Abstract
Aim
To assess the effects of incretin-based therapies on β-cell function in patients with type 1 diabetes mellitus (T1DM).
Methods
We searched the PubMed, Cochrane Library, Embase, and Web of Knowledge databases for eligible randomized clinical trials published up to July 2021. The inclusion criteria were patients with T1DM or latent autoimmune diabetes in adults, patients treated with dipeptidyl peptidase-4 inhibitors or glucagon like peptide-1 receptor agonists, and outcomes included one of the following: fasting plasma glucose, fasting C-peptide, postprandial C-peptide, C-peptide area under the curve (AUC), homeostasis model assessment for β cell function, and insulin resistance. The effects were analyzed using a random effect model with STATA 11.0.
Results
Eight trials including 427 participants were included in the final analysis. A pooled analysis found no significant difference in fasting plasma glucose, fasting C-peptide, postprandial C-peptide, or C-peptide AUC between patients treated with incretin-based therapies and placebo. The two trials that reported changes in 2-hour postprandial C-peptide and two of the four trials that reported changes in C-peptide AUC reported increases after incretin-based therapies.
Conclusion
This meta-analysis showed that incretin-based therapies did not preserve β-cell function in patients with T1DM.
Keywords
Introduction
Type 1 diabetes mellitus (T1DM) is a T cell-mediated autoimmune disease characterized by the gradual loss of pancreatic β-cells, resulting in insulin deficiency and hyperglycemia. 1 However, recent studies showed that 29% to 80% of patients with T1DM, especially those with latent autoimmune diabetes in adults (LADA) or slowly progressive T1DM (SPIDDM), acquired excellent glucose control with little or no exogenous insulin therapy because enough of their β-cells continued to function.2,3 There are two distinct stages in C-peptide decline, as a measure of β-cell function: an initial decrease of 47% per year over 7 years, followed by a stable period. 2 An increasing number of studies have thus focused on preserving residual islet β-cell function in individuals with T1DM.
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) and dipeptidyl peptidase 4 inhibitors (DPP-4Is) are effective antidiabetic medications for type 2 diabetes mellitus (T2DM).4,5 Although GLP-1 RAs and DPP-4Is are both incretin-based drugs, they have different therapeutic mechanisms. The former mainly stimulates glucose-dependent insulin secretion, and inhibits glucagon release and gastrointestinal movement, while the latter prevents the decomposition and inactivation of GLP-1 and glucose-dependent insulin-promoting polypeptide. GLP-1 RAs and DPP-4Is have salient effects on glucose management, resulting in improved β-cell function in patients with T2DM.5–7
Based on their potential beneficial effects and promising results in animal studies, incretin-based drugs have been clinically tested in patients with T1DM or SPIDDM/LADA;8–11 however, their beneficial effects on β-cell function in individuals with T1DM remain unclear. Ahrén et al. 12 found no significant difference in mean plasma C-peptide levels between patients with T1DM with detectable C-peptides treated with any liraglutide dose and placebo at 26 weeks. Johansen et al. 8 found that fasting C-peptide levels increased from baseline at weeks 28 and 52 in linagliptin-treated patients, but decreased in patients treated with glimepiride, with significant between-group differences at 28 and 52 weeks in patients with LADA. Given the limited sample size and mixed results of previous studies, we conducted a meta-analysis to assess the effects of incretin-based therapies on β-cell function compared with placebo or active drugs in patients with T1DM.
Methods
Search strategy
We searched the PubMed, Cochrane Library, Embase, and Web of Knowledge databases for all studies concerning the effects GLP-1RAs and DPP-4Is on β-cell function published in English up to July 2021. The combinations of keywords used for searching were: (“dipeptidyl peptidase-4 inhibitors” OR dutogliptin OR sitagliptin OR “dpp-4 inhibitor” OR alogliptin OR saxagliptin OR linagliptin OR vildagliptin) OR (“glucagon-like peptide 1 receptor agonists” OR dulaglutide OR “glp-1 agonists” OR incretin OR exenatide OR liraglutide)) AND ((“type 1 diabetes” OR t1dm) OR (“latent autoimmune diabetes in adults” OR lada)). Relevant references from the retrieved articles were also identified.
Study selection
Randomized controlled trials (RCTs) that met the following criteria were included: (1) human RCT, (2) patients with T1DM or LADA, (3) RCT comparing the effects on β-cell function of GLP-1RAs or DPP-4Is with other active drugs or placebo, and (4) the outcome of interest included fasting plasma glucose, fasting C-peptide, 2-hour postprandial C-peptide, C-peptide area under the curve (AUC), homeostasis model assessment for β cell function (HOMA-β), and insulin resistance (HOMA-IR). Case reports, abstracts, editorials, unpublished reports, review articles, and letters were excluded. The present study was conducted following the PRISMA guidelines.
Data extraction
Two investigators initially screened the identified studies independently, reviewed the abstracts, examined the full texts, and decided on the final studies for inclusion in the analysis. The following data were extracted from each study: study design, year of publication, study duration, first author, baseline measures, and changes from baseline in fasting plasma glucose, fasting C-peptide, postprandial C-peptide, C-peptide AUC, HOMA-β, and HOMA-IR in the treatment and control groups. The risk of bias was assessed using the Cochrane Collaboration tool 13 (Supplemental Table S1).
Statistical analysis
Standard deviations of changes in fasting C-peptide, postprandial C-peptide, and C-peptide AUC from baseline were obtained from the manuscripts or were calculated using a formula 14 if not reported. The mean difference (MD) and 95% confidence intervals (CIs) were calculated to assess the effects of incretin-based therapies on fasting plasma glucose, fasting C-peptide, and postprandial C-peptide. The standardized MD (SMD) and 95% CIs were calculated to evaluate the effects of incretin-based therapies on C-peptide AUC due to different scales or units. Statistical analysis was conducted using STATA 11.0 (Stata Corp., TX, USA). The pooled MD or SMD was calculated using a random effects model. 14 Ethical approval was not required for this meta-analysis, because all of the included studies had already obtained ethical approval.
Results
A total 1867 studies were identified, of which 1288 were excluded because they were duplicates. A further 531 articles were excluded after reviewing the titles and abstracts, and 40 articles were excluded because they were not RCTs, were reviews, or had incomplete information. The study selection process is shown in Figure 1. The characteristics of the included trials are presented in Table 1. Eight trials with 427 participants were included in the final analysis. Mentions of T1DM in the results refer to classical T1DM.

Flowchart of study selection.
Characteristics of included trials.

DM, diabetes mellitus; RCT, randomized controlled trial; LADA, latent autoimmune diabetes in adults; I, intervention group; C, control group; FCP, fasting C-peptide; 2h-CP, 2-hour postprandial C-peptide; SPIDDM, slowly progressive type 1 diabetes mellitus; OAD, oral medicine; T1DM, type 1 diabetes mellitus.
Fasting C-peptide
Three trials reported changes in fasting C-peptide (n = 201), including one post hoc analysis including data from five previously reported double-blind, randomized, placebo-controlled, 24-week phase 3 studies, 15 and two RCTs lasting 1 year.8,16 Two of these three trials conducted in individuals with LADA reported increases in fasting C-peptide after incretin-based therapies. A pooled analysis of the three trials showed no significant difference in fasting C-peptide levels between incretin-based therapies and placebo. A forest plot of the effect is shown in Figure 2. There was also a high level of heterogeneity among the studies in terms of fasting C-peptide (I2 = 71.1%; P = 0.031).

Change in fasting C-peptide among patients with type 1 diabetes receiving incretin-based drugs versus control.
2-hour postprandial C-peptide
Two trials reported changes in 2-hour postprandial C-peptide (n = 58), both of which were open-label RCTs lasting 1 year. One included patients with LADA 15 and the other included patients with new-onset T1DM. 17 Both trials reported an increase in 2-hour postprandial C-peptide after incretin-based therapies. However, a pooled analysis of the two trials found no significant difference in 2-hour postprandial C-peptide between patients treated with incretin-based therapies and placebo (Figure 3), with high homogeneity (I2 = 0.0%; P = 0.820).
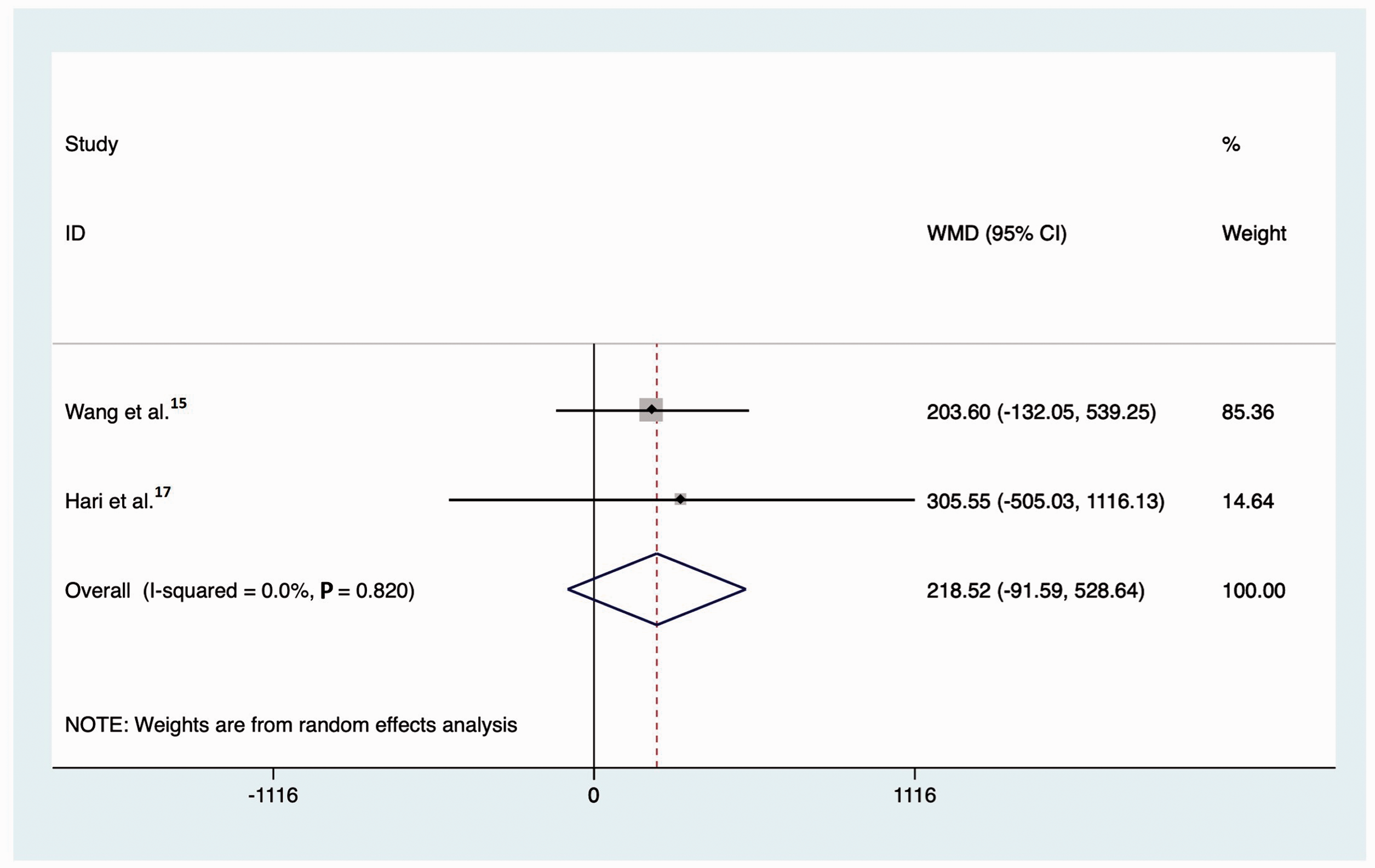
Change in 2-hour postprandial C-peptide among patients with type 1 diabetes receiving incretin-based drugs versus control.
C-peptide AUC
Four trials reported changes in C-peptide AUC (n = 180), including one study of patients with LADA, 16 one of patients with SPIDDM/LADA, 18 and two of patients with T1DM.19,20 The trial duration ranged from 16 weeks to 12 months. Two of the four trials reported an increase in C-peptide AUC after incretin-based therapies. Although C-peptide AUC decreased in the other two, the average decrease was lower than that in the control group. A pooled analysis of the four trials found no significant difference in C-peptide AUC between the incretin-based therapies and control groups. A forest plot of the effect is presented in Figure 4. The studies showed statistical homogeneity for C-peptide AUC (I2 = 0.0%; P = 0.607).

Change in C-peptide area under the curve among patients with type 1 diabetes receiving incretin-based drugs versus control.
HOMA-β and HOMA-IR
Only one trial reported changes in HOMA-β, 16 which showed no significant difference between the incretin-based therapies and control groups in patients with LADA; however, HOMA-β increased in patients receiving incretin-based therapies and decreased in those receiving placebo. No articles on HOMA-IR in T1DM were found.
Fasting plasma glucose
Three trials reported changes in fasting plasma glucose (n = 283). One reported an increase after 52 weeks of incretin-based therapies in patients with T1DM 21 and the other two included patients with LADA treated for 24 and 52 weeks, respectively.15,16 A pooled analysis of the three trials found no significant difference in fasting plasma glucose between patients treated with incretin-based therapy and placebo (Figure 5), with high homogeneity among the studies (I2 = 0.0%; P = 0.926).

Change in fasting plasma glucose among patients with type 1 diabetes receiving incretin-based drugs versus control.
Discussion
In this study, we analyzed the effects of incretin-based drug treatment on β-cell function in patients with T1DM. Eight studies including 427 patients with T1DM were finally included in the meta-analysis. Although the analysis found no significant differences between patients treated with incretin-based and other drugs, 2-hour postprandial C-peptide levels tended to increase after incretin-based therapies in patients with LADA or newly diagnosed classical T1DM, and C-peptide AUC increased in patients with SPIDDM/LADA. The C-peptide AUC decreased in patients with classical T1DM receiving incretin-based therapies, but the mean change was lower than in patients receiving placebo treatments.
Some articles that were not included in the study because of the lack of control group are summarized below. Kuhadiya et al. 22 conducted a retrospective analysis of ten C-peptide-positive patients with T1DM who were treated with a GLP-1RA in addition to insulin. They found a 3.5-fold increase in C-peptide concentrations after receiving GLP-1RA therapy for 12 weeks. They also found that the rise in C-peptide concentration was similar in patients with LADA (n = 6) and classical T1DM (n = 4). Pozzilli et al. 23 showed that HOMA-β increased in glutamic acid decarboxylase antibody (GADA)-positive patients during treatment with dulaglutide (HOMA-β improved by 29.90% at 6 months and 27.52% at 12 months), but there was little or no change in HOMA-IR at 6 or 12 months. Pinheiro et al. 24 reported the cases of two young women with positive GADA and classic clinical manifestations of T1DM, who achieved 4-year clinical diabetes remission and decreased anti-GADA after oral sitagliptin 100 mg and vitamin D3 5000 IU daily. All the above four trials showed that incretin-based therapies improved islet β-cell function in patients with T1DM.
Two RCTs included in this meta-analysis explored the effects of incretin-based therapies on islet β-cell function in T1DM at multiple time points. Johansen et al. 8 showed that fasting C-peptide levels increased gradually in GADA-positive patients treated with linagliptin at 28, 52, and 104 weeks, with significant increases compared with the glimepiride group at 28 and 52 weeks. Awata et al. 18 found that the sum of serum C-peptide levels was consistently higher than baseline for 48 months in patients with non-insulin-dependent SPIDDM/LADA treated with sitagliptin. Although, C-peptide values showed a nonsignificant increase in the sitagliptin group compared with the pioglitazone group, sitagliptin increased C-peptide levels compared with insulin or sulfonylurea based on data from the Tokyo Study, 25 especially in patients followed-up for 48 months. These two trials suggested that incretin-based therapies protected islet β-cell function for at least 2 to 4 years.
There are two possible reasons for the favorable effect of incretin-based therapies on β-cell function. First, improved glycemic control results in decreased glucotoxicity. A meta-analysis in 2019 26 showed that incretin-based therapy improved glycemic control and decreased insulin dose and weight without increasing the occurrence of severe hypoglycemia. Second, incretin-based therapies may also play a crucial role in the immune system. Recent studies showed that DPP-4 inhibitors modulated T cell immunity, promoted a Treg response, and stimulated islet β-cell proliferation and neogenesis, and could even reverse new-onset diabetes in a non-obese mouse model of T1DM.27–29 In addition, Zhang et al. 30 found that continuous transfusion of GLP-1 significantly increased the mass and replication and significantly reduced the apoptotic rate of β-cells in prediabetic NOD mice. Hadjiyanni et al. 31 reported that sustained GLP-1R activation delayed the onset of diabetes by preserving β-cell function in a murine model of T1DM. Recent trials in patients with T1DM or LADA also showed that incretin-based therapies could help to regulate the imbalance of CD4+ T cell subsets at both the cellular and mRNA levels.15,24
This meta-analysis had some limitations. The main limitation was the small sample sizes of the included trials, which substantially reduced the power of the trials. Second, most of the trials were not specifically designed to assess the effects of incretin-based therapies on β-cell function, and the results thus need to be interpreted with caution. Third, the results might have been affected by three included open-label trials due to a lack of blinding. Finally, some comparisons of the effects of incretin-based drugs on β-cell function in patients with T1DM were assessed as low quality according to the Cochrane Collaboration tool, which might also have limited the interpretation of the results.
Conclusion
This meta-analysis found no significant effect of incretin-based therapies compared with other therapies in terms of the preservation of β-cell function in patients with T1DM.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211066306 - Supplemental material for Effects of incretin-based therapies on β-cell function in type 1 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605211066306 for Effects of incretin-based therapies on β-cell function in type 1 diabetes mellitus: a systematic review and meta-analysis by Yucheng Wu, Yu Lu, Shufang Yang and Qingqing Zhang in Journal of International Medical Research
Footnotes
Acknowledgments
We thank all the participants in this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This research was supported by the Taizhou People's Hospital scientific research start-up fund project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
