Abstract
Objective:
The objective of this study is to investigate endothelial dysfunction (ED) and arterial stiffness (AS) and determine the association with diastolic dysfunction in children with type 1 diabetes mellitus (DM).
Methods:
A total of 42 patients without diabetic complications (mean age: 13.21 years) and 40 healthy (mean age: 13.07 years) children were included in this study. AS was assessed with ascending aorta M-mode measurements, diastolic dysfunction with pulsed wave (PW) Doppler and tissue Doppler echocardiography measurements and flow-mediated dilatation (FMD) and carotid intima–media thickness (CIMT) with high-resolution ultrasonography.
Results:
Results of diabetic group and healthy children were compared. In diabetic group, aortic strain (8.40 ± 2.98, 20.12 ± 5.04;
Conclusions:
ED and AS were found in type 1 DM patients without diabetic complications. Additionally, correlation was shown between increased AS and ED and right and left ventricular diastolic dysfunctions.
Introduction
Prolonged life years in diabetes mellitus (DM) have increased the incidence of diabetic complications. Currently, diabetes is the main cause of acquired blindness, dialysis or kidney transplantation among adults. However, clinically evident diabetes-related vascular complications should be rare in childhood and adolescence. Nevertheless, early functional and structural abnormalities may be present a few years after the onset of the disease. Childhood and adolescence is a period during which intensive education and treatment may prevent or delay the onset and progression of complications. 1 The hyperglycaemia in diabetes leads to vascular and neuronal damages by causing non-enzymatic glycosylation, increased oxidative stress and activation of polyol and pyruvate kinase C pathways. 2 The mechanisms triggered by hyperglycaemia result in endothelial dysfunction (ED) and arterial stiffness (AS), which are responsible for the microvascular and macrovascular complications of diabetes.3,4
Cardiovascular risk factors such as type 1 DM cause structural changes in blood vessels, leading to vascular stiffness and decreased vascular compliance. Increased vascular stiffness has been associated with increased cardiovascular mortality and morbidity.4,5 Patients with type 1 DM have extensive AS even before the occurrence of vascular complications. Early occurrence of AS is predictive of diabetic complications. 6 AS is an early finding of atherosclerosis and can be detected using echocardiographic methods prior to the occurrence of coronary artery disease (CAD) and other diabetic complications. 7 A close relation was determined between AS and left ventricle diastolic function. AS-induced left ventricle afterload increase or concurrent damage creation of glycosides by accumulating in myocardial tissue in parallel to AS may be the cause of left ventricle diastolic dysfunction. 8
Cardiovascular disease is an important cause of mortality and morbidity in patients with DM. Intensive glucose control does not necessarily decrease the incidence of cardiovascular events. Thus, there is a need for new approaches to the prevention and management of cardiovascular risk in diabetes.4,9,10 Management of increased AS and ED in addition to intensive glucose control may reduce diabetic complications. Certain experimental drugs have been successfully used for this purpose.10,11 In a pilot study in children, 3-month-long atorvastatin treatment provided decreased AS and improved endothelial function. 12 Angiotensin-converting-enzyme inhibitors have also been used for this purpose and resulted in improved arterial elasticity. 13 A direct renin inhibitor has been used for renin–angiotensin system blockade in another study, which improved systemic vascular function. 14 Early diagnosis and early management of ED and AS may be useful to prevent diabetic complications.
The aim of this study is to investigate ED and AS and determine the association with diastolic dysfunction in children with type 1 DM without microvascular or macrovascular complications.
Materials and methods
This study had a cross-sectional design. A total of 42 patients diagnosed with type 1 DM in Akdeniz University Medical Faculty Pediatric Endocrinology Polyclinics were included in this study as the DM group, and 40 subjects with matched age and body mass index (BMI) were included as the control group. The patients and control subjects included in this study and their parents were informed about the study, and ‘informed consent’ was obtained from each subject; this study was conducted in accordance with the Helsinki Declaration regulating biomedical investigations on humans, which has been revised in 2000.
Children who were found to have murmur at routine paediatrics visits were evaluated at paediatric cardiology polyclinic. The children who diagnosed with innocent murmur and cardiac defect was excluded via electrocardiograph (ECG) and echocardiographic were included in the study as control group. Type 1 DM patients of 7–16 years of age with a disease duration of at least 5 years who had no diabetic complications (neuropathy, nephropathy, retinopathy, CAD) were included in this study. The patients included in this study were evaluated in terms of diabetic complications. Microalbuminuria (30–300 mg/day protein in urine) was tested in 24-h urine to assess nephropathy and include subjects without microalbuminuria (0–30 mg/day), electromyography was performed for diabetic patients to assess neuropathy and to include subjects without neuropathy, retina examination was performed in ophthalmology polyclinic to include subjects without retinopathy and ECG and conventional echocardiography were performed to include subjects without cardiac disease. Patients younger than 7 years of age and older than 16 years of age, subjects with cardiac disease (congenital heart disease, CAD), diabetic complications (retinopathy, nephropathy, neuropathy), hypertension [systolic blood pressure (SBP) and diastolic blood pressure (DBP) levels above 90% percentile], obesity (BMI of 30 and above), subjects receiving medical treatment apart from insulin therapy and smokers were excluded from this study. Age, BMI, pulse, SBP and DBP were recorded in all groups. SBP and DBP were measured from the right brachial artery using a mercury gauge in supine position prior to examination after resting for a minimum of 5 min. BMI was calculated by dividing body weight in kilograms by the square of height in metres (kg/m2).
Echocardiography recordings
M-mode, two-dimensional (2D), Doppler and tissue Doppler echocardiographic recordings (3-MHz transducer; Vivid 7 pro, GE, Horten, Norway) were obtained for all subjects. M-mode and transmitral pulsed wave (PW) Doppler velocity recordings were obtained by evaluating three cardiac cycles. Velocities A and E were defined as the highest values achieved following early diastole and atrial contraction, respectively. Tissue Doppler recordings were obtained based on left ventricular lateral, basal septal segment and right ventricular free-wall segments on apical axis. E′’ wave velocities, A′’ wave velocities, S′’ wave velocities, isovolumic contraction time (IVCT), isovolumic relaxation time (IVRT), systolic time (ST) and myocardial performance index (MPI) were assessed in every segments. MPI is estimated by adding the IVRT and IVCT and dividing the sum by the ST.
Determining ED
Patients were instructed to avoid consuming caffeine and smoking before the procedure as such substances may interfere with endothelial functions. Brachial artery diameter was measured using ultrasonography (12-MHz transducer; Toshiba PowerVision 6000, Japan). Brachial artery was located in antecubital fossa and marked upon the skin in supine position under fasting condition. Brachial artery basal diameter (BABd) was determined following a resting period of 10 min. Subsequently, the clamping sleeve was placed above the measurement site, inflated to reach at least 30 units above the SBP, and approximately 3 min were allowed in this position. The clamping sleeve was then lowered. Brachial artery diameter was re-measured 60 s later. This value was recorded as the maximum brachial artery diameter (BAMd). The images were recorded in a digital setting. Vessel diameters were measured manually upon R-wave, providing synchronization with the ECG wave. Flow-mediated dilatation (FMD) was calculated by dividing the arterial diameter change by the basal arterial diameter (%) 15 (FMD% = [(BAMd − BABd)/BABd] ×100).
Determining elasticity properties of aorta
Following the routine echocardiography examination, patients were placed on their left side in supine position and their ascending aorta recordings were obtained in M-mode using 2D guidance (3-MHz transducer; Vivid 7 pro, GE). This M-mode ascending aorta records were obtained 3 cm above the aortic valve. Aorta diameters were calculated by measuring the distance between the inner edges of the front and back walls in systole and diastole. Systolic diameter of aorta (AoS) was measured in the open position of aortic valve. Diastolic diameter of aorta (AoD) was measured from the site matching the peak of QRS in ECG recordings (Figure 1). The following formulas were used to determine arterial elasticity. 16 Pulse pressure (PP) = SBP − DBP, aortic strain (%) = [(AoS − AoD)/AoD] × 100, distensibility = 2 × [AoS − AoD/AoD] × PP.
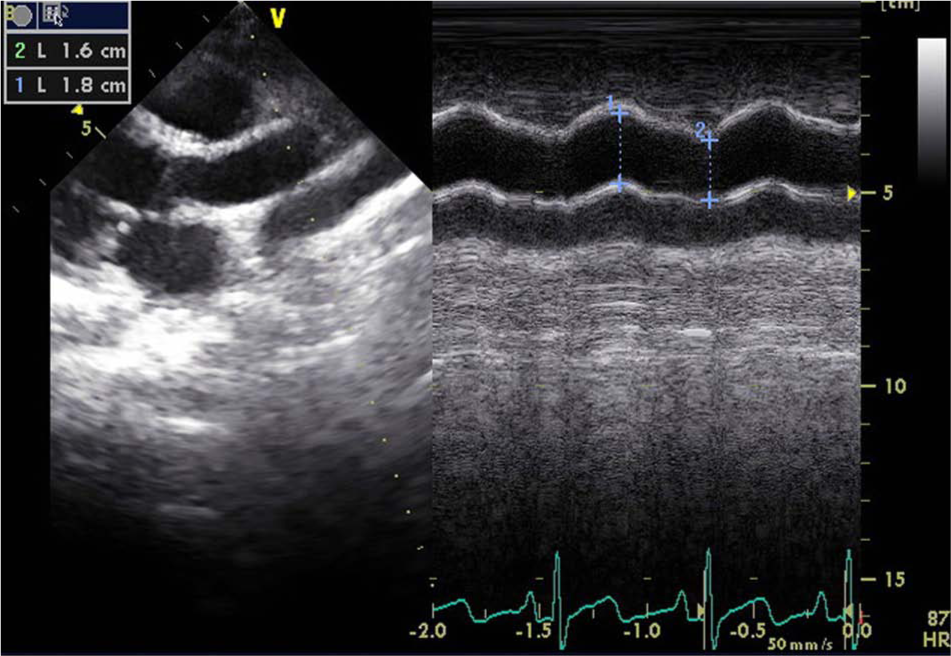
Aortic systolic diameter and aortic diastolic diameter measurements in a case.
Carotid intima–media thickness measurements
A resting period of 10 min was allowed before the ultrasonographic examination, and carotid intima–media thickness (CIMT) was measured using ultrasound B-mode imaging (12-MHz transducer; Toshiba PowerVision 6000). Measurements were performed while the patients were in supine position with slight extension of the head towards the opposite of the carotid artery of interest. Right and left carotid artery images were obtained in predefined scanning positions. The distant wall was focused during image scanning. The images were transferred to a computerized system and analysed. Hence, the proximal wall images were not of good quality during the analysis; measurements were made using the distant wall. Three measurements were performed and a mean value was obtained.
Statistical assessment
The statistical analysis was performed using Statistical Package for Social Sciences (SPSS; SPSS, Inc., Chicago, IL, USA) version 17.0 for Windows. Variables were expressed as mean ± standard deviation (SD). Significance of difference test (Student’s t test) was used when parametric test assumptions were met, and Mann–Whitney U test was used when parametric test assumptions were not met. Pearson correlation test was used to assess the significance of linear association between continuous variables.
Result
Clinical characteristics, results of conventional echocardiography and AS values
The groups were compared regarding age, BMI, SBP and DBP. There was no statistically significant difference between these values. Mean disease duration was 6.95 ± 1.79 years in the diabetic group with a mean haemoglobin A1c (HbA1c) of 9.00 ± 1.40. Conventional M-mode echocardiography parameters were found to be similar. Mitral E wave deceleration time was increased (189.74 ± 38.83 ms, 162.52 ± 32.85 ms;
Demographic characteristics, conventional echocardiography and arterial stiffness values.
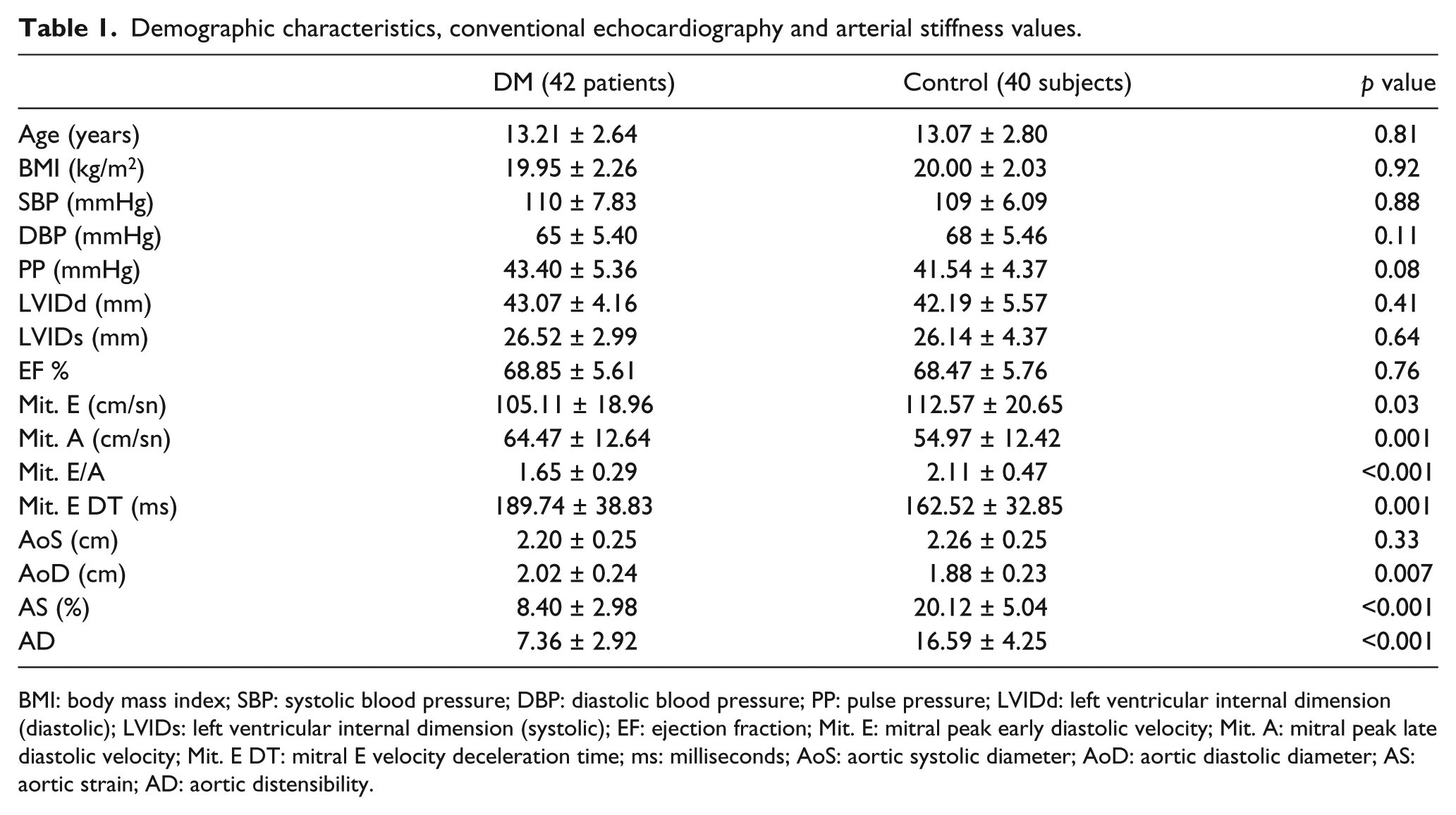
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; PP: pulse pressure; LVIDd: left ventricular internal dimension (diastolic); LVIDs: left ventricular internal dimension (systolic); EF: ejection fraction; Mit. E: mitral peak early diastolic velocity; Mit. A: mitral peak late diastolic velocity; Mit. E DT: mitral E velocity deceleration time; ms: milliseconds; AoS: aortic systolic diameter; AoD: aortic diastolic diameter; AS: aortic strain; AD: aortic distensibility.
Brachial artery FMD and CIMT
The change in brachial artery diameter (0.23 ± 0.08, 0.34 ± 0.09;
Brachial artery flow-mediated dilatation values.
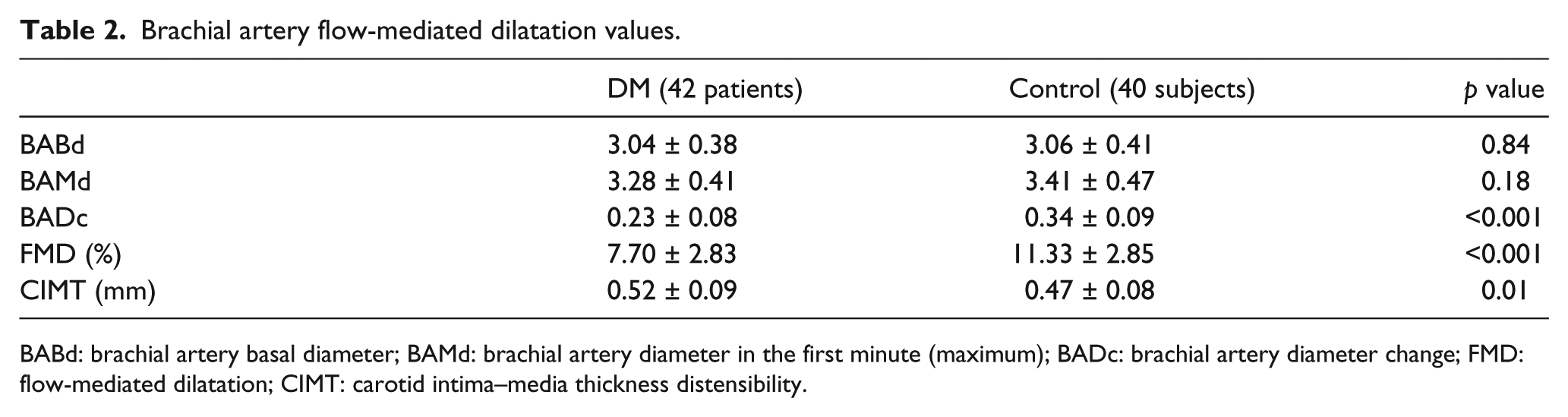
BABd: brachial artery basal diameter; BAMd: brachial artery diameter in the first minute (maximum); BADc: brachial artery diameter change; FMD: flow-mediated dilatation; CIMT: carotid intima–media thickness distensibility.
Right and left ventricular tissue Doppler findings
Left ventricular lateral E′ wave velocities (
Right ventricular and left ventricular tissue Doppler measurements.
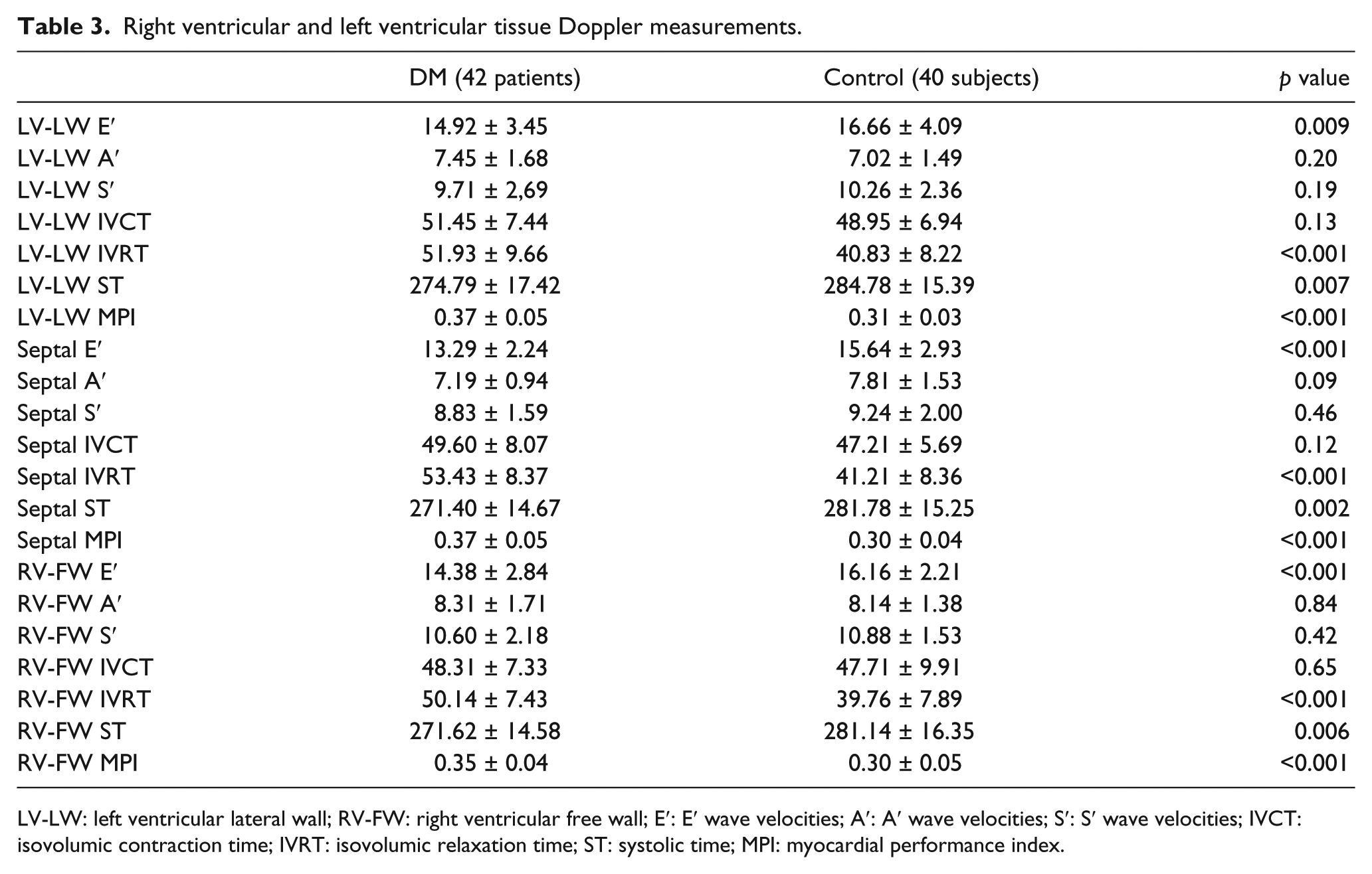
LV-LW: left ventricular lateral wall; RV-FW: right ventricular free wall; E′: E′ wave velocities; A′: A′ wave velocities; S′: S′ wave velocities; IVCT: isovolumic contraction time; IVRT: isovolumic relaxation time; ST: systolic time; MPI: myocardial performance index.
Correlation analyses
Correlation analyses demonstrated positive correlations between FMD and aortic strain (r = 0.496;
Discussion
Prolonged life years in DM have increased the incidence of diabetic complications. Childhood and adolescence is a period during which intensive education and treatment may prevent or delay the onset and progression of complications. 1 However, intensive glucose control alone does not necessarily decrease the incidence of cardiovascular disease. Thus, there is a need for new approaches to the prevention and management of cardiovascular risk in diabetes. The role of increased AS and ED are well established in development of diabetic complications.4,9,10 Management of increased AS and ED in addition to intensive glucose control may be a good choice in order to reduce diabetic complications. And it may even decrease the incidence of cardiovascular disease. Certain drugs have been successfully used for the management of ED and AS and therefore for the prevention of potential DM complications.12–14 Early diagnosis and early management of ED and AS may reduce diabetic complications. In this study, we aimed to detect ED and AS and determine the association with diastolic dysfunction in children with DM who had no complications.
DM leads to AS by causing functional and structural changes in large vessels.17 –19 AS and ED are early findings of atherosclerosis and can be detected using ultrasonography before the occurrence of CAD and other complications of diabetes. 7 Studies have shown an association between ED and segmental diastolic dysfunction. However, majority of these studies were conducted with adult patients in late stages.20,21 In this study, we found ED in paediatric patients with diabetes. Additionally, ED and AS correlated with diastolic dysfunction. Diagnosis and management of ED before the occurrence of diabetic complications may prevent diastolic dysfunction. Furthermore, ED is a risk factor for atherosclerosis and plays a role in early stages of atherosclerosis development. 22 Early diagnosis and management of ED may also prevent potential CAD.
CAD-related mortality is high among patients with type 1 DM, which has been established at the end of 1970s. Framingham Heart Study demonstrated the increase in various cardiovascular disorders such as peripheral vascular disease, congestive heart failure, CAD, myocardial infarction and sudden death in DM. 23 Type 1 DM is an independent risk factor for CAD development. Diabetes is associated with early atherosclerosis.24,25 AS and increased arterial wall thickness may lead to less vascularized wall layer, that is, the intima, to receive less oxygen and other nutrients resulting in early ischaemic damage that may favour atherosclerosis. 6 In patients with type 1 DM but without cardiovascular disease, AS was shown to be increased. And, in these patients, low-grade inflammation has a potential role in AS. 26 Also, in diabetic patients, increase of CIMT was found to be gender specific. 27 Type 1 DM is characterized by diffuse arterial wall stiffening and thickening which progress with the severity of the disease, but these problems may also occur in the absence of any diabetic complications. This suggests that vascular stiffening and thickening are early markers of vascular damage in diabetes. 6 Previous studies have demonstrated the association between diastolic dysfunction and AS in adult patients with type 1 DM.28,29 This study in children also showed increased AS and CIMT in patients with diabetes. Furthermore, AS and CIMT correlated with diastolic dysfunction.
Our study demonstrated decreased E′ wave velocity in left ventricular lateral, basal septal segment and right ventricular free wall and increased IVRT and MPI. Impaired cardiac relaxation mechanism and diastolic dysfunction in right and left ventricles have been found in patients with diabetes before the disruption of systolic functions. Additionally, co-occurrence of cardiac diastolic dysfunction and impaired elasticity of aorta has been shown in patients with DM. Decreased elasticity of aorta may cause increased afterload and eventually increase left ventricular wall tension, playing a causal role in left ventricular diastolic dysfunction. Therefore, early diagnosis or management of AS may be preventive in terms of right and left diastolic dysfunctions.
In diabetic patients, impaired endothelial functions and increased arterial wall thickness potentially lead to diabetic micro- and macrovascular complications. Therefore, it is crucial to maintain ED and arterial elasticity by means of intensive diabetes treatment and improved glycaemic control. Additionally, management of ED and AS with certain experimental drugs and/or some medication used in clinical practice is promising regarding prevention of diabetic complications.12 –14 Furthermore, management of ED and AS is likely to decrease the incidence of cardiovascular disease, which is an important cause of mortality in DM.
In conclusion, our study demonstrated ED and impaired arterial elasticity in paediatric patients without diabetic complications. ED and impairment in elasticity properties in patients with type 1 DM whose diabetes complications have not yet developed and systolic functions were preserved as both a prognosis indicator for possible diabetic complications and atherosclerosis and the cause of diabetic complications. Therefore, prevention of significant complications which may occur in diabetic patients may be aimed with intensive diabetes treatment, improvement in glycaemic control, endothelial function and protection of arterial elasticity properties and/or early treatment of ED. Additionally, correlation was shown between increased ED and AS and right and left ventricular diastolic dysfunctions.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by Scientific Research Projects Management Unit of Akdeniz University with the Project Number 2011.04.0103.025.
