Abstract
Background
Sleep insufficiency is known to negatively impact on glucose metabolism. Consequently, there is interest in determining the impact of improving sleep on glucose metabolism. We conducted a meta-analysis of studies that aimed at improving sleep using cognitive behavioural therapy for insomnia (CBT-I) and/or sleep hygiene or sleep extension on glucose metabolism.
Methods
Searches were performed on MEDLINE, EMBASE, CINAHL and Cochrane. We included studies featuring adults≥18years, a sleep intervention and glycaemic measurements. The pooled mean differences were calculated by the inverse variance method.
Results
24 studies (15 CBT-I and/or sleep hygiene; 9 sleep extension) were included. Meta-analysis of 12 studies (n = 2,044) of CBT-I and/or sleep hygiene demonstrated a significant reduction in HbA1c of 0.27% (95% CI 0.07, 0.47, I2 74%, p = 0.008) compared to control. In T2DM (n = 1,911; 9 studies), HbA1c level decrease was 0.43% (0.19, 0.67, I2 59%, p = 0.0004). There were no significant changes in fasting blood glucose analyses nor in any sleep extension intervention. For quality assessment, only 9 studies had low concern.
Conclusions
Using CBT-I and/or sleep hygiene interventions led to significant reductions in HbA1c levels, which were clinically meaningful in T2DM. Addressing sleep insufficiency should be an integral part of diabetes care.
Registration
PROSPERO Identification number: CRD42022376606.
Keywords
Key messages
• Sleep insufficiency is known to be associated with incident type 2 diabetes mellitus (T2DM) or worse glycaemia control in those with established T2DM. • Hence, there has been much interest in evaluating the impact of improving sleep on glucose metabolism. • Our meta-analysis demonstrated using cognitive behavioural therapy for insomnia and/or sleep hygiene was associated with a significant decrease in HbA1c levels of 0.27% and 0.43% in the total cohort and people with T2DM respectively, compared to control. • These findings suggest more emphasis should be placed on detecting sleep insufficiency in clinical care, particularly for people with T2DM.
Introduction
Sleep insufficiency, either as a function of poor quality, timing, or duration, are known to negatively impact on glucose metabolism.1–4 They increase the risk of type 2 diabetes mellitus (T2DM) in the general population, predict progression to T2DM in people with pre-diabetes and worsen glycaemic control in those with established T2DM.1–4 Consequently, there is much interest in determining the impact of improving sleep on glucose metabolism. Interventions include pharmacological therapies, sleep manipulation techniques including education/ sleep hygiene, cognitive behavioural therapy for insomnia (CBT-I) and/ or sleep extension. A further option is continuous positive airway pressure (CPAP) specifically in the context of obstructive sleep apnoea (OSA).
Sleep intervention techniques can be applicable to a range of sleep disturbances and behaviours and do not involve taking medications or wearing equipment. If these interventions were demonstrated to have a beneficial impact on glucose metabolism, they could be used to complement existing lifestyle-based interventions that are already recommended for people with pre-diabetes and T2DM. 5 As the prevalence of T2DM and its complications increase, there is a need for strategies to prevent T2DM and reduce its health and economic burdens.
Two previous meta-analyses did not find any significant impact of CBT-I and/or sleep hygiene or sleep extension on glucose metabolism.6,7 However, the results were not analysed according to baseline glycaemic categories (i.e. people with and without T2DM), possibly due to the low number of studies available at the time. As such, it is unknown if these interventions have a differential impact on specific glycaemic categories.
Given the increasing number of published studies in this area, the aim is to address this issue by analysing results for each intervention according to glycaemic categories. We hypothesised that in people with T2DM, whom have a stronger degree of glucose intolerance, the observed changes in glucose metabolism through use of sleep interventions would be larger than in people without T2DM, mirroring the observed effects of using diet and exercise lifestyle programs in people with and without T2DM, though some changes would be expected in both groups.8–10
The aim of this systematic review and meta-analysis was to assess the impact of non-pharmacological sleep interventions on glucose metabolism according to baseline glycaemic category.
Methods
This systematic review was registered on International Prospective Registry of Systematic Reviews (PROSPERO Identification number: CRD42022376606) database and meets the requirements of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 11
Data sources, search strategy and study selection
The search strategy was developed by SM and AT, in collaboration with an experienced local hospital health sciences librarian, to identify relevant studies using MEDLINE, Embase, CINAHL and Cochrane bibliographic databases. Databases were searched using a combination of appropriate subject headings for indexed articles, tailored to individual bibliographic databases, where required, and free-text terms relevant to the framed question of this review, which were combined using Boolean operators. The full electronic search strategy used for one database is included in Appendix 1. The first literature search was performed from inception up to 16th November 2022; an additional search was performed until 19th January 2024.
After conducting the electronic database searches, duplicates were removed and two investigators (SM and WH) independently screened and assessed titles and abstracts of identified studies for eligibility. The full text of these papers was retrieved and independently assessed for inclusion by the same two reviewers. Discrepancies were resolved through a third investigator (AT) where necessary until a consensus was reached. The reference lists of review articles and of included original publications were also screened for potentially relevant studies. For any relevant trial protocols derived from the database searches, the corresponding publication was searched for separately. We included studies of any language. We focused on studies published as full text papers, but we considered abstract publications, if further information/ data could be provided by the original authors, following methods used in a similar systematic review. 6
We included intervention studies of adults ≥18 years old at baseline that were (a) randomised or non-randomised trials, quasi experimental, laboratory or single-arm intervention studies, (b) consisted of a sleep intervention as the exposure (sleep extension, education, hygiene and/ or cognitive behaviour therapy) and (c) measured either blood glucose levels (glycated haemoglobin, HbA1c, fasting blood glucose, FBG, and/ or two-hour blood glucose), glycaemic metrics/ measures derived from electronic devices (e.g. continuous glucose monitoring devices for time in target glucose range) or surrogate markers of glycaemia (e.g. insulin level), at both baseline and follow-up.
In the case of a two-arm study, the comparator was considered as either a control group (i.e. no intervention), a lifestyle program or advice session consisting of diet and exercise modifications (considered as standard care in pre-diabetes and T2DM), or certain other interventions (e.g. mindfulness, meditation). If the sleep intervention was part of a multifactorial lifestyle intervention, the study was included only if control arm consisted of the same lifestyle intervention without the sleep component. For this reason, in the case of any three-arm studies, we only included data from two arms.
The exclusion criteria consisted of studies that (1) did not feature a sleep intervention (2); consisted of interventions of CPAP for OSA, bright/ dim light exposure, pharmaceutical therapies or laboratory based sleep restriction/ deprivation (unless as a part of CBT-I) as the exposure; (3) did not report measures of the same glucose metabolism marker at both baseline and follow-up or (4) studies of type 1 diabetes mellitus or (5) studies where reported glucose marker may not be accurate (e.g. HbA1c levels in dialysis studies of end-stage renal failure).
If multiple published reports from the same study were available, we aimed to include the report with the most detailed information for both exposure and outcome, unless the essential outcomes were presented in different publications.
Data extraction and quality assessment
One investigator (SM) extracted the data and the second investigator (WH) independently checked for consistency. Any discrepancies that existed were discussed in arbitration (SM, WH and AT) until a consensus was reached. Data was extracted on the following: first author’s surname, year of publication, country of origin of the population studied, type of study (e.g. randomised trial), sample size of intervention and control arms, population demographic characteristics (e.g. age, sex, body mass index), baseline glycaemic status of the population, sleep variable(s) under analysis (subjective and/ or objective measures), type of sleep intervention, length of follow-up, measure(s) used to assess change in sleep variable, type of glycaemia marker(s) measured, and the changes in the levels of the sleep variable and glycaemia at baseline and follow-up.
Sleep interventions were categorised as either (a) CBT-I and/ or sleep hygiene, in which the sleep outcomes tend focus on sleep quality or (b) sleep extension, which focuses more on sleep duration (SLD) or time in bed (TIB). These two categories also follow those that were used in a previous systematic review. 6
Study quality assessment was performed independently for parallel arm or cross-over studies by two reviewers (SM and WH), according to Cochrane risk-of-bias tool for randomized trials version 2 (ROB-2), as this is commonly used and recommended tool for assessing risk of bias in randomised trials. 12 Reviewers met to compare results and reach consensus. ROB2 includes five assessments of possible bias including randomisation process, deviation from proposed interventions, missing outcome data, outcome measurement, and result reporting. Studies were judged to have low, moderate or high risk of bias for each assessment.
Statistical analysis
Data was analysed quantitatively using meta-analysis if there were three or more studies that used similar sleep interventions and reported the same outcome variable. Otherwise, we proceeded with summarising the data qualitatively/ narratively. Single-arm interventions were excluded from meta-analysis as no comparator group was available.
Firstly, data was analysed in the total population and then according to glycaemic category (those with T2DM compared to non-T2DM). Where sufficient data existed, we used categories of normal glucose tolerance, pre-diabetes and T2DM. In cases where cohorts had a mixture of glycaemic categories, if one category was vastly more prevalent over others, it would be placed in this category. For example, a cohort of 90% with T2DM and 10% with pre-diabetes would be categorised under T2DM; in contrast a cohort with a mean HbA1c level of 5.5% (standard deviation, SD, 0.4) would be placed under non-T2DM.
Where data in the original publication was reported as both intention-to-treat and per protocol analyses, we selected the former to reduce the impact of bias.
Where relevant data was not available in the original publication, but was published in subsequent meta-analyses, we extracted the data from the latter to ensure a complete dataset and for better comparison with previous systematic reviews. Otherwise, we contacted the first author of the original publication for the most essential results.
The outcome variables were reported as mean (SD). When the original study reported outcomes as median (interquartile range, IQR) or mean (95% CI), these were converted to mean (SD) using accepted methods. 13
We estimated the pooled mean differences (and 95% CI) of the variables of interest, considered as the effect size for all studies. These were calculated using the inverse variance method and through using a random-effects model, as we anticipated there would be some degree of study-level variability in the exact interventions used. We conducted a brief sub-analysis using fixed effects model. Heterogeneity among studies was quantified by I2 statistic. We considered low, moderate, and high I2 values to be 25, 50, and 75%, respectively. 14
All statistical analyses for the meta-analysis were performed with Review Manager (RevMan) software Version 5.4.1 (Copenhagen, Cochrane Collaboration) and all tests were two-sided with p-level of p < 0.05 considered as statistically significant.
Results
Identification of studies
There were 901 abstracts derived from the search strategy, from which 33 met inclusion criteria for full text analysis; of these 24 studies were included in the systematic review (Figure 1 including the reasons for exclusion;15–38,
41
). Flow chart to demonstrate the process of study selection. Key: CBT-I = cognitive behaviour therapy for insomnia, n= number. * For reasons excluded, there may have been more than 1 reason, however the allocated category represents the first obvious reason for exclusion.
Summary of included studies and study characteristics
Baseline characteristics of studies featuring sleep interventions.

Continuous variables represented as mean (SD) or median (IQR), except for.
aRepresents the geometric mean with 95% CI.
bUnits for variables: T2DM dx: years, FBG, MDG and 2hr-PG: mmol/l, HbA1c: %, SLD, TIB and TST in minutes.
Assessment of study quality
From the 20 parallel-arm or cross-over studies considered for study quality, only nine studies had low concern throughout all five assessment categories, with most other studies having moderate concern in one to three of the categories, Appendix 2.
Interventions of CBT-I and/ or sleep hygiene
The interventions varied between CBT-I in 7 studies (Alshehri, Groeneveld, Tannas, Zhang, Zuo, Cain and Carroll) and sleep education/hygiene/advice in 8 studies (Cheung, Garcia-Serrano, Gunduz, Li, Pandey, Pereira, Chelsa and Duncan). The length and frequency of the interventions also varied (Table 1). All studies reported a positive impact on sleep measures, particularly for PSQI but some also reported improvements on the Insomnia Severity Index (ISI) (Alshehri, Groeneveld, Tannas); the only exception was a non-randomised trial (Chelsa) of general sleep advice.
In studies of people with T2DM, there were reductions in HbA1c levels observed in seven studies (Li, García-Serrano, Gunduz. Pandey, Tannas, Zuo and Zhang) but not in others (Alshehri, Groeneveld, Cheung and Pereira). García-Serrano et al. observed 29.4% of the intervention arm achieved a post-intervention HbA1c level decrease of >0.5%, considered a clinically significant HbA1c level reduction. FBG reductions were reported in some studies (Li and Cheung) but not others (Garcia). Cheung et al., conducted a long-term follow-up of median 6.2 years and reported a significant reduction in FBG levels in the intervention arm (6.9 mmol/l, 6.1-6.8, vs 7.1, 6.6-7.8, p = 0.027), with the authors suggesting a legacy effect of their sleep intervention on glucose metabolism.
In people without T2DM, none of the studies reported any changes in glucose markers of FBG or HbA1c levels where measured.
Sleep extension studies
All nine studies were classified as non-T2DM and were generally able to improve various sleep measures, but no study reported post-intervention lower levels of FBG. However, Reutrakul et al. reported a lower increase in mean FBG levels in intervention participants compared to control. Also, some studies reported post-intervention changes in glycaemic pre-cursor markers that favoured a lower diabetes risk: Hartescu et al. observed lower HOMA_IR levels; Killick et al. reported lower levels of fasting insulin and HOMA-IR and higher QUICKI levels; Matthew et al. reported lower glucose area under the curve levels, So-ngern et al. reported lower HOMA-IR levels, insulinogenic index and disposition index in those who extended SLD to greater than 6.0 hours/night.
The RCT of Cizza et al., was stopped early, as in the 2.7 months between baseline and screening, 125 participants significantly increased their SLD before undertaking any intervention, which led to significant reductions in levels of FBG, fasting insulin and an increase in Quantitative Insulin Sensitivity Check Index (QUICKI).
Meta-analysis
Data from these studies were extracted to conduct meta-analyses (Figures 2 and 3), where sufficient data was available. Zuo et al., and Zhang et al., were confirmed as separate studies hence both were included (personal communication from Professor Peian Lou, Xuzhou Center for Disease Control and Prevention, Jiangsu, China). We were unable to confirm if Li et al. and Cheung et al. contained some similar participants and as the latter was available as an abstract publication, we included Li et al. only. The effect of CBT-I and/or sleep hygiene on variables: (a) HbA1c levels in (i) Total cohort, (ii) T2DM and (iii) non-T2DM; (b) fasting blood glucose levels in (iv) Total cohort, (v) T2DM and (vi) non-T2DM; (c) PSQI in (vii) Total cohort, (viii) T2DM and (ix) non-T2DM; ISI in total cohort. The effect of sleep extension in people without T2DM on (a) Fasting blood glucose, (b) HOMA-IR, (c) Sleep duration and (d) PSQI.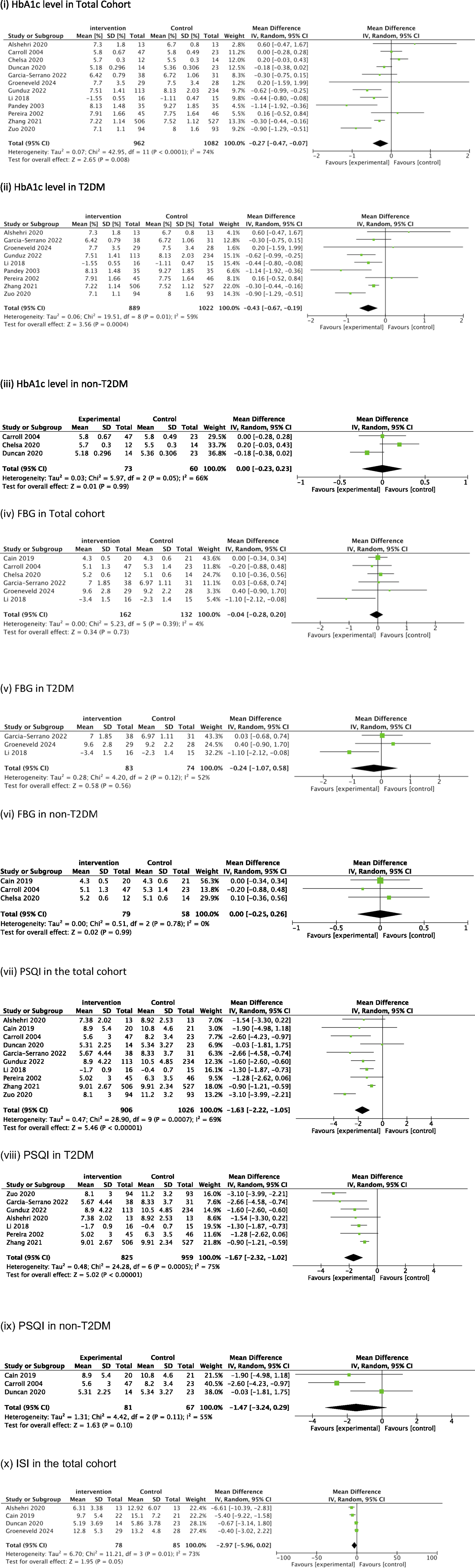

For interventions of CBT-I and/ or sleep hygiene, from 12 studies of 2,044 people, use of the intervention was associated with a significant reduction in HbA1c levels of 0.27% (95% CI 0.07, 0.47) p = 0.008, I2 for heterogeneity (I2) = 74%, compared to control. Considering the sub-analysis for 1,911 people with T2DM from 9 studies, use of the interventions demonstrated an HbA1c level decrease of 0.43% (0.19, 0.67), p = 0.0004, I2 = 59%. In contrast, for 133 people without T2DM from three studies, the HbA1c level did not change, 0.0% (−0.23, 0.23), p = 0.99, I2 = 66%.
Using a similar analysis where the sufficient data was available, we found use of the interventions did not reduce FBG levels in the total cohort (n = 6 studies) nor in those with or without T2DM (n = 3 studies).
Furthermore, use of the interventions was associated with a decrease in PSQI scores of 1.63 (1.05, 2.22), p < 0.00001, I2 = 69%, 1.67 (1.02, 2.32), p < 0.00001, I2 = 75% and 1.47 (−0.29, 3.24), p = 0.11, I2 = 55% in the whole cohort and in those with and without T2DM, where the number of included studies were 10, 7 and 3 respectively.
For sleep extension studies, from five studies with 106 people, use of the intervention did significantly change FBG levels, −0.01 mmol/l (−0.20, 0.17), I2 = 13%, p = 0.90. This was despite increasing SLD by 48.38 minutes (18.6, 78.16), I2 = 82%, p = 0.001 and decreasing PSQI score by 1.06 (0.13, 1.98), I2 = 53%, p = 0.02 in three and four studies respectively. HOMA-IR levels did not significantly change −0.21 (−0.45, 0.04), I2 = 18%, p = 0.1.
Using fixed effects models in place of random effects models, did not change the significance for associations.
Discussion
This systematic review and meta-analyses of non-pharmacological interventions is the first to demonstrate use of CBT-I and/ or sleep hygiene education leads to a significant reduction in glycaemic control in people with insufficient sleep. In people with T2DM the HbA1c level was 0.43% lower, a clinically meaningful reduction. Considering all individuals irrespective of their glycaemic status, the HbA1c level was reduced significantly by 0.27%, however there were only 133 people without T2DM, suggesting this result needs further investigation, especially for pre-diabetes.
Considering sleep extension interventions, despite successfully extending sleep by 48 min, on average, there was no change on FBG or HOMA-IR levels. Whilst epidemiological studies have associated short sleep duration and diabetes, our results suggest that sleep extension does not impact on glycaemic control, that is this was a true effect. However, we should also consider that these studies were conducted over shorter time frames, suggesting there was less time for interventions to exert their effect on glucose metabolism. Also, within the systematic review the sleep extension interventions demonstrated improvements in some surrogate markers of glucose metabolism that favour early changes towards lowering glucose levels. Secondly, the sleep extension studies consisted exclusively of people without T2DM; our hypothesis suggested that larger glucose metabolism changes would occur in those with T2DM, as they possess a more severe degree of glucose intolerance, which appeared to be true for CBT-I and/or sleep hygiene interventions.
There were potential causes of heterogeneity in the meta-analysis impacting on the results. For the study designs, the interventions varied in length of time, number of sessions, the length of the subsequent follow-up and support given during the follow-up period. Other possible factors included the baseline population characteristics such as age, gender, BMI, and in those with T2DM, the level of glycaemic control and length of T2DM duration.
Meta-analyses of people with T2DM have demonstrated that poor sleep quality is associated with worsening of glycaemic control by an HbA1c level of 0.35% (95% CI 0.12-0.58). 4 Insufficient sleep is also known to be associated with future T2DM in the general population and also predict progression from pre-diabetes to T2DM.1–4 The plausible mechanisms explaining these associations centre on changes in beta-pancreatic cell function and insulin resistance/ sensitivity and include changes in hormones levels for catecholamines giving a sympathetic predominance, melatonin, cortisol and appetite regulating hormones such as ghrelin, leptin, endocannabinoids and glucagon-like peptide-1 and growth hormone. 39 The results of the present study suggest there may be potential to reverse the impact of these adverse sleep behaviours and disorders on glucose metabolism through CBT-I and/ or sleep hygiene interventions. The HbA1c reduction of 0.43% in T2DM from our meta-analysis is larger than that of other sleep interventions such using CPAP in people with T2DM and OSA of 0.24% (95% CI 0.06, 0.43), according to a recent meta-analysis. 40 Furthermore, meta-analyses of lifestyle interventions focusing on diet and/ or lifestyle in T2DM has shown variable impact on HbA1c reduction ranging between 0.37% (0.14, 0.59) to 0.82% (0.53, 1.11).8,9
Regarding limitations of the study, the quality of methods used in studies was variable as most studies were pilot trials. For example, one RCT of people with T2DM was conducted in a real-world setting and collected final follow-up data within 6 months of the first appointment, based on whenever the participant would return for their next clinical appointment. 10 Secondly, only some studies accounted for those people with T2DM who changed their diabetes medications during the trial. However, one reason for this may be because many studies were conducted over shorter periods in which medications could not have been changed from baseline. Thirdly, some studies analysed their data as per protocol and others as an intention to treat, only three studies provided both sets of data.33,38,41 Fourthly, for sleep extension studies used in the meta-analyses, some data was only available as mean change in the variable, however, this was also observed in two previous meta-analyses in this area.6,7
The strengths of this study are the systematic approach and robust methods used to obtain our results, ensuring they met accepted standards for meta-analyses. We considered a wide range of sleep behaviours and disorders as causes for insufficient sleep, as well as glucose related terms, which were used in the comprehension list of search terms in the search strategy. We analysed full-text articles and some abstracts for studies, featuring people with and without T2DM, allowing us to detect as many eligible studies as possible.
Regarding potential implications for clinical practice from the present study, we found use of CBT-I) and/or sleep hygiene in people with T2DM was associated with an HbA1c reduction of 0.43%, which can be considered clinically meaningful. As we excluded studies of OSA as this disease has a specific treatment (CPAP), detecting individuals with other forms of sleep insufficiency, followed by implementing interventions to improve their sleep, could be a cost-effective approach to improving glycaemic control without use of medications. Commonly used sleep assessment tools include the Insomnia Score Index and Pittsburgh Sleep Quality Index.42,43 Sleep interventions could be a useful adjunct to lifestyle advice and recommendations which traditionally focus on modification of diet and physical activity only. However more research is required to answer these questions and also if CBT-I and/or sleep hygiene can aid the prevention of T2DM in high-risk people with pre-diabetes.
In conclusion, use of CBT-I and/ or sleep hygiene interventions led to a clinically significant reduction in HbA1c levels of 0.43% in people with T2DM and insufficient sleep. Future research needs to explore the role of using sleep interventions in sleep insufficiency as a potential target to aid current measures of preventing T2DM complications and also in prevention of this disease in high risk people with pre-diabetes.
Supplemental Material
Supplemental Material - The effect of non-pharmacological sleep interventions on glycaemic measures in adults with sleep disturbances and behaviours: A systematic review and meta-analysis
Supplemental Material for The effect of non-pharmacological sleep interventions on glycaemic measures in adults with sleep disturbances and behaviours: A systematic review and meta-analysis by Samiul A Mostafa, Wasim Hanif, Francesca Crowe, George Balanos, Krishnarajah Nirantharakumar, Jason G. Ellis and, Abd A Tahrani in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
We would like to thank University Hospitals of Birmingham (a) Library team (especially Jennifer Davis-Manders and Anita Phul) for their support with developing and modifying the final search terms, conducting the searches and also for providing access to their systemic review training courses and resources (b) for statistical support through Mr James Hodgson, research statistician.
Author contributions
SAM – conceived the idea for the review, designed the study, developed the search strategy, conducted the literature searches, screened articles for inclusion, selected and retrieved relevant papers, determined inclusion/exclusion of papers, conducted data extraction, performed the analysis, performed the critical analysis and wrote the manuscript. SM is the guarantor of the data. WH - screened articles for inclusion, selected and retrieved relevant papers, determined inclusion/exclusion of papers, verified data extraction and drafted the manuscript. FC – drafted the manuscript. GB - drafted the manuscript. KN - drafted the manuscript. JE - drafted the manuscript. AAT - conceived the idea for the review, designed the study, developed the search strategy, supervised the meta-analyses and drafted the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declarations
SM undertook this research work through receiving a Clinical Research Network West Midlands Health and Care Research Scholarship. He has previously received research support from Novo Nordisk Research Foundation UK and Academy of Medical Sciences. AAT is currently an employee and share-holder of Novo Nordisk. Novo Nordisk had no role in this manuscript. The views expressed in this manuscript are those of the authors and not Novo Nordisk.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
