Abstract
Objective:
The aim of this study was to assess the association between sodium-glucose cotransporter-2 (SGLT2) inhibitors and the risk of orthostatic hypotension (OH) in patients with type 2 diabetes mellitus (T2DM).
Method:
A systematic literature retrieval was performed using PubMed, Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) from inception up to 16 October 2019. Data for study characteristics and outcomes of interest were extracted from each eligible study. Pooled risk ratios (RRs) with 95% confidence intervals (CI) for OH were calculated using a random-effects model.
Result:
A total of 16 studies (
Conclusion:
This meta-analysis suggested that, in general, SGLPT2 inhibitors did not increase the risk of OH in patients with T2DM. The possibility of OH should be, therefore, considered on an individual basis, especially in patients with a history of OH, long duration of T2DM, or comorbidities.
Bulleted novelty statement
Sodium-glucose cotransporter-2 (SGLT2) inhibitors have an impact on blood pressure, as there have been reports of orthostatic hypotension relevant adverse events in RCTs. There has been a lack of analysis to assess the risk of orthostatic hypotension with use of SGLT2 inhibitors; thus, it was reasonable to provide this information for practitioners in clinical practice.
There was no evidence of SGLT2 inhibitors increasing the risk of orthostatic hypotension in patients with T2DM (RR, 1.17 95% CI: 0.65–2.09), when stratified by category, age, duration of T2DM, or placebo-control or active-control and baseline blood pressure.
Introduction
Orthostatic hypotension (OH) is a frequent disorder among patients with diabetes. 1 Blood pressure (BP) maintains the homeostasis ascribed to adaptive compensatory adjustments, which are regulated by the autonomic nervous system, occurring on the upright position in the normal population, 2 whereas the functional impairment of the autonomic nervous system via diabetes shows an increased incidence of OH. 3 The presence of OH independently predicts the mortality and incidence of myocardial infarction, stroke, heart failure, and atrial fibrillation.4–6 Not only is OH the second most common cause of syncope, it also predicts mortality from coronary events, congestive heart failure (CHF), and cardiovascular disease, and the concurrent development of OH in patients with diabetes mellitus (DM) and CHF predicts poorer outcomes for these disease states.7–10 OH management is not satisfactory due to asymptomatic patients or cases with minimal symptoms. 11 Exact diagnosis and well management of OH can improve quality of life and minimize cardiovascular risk.
Sodium-glucose cotransporter-2 (SGLT2) inhibitors are a novel class of oral antidiabetic drugs that reduce plasma glucose concentration by enhancing glycosuria. 12 They also lower blood pressure through osmotic diuresis and natriuresis. 13 Given their particular insulin-independent modes of action, SGLT2 inhibitors have marked benefits on cardiovascular-renal outcomes in moderate-to-high risk patients with type 2 diabetes mellitus (T2DM).14,15 However, the potential adverse effects related to volume depletion, such as orthostatic hypotension (OH) and dehydration, should not be neglected. Moreover, orthostatic hypotension is associated with diabetes mellitus. 16 Patients with T2DM and OH often suffer a higher incident rate of transient, posture-mediated cognitive deficits than those without OH. 17
The present meta-analysis was performed to appraise OH risk in patients with T2DM treated with SGLT2 inhibitors compared with placebo or active control treated patients.
Method
Literature search
A systematic and comprehensive literature search was conducted using PubMed, Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) from the inception of this study up to 16 October 2019. The search strategy combined both the Medical Subject Heading and the text words canagliflozin, dapagliflozin, empagliflozin, ipragliflozin, remogliflozin, ertugliflozin, sergliflozin, luseogliflozin, sotagliflozin, tofogliflozin, Sodium glucose co-transporter, SGLT2, SGLT-2, and SGLT 2. These terms were adjusted to conform to the searching principals of each database, and we also conducted an additional literature search in each aforementioned database on 22 December 2019. Citations were limited to randomized controlled trials (RCTs) published in English and did not contain any studies with animals.
Study selection
Two authors (X.L. and Q.G.) independently reviewed all potentially relevant studies according to a prespecified criteria. Inclusion criteria were: (1) RCTs that included adult patients with T2DM; (2) SGLT2 inhibitors compared with placebo or active drugs regardless of back-ground treatments; (3) a duration of follow-up of at least 12 weeks; and (4) reports on data on orthostatic hypotension, which was identified using preferred terms from the Medical Dictionary for Regulatory Activities (MedDRA) or patient self-report defined by investigators. In specific, orthostatic hypotension was defined as a decrease from supine to standing blood pressure of >20 mmHg in systolic blood pressure or >10 mmHg in diastolic blood pressure. Lists of preferred terms as follow: Blood osmolarity increased, blood pressure ambulatory decreased, blood pressure decreased, blood pressure diastolic decreased, blood pressure immeasurable, blood pressure orthostatic abnormal, blood pressure orthostatic decreased, blood pressure systolic decreased, blood pressure systolic inspiratory decreased, central venous pressure decreased, circulatory collapse, decreased ventricular preload, diastolic hypotension, hypotension, mean arterial pressure decreased, orthostatic heart rate response increased, orthostatic hypotension. Data from completed published manuscripts were considered for inclusion in this analysis.
Data extraction and validity assessment
Two researchers (X.L. and Q.G.) independently screened and extracted the data using an a priori defined standardized Microsoft Excel sheet. The following information was extracted from each eligible trial: first author, year of publication, trial identifier, study duration, intervention drug, control drug, sample size, patient characteristics, duration of T2DM, and incident orthostatic hypotension events. These data were further examined by another investigator (X.R.), and any discrepancies resolved by discussion and consensus. If orthostatic hypotension events were not reported in the published paper, then these data were instead extracted from the trial register website. If the trial register website also did not provide the data of orthostatic hypotension events, then the incidence of such events was assumed to be zero. If the study did not design to observe any adverse events, this study was excluded. Two reviewers independently applied the Cochrane risk-of-bias tool 18 to assess the quality of included RCTs based on the following domains: random sequence generation, allocation concealment, blinding of study participants and personnel, incomplete outcome data, selective reporting, and other biases. The quality of trials was evaluated in terms of low, unclear, or high risk of bias, with disagreements being resolved through discussion.
Data synthesis and statistical analysis
Most of analyses were performed using RevMan (version 5.3.5; Cochrane Collaboration). For dichotomous data, risk ratios (RRs) and 95% confidence intervals (CI) were calculated to appraise the risk of orthostatic hypotension with SGLT2 inhibitors. Furthermore, subgroup analyses were conducted on the category of SGLT2 inhibitors, patients’ age, duration of T2DM, placebo-control or active-control and baseline blood pressure, baseline HbA1c (%), and patients with or without established cardiovascular disease to evaluate whether the risk of OH could be modified by clinical variables. A chi-square test (χ2) and
Results
The results of our retrieve methodology and literature screening process are outlined in Figure 1. In brief, we initially screened 2163 citations, but after exclusion of 968 duplications and exclusion of 290 articles based on their titles and abstracts, 909 full-text articles were further assessed. Of these 909 papers, 553 were excluded for being non-randomized controlled trials, 59 were excluded for enrolling patient without T2DM, 56 were excluded for having a follow-up less than 12 weeks, 48 citations were excluded as they are trial protocols, 34 were excluded for not designed to observe any adverse events, and 23 and 16 were excluded for being conference abstracts and animal trials, respectively. Another three papers were included on 22 December 2019. Finally, a total of 16 eligible RCTs involving 12,749 patients with T2DM were included in our meta-analysis.20–35 Six RCTs (
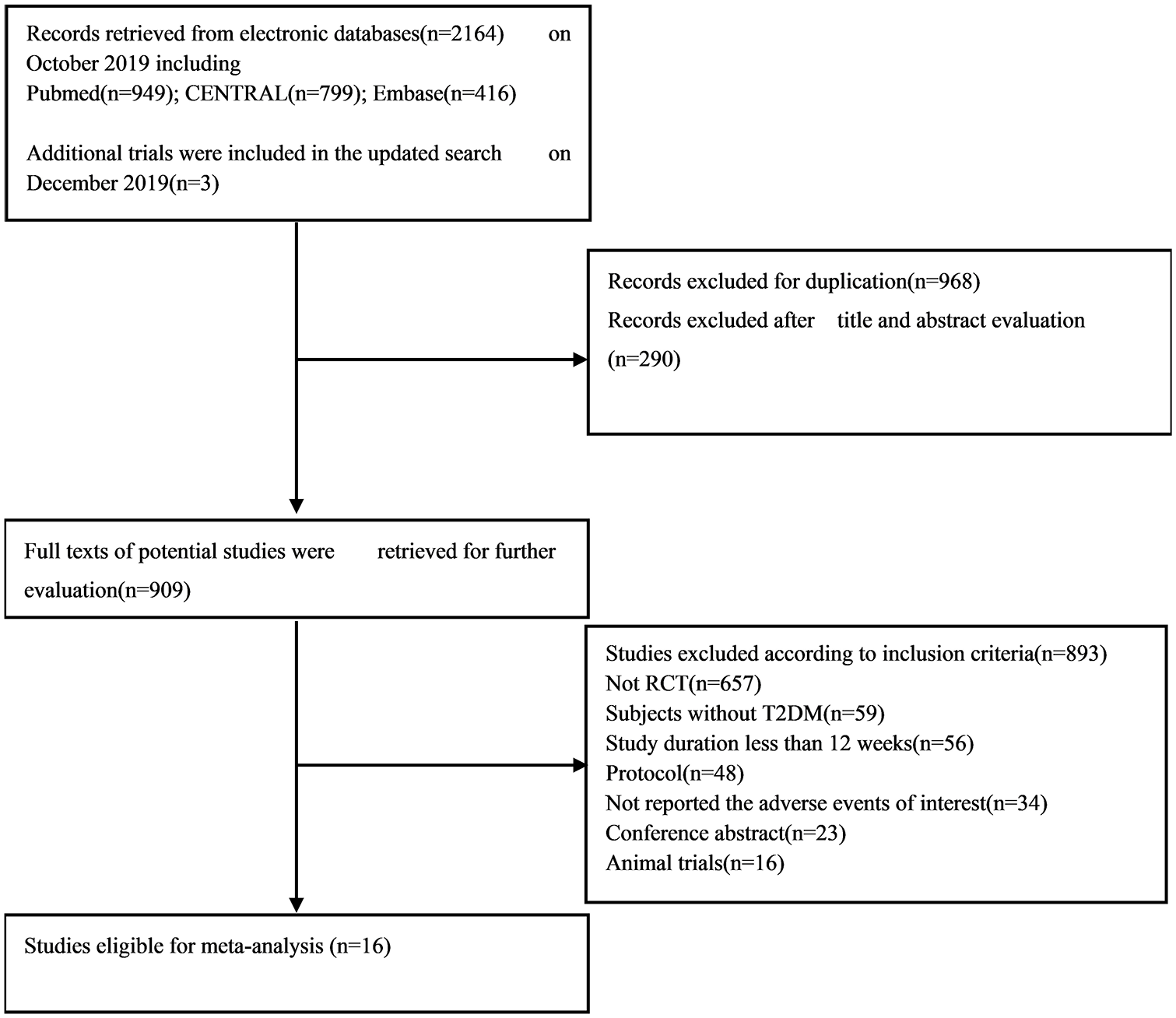
Flow chart of study selection for analysis.
Characteristics of the randomized controlled studies (RCTs) included in the meta-analysis.
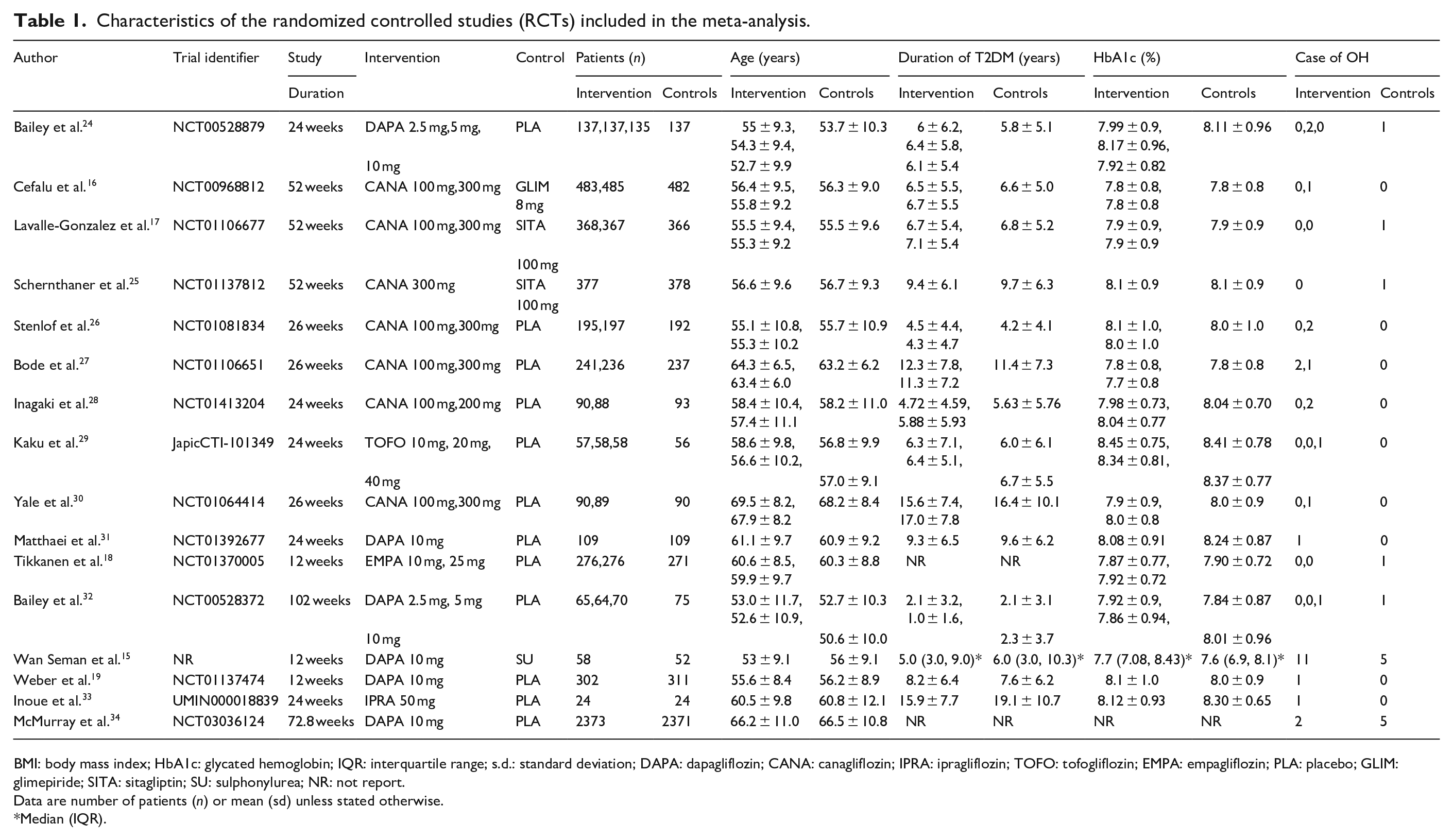
BMI: body mass index; HbA1c: glycated hemoglobin; IQR: interquartile range; s.d.: standard deviation; DAPA: dapagliflozin; CANA: canagliflozin; IPRA: ipragliflozin; TOFO: tofogliflozin; EMPA: empagliflozin; PLA: placebo; GLIM: glimepiride; SITA: sitagliptin; SU: sulphonylurea; NR: not report.
Data are number of patients (
Median (IQR).
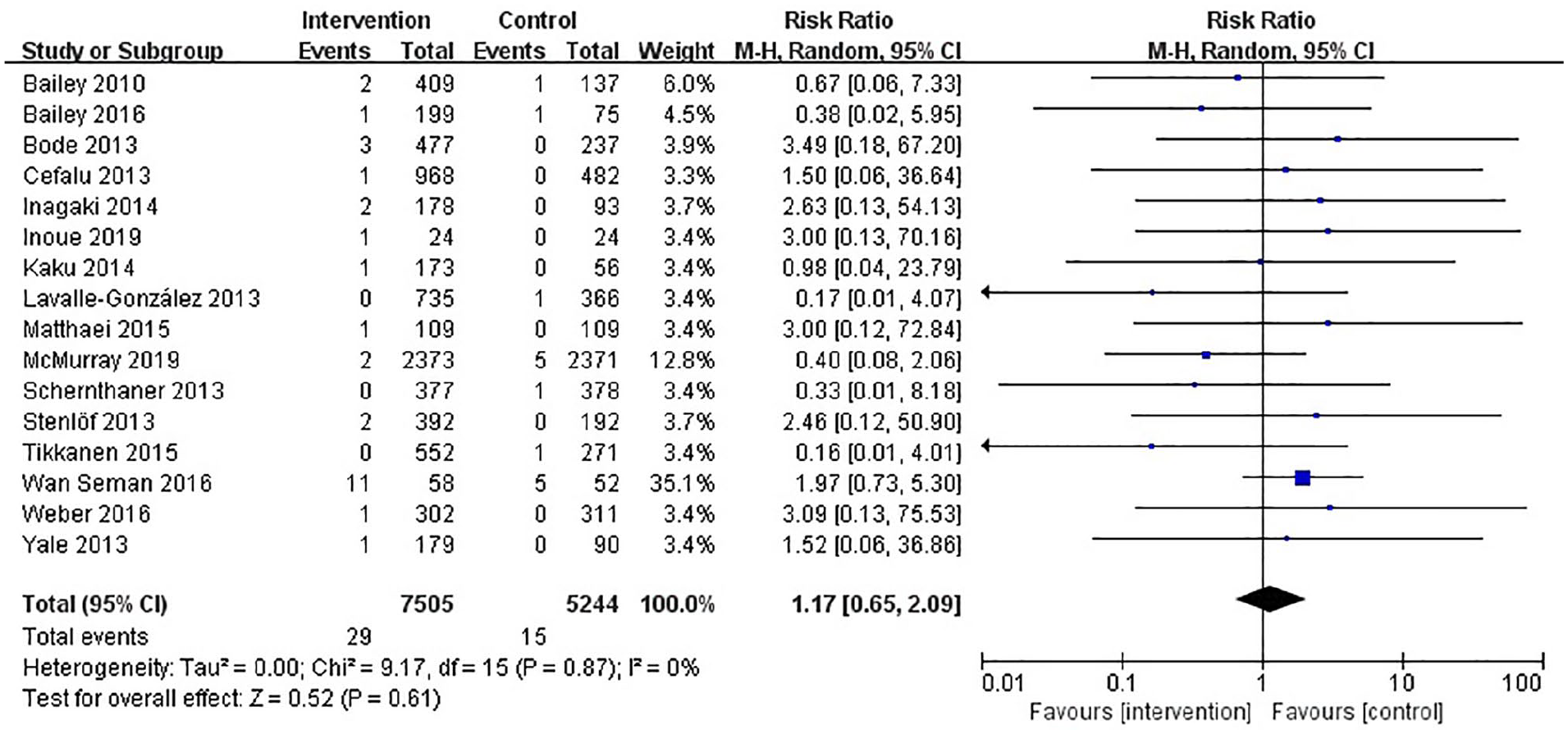
Forest plot of SGLT2 inhibitors versus control for risk of orthostatic hypotension.
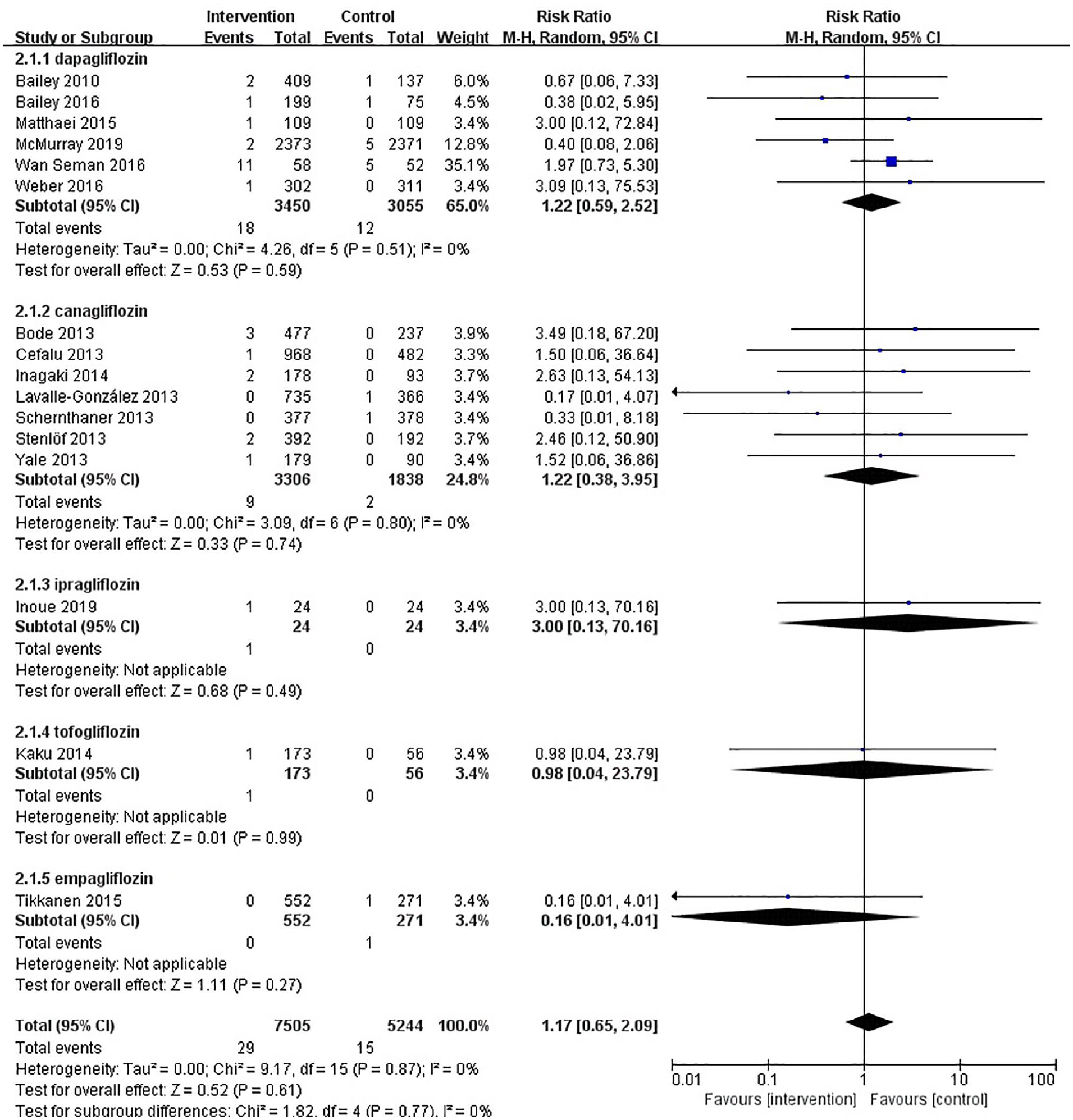
Forest plot of different SGLT2 inhibitors versus control for orthostatic hypotension.
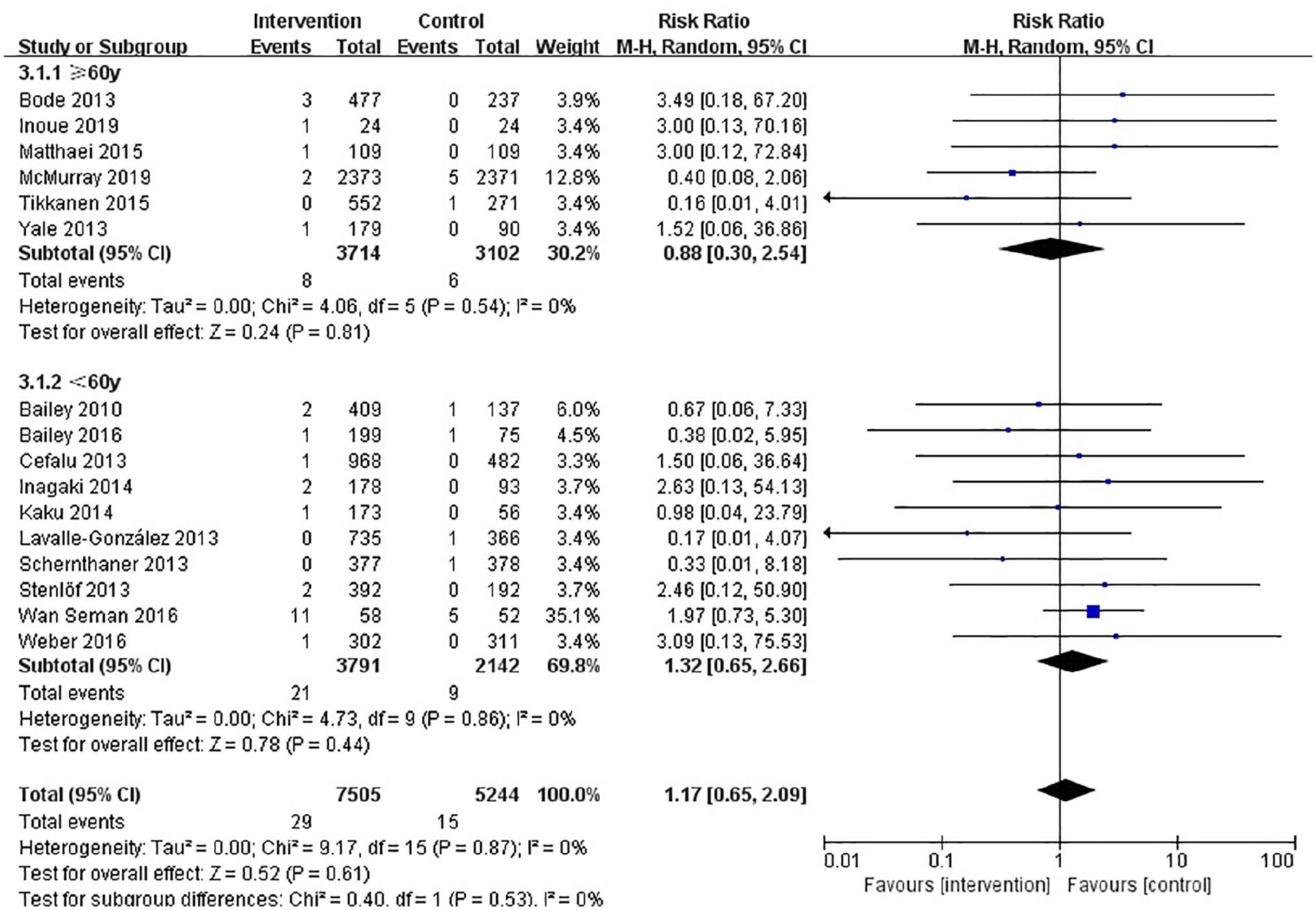
Forest plot of the effect of different age for orthostatic hypotension.
Discussion
The present meta-analysis of 16 RCTs involving proximately 12,000 patients demonstrated that SGLT2 inhibitors did not increase the risk of orthostatic hypotension. There was no evidence that the specific category of SGLT2 inhibitors used was associated with a greater risk of orthostatic hypotension compared with each other. When stratified by age there was a slight increase in risk with ages less than 60 years, and the same result was observed when the duration of T2DM was greater or equal to 9 years. When stratified by baseline blood pressure, SGLT2 inhibitor treatment with a baseline BP < 130/80 mmHg appeared to have beneficial effects for preventing orthostatic hypotension. However, if the baseline BP was ⩾130/80 mmHg, there was an increasing risk of orthostatic hypotension with SGLT2 inhibitor treatment. Additionally, there was a higher RR for patients with a baseline HbA1c ⩾ 8%, as well as patients with established cardiovascular diseases.
As they have been strongly recommended by ESC/EASD 36 and ADA, 37 SGLT2 inhibitors have attracted much attention from researchers. There have been numerous meta-analyses of efficacy and safety. 38 But to date there has been a lack of meta-analysis of the risk of orthostatic hypotension. As far as we know, the present meta-analysis is a novel review on the risk of orthostatic hypotension associated with SGLT2 inhibitors. Our conclusions, however, are consistent with the results of a previous meta-analysis that suggested that SGLT-2 inhibitors did not increase the risk of orthostatic hypotension. 17 It should be noted, however, that it focused on assessing the BP lowering ability of SGLT2 inhibitors. Moreover, it did not distinguish the risk of orthostatic hypotension from hypotension. We further performed subgroup analysis to investigate the effect of category, age, duration of T2DM, placebo/active control, baseline blood pressure, baseline HbA1c (%), and patients with or without established cardiovascular disease on the risk of orthostatic hypotension. The increasing risk of orthostatic hypotension with less than 60 years of age, however, seemed to be contradictory to the pathophysiology of orthostatic hypotension. Comprehensively, there was no significant difference between ⩾60 years and <60 years. The finding that with a longer duration of T2DM came a greater risk of orthostatic hypotension can be explained by the pathophysiology of orthostatic hypotension. 39 However, orthostatic hypotension is a major finding in patients with diabetic autonomic neuropathy, a complication that may be more frequent in older patients as well as in those with long-standing T2DM and poorly controlled diabetes. Such an assumption could explain why age >60 years and duration >9 years were predictors of OH. When taking into account the baseline blood pressure, patients with BP ⩾ 130/80 mmHg tended to have a higher risk compared with those with a BP < 130/80 mmHg. We speculate that a possible reason is that a larger proportion of concomitant therapy is conducted with antihypertensives among patients with BP ⩾ 130/80 mmHg and this was also observed in the ACCORD trial. 16 This meta-analysis also suggested that SGLT2 inhibitor-treated patients with HbA1c ⩾ 8% or with established cardiovascular diseases may be predisposed to OH. Moreover, sympathetic hyperactivity is a characteristic of type2 diabetes. 40 Abnormal sympathetic nervous activity plays a pivotal role in the pathogenesis of OH. Previous studies have indicated that SGLT2 inhibitors decrease BP without a compensatory increase or notable changes in heart rates, 41 implying the sympathoinhibitory effects of SGLT2 inhibitors.42,43 SGLT2 inhibitor-induced reduction of sympathetic activity may be an important reason why SGLT2 inhibitors do not increase the risk of OH in patients with T2DM.
Study limitations
This meta-analysis had certain limitations. First, the assessment of orthostatic hypotension was based upon a predefined list of preferred terms. This strategy was conservative, as orthostatic hypotension can be asymptomatic. Second, we excluded RCTs that reported hypotension adverse events in consideration of the different definitions of hypotension. Third, there were insufficient synthesized data in order to detect the effect of estimated glomerular filtration rates (eGFR) and concomitant antihypertensives on the risk of orthostatic hypotension. Last, SGLT2 inhibitor reduction of sympathetic activity may be important for their cardioprotective effects. The sympathoinhibitory effect of SGLT2 inhibitors could have an impact on the risk of OH. Although our meta-analysis included 16 studies, there was not sufficient information to assess the influence on the circadian rhythms of BP or the sympathetic activity of SGLT2 inhibitors. Further studies are necessary to test the hypothesis of the sympathoinhibitory effects of SGLT2 inhibitors.
Conclusion
The present meta-analysis demonstrated that SGLT2 inhibitors do not increase the risk of orthostatic hypotension in patients with T2DM. However, these results need to be interpreted with caution, due to the possibility of underestimating risk of asymptomatic orthostatic hypotension. Large-scale RCTs with prespecified, well-measured orthostatic hypotension are now called for to assess the impact of SGLT2 inhibitors on orthostatic hypotension. Thus far, it is important to pay attention to the risk of orthostatic hypotension associated with SGLT2 inhibitors, especially in patients with a long duration of T2DM and comorbidities of hypertension.
Supplemental Material
supplemental_File – Supplemental material for Risk of orthostatic hypotension associated with sodium-glucose cotransporter-2 inhibitor treatment: A meta-analysis of randomized controlled trials
Supplemental material, supplemental_File for Risk of orthostatic hypotension associated with sodium-glucose cotransporter-2 inhibitor treatment: A meta-analysis of randomized controlled trials by Xi Rong, Xinran Li, Qiling Gou, Kai Liu and Xiaoping Chen in Diabetes & Vascular Disease Research
Supplemental Material
supplemental_File_2 – Supplemental material for Risk of orthostatic hypotension associated with sodium-glucose cotransporter-2 inhibitor treatment: A meta-analysis of randomized controlled trials
Supplemental material, supplemental_File_2 for Risk of orthostatic hypotension associated with sodium-glucose cotransporter-2 inhibitor treatment: A meta-analysis of randomized controlled trials by Xi Rong, Xinran Li, Qiling Gou, Kai Liu and Xiaoping Chen in Diabetes & Vascular Disease Research
Footnotes
Author contributions
X.R. was involved in designing the study, as well as the data analysis and manuscript preparation. X.L., Q.G. were involved in extracting data, K.L. provided suggestion on carrying out the study and X.C. were involved in manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
