors may modulate cerebral vascular function and reduce susceptibility to ischemic injury.
Large cardiovascular outcome trials, such as EMPA-REG OUTCOME and the integrated CANVAS and CANVAS-R analyses, have demonstrated significant reductions in major adverse cardiovascular events in patients with T2DM and established cardiovascular disease.11,33 While these trials primarily focused on composite cardiovascular outcomes, our study extends existing evidence by specifically evaluating recurrent cerebrovascular events across a broad clinical spectrum, including severe cases, thereby providing a more comprehensive assessment of cerebrovascular protection associated with SGLT2 inhibitor use.
Our analysis also identified significant interactions between DPP4 inhibitors and cerebrovascular event recurrence. In the SGLT2 inhibitor group, concomitant use of DPP4 inhibitors was associated with a lower risk of recurrence (aHR 0.69, 95% CI 0.52-0.92). Conversely, in the metformin group, DPP4 inhibitor use was linked to a higher risk of cerebrovascular event recurrence (aHR 1.26, 95% CI 1.08-1.48, p=0.011). While DPP4 inhibitors have been shown to be cardiovascularly safe in major randomized controlled trials, they have generally yielded neutral results regarding stroke reduction, in contrast to the superior outcomes observed with SGLT2 inhibitors or GLP-1 receptor agonists.34,35 Despite these neutral findings in monotherapy trials, our results suggest that the combination of SGLT2 inhibitors and DPP4 inhibitors may exert a synergistic effect on the cerebrovascular system, potentially through complementary pathways involving both hemodynamic stability and endothelial protection.
Similarly, the higher recurrence risk observed in patients receiving P2Y12 antagonists or NOACs within the metformin group likely underscores a higher baseline cardiovascular risk in these individuals, rather than a lack of efficacy of these antithrombotic agents. Interestingly, this trend was less pronounced in the SGLT2 inhibitor group, suggesting that SGLT2 inhibitors might provide a more robust protective backdrop in high-risk secondary prevention settings.
In the present study, patients in the SGLT2 inhibitor group more frequently received lipid-lowering and blood pressure–lowering therapies compared with those in the metformin group. These differences likely reflect a higher baseline cardiovascular risk and more intensive secondary prevention strategies in patients selected for SGLT2 inhibitor therapy. However, in multivariable analyses, neither antihypertensive agents nor lipid-lowering therapies were independently associated with recurrent cerebrovascular events. This suggests that the observed reduction in recurrence risk among SGLT2 inhibitor users is unlikely to be solely explained by differences in concomitant cardiovascular medications. Rather, the benefit appears to persist after adjustment for these background therapies, supporting a potential independent cerebrovascular protective effect of SGLT2 inhibitors beyond conventional risk factor management.
The strengths of this study include the large-scale real-world data analysis, stringent patient selection criteria, and robust statistical adjustments to minimize confounding. However, several limitations should be acknowledged. First, as this was a retrospective observational study, causality cannot be definitively established. Although propensity score matching was applied, residual confounding may still exist. Second, treatment selection in real-world clinical practice is influenced by patients’ overall health status, comorbidity burden, and perceived cardiovascular risk, factors that may not be fully captured in the database. In particular, patients receiving SGLT2 inhibitors were more frequently treated with lipid-lowering and blood pressure–lowering therapies, reflecting more intensive secondary prevention strategies. However, these concomitant medications were not independently associated with recurrent cerebrovascular events in adjusted analyses, suggesting that the observed risk reduction associated with SGLT2 inhibitor use is unlikely to be fully explained by differences in background cardiovascular therapies alone. Third, information on medication adherence, lifestyle factors, laboratory parameters, and stroke severity was not available, which may have influenced outcomes. Lastly, the study population was limited to patients in Korea, potentially limiting the generalizability of our findings to other populations with different genetic backgrounds and healthcare systems.
In conclusion, our findings suggest that SGLT2 inhibitors may offer a protective effect against the recurrence of cerebrovascular events in patients with T2DM compared to metformin. Clinical implications of these findings highlight the potential for SGLT2 inhibitors to be considered as a primary therapeutic strategy in T2DM patients at elevated risk for recurrent cerebrovascular events. Further prospective studies and randomized controlled trials are warranted to validate our findings and explore the underlying mechanisms driving these associations.
Supplemental material
Supplemental Material - Association between SGLT2 inhibitors and recurrence of cerebrovascular events in patients with type 2 diabetes: A population-based cohort study
Supplemental Material for Association between SGLT2 inhibitors and recurrence of cerebrovascular events in patients with type 2 diabetes: A population-based cohort study by Mee Hyeon Kim, Jin Yeon Gil, Kyung Hyun Min, Jun Hyeob Kim, Jun Hyuk Park, Sunny Park, Ji Min Han, Kyung Eun Lee in Diabetes & Vascular Disease Research.
Footnotes
Author contributions
All authors have made substantial contributions to the conception and design of the study. All authors have been involved in drafting and revising the manuscript. All authors read and approved the final manuscript. Lee KE managed and coordinated research activity planning and execution. Kim MH wrote original draft and performed analysis. Gil JY, Kim JH, Min KH, and Park JH performed data curation and analysis. Han JM and Park S validated results and reviewed manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (RS-2025-16069428).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the National Health Insurance Sharing Service of South Korea but restrictions apply to the availability of these data because of domestic laws and regulations that prohibit the distribution or release of an individual’s data to the public, and so are not publicly available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
