Abstract
Background
The SUSTAIN-6 trial showed the cardiovascular disease (CVD) benefits of semaglutide among patients with type 2 diabetes mellitus (T2DM). We estimated the US population eligibility and preventable CVD events from semaglutide.
Methods
US adults with T2DM were selected from the National Health and Nutrition Examination Survey 2011–2020 based on SUSTAIN-6 eligibility criteria. We estimated composite and secondary CVD events from SUSTAIN-6 treated and placebo published event rates multiplied by the weighted US eligible population, the difference being the number of preventable events which was then divided by 2.1 years to provide annualized estimates.
Results
Among 4755 (projected to 33.6 million [M]) adults with T2DM, 1132 (6.9 million) fit SUSTAIN-6 criteria. Compared to SUSTAIN-6, our sample more likely had female and black patients, shorter diabetes duration, and was less likely with prior CVD. We estimated 75,681 primary outcome CVD events, 128,329 expanded composite CVD events, 72,390 combined nonfatal MI, nonfatal stroke, and all-cause deaths, 36,195 non-fatal stroke events, 85,552 revascularizations, and 75,681 cases of nephropathy could be prevented annually if eligible T2DM subjects were on semaglutide.
Conclusion
Semaglutide may prevent over 75,000 CVD events annually if provided to eligible T2DM US adults. Efforts to improve use of these therapies are needed.
Diabetes mellitus (DM) is a global issue that affects over 500 million people worldwide, 1 and over 38 million people in the United States. 2 Cardiovascular disease (CVD) is the leading cause of death in persons with DM, including coronary heart disease (CHD), stroke, heart failure, and peripheral arterial disease. GLP-1 RA therapy has been shown to improve CVD risk factors, including lipids, blood pressure, and body weight. 3 Multiple studies have shown the efficacy of GLP1-RA drugs (semaglutide, liraglutide, albiglutide) in lowering CVD risk.4–8 Dual GIP-GLP1-RA therapies such as tirzepatide also show promise in reducing CVD outcomes given their beneficial risk factor effects. 9 The Trial to Evaluate Cardiovascular and Other Long-term Outcomes with Semaglutide in Subjects with Type 2 Diabetes (SUSTAIN-6) trial showed over 2.1 years a relative risk reduction of 26% for the primary composite CVD outcome with a 39% reduction in non-fatal stroke. 8
This purpose of this study is to determine the number of US adult type 2 diabetes mellitus (T2DM) patients who are eligible for semaglutide based on SUSTAIN-6 eligibility criteria as well as based on US Food and Drug Administration (FDA) indications, and to estimate based on applying the SUSTAIN-6 trial risk reductions the preventable primary and secondary cardiovascular (CV) events from treatment with semaglutide in eligible adults with T2DM.
Materials and methods
Sample selection
Our study samples were selected from the US National Health and Nutrition Examination (NHANES) Surveys 2011 to March of 2020. NHANES is a cross-sectional survey that gathers data biennially on approximately 10,000 US individuals representative of the US population, recording their demographic characteristics, medical history, clinical and laboratory measurements, and prescription medication use. The methodology of NHANES data collection has been described previously. 10 Due to the impact of the COVID-19 pandemic on safe data collection, complete data were available through March 2020 which forms the basis of our analysis. Two separate samples were created for semaglutide eligibility, one based on SUSTAIN-6 criteria, and the other based on FDA criteria. This study utilized de-identified publicly available data and was exempt from institutional review board review.
SUSTAIN-6 eligibility criteria required all patients to have T2DM, defined by having at least one of the following conditions in NHANES: 1) hemoglobin A1c (HbA1c) ≥ 6.5% 2) fasting glucose ≥ 126 mg/dL; 3) non-fasting glucose ≥ 200 mg/dL; 4) currently taking anti-diabetic medication or insulin; 5) ever diagnosed with diabetes by a doctor; those with Type 1 DM based taking insulin within one year of diagnosis as previously defined in NHANES 11 were excluded. Furthermore, patients were also required to have a HbA1c of at least 7.0% and be greater than age 50. In addition, those aged 50–59 needed to have either established CVD (ischemic heart disease, myocardial infarction, heart failure, or stroke), or an estimated glomerular filtration rate (eGFR) of < 60 mL/min/1.73 m2. Those aged ≥ 60 years needed to have either established CVD, microalbuminuria (albumin 30-299 mg/g), proteinuria (albumin ≥300 mg/g), hypertension (SBP/DBP ≥ 140/90 mmHg), or eGFR < 60 mL/min/1.73 m2. FDA indications include patients aged 18 years and over and to have T2DM with established CVD. Other inclusion and exclusion criteria variables were not available in NHANES dataset.
Statistical analysis
Descriptive statistics were examined in our study that corresponded with the SUSTAIN-6 trial: age, sex, body weight (kg), race/ethnicity, body mass index (BMI, kg/m2), duration of diabetes, glycated hemoglobin, systolic blood pressure (SBP) (mmHg), diastolic blood pressure (DBP) (mmHg), low-density lipoprotein cholesterol (LDL-C) (mg/dl) and proportion of patients who have never smoked. Further information on prior cardiovascular-related conditions including myocardial infarction (MI), coronary heart disease (CHD), and stroke (overall) were also available in NHANES and tabulated alongside similar information from the SUSTAIN-6 trial placebo group (both the 0.5 mg and 1.0 mg semaglutide groups) to show comparability of our study population.
After identifying the raw number of eligible participants in NHANES 2011–2020, a 9.3-year weighted variable based on the NHANES 2-year sample weights was created which extrapolated that raw number to the US population. This provided an estimated number of T2DM subjects (in millions) who were eligible for semaglutide based on SUSTAIN-6 criteria and was repeated for FDA criteria.
Lastly, the primary composite and expanded composite events, combined (nonfatal MI, nonfatal stroke, death) events, non-fatal stroke events, revascularization events, and nephropathy events were calculated for the placebo, the SUSTAIN-6 sample, and the FDA sample. This was done by multiplying both our sample sizes by the SUSTAIN-6 published treated and placebo event rates, respectively, for each of the outcomes, with the difference between the two defining the number of preventable events. While not all patients in SUSTAIN-6 were taking the same dosage of semaglutide (0.5 mg and 1.0 mg), the event rates published were from a pooled cohort combining the results of both dosages. The primary composite outcome was defined as first occurrence of CV death, nonfatal MI, or nonfatal stroke. The expanded composite outcome was defined as death from CV causes, nonfatal MI, nonfatal stroke, revascularization (coronary or peripheral), composite CVD events and hospitalization for unstable angina or heart failure (HF). Furthermore, preventable events for the primary composite outcome were also estimated by sex, race/ethnicity (White, Asian, Black, Hispanic, Other) and BMI (≤30 kg/m2 vs >30 kg/m2), based on the sex and ethnic-specific CVD risk reductions reported in the supplementary material of the SUSTAIN-6 publication. 6 Annualized preventable events were calculated by dividing by the mean follow-up time of 2.1 years. All analyses were done using SAS Studio (version 3.82; SAS Institute, Cary, NC, USA).
Results
A total of 45,462 individuals were identified in the NHANES 2011–2020 data. Of those, 42,995 had population weighting information available, reflecting a population of 315.2 million (M). Of those individuals, 26,239 (240.6 M) adults were identified, which included 4755 adult patients with T2DM (33.6 M). Following the additional inclusion criteria, 1132 (6.9 M) patients fit SUSTAIN-6 criteria (Figure 1). For the FDA sample, patients with T2DM needed to be aged 18 years or over and have prior CVD. This amounted to 1200 (8.4 M) patients in the FDA sample. SUSTAIN-6 eligible sample selection from the national health and nutrition examination surveys 2011–2020.
Descriptive statistics among SUSTAIN-6 trial and NHANES sample participants.

aData for Race from the SUSTAIN-6 Trial included White, Asian, Black, and Other race patients, with a non-mutually exclusive category for all Hispanic patients. The NHANES Samples included Non-Hispanic White, Asian, Black, and Other race patients, with a mutually exclusive category for only Hispanic/Latino patients.
bOnly combined Ischemic and Hemorrhagic stroke data was available in NHANES 2011-2020.
Estimated events, based on SUSTAIN-6 data, SUSTAIN-6 criteria, pooled semaglutide group (N = 1132, 6.9 M).

Note: Estimates combining strata may not total overall due to rounding error.
aAnnualized preventable events are based on a median follow-up time of 2.1 years.
Estimated primary composite events, based on SUSTAIN-6 data, SUSTAIN-6 criteria, by sex and race/ethnicity (N = 1132, 6.91 M).

Note: Estimates combining strata may not total overall due to rounding error.
aAnnualized preventable events are based on a median follow-up time of 2.1 years.
Estimated primary composite events, based on SUSTAIN-6 data, SUSTAIN-6 criteria, by BMI group (N = 1097, 6.73 M).
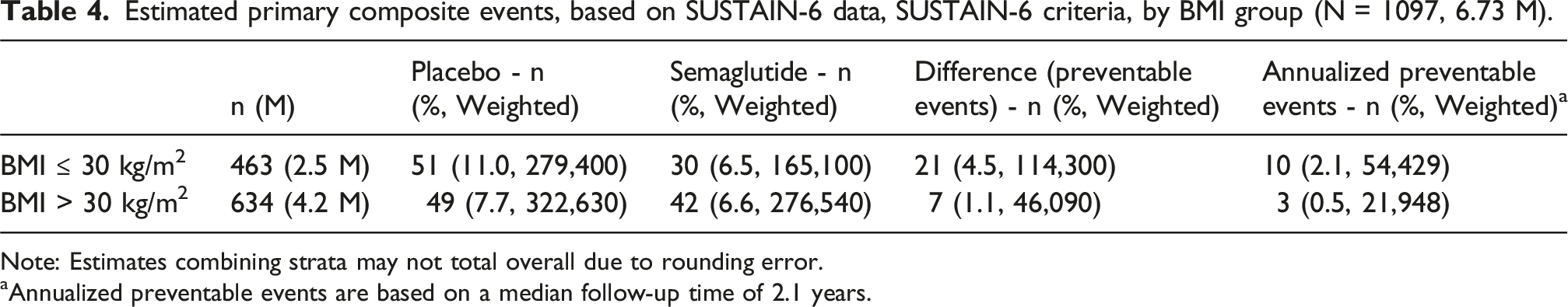
Note: Estimates combining strata may not total overall due to rounding error.
aAnnualized preventable events are based on a median follow-up time of 2.1 years.
Estimated events, based on SUSTAIN-6 data, FDA criteria, pooled semaglutide group (N = 1200, 8.36 M).

Note: Estimates combining strata may not total overall due to rounding error.
Estimated primary composite events, based on SUSTAIN-6 data, FDA criteria, by sex and race/ethnicity (N = 1200, 8.36 M).

Note: Estimates combining strata may not total overall due to rounding error.
aAnnualized preventable events are based on a median follow-up time of 2.1 years.
Estimated primary composite events, based on SUSTAIN-6 data, FDA criteria, by BMI group (N = 1137, 8.01 M).

Note: Estimates combining strata may not total overall due to rounding error.
aAnnualized preventable events are based on a median follow-up time of 2.1 years.
Discussion
Our study is unique in estimating semaglutide eligibility and preventable CVD events to the US T2DM population, based on both SUSTAIN-6 eligibility and FDA indications. 23.8% (by SUSTAIN-6 Criteria) and 25.2% (by FDA criteria) of the estimated 33.6 million adult T2DM population are eligible for semaglutide. Importantly, we estimate that semaglutide if given to SUSTAIN-6 eligible and FDA indicated patients can prevent more than 75,000 and 90,000 primary composite events annually, respectively. Moreover, we show more preventable CVD events will occur in males compared to females (over 58,000 vs 14,000 annually) due to their higher absolute event rates as well as SUSTAIN-6 findings 6 that male compared to female subjects have a greater relative risk reduction for the primary endpoint (31% compared to 15%). Also, with SUSTAIN-6 showing differences in efficacy among race/ethnicity (23%, 41%, 26%, 32%, and 54% relative risk reductions observed for White, Asian, Black, Hispanic, and other patients, respectively), we estimate proportionately more preventable CVD events among Hispanic and Asian patients. Our study documents the potential US population-wide impact of semaglutide in reducing CVD outcomes among US adult patients with T2DM.
Multiple studies have shown the benefits of GLP-1 RA drugs in improving cardiovascular health in those with and without T2DM and with estimations of population benefit assessed. For example, we previously estimated 4.2 million US adults with T2DM would fit LEADER trial 4 eligibility criteria with the potential for liraglutide to prevent 21.2 K cardiovascular events annually. 12 Among those without DM, but with obesity or overweight, we showed applying STEP-1 trial 7 eligibility criteria, that 93 million US adults might be eligible for semaglutide 2.4 mg, with the potential to prevent an estimated 1.5 million CVD events over 10 years reflecting a relative risk reduction of 18% compared to placebo, 13 which compared favorably to the actual 20% risk reduction observed in the SELECT trial examining the efficacy of semaglutide 2.4 mg in reducing CVD outcomes in persons with overweight or obesity and a history of cardiovascular disease. 8 Further, from applying the SURMOUNT-1 trial 9 eligibility criteria and CVD risk factor effects, we have subsequently estimated 93 million US adults would be eligible for tirzepatide based on SURMOUNT-1 eligibility criteria, with the potential to prevent 2.0 million CVD events over 10 years with an estimated relative risk reduction of 24% compared to placebo. 14
There are strengths and limitations to our study. The principal strength of our use of NHANES data is the ability to extrapolate to the general US population, estimating the eligible individuals and preventable CVD events on a population-wide basis. However, certain inclusion/exclusion criteria of the SUSTAIN-6 trial were unavailable in NHANES. For example, information regarding prior organ transplants, malignant neoplasm, endocrine neoplasia, non-familial medullary thyroid carcinoma, hypersensitivity to semaglutide, left ventricular hypertrophy by ECG, left ventricular systolic or diastolic dysfunction by imaging, and ankle-brachial index are not collected in our included NHANES surveys, which may slightly impact the number of semaglutide eligible patients based on SUSTAIN-6 eligibility criteria. There were also key differences between the descriptive statistics of our study samples and the SUSTAIN-6 placebo sample. Compared to the SUSTAIN-6 trial placebo, both of our samples had higher proportions of black patients, and less white patients. Furthermore, our samples had a lower mean duration of diabetes. These differences may limit the applicability of the SUSTAIN-6 placebo and semaglutide group event rates to our sample. Moreover, we were limited in our capture of CVD history being self-reported and limited to MI, CHD, and stroke, without information on stroke type (ischemic vs hemorrhagic) or revascularizations, which may also have affected our estimated eligible population. However, self-reported information captured in NHANES provides reasonable reliability, 15 While our estimates are based on the 2.1-year duration of the SUSTAIN-6 trial and we cannot be certain the same risk reduction rates would hold for a longer treatment duration; however, if this assumption were to be made, there would be the potential to prevent approximately 750,000 CVD events over 10 years of treatment.
Overall, our study shows nearly 7 million US adults with T2DM would be potentially eligible for semaglutide based on the SUSTAIN-6 trial with an estimated more than 75,000 preventable CVD events from its use annually. However, given uncertainty regarding long-term efficacy and adherence, evaluation of real-world data on actual observed outcomes is needed to validate our projections. With current use of these therapies among higher risk persons with DM still suboptimal in real-world settings,16,17 efforts to improve awareness of the CVD benefits of these therapies will hopefully lead to their greater use and realization of the CVD benefits we have projected from their use.
Footnotes
Author note
This project was previously presented at the American Heart Association scientific sessions, Chicago, Illinois, USA November 2024.
Ethical considerations
This project involved publicly accessible de-identified data and thus did not meet our University’s definition for human subject research.
Author contributions
Mr. Karthikeyan and Dr. Wong wrote the paper, Mr. Karthikeyan conducted the analysis, and Dr. Fan provided critical revision and comment.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This independent research was supported by unrestricted ISS grant funding from Novo Nordisk Inc. to the University of California, Irvine.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Wong receives research support from Amgen, Novartis, Novo Nordisk, and Regeneron and is an advisory board member for Amgen and consultant for Heart Lung, Novartis and Ionis. No other authors have any disclosures.
