Abstract
Introduction & Objective
Latent autoimmune diabetes mellitus (LADA), a heterogeneous disease, is much more common in society than thought. Although it has been claimed that LADA is similar to type 2 diabetes mellitus (T2DM) from a cardiovascular perspective, there is no clear consensus. In this context, the objective of this study is to assess subclinical dysfunction in the left and right ventricles in patients with LADA using novel tissue Doppler imaging (TDI) parameters.
Materials &Methods
The sample of this observational case-control study consisted of 57 consecutive patients aged between 30 and 70 years who applied to the endocrinology and metabolism outpatient clinics, were diagnosed with LADA, and were referred to the cardiology clinic for echocardiographic (ECHO) evaluation. The control group consisted of 60 healthy volunteers. Participants’ demographic and clinical characteristics and laboratory findings were recorded. All participants underwent ECHO using conventional ECHO and TDI. Subclinical left ventricular dysfunction was assessed using the myocardial performance index (MPI) and isovolumic myocardial acceleration (IVA).
Results
There were no significant differences between the patient and control groups in terms of conventional ECHO parameters. The left ventricular (LV) MPI was significantly higher in the patient group than in the control group (0.54 ± 0.11 vs 0.47 ± 0.07, p = 0.008). There was no significant difference between the groups in right ventricular (RV) MPI (0.49 ± 0.10 vs 0.46 ± 0.70, p = 0.217). IVA was decreased in both ventricles (IVA mitral: 3.03 ± 1.44 vs 3.78 ± 1.66, p = 0.008 and IVA tricuspid: 2.67 ± 0.88 vs 3.42 ± 0.97, p = 0.034). Both LV IVA and RV IVA were found to be significantly correlated with glutamic acid decarboxylase antibodies’ (GADA) levels in the negative direction (R = −0.290, p = 0.005 and R = −0.340, p = 0.001).
Conclusions
It was observed that LADA negatively affected the systolic and diastolic functions of both ventricles, with its effect being more pronounced in the left ventricle. Glycemic control and autoantibody titers were found to be correlated with TDI parameters, emphasizing their relevance in assessing cardiac dysfunction.
Keywords
Introduction
Latent autoimmune diabetes mellitus (LADA), a heterogeneous disease, presents more like type 2 diabetes mellitus (T2DM) but continues more like type 1 diabetes mellitus (T1DM) after diagnosis. Approximately 10% of patients diagnosed with T2DM actually suffer from LADA, 1 indicating that LADA is much more common in our society than thought. LADA generally has less genetic load and metabolic decompensation than T1DM. 2 There is a consensus on the cardiovascular (CV) complications of T1DM and T2DM, their effects on the myocardium, and the microvascular diseases they cause. However, the pathophysiology or CV complications of LADA have not yet been fully elucidated. T2DM is considered the most general risk factor for CV diseases in adults. T1DM has been shown to be an important risk factor for both micro- and macrovascular diseases in long-term prospective studies. 3
Patients diagnosed with LADA are generally younger and less likely to suffer from comorbidities than patients with other types of diabetes. Although several studies have reported the CV risks caused by LADA is similar to those caused by T2DM, the fact that the patients with LADA are healthier than those with T2DM makes the level of CV risk posed by LADA unclear. 4
The prevalence of subclinical left ventricular (LV) dysfunction in patients with T2DM ranges between 25% and 60%. 5 The first step in myocardial dysfunction is subclinical systolic and diastolic dysfunction, which have been shown in recent echocardiography (ECHO) studies to be risk factors for the development of cardiomyopathy despite good glycemic control. 6 However, there are a limited number of studies on ventricular functions in T1DM, and while no difference was observed in terms of ventricular functions between T1DM patients and healthy individuals in conventional ECHO studies, significant differences were observed in terms of left ventricular diastolic functions and tissue deformation. 7
Tissue Doppler imaging (TDI) has been used to assess subclinical dysfunction noninvasively. Myocardial performance index (MPI), also called the Tei-Doppler index, was defined by Chuwa Tei in 1995 as a Doppler ECHO parameter to assess left ventricular systolic and diastolic functions in patients with primary myocardial systolic dysfunction. 8 MPI can be calculated separately for both ventricles. MPI has been extensively researched in the context of metabolic syndrome and its subtypes as it provides global information about myocardial performance, is easy to apply, and is feasible. 9 Isovolumetric myocardial acceleration (IVA) is a new TDI parameter that allows early detection of deterioration in both LV and right ventricular (RV) functions. 10
In view of the foregoing, the objective of this study is to assess LV and RV subclinical dysfunction in patients with LADA using novel TDI parameters.
Material and methods
Study design
This study was designed as an observational case-control study. The study protocol was approved by the Local Ethics Commission of Siirt University with reference number 2024/12/01/07. The study was conducted in line with the ethical considerations set forth in the Declaration of Helsinki and Good Clinical Practice Guidelines. Informed consent was obtained from all participants included in the study.
Population and sample
The study population consisted of 68 consecutive patients who had applied to the endocrinology and metabolism clinics, had been diagnosed with LADA, and had been referred to the cardiology clinic for ECHO evaluation. Participants with ischemic heart disease, congenital heart disease, acute coronary syndromes, previous systolic or diastolic ventricular dysfunction, history of myocarditis or pericarditis, history of bariatric or diabetic surgery, gestational diabetes, history of antidiabetic use for weight loss, chronic organ failure, oncological conditions, drug abuse, patients who were breastfeeding, receiving chemotherapy were excluded from the study. In the end, 57 patients aged between 30 and 70 years who agreed to participate in the study and met the study’s inclusion criteria were included in the patient group, and 60 healthy volunteers were included in the control group.
Data collection
Participants’ demographic and clinical characteristics ((age, gender, body mass index (BMI) value, duration of diabetes)) and laboratory findings (fasting blood sugar, glycated hemoglobin, and plasma creatinine levels at least 8 h of fasting) were recorded. All participants underwent ECHO using conventional ECHO and TDI. Subclinical LV dysfunction was assessed using MPI and IVA.
LADA diagnosis
The patients who did not develop diabetic ketoacidosis within 6 months after diagnosis, did not require insulin, and had insulin autoantibodies were considered to have LADA. The patients who had diabetic autoantibodies but were insulin-dependent and/or developed diabetic ketoacidosis after diagnosis were considered to have T1DM. 11 The patients who were determined not to have insulin autoantibodies according to standard criteria were considered to have T2DM. Participants’ glutamic acid decarboxylase antibodies (GADA) and islet antigen 2 (IA2) antibodies were analyzed using enzyme-linked immunosorbent assay (ELISA) kits. The cut-off value of 10 u/mL for both (GADA) and (IA2) antibodies was based on the threshold recommended by the ELISA kit manufacturer (Biomerica, USA), ensuring optimal assay performance.
Echocardiographic assessment
The participants underwent transthoracic ECHO using the Philips Affiniti 50 ECHO system with S5 transducer in left lateral and supine positions in two-dimensional (2D), M-mode, color Doppler, and pulsed wave Doppler modes. All ECHO examinations were carried out under single-lead electrocardiography. Parasternal long axis, short axis, and apical four and five chambers view were used. Left ventricular ejection fraction (LVEF) was calculated using the modified Simpson’s method. The mitral inflow velocities, E and A waves, as well as their ratio (E/A), were assessed using pulsed wave Doppler from the apical four-chamber view, with the Doppler probe positioned over the mitral valve leaflets during the diastolic phase. Every effort was made to ensure that the probe cursor was vertically positioned. Dimensions of the left atrium (LA), right atrium (RA), and both LV end-diastolic and LV end-systolic measurements were measured from the parasternal long-axis view, whereas the basal RV dimensions were measured from the apical four-chamber view. LA volumes were measured at the LV end-systole from both the apical four-chamber and two-chamber views and calculated using the biplane-modified Simpson’s method. Tricuspid annular plane systolic excursion (TAPSE), an indicator of RV systolic function, was measured using M-mode ECHO at the lateral tricuspid annulus during systole. All measurements were conducted in accordance with the guidelines published by the American Society of Echocardiography and the European Association of Cardiovascular Imaging. 12
The peak myocardial velocity during isovolumic contraction, the acceleration time of this peak myocardial velocity (AT), peak myocardial systolic velocity (Sa), and the peak early and late diastolic velocities (E′ and A′) were assessed. Additionally, isovolumic contraction time (IVCT), isovolumic relaxation time (IVRT), and ejection time (ET) were recorded. MPI was calculated by dividing the sum of IVCT and IVRT by ET. IVA was calculated by dividing the isovolumic velocity (IVV) by AT. IVA was calculated by dividing the peak isovolumic myocardial velocity by the acceleration time during the isovolumic contraction phase. 13 All ECHO images were digitally saved and analyzed offline, ensuring that all measurements adhered to the recommended standards for reproducibility and accuracy. Three consecutive cycles were averaged for each parameter to minimize variability. Observers performed a subset of repeat measurements to evaluate intra-observer variability in ECHO parameters.
Statistical analysis
The statistical analyses of the collected data were conducted using the SPSS 27.0 (Statistical Product and Service Solutions for Windows, Version 27.0, IBM Corp., Armonk, NY, U.S., 2020) software package. The results of the statistical analyses were expressed using descriptive statistics, i.e., mean ± standard deviation values in the case of continuous variables determined to conform to the normal distribution, median with interquartile ranges (IQR) in the case of continuous variables determined to not conform to the normal distribution, and frequencies (n) and percentage (%) values in the case of categorical variables. The normal distribution characteristics of numerical variables were analyzed using the Shapiro-Wilk test. In comparing the differences in numerical variables between two independent groups, an independent samples t-test was used for numerical variables determined to conform to the normal distribution, and the Mann-Whitney U test was used for numerical variables determined to not conform to the normal distribution. Additionally, chi-square tests were used to compare the differences in categorical variables between the groups. Pearson and Spearman’s rank correlation coefficients were used to assess the relationships between continuous and non-parametric variables, respectively. The effect size of this study was 0.76 (Cohen’s D). Achieved power was 98.3%. Two-tailed probability (p) statistics of < 0.05 were deemed to indicate statistical significance in all tests.
Results
Demographic and clinical characteristics and laboratory findings
Distribution of demographic and clinical characteristics in LADA and control groups.

BMI: Body Mass Index, DBP: Diastolic Blood Pressure, SBP: Systolic Blood Pressure, GADA: Glutamic Acid Decarboxylase Antibodies, Hba1c: Hemoglobin A1c, HDL-C: High-Density Lipoprotein Cholesterol, Tg: Triglycerides, Total-C: Total Cholesterol, LDL-C: Low-Density Lipoprotein Cholesterol, GLP-1RA: Glucagon like peptide 1 receptor agonist, SGLT -2: sodium-glucose cotransporter-2 inhibitor.
LV ECHO findings
There was no significant difference between the groups in conventional ECHO findings. There was also no significant difference between the groups in LV functions assessed by conventional techniques. Early diastolic mitral velocity and E/A ratio were found to be significantly decreased in the patient group compared to the control group (8.46 ± 3.87 cm/s vs 9.70 ± 3.40 cm/s, p = 0.032 and 0.83 ± 0.42 vs 0.93 ± 0.40, p = 0.022, respectively). On the other hand, there was no significant difference between the groups in late diastolic atrial contraction velocity. Isovolumic relaxation time (IVRT) and isovolumic contraction time (IVCT) were found to be more prolonged, although not significantly in the patient group than in the control group. The acceleration time of the mitral valve was significantly more prolonged, whereas the ejection time was significantly shorter in the patient group than in the control group (p = 0.010 and 0.027, respectively). MPI was significantly higher in the patient group than in the control group (0.54 ± 0.11 vs 0.47 ± 0.07, p = 0.008). IVA was significantly lower in the patient group than in the control group (3.03 ± 1.44 m/s2vs 3.78 ± 1.66 m/s2, p = 0.008) (Table 2).
RV ECHO findings
Left ventricle echocardiographic findings.
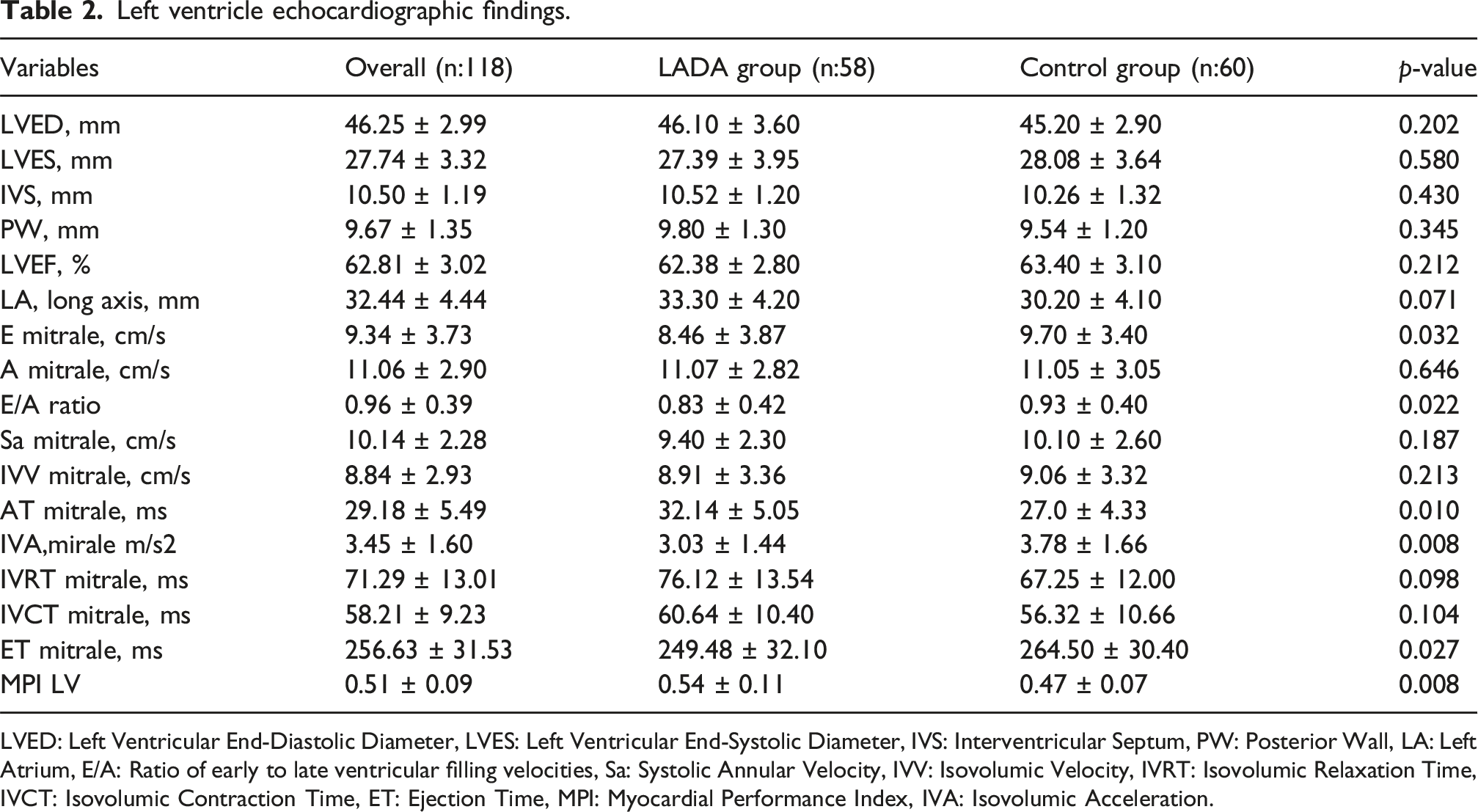
LVED: Left Ventricular End-Diastolic Diameter, LVES: Left Ventricular End-Systolic Diameter, IVS: Interventricular Septum, PW: Posterior Wall, LA: Left Atrium, E/A: Ratio of early to late ventricular filling velocities, Sa: Systolic Annular Velocity, IVV: Isovolumic Velocity, IVRT: Isovolumic Relaxation Time, IVCT: Isovolumic Contraction Time, ET: Ejection Time, MPI: Myocardial Performance Index, IVA: Isovolumic Acceleration.
Right ventricle echocardiographic findings.

LVED: Left Ventricular End-Diastolic Diameter, LVES: Left Ventricular End-Systolic Diameter, IVS: Interventricular Septum, PW: Posterior Wall, LA: Left Atrium, E/A: Ratio of early to late ventricular filling velocities, Sa: Systolic Annular Velocity, IVV: Isovolumic Velocity, IVRT: Isovolumic Relaxation Time, IVCT: Isovolumic Contraction Time, ET: Ejection Time, MPI: Myocardial Performance Index, IVA: Isovolumic Acceleration.
Correlations between IVA and laboratory findings in patients with LADA
Correlation between left or right ventricular IVA and.

LV: left ventricular, RV right ventricular, GADA glutamic acid decarboxylase.

Echocardiographic Comparisons of Myocardial Performance Index and Isovolumic Acceleration in LADA. Bar graph illustrating group comparisons between LADA patients and healthy controls for key tissue Doppler imaging parameters. The left ventricular myocardial performance index (LV MPI) was significantly higher in the LADA group, while isovolumic acceleration (IVA) values were significantly lower for both mitral (LV IVA) and tricuspid (RV IVA) annuli, suggesting subclinical systolic dysfunction. LADA: Latent Autoimmune Diabetes in Adults; MPI: Myocardial Performance Index; IVA: Isovolumic Acceleration; LV: Left Ventricle; RV: Right Ventricle.
Discussion
This study demonstrated subclinical LV dysfunction in patients with LADA through new TDI parameters, including IVA and MPI. LADA causes subclinical LV and RV systolic dysfunction and also LV diastolic dysfunction. TDI findings were found to be strongly correlated with LADA severity. IVA, a recently defined TDI parameter, correlated well with other cardiac performance parameters.
LADA is a unique type of diabetes disease that exhibits some features of both T1DM and T2DM. LADA presents more like T2DM, but after diagnosis, it becomes more and more like T1DM as progressive autoimmune destruction alters the properties of pancreatic beta cells. Approximately 1 in 10 patients diagnosed with T2DM are diagnosed with LADA in the long-term. 1 The Botnia study conducted in western Finland demonstrated that patients followed up with LADA were less likely to exhibit features of metabolic syndrome, such as abdominal obesity, hypertension, or hyperlipidemia yet, but the prevalence of CV events was not lower in patients with LADA compared to patients with other types of diabetes. 14 A cohort study found a higher incidence of carotid atherosclerosis in patients with LADA compared to propensity score-matched patients with T2DM. 15
Glycemic control plays a central role in the progression of myocardial dysfunction in patients with diabetes. Elevated fasting glucose and glycated hemoglobin levels were found to be correlated with deteriorated diastolic functions and reduced myocardial velocities, even in asymptomatic individuals. 16 Additionally, the role of autoimmunity, reflected in serum GADA levels, points to another dimension of the pathophysiology of LADA-induced cardiac dysfunction. A study by Maddaloni et al. highlighted the interplay between glycemic control and autoimmunity in influencing CV outcomes in patients with LADA. 2 Our correlation analysis revealed a significant negative relationship between LV IVA and both glycemic markers, i.e., fasting glucose and HbA1c levels and GADA titers, suggesting that poor glycemic control and autoimmunity synergistically contribute to subclinical ventricular dysfunction. Such findings underscore the need for tailored CV risk stratification in patients with LADA.
Subclinical LV dysfunction is an early indicator of diabetic cardiomyopathy, often preceding symptomatic cardiac disease. Our study demonstrated a significant reduction in LV IVA in patients with LADA compared to healthy control subjects, consistent with previous studies that linked reductions in IVA to impaired myocardial contractility due to metabolic disorders. 10 Prolonged AT and increased MPI further confirmed the presence of early myocardial dysfunction in patients with LADA. These parameters emphasize the utility of TDI in detecting subtle changes in cardiac performance. 5
The MPI index introduced by Tei et al. has been widely used to evaluate global ventricular performance in both systolic and diastolic functions. 8
Previous studies have reported that the increases in MPI in patients with metabolic syndrome and diabetes may serve as an early indicator of subclinical myocardial damage. In line with the literature data, we found that MPI values were elevated in the patient group than in the control group. While we found that LV MPI was significantly higher in the patient group compared to the control group, we did not find a significant difference in RV MPI between the groups. In a study conducted with patients with metabolic syndrome, Ertürk et al. found that both LV and RV MPI were elevated and correlated with IVA in the negative direction. 10 Although they identified abnormalities in RV MPI, they found no significant difference in RV function using conventional methods. Similarly, in another study conducted with patients with metabolic syndrome, Karakurt et al. reported significant differences in both TAPSE and MPI. 17 In contrast, we found no significant difference in RV function between the patient and control groups using conventional ECHO or TDI. Notably, although RV dysfunction was less pronounced, the reductions in tricuspid IVA and prolonged AT in the patient group suggest that RV function was not completely preserved. These findings are consistent with previous studies demonstrating the potential systemic effect of metabolic dysregulation on both ventricles. IVA, a novel TDI-derived parameter, provides direct insight into myocardial contractility and has shown promise in detecting early dysfunction in various metabolic and cardiovascular disorders. 18 We found that LV IVA was significantly reduced in patients with LADA compared to healthy control subjects, highlighting the presence of subclinical LV dysfunction. Our finding is consistent with those of previous studies reporting a decrease in IVA and a strong negative correlation between IVA and MPI in patients with metabolic syndrome.10,19In sum, IVA emerges as a critical parameter for assessing subclinical ventricular dysfunction in LADA. Its noninvasive nature and sensitivity to contractile changes make it a practical tool for CV risk stratification and long-term monitoring in this unique diabetic subgroup.
Limitations of the study
This study had several limitations. First, its observational design limited the ability to establish causal relationships between LADA and subclinical ventricular dysfunction. Secondly, while adequately powered, the sample size may not have fully captured the heterogeneity of LADA. Thirdly, while IVA and MPI provide sensitive measures of ventricular performance, incorporating additional imaging modalities, such as strain ECHO or cardiac magnetic resonance imaging (MRI), could enhance the understanding of myocardial mechanics in LADA. Fourthly, the fact that parameters such as the time since the diagnosis of diabetes, pre-diagnosis period, and treatment compliance were not addressed may not have allowed for a more holistic conclusion to be reached. Lastly, clinical follow-up of the patients and consecutive ECHO evaluations could have increased the impact of the study.
Conclusions
This study revealed significant subclinical LV dysfunction in patients with LADA, characterized by a decrease in IVA, prolongation of AT, and increase in MPI. These findings highlight the need for comprehensive CV evaluation in patients with LADA, even in the absence of overt symptoms. TDI parameters, particularly IVA and MPI, are valuable in detecting early myocardial dysfunction, emphasizing the importance of tailored risk management strategies in this patient population. Despite these insights, research on ventricular dysfunction in patients with LADA remains limited, with most studies focusing on T1DM and T2DM populations. This study fills this gap to some extent by incorporating advanced ECHO techniques involving parameters such as IVA and MPI to elucidate the CV profile of LADA.
Footnotes
Acknowledgments
We thank the cardiology and endocrinology department staff for their assistance with patient recruitment and data collection. Special appreciation to our echocardiography laboratory technicians for their expertise in image acquisition.
Ethical statement
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during the current study are available from the corresponding author upon reasonable request, following institutional and ethical guidelines.
IRB information
The study protocol was approved by the Siirt University Ethics Committee with approval number: 2024/12/01/07.
Novelty statement
This study pioneers the application of novel tissue Doppler parameters in LADA patients, establishing their utility in detecting subclinical cardiac dysfunction and its relationship with autoimmunity markers.
Clinical relevance statement
The findings support incorporating advanced echocardiographic screening in LADA patient care, potentially enabling earlier intervention before clinical manifestation of cardiac dysfunction.
