Abstract
Our study evaluates the interconnection between diabetes treatment in the community setting and diabetes treatment regimens in these patients during hospitalization.
Methods
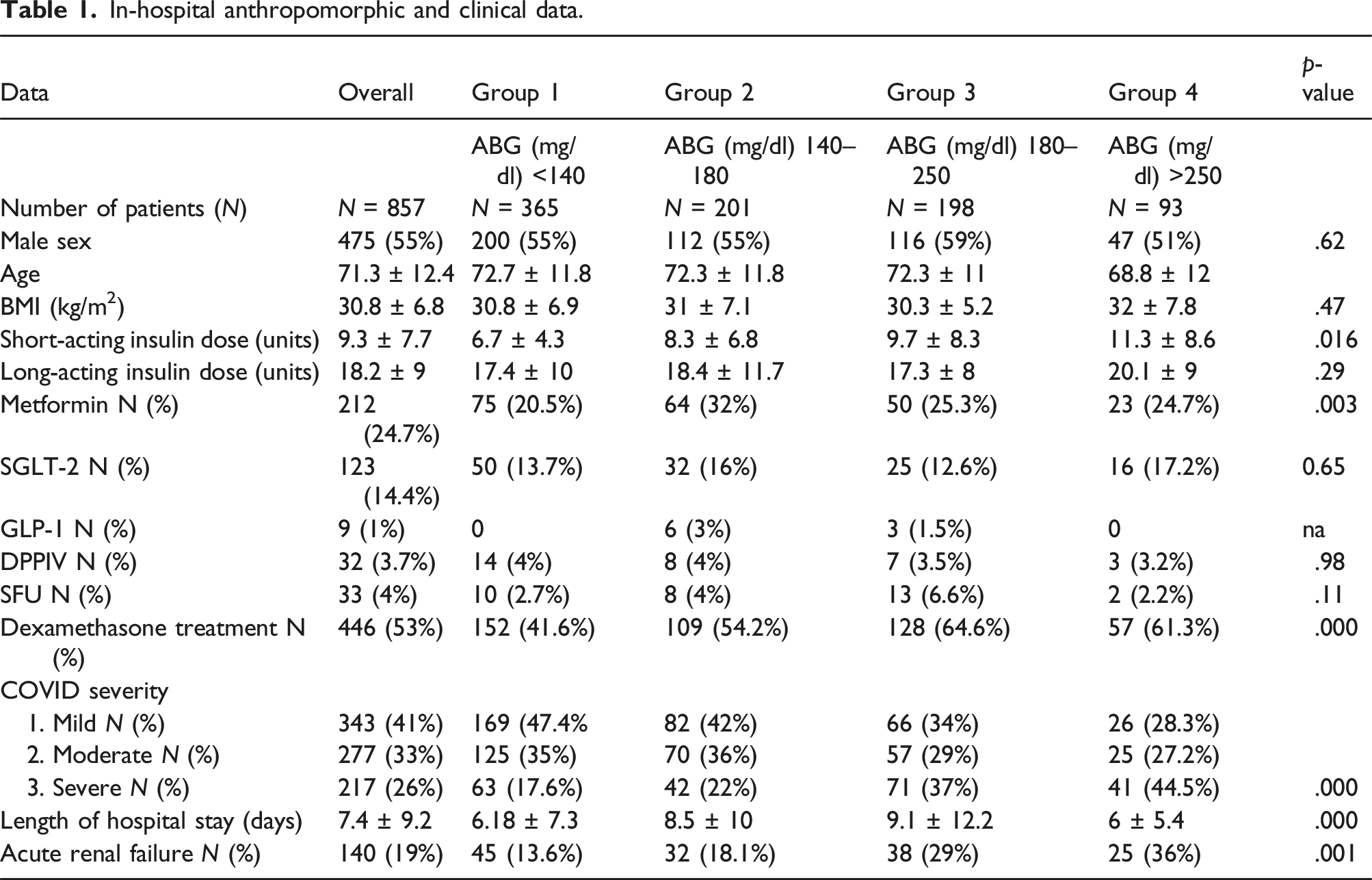
The patients were divided into four groups according to blood glucose level during hospitalization. Group 1 included patients with an average blood glucose ≤140 mg/dl, Group 2 included patients with an average blood glucose level between 140 and 180 mg/dl, Group 3 included patients whose blood glucose level was between 180 and 250 mg/dl, and Group 4 included patients with average blood glucose >250 mg/dl. In all subjects, we assessed preadmission diabetes treatment and prior diagnoses of major comorbidities (atherosclerotic cardiovascular disease, congestive heart failure, chronic renal disease, chronic pulmonary diseases, and dementia.
Results
Significant predictors of mortality were the severity of COVID-19 (OR 62, CI 95%; 18–235, p < .000), acute renal failure (OR 3.2, CI 95% - 1.26–8.2; p = .015), and a diagnosis of congestive heart failure before hospitalization (OR 2.6; CI 95% 1.14–6.8; p = .024). Lower preadmission HbA1c levels, insulin treatment in the hospital, SGLT-2 treatment before, and absence of acute renal failure are significant predictors of good glycemic control during hospitalization.
Conclusions
In patients with type 2 diabetes hospitalized with COVID-19, poor long-term glycemic control is associated with the level of hyperglycemia during hospitalization.
Key messages
Acceptable glycemic control in outpatient settings is an essential factor in mitigating deleterious outcomes of COVID-19 disease in hospitalized patients with type 2 diabetes. Acute renal failure during hospitalization and prior diagnosis of congestive heart failure are significant predictors of unfavorable outcomes in these patients.
Introduction
Diabetes is a prevalent condition among hospitalized COVID-19 patients.1,2 Ensuring effective management of glycemic control throughout the hospital stay results in less severe complications, notably acute respiratory distress syndrome and acute kidney injury. 2 A previous study showed that maintaining optimal glucose levels in hospitalized patients (average blood glucose level of 115 mg/dL) compared to inadequate glucose management (average blood glucose of 197 mg/dL) is linked to a reduced hazard ratio (HR) for mortality (HR 0.13; 95% confidence interval [CI], 0.04–0.44; p < .0001) and a decreased risk of complications from COVID-19. 3 Novel antihyperglycemic medications, including GLP-1 receptor agonists and SGLT-2 inhibitors, as well as metformin and DPPIV inhibitors, could be safely continued in hospital settings provided there are no contraindications. 4 Preadmission HbA1C level influences the length of hospital stay, rate of complications, and long-term mortality in non-COVID patients. 5 Our study investigates the correlation between glucose control before and during hospitalization and the mortality rates within 30 days of admission.
Methods
The current study provides data from Clalit Health Services using a data-sharing platform powered by MD clone. The study included data on patients hospitalized with COVID-19 between April 1, 2020, and March 31, 2021.
Inclusion criteria were type 2 diabetes mellitus, COVID-19 diagnosis, age ≥18 years. Exclusion criteria were pregnancy, type 1 diabetes, and diabetic ketoacidosis.
The baseline characteristics included gender, age, BMI, glomerular filtration rate (GFR), preadmission diabetes treatment, and specific comorbidities related to diabetes or that could influence COVID-19 outcomes.
We divided the patients into four groups according to average blood glucose (ABG) during hospitalization. The first group included patients with ABG <140 mg/dl, the second group with ABG 140–180 mg/dl, the third group with ABG 180–250 mg/dl, and the fourth group with ABG >250 mg/dl during hospitalization.
Preadmission diabetes treatment was retrieved and included medication refills within 3 months before hospitalization: a combination of long-acting and short-acting insulin, SGLT-2 inhibitors, GLP-1 agonists, DPPIV inhibitors, and sulfonylurea.
The most recent HbA1c for each patient (within 6 months) before and after hospitalization was retrieved. ABG during hospitalization was determined as a sum of fasting and postprandial glucose readings divided by the number of measurements. COVID-19 severity was determined according to NIH criteria. 5
Statistical analysis
We used ANOVA analysis for continuous variables (BMI, HbA1C, insulin dosages, length of hospital stay). Categorial variables (sex, HbA1C ranges <7, 7–9, >9, BMI ranges) were compared using the χ2 test.
The parameters included in logistic regression analysis related to death prediction included essential variables such as age, sex, BMI and the variables associated with COVID-19 mortality included length of hospitalization, COVID-19 severity, comorbidities (congestive heart failure, atherosclerosis, chronic kidney disease), insulin treatment during hospital stay, metformin treatment during hospital stay, SGLT-2 treatment during hospital stay, treatment with dexamethasone, CRP level, presence of acute kidney injury. The method of inserting variables into models is as a single piece (ENTER). The odds ratio index was calculated to examine the likelihood of the outcome in the entered parameters.
Kaplan -Meier survival analysis tested the differences in the probability of survival between two dependent variables: the probability of death 30 days after admission and the group of blood glucose (from group 1 to group 4).
Ordinal regression analysis was used to predict diabetes control with the set of independent variables. The parameters included in this multivariate analysis were age, sex, BMI, length of hospitalization, COVID-19 severity, comorbidities (congestive heart failure, atherosclerosis, chronic kidney disease), insulin treatment during hospital stay, metformin treatment before and during hospital stay, SGLT-2 before and during hospital stay, and treatment with dexamethasone. A p-value of <.05 was considered statistically significant. Analyses were performed with SPSS software (version 24.0; IBM).
Results
A total of 857 patients met the inclusion criteria.
In-hospital anthropomorphic and clinical data.
Relationship between average blood glucose and outcomes
The ABG level during hospitalization was 166.7 ± 64.7 mg/dl and varied from 112.7 ± 16.5 in Group I to 303 ± 45 in Group IV (p = .000). Ordinal regression model for prediction of blood glucose control during hospitalization (R2 = 0.957 p = .000) revealed that preadmission HbA1C level (OR 1.8; CI 95% 1.5–2.1, p < .001), SGLT-2 treatment before hospitalization (OR 0.47; CI 95% 0.28–0.79, p = .005), insulin treatment in the hospital (OR 0.2; CI 95% 0.12–0.3, p < .000), and presence of acute renal failure (OR 2; CI 95% 1.18–3.43; p = .01) are significant predictors of blood glucose control during hospitalization.
Kaplan-Meier survival analysis revealed a significantly higher survival probability within 30 days in groups 1 and 2 patients than in groups 3 and 4 (p = .005, Figure 1). The distinction arises from the results observed in Group 4. (Figure 1). The Kaplan-Meier survival probability during 30 days admission according to the groups of glucose control.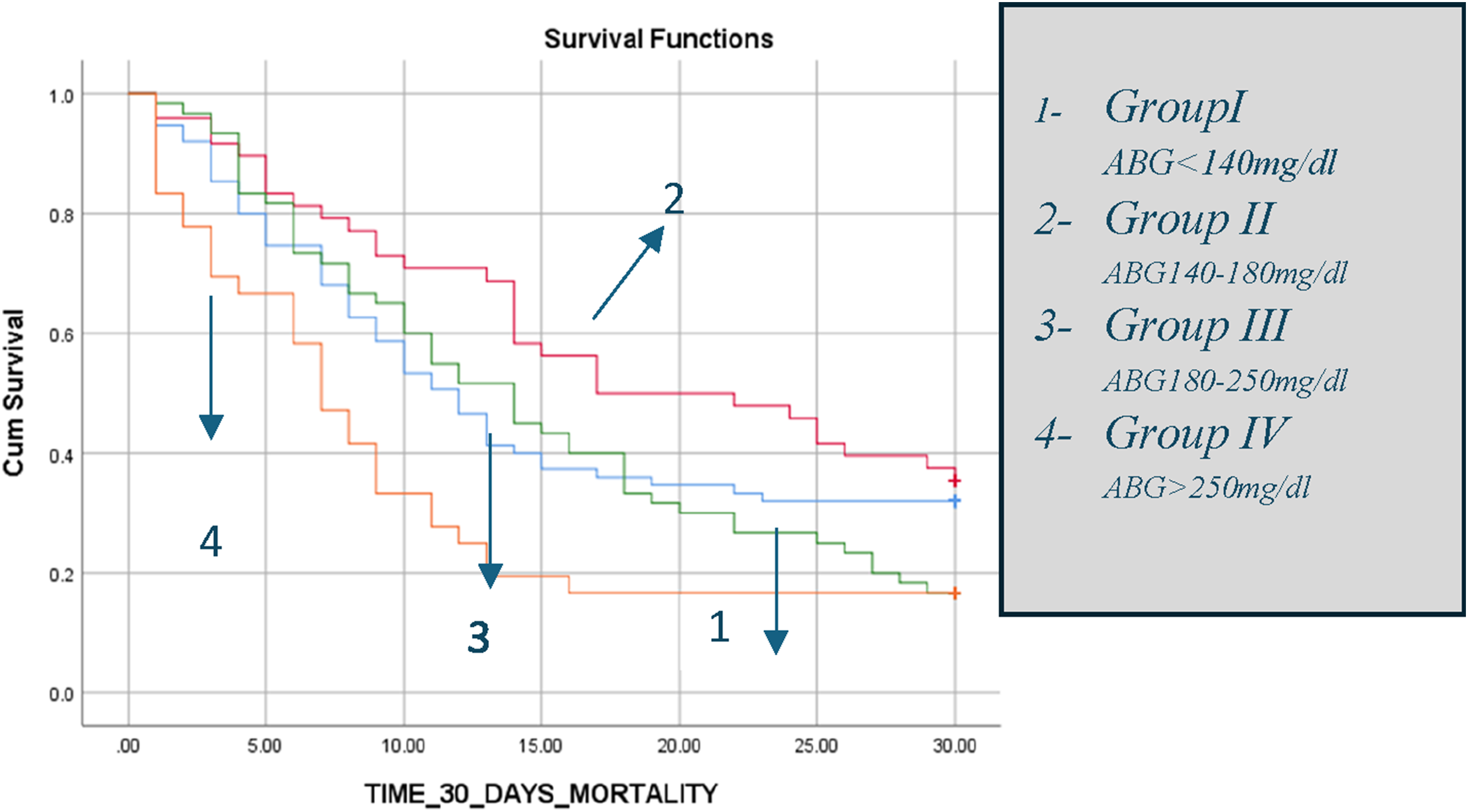
Effects of COVID severity and associated comorbidities on outcomes
Among the 837 patients with an assessment of COVID-19 severity, 343 patients (41%) had mild disease, 277 (33%) had moderate disease, and 217 patients (26%) had severe disease.
440 patients (51.3%) had cardiovascular disease before hospitalization, 264 patients (31%) had chronic pulmonary disease, 319 (37%) had chronic kidney failure, and 244 (28.5%) had congestive heart failure diagnosis.
Acute deterioration of renal function developed in 19% of patients overall, but significantly more patients in the uncontrolled group developed acute renal failure during hospitalization. Significantly more patients in groups 3 and 4 had severe COVID-19 compared with the two groups of patients (1 and 2), with better glucose control (37% and 45.5% vs17.6% and 22%, respectively, p = .000). A logistic regression model for predicting the death probability at 30 days since admission was significant (R2 66%, p = .000). The severity of COVID (OR 12.4, 95% CI: 6.53–25.7; p = .001), a preadmission diagnosis of congestive heart failure (OR 2.43, 95% CI 1.04–5.82; p = .04), and acute renal failure (OR 3.85, 95% CI 1.52–10.1; p = .005) were found to be significant predictors.
The treatment with dexamethasone was not significantly correlated with mortality (OR 0.79; CI 0.28–2.18; p = .64) and glucose control (OR 0.81; CI 95% 0.49–1.3, p = .39).
Impact of antihyperglycemic therapy before hospital admission on glucose control
Before hospitalization, 165 patients (19%) were treated with long-acting insulin, and 60 patients (7%) received short-acting insulin. 342 patients (40%) received metformin, 86 patients (10%) had DPPIV, and 33 (4%) used SFU. Eighty-eight patients (10.3%) received GLP-1 agonists, while 102 (12%) had SGLT-2. (Table 1).
Preadmission HbA1C levels were significantly different in all study groups, with the lowest level in Group 1 and the highest in Group 4 (6.6 ± 1.16 vs 8.6 ± 1.9 mg%, p < .0001).
The ordinal regression model related to glucose control during hospitalization was statistically significant, χ2 (df) = 13, p < .001. SGLT-2 inhibitors (OR 2.1, 95% CI 0.22–1.22, p = .005) and HbA1C levels before hospitalization (OR 2.2, 95% CI 1.76–2.7; p < .001). were related to blood glucose control in the hospital.
Discussion
According to our research findings, elevated blood glucose levels are related to lower survival probability in hospitalized COVID-19 patients. Patients with average blood glucose levels <140 mg/dl and 140–180 mg/dl (groups 1 and 2) had a better survival probability in comparison to non-controlled groups (groups 3 and 4). These data confirm the results from previous studies related to COVID-19 infection.2,6 The ordinal regression model related to predicting blood glucose control during hospitalization was statistically significant and included metformin. However, preadmission HbA1c levels, SGLT-2 treatment before hospitalization, and insulin treatment during hospitalization were better predictors of controlled glucose levels. A correlation between insulin treatment in COVID-19 patients and increased mortality was found in some, but not all, studies.7,8 This finding aligns with a recent study, 7 which identified a notable reduction in mortality rates associated with all anti-diabetic medications administered before hospitalization, including insulin therapy.
Our findings indicate that insulin treatment during hospitalization is not associated with increased mortality (OR 1.8, CI 95% 0.69–4.9, p = .2). However, it was related to better glucose control during hospitalization (OR 0.2, CI 95% 0.12–0.31, p < .0001). The adverse outcomes attributed to insulin treatment 8 probably reflect the fact that patients with insufficient glucose control before hospitalization had more comorbidities because of long-standing diabetes with relative insulin insufficiency. Indeed, 34% of patients in Group 3 and 41% in Group 4 were treated with long-acting insulin versus 9.6% and 16% in Group 1 and 2, respectively (p = .000).
Treatment with SGLT-2 inhibitors is a gold standard for patients with heart and kidney failure with or without diabetes. 9 Still, the benefit of SGLT-2 treatment during hospitalization remains obscure. A recent meta-analysis, based on 17 studies, showed that treatment with SGLT-2 inhibitors before admission was associated with survival benefits and reduced severity of COVID-19 in patients (OR 0.69; 95 % CI: 0.56 – 0.87, p = .001. 10 Interestingly, this effect was not connected to heart failure. 8 Nevertheless, a randomized placebo-controlled study that included 1250 patients with at least one cardiometabolic risk factor did not find a benefit from dapagliflozin treatment in terms of death and complication in COVID-19 hospitalized patients. 10 In line with a recent meta-analysis data, 8 our study found that SGLT-2 treatment before hospitalization was associated with a positive effect on blood glucose during hospitalization (OR 0.48, CI 95% 0.27–0.79, p = .005). Two randomized control studies investigated early treatment with metformin in ambulatory COVID-19 patients with and without diabetes.11,12 No additional benefit was found regarding the incidence of cardiovascular, kidney, respiratory complications or all‐cause mortality, and no clinical recovery improvement compared to the standard treatment.
In contrast to these studies, a comprehensive meta-analysis examining the effects of metformin on COVID-19 outcomes synthesized data from 20 studies. 11 studies demonstrated a statistically significant association between metformin treatment and improved survival rates in COVID-19 patients. 8 Consistent with previous studies reporting the benefits of metformin, our findings indicate that pre-hospitalization metformin use is significantly associated with reduced mortality (OR 0.3; 95% CI 0.11–0.23, p = .013). However, our data showed no statistically significant correlation between in-hospital metformin use and glucose control (OR 0.71; 95% CI 0.39–1.3, p = .27).
In the same way, we found that metformin in-hospital use did not impact survival rates. Conflicting data existed on GLP-1 agonists and DPPIV inhibitors regarding COVID-19 outcomes.8,13,14 In a meta-analysis of 61 studies, only preadmission antidiabetic medications were included. DPPIV inhibitors were found to be related to increased OR of death (OR 1.23; CI 95% 1.07–1.42), whereas GLP-1 agonists had a beneficial effect (OR 0.51; CI 95% 0.37–0.69). 14 In our study, about 10% of patients were treated with DPPIV and GLP-1 before hospital admission, and only a few received DPPIV and GLP-1 during hospitalization. We did not find that GLP-1 agonists or DPPIV inhibitors were related to glucose control or death probability.
Diabetes-related comorbidities have been associated with unfavorable COVID-19 outcomes. Many studies showed a significant increase in hospitalization rates, severity of COVID-19, and death in patients with diabetes and preexisting comorbidities.2,15 Higher GFR levels during hospitalization have also been found to correlate with better outcomes. 7 A recent study examined the COVID-19 outcomes in patients with both type 2 diabetes mellitus and cardiomyopathy, comparing them to a broader cohort of type 2 diabetes patients. The findings revealed a significantly higher mortality rate among those with both conditions compared to the general diabetes population (2.6% vs 1.2%, p < .001). 16 In the same way, in our study, congestive heart failure diagnosis before hospitalization significantly correlated with 30-days mortality.
We also examined the effects of dexamethasone on our COVID-19 patients.
Dexamethasone is beneficial in severely affected patients requiring invasive mechanical ventilation or oxygen therapy. 17 In our patient’s cohort, out of 835 patients, 446 (53%) received dexamethasone treatment, with 217 classified as having severe disease. Contrary to some previous studies,17,18 this research found no significant correlation between dexamethasone treatment and mortality risk (OR 0.79; 95% CI 0.28–2.18; p = .64). The maximal benefit of dexamethasone treatment was observed early in the COVID-19 pandemic, before widespread immunization and antiviral treatment that can mitigate excessive inflammation. Dexamethasone was also included in the ordinal regression model related to glycemic control during hospitalization. Treatment with dexamethasone was not correlated with glucose control during hospitalization (OR 0.81; CI 95% 0.49–1.3, p = .39). We speculate that short-term use of dexamethasone use in relatively stable patients may not significantly impact glucose levels. However, in severely ill, poorly controlled patients, the extreme glucose fluctuations may mask any effects of dexamethasone.
In conclusion, we conducted a retrospective real-data study to determine the impact of diabetes control before and during hospitalization on outcomes of patients hospitalized with COVID-19. In line with the previous knowledge, we found that in COVID-19 patients, adequate diabetes control before hospitalization is related to better blood glucose control in the hospital and less severe COVID-19. 3 However, a higher survival probability at 30 days is associated with better glucose control during hospital stay. Probably, blood glucose may be a more relevant predictor for mortality because it may be interpreted as a mirror of systemic inflammation related to the severity of COVID-19. Hospital treatment with insulin achieved better blood glucose control but did not lead to better survival. Pre-hospital treatment with metformin, but not in-hospital use, correlated with better survival rates. SGLT-2 inhibitor use before hospital admission was correlated with improved glucose control but did not affect survival.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
