Abstract
Sodium–glucose cotransporter 2 inhibitors are antihyperglycaemic medications with an emerging evidence base for cardiovascular and kidney disease risk reduction. Sodium–glucose cotransporter 2 inhibitors medications lower plasma glucose by inhibiting glucose reabsorption in the proximal tubule of the kidney independent of insulin. Furthermore, they reduce intraglomerular pressure by restoring tubuloglomerular feedback. Large cardiovascular outcome trials of both empagliflozin and canagliflozin have consistently shown beneficial kidney effects that go beyond glycaemic control, such as reducing risk for incident nephropathy and progression of chronic kidney disease. The mechanisms by which sodium–glucose cotransporter 2 inhibitors improve kidney outcomes are not clear.
Proposed hypotheses underpinning the kidney benefits include kidney-specific effects such as decreased intraglomerular pressure, activation of angiotensin-(1-7) and the Mas receptor leading to decreased inflammation, decrease in overall kidney oxygen consumption, rise in erythropoietin levels, inhibition of the renal sodium–hydrogen exchanger and secondary kidney effects related to improvements in HbA1c and blood pressure.
This review will focus on describing the mechanisms of action of sodium–glucose cotransporter 2 inhibitors in the kidney, clinical efficacy data on their use in patients with chronic kidney disease, postulated physiologic underpinnings of kidney protection observed with sodium–glucose cotransporter 2 inhibitors and the promise and potential pitfalls for their use in patients with chronic kidney disease.
Keywords
Introduction
Sodium–glucose cotransporter 2 inhibitors (SGLT2i) are antihyperglycemic medications with effects that extend well beyond glycaemic control. Canagliflozin, dapagliflozin, and empagliflozin are approved by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for use in patients with type 2 diabetes mellitus (T2DM), and ertugliflozin is in Phase IIIb of clinical investigation, as is sotagliflozin – a dual SGLT 1 and 2 inhibitors. As the name implies, this class of medications works by targeting Na⁺/glucose cotransporter 2 located in the S1 segment of the proximal tubule, inhibiting reabsorption of filtered glucose and thereby increasing glucosuria and lowering plasma glucose independently of insulin. Pooled data from several phase II/IIIa studies have shown that canagliflozin, dapagliflozin and empagliflozin lower glycated haemoglobin (HbA1c) when used as a monotherapy or in combination with metformin, sulfonylureas, inhibitors of dipeptidyl peptidase 4 (DPP4i), glucagon-like peptide-1 receptor agonists (GLP-1RA) or insulin.1–5 These medications are associated with negligible risk of hypoglycaemia, except for small incremental risks when used in combination with sulfonylureas and insulin.2,6–8 In addition, phase III efficacy and safety trials have shown significant decreases in body weight2,6,9,10 and systolic blood pressure (SBP)6,11 at the expense of a small increase in risk of developing urinary tract infections and a modest incremental risk for genital mycotic infections in patients using these medications.2,12,13 In addition, canagliflozin was shown in the CANVAS Programme to double the risk of lower limb amputation when compared with placebo, a finding not yet amply elucidated. Similarly, a numerical imbalance in rates of bladder cancer between treatment groups during the new drug application to the FDA for dapagliflozin was noted and this issue is being prospectively evaluated. The CREDENCE (canagliflozin, NCT02065791) and DECLARE (dapagliflozin, NCT01730534) trials are expected to help clarify these safety concerns.
Cardiovascular (CV) outcome trials of both empagliflozin and canagliflozin have consistently shown beneficial effects that go beyond glycaemic control. The EMPA REG OUTCOME trial was a randomized, controlled clinical trial comparing the effect of empagliflozin versus placebo on CV outcomes in patients with established atherosclerotic CV disease, demonstrating superiority of empagliflozin. 14 Empagliflozin also significantly reduced the risk for the composite outcomes of CV mortality, all-cause mortality and hospitalizations for heart failure.
In consonance, results of the CANVAS and CANVAS-R trials have been published in combination as the CANVAS Programme, 15 pooling data from over 10,000 participants randomized to canagliflozin or placebo, demonstrating superiority of canagliflozin for major adverse CV events (Table 1).
Characteristics of outcome trials with kidney endpoints involving SGLT2 inhibitors.
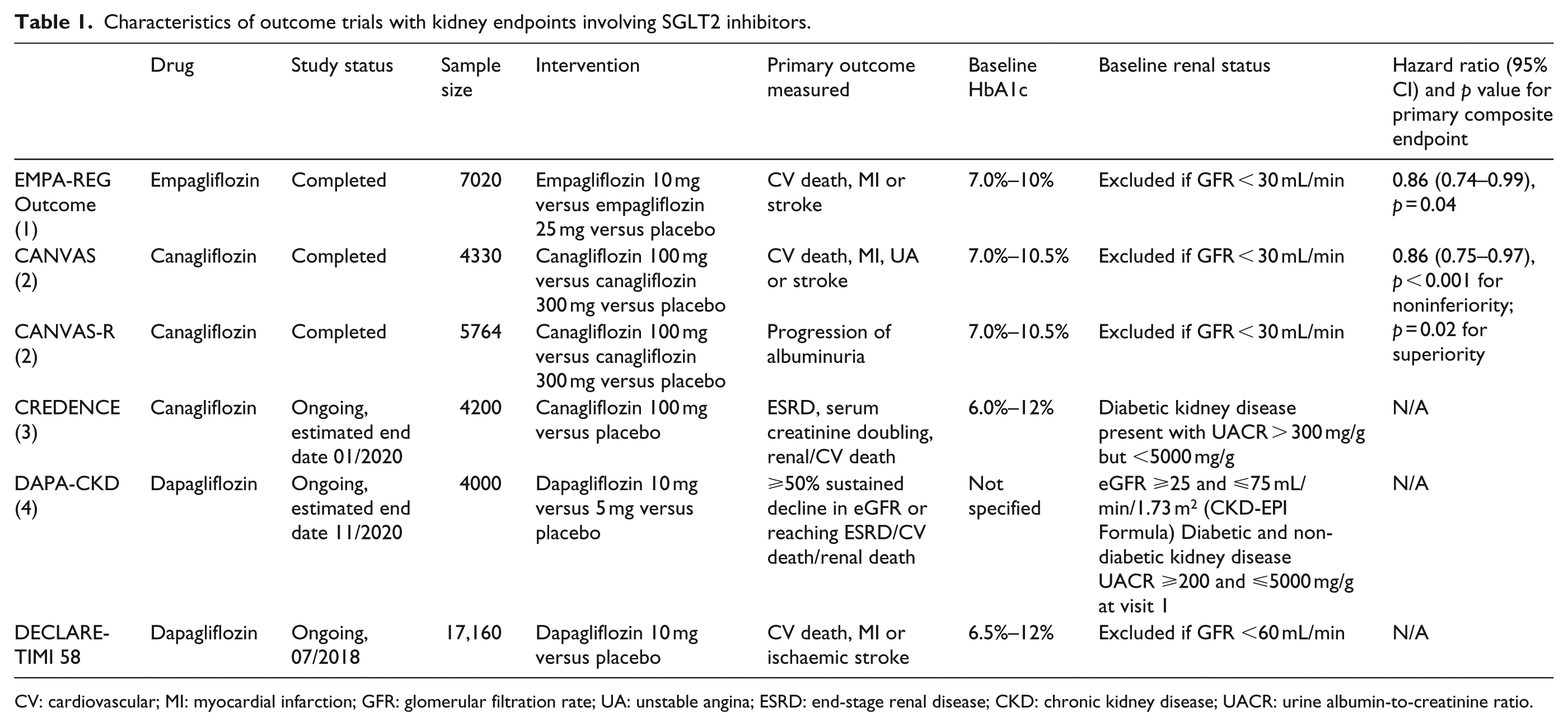
CV: cardiovascular; MI: myocardial infarction; GFR: glomerular filtration rate; UA: unstable angina; ESRD: end-stage renal disease; CKD: chronic kidney disease; UACR: urine albumin-to-creatinine ratio.
Less is known about the long-term kidney effects of SGLT2i. Below, we focus on describing the mechanisms of action of SGLT2i in the kidney, clinical efficacy data on their use in patients with chronic kidney disease (CKD), postulated physiologic underpinnings for kidney protection observed with SGLT2i and potential and pitfalls for their use in patients with CKD.
Kidney effects of SGLT2i medications: observations from large-scale outcome trials
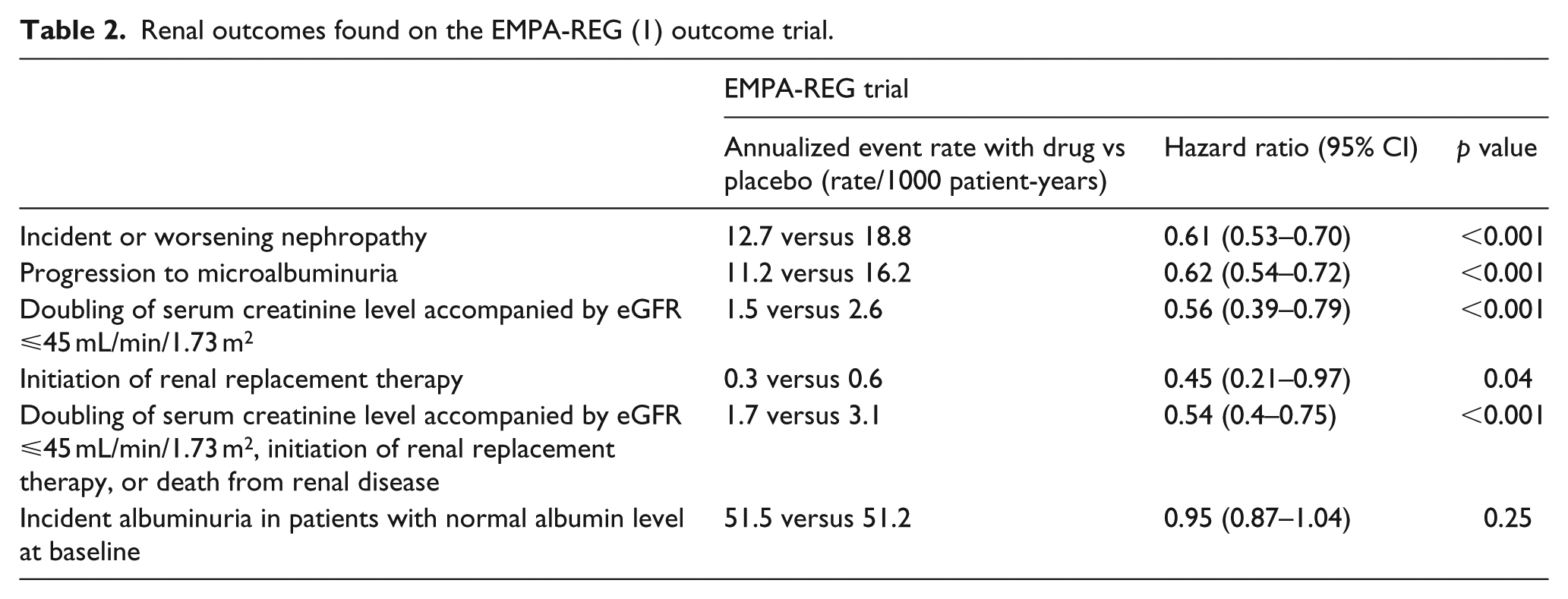
The long-term effects of empagliflozin on kidney outcomes were part of secondary prespecified analyses of the EMPA REG OUTCOME trial. 16 Patients with T2DM and estimated glomerular filtration rate (eGFR) of at least 30 mL/min/1.73 m2 were randomized to receive either empagliflozin 10 mg, 25 mg or placebo once daily. The measured kidney outcomes included incident albuminuria and incident or worsening nephropathy, which was defined as progression to incident microalbuminuria, doubling of serum creatinine, initiation of renal-replacement therapy or death from kidney disease. The authors found a 39% lower risk of nephropathy incidence and progression with empagliflozin (both arms pooled) versus placebo [hazard ratio (HR): 0.61; 95% confidence interval (CI): 0.53 to 0.70; p < 0.001] and a 46% reduction in the composite kidney outcome of doubling of serum creatinine, initiation of renal replacement therapy or death due to kidney disease (HR: 0.54; 95% CI: 0.40 to 0.75; p < 0.001). Patients in the empagliflozin group also had a significantly lower risk of progression from micro- to macroalbuminuria, although no significant between-group difference in the rate of incident albuminuria was reported. Patients in the empagliflozin group had a small decrease in eGFR after starting the drug, with a similar pattern observed in patients with eGFR of ⩾60 mL/min/1.73 m2 and <60 mL/min/1.73 m2 (Table 2). The initial drop in eGFR was completely reversed after cessation of the drug at the end of the trial (consistent with a lowering of intraglomerular pressure after drug initiation and return to baseline after cessation, as opposed to ‘kidney injury’). More importantly, eGFR stabilized over time in those randomized to empagliflozin, in contrast with the placebo group wherein eGFR declined steadily over the course of the trial notably in the context of overall excellent blood pressure control and highly prevalent use of angiotensin-converting enzyme inhibitors (ACEi) or angiotensin II receptor blockers (ARBs) in the overall trial cohort. Similar to the beneficial effects on CV outcomes, the Kaplan–Meier curves for kidney outcomes in this trial began to diverge early (by 3 months) favouring empagliflozin with continued divergence throughout the trial observation. There was no heterogeneity in the effect seen with empagliflozin on the primary end-point of major adverse cardiovascular events (MACE) and CV death among CKD patients when compared with patients with normal kidney function at baseline in the trial (interaction p = 0.20 and 0.15, respectively).14,17 The infrequent occurrence of doubling of serum creatinine and initiation of renal replacement therapy points to the fact that most patients had no kidney disease or very mild disease at enrolment. This was confirmed by the fact that only about 30% of the participants had eGFR < 60 mL/min/1.73 m2, while half had albuminuria at baseline. Nevertheless, the kidney-specific benefits were seen even in the group with baseline CKD. 18
Renal outcomes found on the EMPA-REG (1) outcome trial.
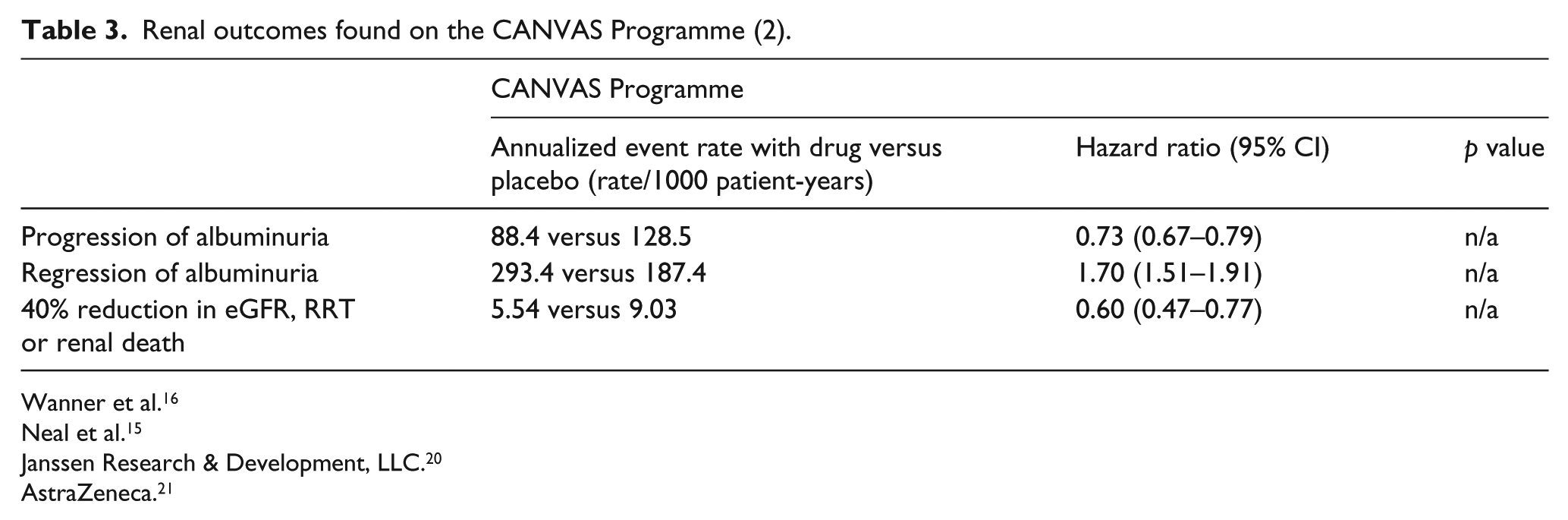
Kidney outcomes reported from the CANVAS programme were qualitatively similar, with a 40% relative reduction in the composite kidney outcome of sustained 40% reduction in eGFR, need for renal-replacement therapy or death from renal causes for the canagliflozin group compared with placebo (5.5 vs 9.0 participants per 1000 patient-years with the outcome, HR: 0.60; 95% CI: 0.47 to 0.77), with no difference observed between the CANVAS and CANVAS-R trials. Progression of albuminuria occurred less frequently in the canagliflozin group when compared with placebo, while regression of albuminuria was found more frequently in the treatment group. 15 Only 30% of the participants had albuminuria at baseline and the mean eGFR was 76.5 mL/min/1.73 m2, demonstrating a low kidney-risk group. Unlike empagliflozin, however, the CV and kidney benefits with canagliflozin came at the cost of a doubling of risk of amputations associated with canagliflozin versus placebo (6.3 vs 3.4 participants per 1000 patient-years; HR: 1.97; 95% CI: 1.41 to 2.75; Figure 1, Table 3). Although mechanistic explorations are not the objective of clinical outcome trials, these results are provocative and further studies in populations with CKD both with and without diabetes are underway (such as the EMPRESS trial with empagliflozin). 19
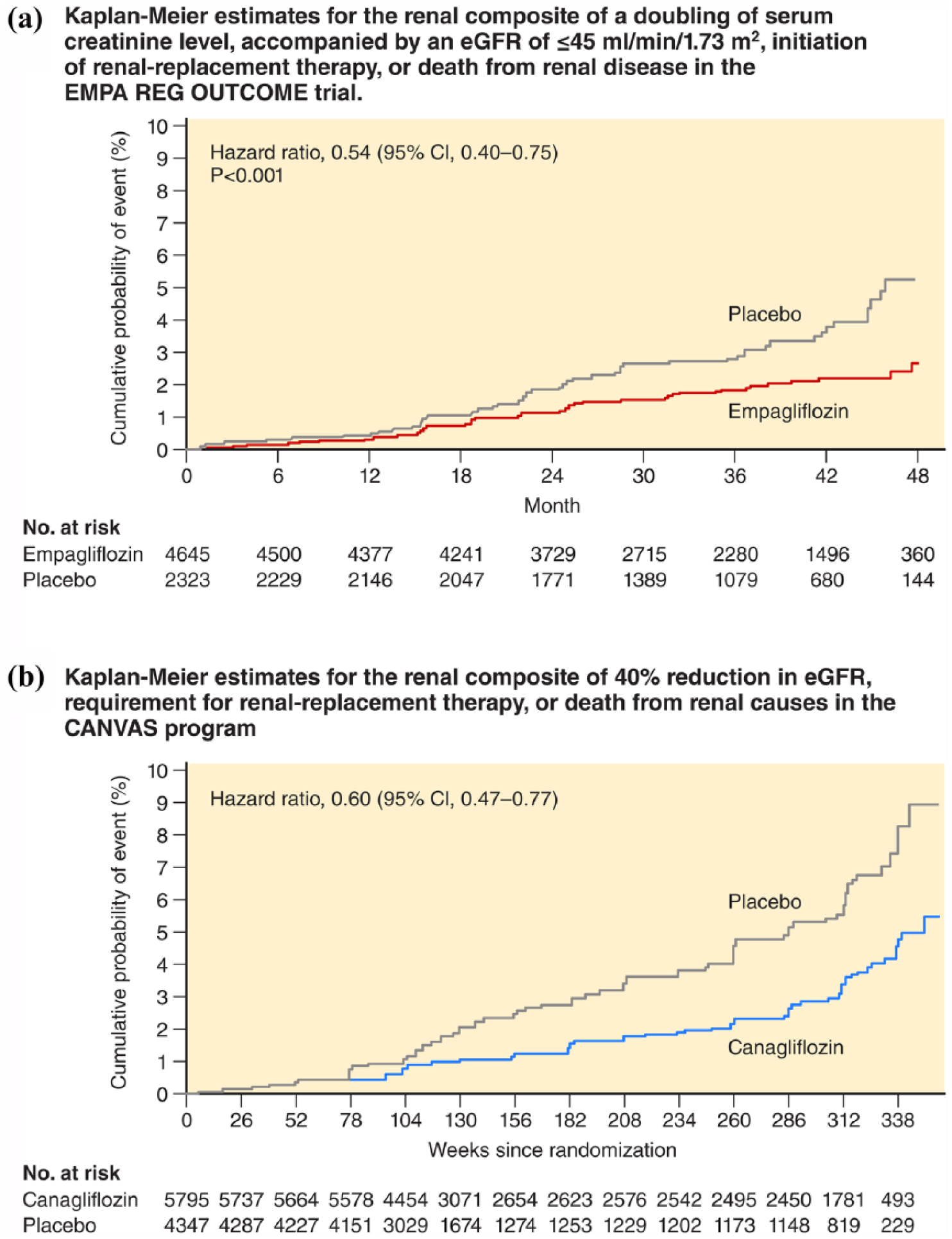
Kaplan–Meier estimates depicting the effects of (a) empagliflozin and (b) canagliflozin on composite kidney outcomes from the EMPA-REG OUTCOME trial and the integrated Canvas Programme, respectively (adapted from REG OUTCOME 16 and CANVAS Programme 15 ). Doubling of serum creatinine on the EMPA-REG OUTCOME represents a 57% drop in eGFR.
Renal outcomes found on the CANVAS Programme (2).
Wanner et al. 16
Neal et al. 15
Janssen Research & Development, LLC. 20
AstraZeneca. 21
The kidney as the gatekeeper of SGLT2 inhibitor efficacy
The beneficial effects of SGLT2i medications rely profoundly on kidney function. In a healthy patient without diabetes, virtually all glucose filtered at the glomerulus is reabsorbed in the proximal tubule, up to limit of about 180 mg/dL. Above this level, the maximal reabsorptive capacity of the kidneys (tubular max for glucose, TmG) is achieved and excess glucose is excreted in the urine. In this setting, SGLT2 is responsible for most of the reabsorption (80%–90%) and SGLT1, located more distally in the tubule, absorbs the remainder (10%–20%). In patients with diabetes, SGLT2 expression and capacity are paradoxically increased in the proximal tubule by mechanisms not yet fully understood,22,23 increasing TmG by as much as 20%.24,25 Consequently, in diabetes, a vicious cycle is created with an increase in the capacity of the kidneys to reabsorb urinary glucose, maintaining and even exacerbating hyperglycaemia.
In patients without diabetes, glomerular filtration is steadily maintained, despite variations in blood pressure and plasma volume, by modulation of pre-glomerular (afferent) and post-glomerular (efferent) arteriolar tone in response to vasoactive mediators and the tubuloglomerular feedback (TGF) mechanism. This balance is disrupted by hyperglycaemia in patients with diabetes, with increased proximal glucose reabsorption coupled with increased proximal sodium reabsorption via SGLT2 and reduced sodium (and chloride) delivery to the macula densa, mimicking kidney hypoperfusion. This leads to stimulation of the TGF mechanism affecting afferent arteriolar dilation, as well as efferent arteriolar constriction via other pathways, resulting in increased intraglomerular pressure (i.e. glomerular hypertension) and hyperfiltration, both of which have been implicated in the genesis and progression of CKD in diabetes.26–30 With SGLT2i, the reabsorption of glucose and sodium is decreased in the proximal tubule, and as a consequence, more sodium is delivered to the macula densa restoring normal TGF tone, reversing afferent artery dilation, reducing intraglomerular pressure and decreasing hyperfiltration and single nephron GFR. Of note, this decline in GFR is a hemodynamic phenomenon (similar to that seen with ACE inhibitors and ARBs) and does not reflect glomerular/kidney injury or pathologic change in GFR.
Thus, SGLT2i medications are mechanistically able to acutely improve glycaemic control and decrease glomerular hyperfiltration.31–33 Concurrently, SGLT2i causes a natriuretic and osmotic diuretic effect and resultant contraction in plasma volume. It is estimated that SGLT2i can reduce plasma volume by about 7% due to the diuretic effects, which in contrast to the effects of thiazides where plasma volume effects are short-lived during chronic treatment, a plasma volume reduction that remains relatively constant during exposure to SGLT2i medications. 11
Phase III trials have consistently shown small, but significant decreases in SBP and diastolic blood pressure (DBP) with SGLT2i medications,6,11 which may also help maintain kidney function over time. The exact mechanisms by which SGLT2i medications lower blood pressure remains a matter of debate, but cannot be attributed to their diuretic effects. This is best demonstrated by the consistent observation of greater BP reductions associated with decreasing eGFR for all SGLT2i medications,26,34 even when adjusting for baseline blood pressure and in the setting of diminishing effects with waning eGFR for changes in HbA1c. The effect of SGLT2i medications on SBP and DBP is fairly constant across the class and is not altered by background anti-hypertensive medications.34,35 SGLT2i medications have a moderate diuretic effect, leading to both acute and sustained intravascular volume contraction.11,26,36–38 Interestingly, this effect is not followed by a compensatory increase in heart rate, displaying possible indirect evidence for inhibition of sympathetic nervous system activity, perhaps as a downstream effect of restoration of normal TGF activity. The impact of weight loss on long-term BP reduction with this class of medications has been estimated to be around 30%–40%,39–41 indicating that although significant, it is not the most prominent contributor. Furthermore, studies have shown that the decrease in BP with SGLT2i is observed significantly earlier than the observed weight reduction, suggesting that weight loss might have a latent effect on BP, but not an acute effect. 35
Mechanisms of kidney protection
The mechanisms by which empagliflozin and canagliflozin impact kidney outcomes are not fully understood. Several hypotheses have been proposed, including better BP control, improvement in HbA1c, decreased glomerular hyperfiltration and/or glomerular hypertension, weight reduction, an overall decrease in renal oxygen consumption, shift in renal metabolism from free fatty acid to ketone use (which is a more efficient metabolic substrate), 42 inhibition of sodium hydrogen exchanger and possibly an increase in erythropoietin production by special fibroblasts located at the juxtamedullary apparatus resulting in increased red-cell mass and oxygen-carrying capacity (Figure 2). The small but significant decrease in BP with SGLT2i medications is unlikely to fully explain the robust beneficial effects on kidney outcomes, especially in the context of a very high prevalence of background ACE inhibitor/ARB therapy in the Cardiovascular Outcome Trials (CVOTs) reported to date. Similarly, improvements in glucose control is an unlikely explanation given the fact that historically, improvements in measures of kidney health associated with intensification of glucose control have taken a decade or more to manifest, 43 and kidney benefits were evident as early as three months in EMPA REG OUTCOME. A pooled analysis comprising patients with T2DM who participated in one of five phase III trials with empagliflozin found overall improvements in microalbuminuria (–32%) and macroalbuminuria (–41%), which was only partially (~50%) explained by changes in HbA1c, weight and BP, variables known to be associated with albuminuria. 44
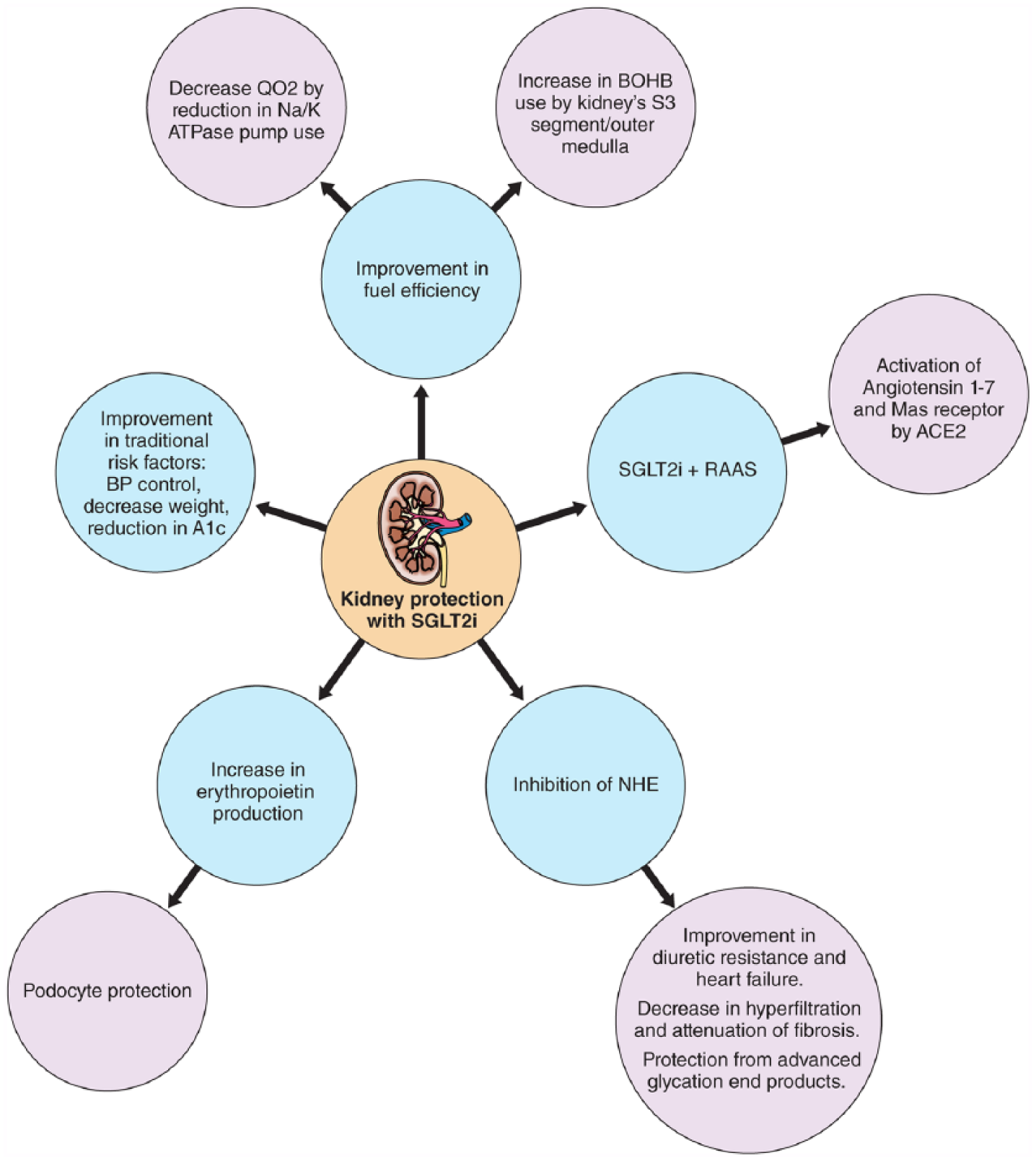
Potential mechanisms underpinning the kidney-specific benefits of SGLT2i.
The kidney protection observed in the EMPA REG OUTCOME study and CANVAS programme, where roughly 80% of patients were on background ACE/ARB in addition to SGLT2i versus placebo, may be explained by a shift to the non-classical renin–angiotensin–aldosterone system (RAAS) pathway (Figure 3). 45 Studies have shown an increase in circulating RAAS mediators such as renin, angiotensin II and aldosterone with SGLT2i,11,39 perhaps due to natriuresis and volume depletion. RAAS functions via two possible pathways. The classical pathway leads to the production of angiotensin II and activation of the AT1 receptor with consequent glomerular efferent artery vasoconstriction, sodium retention and inflammation, cell proliferation, fibrosis and apoptosis in pathologic conditions. 46 ACE inhibitors and ARBs act on this pathway and are known to confer kidney protection by preventing efferent artery constriction, subsequently reducing diabetic hyperfiltration. The non-classical RAAS pathway is thought to oppose the actions of the angiotensin II-AT1 receptor axis, contributing to the therapeutic effects of blockade of the classical pathway. The non-classical axis relies on ACE2 to produce angiotensin-(1-7), which activates the Mas receptor, leading to systemic arteriolar vasodilation, natriuresis, diuresis, reduced oxidative stress and antiproliferative activity by means of nitric oxide and prostaglandin upregulation.46–48 In addition, through the Mas receptor, angiotensin-(1-7) has been shown to reduce signalling pathways thought to be responsible to fibrogenesis and chronic inflammation. 49 Animal studies have shown an additive benefit of SGLT2i to angiotensin-converting enzyme inhibitors (ACEi) in slowing the progression of diabetic nephropathy 50 and lowering BP,51–53 an effect that might be linked to upregulation of the non-classical pathway.
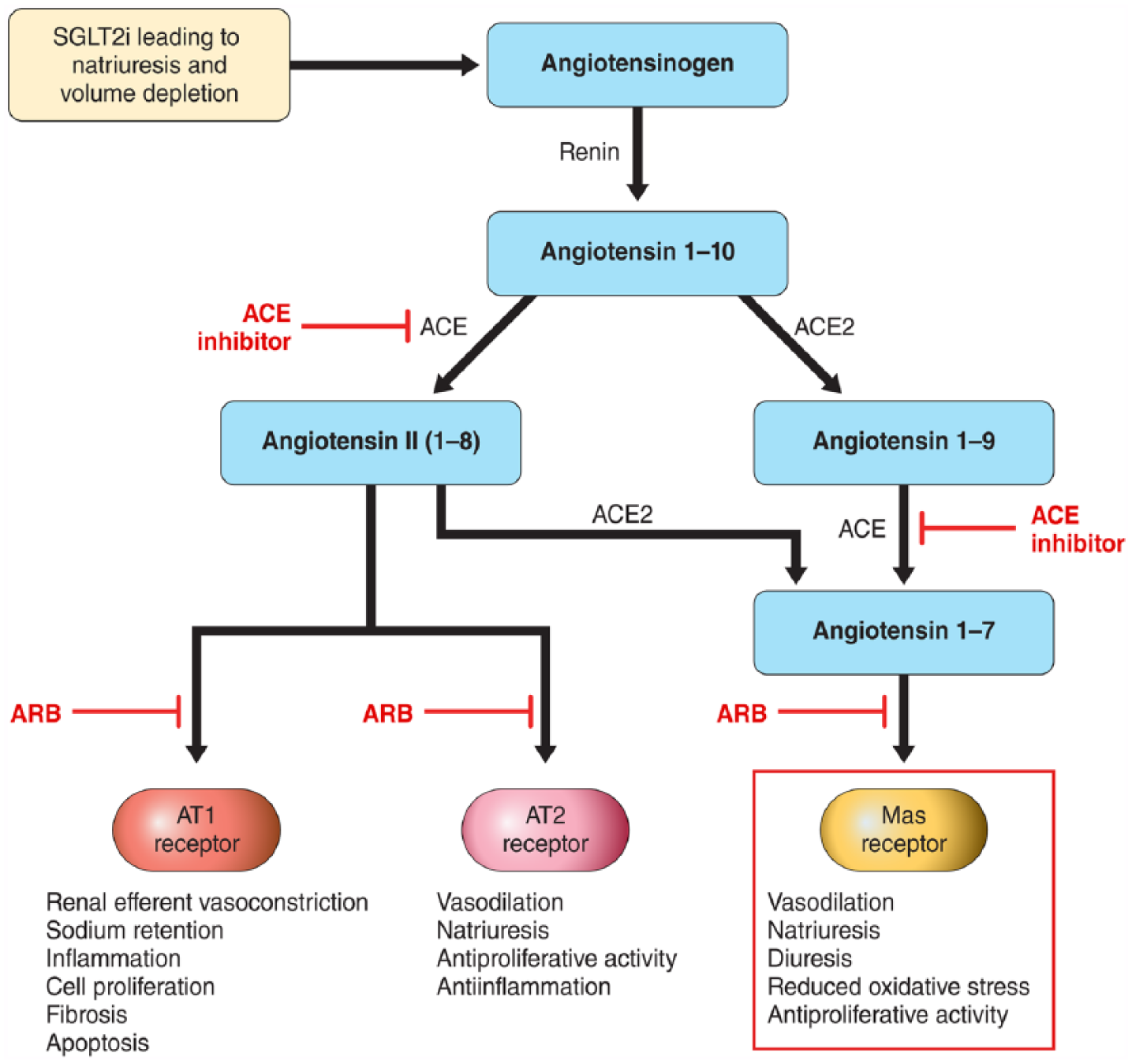
Potential explanation for kidney protection with SGLT2i when combined with ACEi/ARB by favouring activation of AT2 and Mas receptors. SGLT2i lead to natriuresis and volume depletion that activate the RAAS system. When ACEi is present, ACE2 predominates, leading to the formation of angiotensin 1–7 and activation of the MAS receptor.
Another hypothesis that has been proposed to explain the improved kidney outcomes with SGLT2i relies on the optimization of kidney oxygen consumption and shifts in substrate energetics 42 away from energy-inefficient metabolic substrates such as free fatty acids and towards ‘super fuels’ such as ketone bodies. Based on the TGF mechanism described previously, GFR is increased in diabetes leading to high luminal sodium and glucose concentrations and a resulting excess of proximal tubule intracellular sodium, activation of the Na+/K+ATPase pump and consequent increase in kidney oxygen consumption (QO2).54,55 In a study utilizing a rat single-nephron model, chronic SGLT2 inhibition lowered renal cortical QO2 by about 30%. However, due to a consequent increase in sodium reabsorption by SGLT1 located at the S3 segment, QO2 at the renal medulla was increased by 26%. 56 Interestingly, the reduction in filtration rate and its impact in lowering paracellular and transcellular sodium reabsorption in the proximal tubule more than counterbalance the upstream increase in QO2, leading to an overall positive balance. It is plausible that the substantial decrease in overall kidney QO2 leads to less hypoxic stress and could lead to long-term beneficial kidney effects. 42
The outer medulla of the kidney is an area susceptible to ischaemic injury due to the lower oxygen tension found in this area when compared to the cortex. As mentioned above, SGLT2 inhibition leads to an increase in QO2 at the kidney inner cortex/outer medulla level due to an increase in SGLT1 activity. Knowing that renal hypoxia is considered one of the mechanisms leading to CKD, this finding can be worrisome. However, chronic SGLT2 blockade with empagliflozin and canagliflozin did not translate into worse kidney outcomes as would be expected with medullary hypoxic injury. It is hypothesized, but not yet fully explored, that this rise in QO2 by the outer renal medulla is balanced by increased beta-hydroxybutyrate (BHOB) metabolism in that region. This might be a simplistic explanation as substrate metabolism in the kidney is complex and involves different substrate sources depending on fasting or fed state as well as substrate availability. It is known that all regions of the renal tubule are capable of metabolizing ketone bodies, specially under high plasma concentration 57 (such as noted with SGLT2 inhibition). 58 Further studies are needed to precisely measure kidney glucose, ketone bodies and free fatty acid utilization in the setting of SGLT2 inhibition.
Another possible explanation is that this increase in QO2 at the corticomedullary junction could lead to a decrease in renal cortical oxygen tension and therefore be the stimulus for the production of erythropoietin as has been reported in two recent studies. An unexpected rise in haematocrit (out of proportion to the degree of plasma volume contraction) with empagliflozin observed in the EMPA REG OUTCOME trial raised the possibility that increased oxygen carrying capacity and oxygen supply to both myocardium and to kidney could contribute to the beneficial effects observed in the trial. 59 Similar observations have been made with dapagliflozin. 11 Exposure to dapagliflozin was accompanied by a transient increase in reticulocyte and serum erythropoietin levels, with erythropoietin levels returning to stable baseline in the context of a ‘reset’ increased haematocrit. These observations have engendered the hypothesis that SGLT2i can directly increase red cell mass by enhancement of erythropoiesis. It is important to note that red blood cell mass was not directly measured in any of the prior studies, and these findings have not been reproduced to date. Nevertheless, this idea highlights the concept of the kidney possessing a ‘critmeter’ located in the juxtamedullary region, in which erythropoietin production is triggered by changes in oxygen tension and may be affected by SGLT2 inhibition. 60 Furthermore, based on experimental studies in rats and mice in which erythropoietin was shown to directly protect podocytes from diabetic nephropathy damage, it is possible that erythropoietin can not only be a mediator of erythrocytosis and modulator of red cell mass, but also one of the mediators of direct kidney protection caused by SGLT2i.61,62 The relevance of the increase in oxygen consumption by the outer medulla and clinical impact of the rise in erythropoietin level is yet to be fully explored.
Finally, another recently proposed theory to explain the kidney benefits of SGLT2i is based on the known interaction between SGLT2 and the sodium–hydrogen exchanger (NHE). The isoforms NHE1 and NHE3 are present in the kidneys, and their expression is increased by hyperglycaemia and hyperinsulinemia. Both isoforms of NHE contribute to the known features of diabetic nephropathy such as hyperfiltration, sodium reabsorption and mesangial cell proliferation 63 and their increased activity is related to vascular injury associated with hyperglycaemia.64,65 Kidney NHE activity is increased in patients with heart failure, possibly contributing to diuretic resistance. In vitro studies in proximal tubule cells have demonstrated an overexpression of NHE1 in the setting of high glucose followed by pH dysregulation and that inhibition of NHE1 restores pH and decreases cell apoptosis induced by high glucose. Furthermore, inhibition of NHE1 by cariporide was shown to normalize the redox status induced by advanced glycation end products (AGE) and prevented glomerular fibrosis. 65 Importantly, both NHE1 and NHE3 expression in the kidney are upregulated by angiotensin II receptor type 1 and agonism of type 2 receptors inhibits NHE activity. 63 Thus, it is hypothesized that such as with inhibitors of the rennin–angiotensin–aldosterone system, SGLT2i may decrease NHE expression and activity in the kidney, contributing to decreased hyperfiltration and attenuating the related fibrotic and proliferative action. 63
Promises and pitfalls of SGLT2i use in patients with CKD
The EMPA REG OUTCOME trial and the CANVAS Programme showed no heterogeneity of effect in the primary outcome of MACE, CV death, heart failure and all-cause mortality among patient subgroups stratified by normal kidney function versus those with CKD. Both trial programmes also showed consistent kidney protective effects in participants with baseline CKD when compared with individuals with normal kidney function.14,15 This raises the speculation of whether SGLT2i confers kidney protection independently of ambient glucose and GFR, which could potentially expand their use to patients with CKD without diabetes. 66 In both trial programmes, the average baseline eGFR was within normal range (EMPA REG OUTCOME 74 mL/min/1.73 m2 and in CANVAS 76.5 mL/min/1.73 m2) and only one-third of patients had albuminuria at baseline, indicating that most patients had either mild kidney disease or normal kidney function. For this reason, the number of events ascertained in the assessment of kidney outcomes was very low in both trial programmes, and extrapolation of these effects to a more advanced kidney disease population may be premature. Further studies designed specifically for the CKD population are needed prior to concluding that SGLT2i should be used with the specific kidney protection intent. Such studies are underway (e.g. the CREDENCE trial with canagliflozin-NCT02065791; the EMPRESS trial with empagliflozin; 19 and the DAPA-CKD trial-NCT03036150).
Although SGLT2i shows promise in kidney protection, the antihyperglycemic mode of action is dependent on filtered glucose load. Consequently, the use of SGLT2i in patients with more advanced CKD is restricted by medication product labels. In the United States, dapagliflozin is approved for use in patients with eGFR > 60 mL/min/1.73 m2, canagliflozin > 45 mL/min/1.73 m2 with dose adjustment for eGFR between 45 and 60, and empagliflozin > 45 mL/min/1.73 m2. The EMPA REG OUTCOME study and the CANVAS Programme have enrolled patients with lower eGFR (>30 mL/min/1.73 m2) with no evident kidney-related safety signals and with important improvements in blood pressure, kidney and CV outcomes despite smaller changes in HbA1c in the subgroups with compromised kidney function, supporting optimism that these medications might be accessible to CKD patients in the future. A recent subgroup analysis with participants with GFR < 60 of the EMPA-REG OUTCOME study showed that the beneficial outcomes of empagliflozin were no different from those with normal baseline kidney function and that the adverse event profile was also similar. 67 The CREDENCE trial (Evaluation of the Effects of Canagliflozin on Renal and Cardiovascular Outcomes in Participants with Established Diabetic Nephropathy – NCT02065791) and the DAPA-CKD trial (NCT03036150) will hopefully elucidate key clinical kidney effects of canagliflozin and dapagliflozin (Table 1), as they both have kidney primary composite outcomes. Both are expected to publish results in 2020. Kidney effects of dual SGLT1/2 inhibition with sotagliflozin, currently in CVOT development, will be evaluated in the SCORED clinical trial (NCT03315143).
The FDA issued a warning in June 2016 intensifying the previous labelled warnings about the risk of acute kidney injury (AKI) in patients using canagliflozin or dapagliflozin (but not empagliflozin). This warning states that from March 2013 to October 2015, the FDA has received reports of over 100 confirmable cases of AKI, with approximately half of them occurring within 1 month of starting the drugs. Some cases required hospitalization and dialysis and most showed improvement in kidney function after discontinuation of the medication. Some of the reported cases involved patients who were concomitantly taking other drugs known to affect kidney function and some had signs of dehydration. This warning did not include any AKI cases involving patients taking empagliflozin.
Conclusion
The SGLT2is have emerged as paradigm-shifting medications in the modern type 2 diabetes era. Their kidney protection outcomes, on the backdrop of best available evidence-based treatment, are unprecedented and seem to be a class-wide effect. Further studies with prespecified kidney endpoints as primary outcomes are in progress and will help clarify the effects and safety of SGLT2is in patients with CKD. Questions remain as to whether their GFR-independent effects leading to decreased hyperfiltration and intraglomerular pressure would benefit patients without diabetes. Studies such as the DAPA-CKD (NCT03036150), EMPEROR reduced (NCT03057977) and EMPEROR preserved (NCT03057951)68,69 are recruiting patients with and without diabetes and have changes in eGFR as a secondary outcome. Similarly, the EMPRESS trial will recruit patients with CKD, with and without diabetes, to be randomized to treatment with empagliflozin versus placebo. 19 These trials will explore the hypothesis of kidney protection in the absence of diabetes and open the scenario for alternative uses of SGLT2i.
Footnotes
Author’s contributions
NDAR, PAM, IJN, RDT and DKM have contributed with data interpretation, manuscript writing and editing for intellectual content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Dr Neeland is supported by grant K23DK106520 from the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institute of Health and by the Dedman Family Scholarship in Clinical Care from UT Southwestern.
Relationship with industry
N.D.A.R. and P.A.M. have nothing to disclose. I.J.N. has received research support from Boehringer Ingelheim. R.D.T. has served as an advisor or consultant for Amgen, Inc.; AstraZeneca; Boehringer Ingelheim Pharmaceuticals, Inc.; Relypsa, Inc.; ZS Pharma; Novo Nordisk, Inc; Bayer Inc and Reata Pharmaceuticals, Inc. D.K.M. has received support for clinical trial leadership from AstraZeneca, Sanofi Aventis, Janssen, Boehringer Ingelheim, Merck & Co, Novo Nordisk, Lexicon, Eisai, GlaxoSmithKline and Esperion and honoraria for consultancy from AstraZeneca, Sanofi Aventis, Lilly US, AstraZeneca, Boehringer Ingelheim, Merck & Co, Pfizer, Novo Nordisk and Metavant.
Search strategy and selection criteria
References for this review were identified through searches of PubMed for articles published from 1980 to 2017 by use of the terms ‘SGLT2 inhibitors’, ‘Empagliflozin’, ‘Canagliflozin’ and ‘Dapagliflozin’ in combination to the term ‘diabetes’, ‘kidney’ and ‘renal’. Articles resulting from these searches and relevant references cited in those articles were reviewed. Articles published in English were included.
