Abstract
The newfound knowledge in type 2 diabetes (T2D) during the past decade for the sodium-glucose cotransporter-2 inhibitors (SGLT-2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA) is wealthy in favorable results for key patient-important outcomes including morbidity, mortality and health-related quality of life (HRQoL). The SGLT-2i and GLP-1RA offer cardiovascular and renal protection beyond their glucose lowering effect, reduce body weight and hypoglycemia and improve diabetes-related distress, physical function and HRQoL. Along with the fixed-ratio combinations of basal insulin/GLP-1RA, they make feasible a regimen simplification and de-escalation from high dose and multiple injections of insulin reducing treatment burden. Besides cardiorenal risk reduction, the SGLT-2i and GLP-1RA reduce the incidence of depression, cognitive decline, respiratory disease, gout, arrhythmias and other co-occurring conditions of T2D, namely multimorbidity, which frequently complicates T2D and adversely affects HRQoL. The alleviation of multimorbidity by the pleiotropic effects of the SGLT-2i and GLP-1RA, could improve patients’ HRQoL. The use of the SGLT-2i and GLP-1RA should be increased within a shared decision-making in which they are reframed as cardiorenal risk-reducing medications with the potential to lower blood glucose. By improving outcomes that patients may highly perceive and value, the SGLT-2i and GLP-1RA may facilitate the contemporary person-centered management of T2D.
Keywords
Key messages
• The sodium-glucose cotransporter-2 inhibitors (SGLT-2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA) correspond to key patient-important outcomes in type 2 diabetes (T2D), beyond their glucose lowering effect. • The SGLT-2i and GLP1-RA reduce mortality, treatment burden, hypoglycemia and body weight, whilst improve physical function and health-related quality of life (HRQoL). • The SGLT-2i and GLP1-RA reduce metabolic, obesity-related and general multimorbidity (conditions co-occurring with T2D), which adversely affects HRQoL. • By alleviating MM, the SGLT-2i and GLP1-RA could improve the HRQoL of patients living with T2D.
Introduction
The glucose-centric management of type 2 diabetes mellitus (T2D), has recently been challenged if it corresponds to outcomes that patients perceive and value, namely patient-important outcomes (PIO). 1 PIO in T2D are related to the reduction of clinical events and mortality, the improvement of symptoms profile, functioning and quality of life (QoL) as well as the reduction of treatment burden and hypoglycemia. 2 Evidence from meta-analyses of randomized controlled trials (RCT) supports that intensive, compared with moderate glycemic control, exhibits no meaningful benefit for microvascular and macrovascular PIO except of a reduced rate of non-fatal myocardial infarction; instead increases adverse PIO such as the risk of severe hypoglycemia, weight gain and treatment burden. 1 Furthermore, the recent cardiovascular and renal outcomes trials in T2D showed that the glucose-lowering medications (GLM) sodium-glucose cotransporter-2 inhibitors (SGLT-2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA), offer cardiovascular and renal protection irrespectively of a substantial lowering of glycated haemoglobin (HbA1c), suggesting that it is the use of the specific medications and not intensive glycemic control that matters with respect to improving PIO. 1
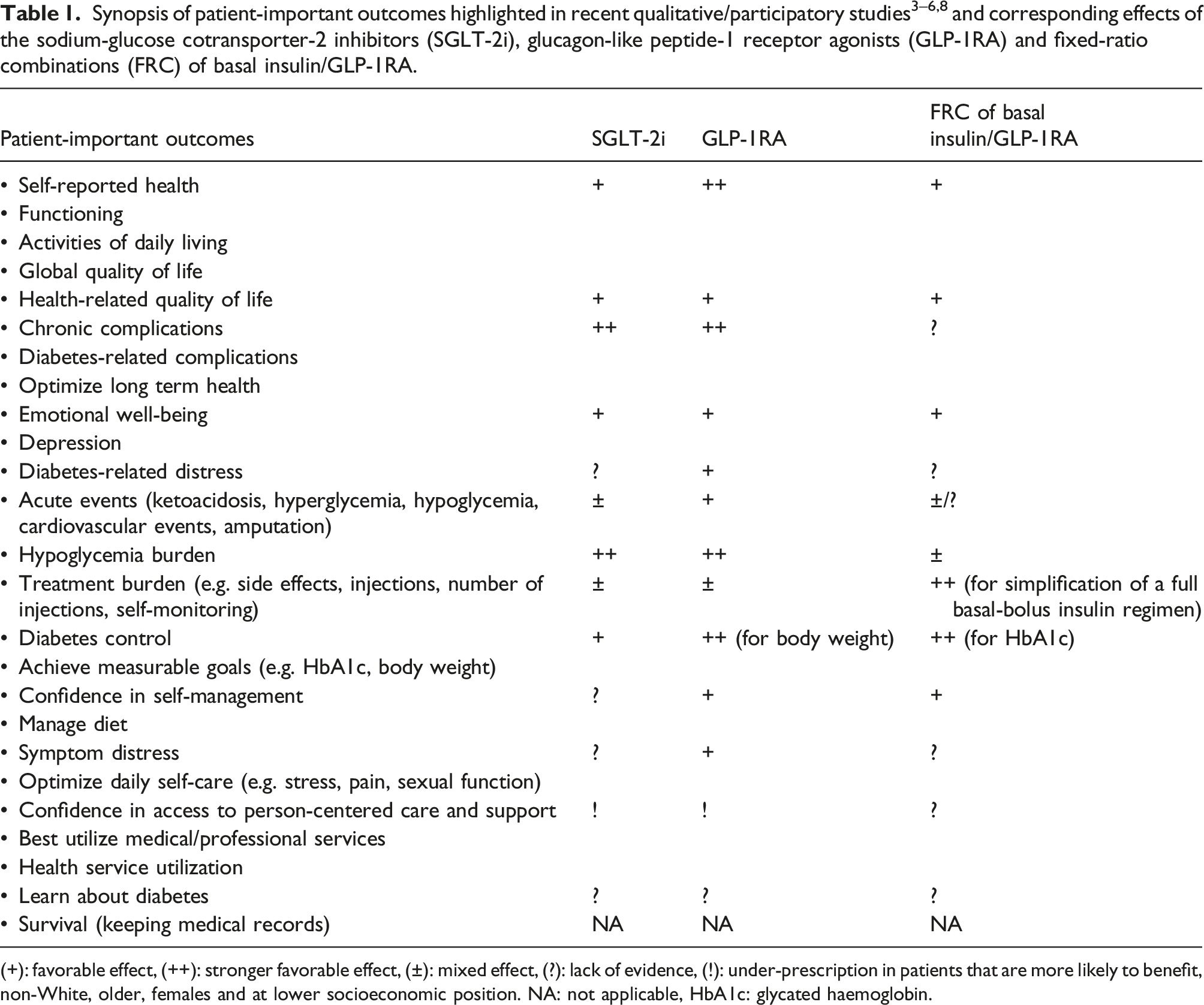
Patient-important outcomes from participatory/qualitative studies
Consequently, the contemporary paradigm of the management of T2D has shifted from a glucose-centric toward a person-centered approach that addresses the totality of patient’s medical, psychological and social needs with the goal of preventing the acute and chronic complications that really matter as PIO. 1 Many studies have recently tried to give voice to patients with diabetes by an equal participation in scientific panels or by qualitative methods, in order to identify core sets of PIO. For example, an international consortium of patients with diabetes, clinicians and researchers identified a standard set of person-centered outcomes related with diabetes control (e.g. HbA1c, body weight), acute events (e.g. hypoglycemia, ketoacidosis), chronic complications, health service utilization (e.g. rate of hospitalizations) and survival, while prioritized the routine assessment of emotional well-being, depression and diabetes-related distress (DRD). 3 Another consensus among patients with type 2 diabetes (PWD), clinicians, researchers and policy makers highlighted core outcomes related with diabetes care, global QoL/activities of daily living and diabetes-related complications. 4 A participatory research methods study involving patients with diabetes and family members, gave prominence to the following core constructs of patient-reported outcomes to be addressed within a person-centered diabetes care, at a national level: self-reported health/functioning, psychological well-being, DRD, impact on general QoL, symptom distress (e.g. pain, sexual function), treatment burden (e.g. medication taking, side effects), hypoglycemia burden and confidence in self-management/access to person-centered care and support. 5 Another qualitative study interviewing poorly-controlled patients with diabetes, in collaboration with patients’ advocates, grouped PIO into 7 broad domains: 3 were more medically defined (achieve measurable goals (e.g. lose weight), best utilize medical/professional services and manage medications (e.g. reduce medicines, insulin or injections)), but most (4/7) represented personal values and life goals: optimize long term health (e.g. prevent complications, maintain functional independence), optimize daily self-care (e.g. improve mental and sexual health), manage diet and learn about diabetes. 6
Implication of the SGLT-2i and GLP-1RA with PIO
(+): favorable effect, (++): stronger favorable effect, (±): mixed effect, (?): lack of evidence, (!): under-prescription in patients that are more likely to benefit, non-White, older, females and at lower socioeconomic position. NA: not applicable, HbA1c: glycated haemoglobin.
Impaired physical function (inability to perform basic activities necessary for daily living) and frailty (increased vulnerability to physical or psychological stressors) emerge as complications of T2D, besides microvascular and macrovascular disease 9 and their prevention as a PIO.4–6 Individual favorable biological effects of the SGLT-2i and GLP-1RA on inflammation and oxidative stress, endothelial function and cellular metabolism, erythropoiesis and vasculature, could improve cardiac and skeletal muscle function, serving physical function. 9 In particular, an observational study showed that older PWD treated with a fixed ratio combination (FRC) of basal insulin/GLP-1RA (glargine/lixisenatide), increase appendicular skeletal muscle mass compared with glargine alone. 10 A meta-analysis showed that the GLP-1RA, especially those associated with greater weight loss, improve patient-reported physical function 9 and interestingly, a recent review envisioned the SGLT-2i as novel anti-frailty drugs due to their favorable effects on several cardiorenal co-occurring conditions of T2D. 11 Perhaps the clinically important weight loss, alongside the cardiorenal benefits of the SGLT-2i and GLP-1RA, could improve physical function and prevent frailty.9,11
The results of recent studies regarding the SGLT-2i, GLP-1RA and FRC of basal insulin/GLP-1RA further facilitate a transition from an insulin-centric toward the person-centered approach in the management of T2D favoring PIO. For example, a simplification of treatment from a full basal-bolus insulin regimen to either a FRC of basal insulin/GLP-1RA or a combination of SGLT-2i plus basal insulin resulted in similar glycemic control with fewer daily insulin dose and injections (1 vs 4) and less hypoglycemia. 12 Another study comparing a full basal-bolus insulin regimen with a FRC of deglutec/liraglutide (iDeg/Lira), showed that PWD treated with iDeg/Lira achieve non-inferior glycemic control, weight reduction and less hypoglycemia, with fewer dose adjustments and daily injections (1 vs ≥3), while they experience a significant reduction in treatment burden and improvement in the mental component of HRQoL. 13 Although data for trending use is lacking and official guidance on initiation is unclear, the FRC of basal insulin/GLP-1RA represents a “well-balanced therapy” that maximizes the efficacy, while minimizes the disadvantages of the two injectable mono-components, improving patient-reported outcomes and glycemic control of inadequately controlled PWD. 14
The psychological burden of T2D is an important aspect of daily clinical practice. 1 Depression and DRD are patient-reported outcomes that frequently complicate the lives of PWD and compromise the ability to deal with the multimorbidity of T2D or T2D per se respectively, affecting QoL and health outcomes. 1 In a recent large cohort-study, PWD had increased risk of depression (14%) compared with the controls without T2D. 15 In this study, patients with T2D using SGLT-2i had lower risk for new-onset depression compared with patients using other GLM or even with controls without T2D. 15 The use of GLP-1RA, dipeptidyl peptidase-4 inhibitors (DPP-4i) (that act on the GLP-1 axis) and low dose of metformin were also associated with lower risk for new-onset depression, while high dose of metformin, insulin and sulfonylureas exhibited the highest risk. 15 The neuroprotective effects of the SGLT-2i and GLP-1RA were also showed in a meta-analysis comparing GLM on cognitive outcomes in T2D. 16 In this study, the SGLT-2i and GLP-1RA exhibited equally lower risk for cognitive decline, vascular and Alzheimer’s dementia compared with other GLM, whereas sulfonylureas exhibited the highest risk, perhaps due to the risk of hypoglycemia. 16 The neuroprotective and anti-inflammatory biological effects of the SGLT-2i and GLP-1RA and the specific ketogenic properties of the SGLT-2i, alongside the lower risk of hypoglycemia, could protect from depression and dementia.15,16 Very recently, a large comparative effectiveness trial, examined the impact on emotional distress, of liraglutide, sitagliptin, glimepiride or insulin glargine added on metformin. 17 Liraglutide and -counter to what was expected-insulin glargine, improved DRD after 1 year of treatment, while no difference was observed for depressive symptoms at a 3-years follow-up. 17
Implication of the SGLT-2i and GLP-1RA with HRQoL
PWD usually live with multimorbidity (MM), namely the co-existence of at least one other chronic condition besides T2D. 1 Very often, MM of T2D is complex, with many co-occurring conditions that obligate patients to take a lot of medications and follow several instructions for diverse diseases, that are time consuming and sometimes contradictory. 1 A large multi-cohort study highlighted the negative role of obesity for the development of complex MM (four or more co-occurring diseases). 18 The participants with obesity were at higher risk for developing complex MM in a dose-response relationship and particularly 20 years earlier (by age of 55 years vs 75 years), compared with the participants with healthy weight. 18 This study places the treatment of excessive body weight at the core of an early prevention of complex MM and its burden 18 and perhaps the clinically significant weight reduction by the SGLT-2i and GLP-1RA, could also prevent complex MM in T2D. Specifically, weight loss has recently been proposed as a primary treatment goal in the care of T2D shifted from a glucose-centric, downstream approach that focuses on T2D’s complications, toward an upstream, weight-centric approach that improves cardio-metabolic MM and QoL. 19
Health-related QoL (HRQoL) is a PIO on its own right
20
and together with physical function, are consistently across the literature, inversely related with MM in T2D.20,21 A growing body of evidence indicates that, in addition to the cardiorenal and neural protection, the SGLT-2i and GLP-1RA protect against the burden of MM by reducing for example overall respiratory diseases,
22
gout,
23
sexual dysfunction
24
and other chronic co-occurring conditions of T2D. Consequently, the alleviation of MM by the favorable physical and psychological pleiotropic effects of the SGLT-2i and GLP-1RA, could prevent frailty
11
and improve physical function and HRQoL of PWD. (Figure 1) In particular, a large real-world prospective study, examined the impact on HRQoL of the SGLT-2i, GLP-1RA, DPP-4i or sulfonylureas added on metformin.
25
Although all four examined second-line GLM showed similar reductions in HbA1c, the SGLT-2i, GLP-1RA and DPP-4i reduced body weight and risk of hypoglycemia compared with the sulfonylureas.
25
Further, the SGLT-2i consistently improved, while the sulfonylureas worsened HRQoL during 36 months of treatment.
25
Very recently, another large comparative effectiveness trial, examined the impact on HRQoL, of liraglutide, sitagliptin, glimepiride or insulin glargine added on metformin.
26
Only liraglutide improved HRQoL at year 1 but not 2 and 3, while the improvement was related with weight loss and higher baseline body-mass index.
26
Finally, a meta-analysis of RCT of all the available GLM, showed that the SGLT-2i, GLP-1RA and tirzepatide (a dual glucose dependent insulinotropic poly-peptide/GLP-1), slightly improve HRQoL.
6
Possible mechanism through which the sodium-glucose cotransporter-2 inhibitors (SGLT-2i), glucagon-like peptide-1 receptor agonists (GLP-1RA) and fixed ratio combinations (FRC) of basal insulin/GLP-1RA could improve health-related quality of life (HRQoL) of patients living with type 2 diabetes.
Future implications
In order to improve PIO of PWD at high risk for cardiovascular and renal disease, the current cardiology guidelines reframe the SGLT-2i and GRP-1RA as cardiorenal risk-reducing medications beyond their role in lowering glucose, prioritizing their use for the treatment of PWD with established ASCVD and CKD, independent of glycemic control. 27 Despite this, real-world data demonstrate a paradox between evidence-based benefit and current use of the SGLT-2i and GLP-1RA. 28 For example, a study showed that PWD with cardiovascular disease, heart failure or nephropathy, that are most likely to benefit from the SGLT-2i and GLP-1RA, are less likely to be prescribed, while prescription is lower among non-White and older PWD. 28 Although insurance restrictions and high cost may limit the use of the SGLT-2i and GLP-1RA, 1 a study showed that, even in a universal health coverage system with limited GLM cost, lower socioeconomic status is associated with lower initiation of the SGLT-2i or GLP-1RA. 29 Perhaps patient and clinician education could increase evidence-based prescription of the SGLT-2i and GLP-1RA and reduce ethnic and socioeconomic disparities associated with under-prescription, in order to avoid future gaps in cardiovascular and renal outcomes.28,29 Finally, future RCT of the SGLT-2i, GLP-1RA and FRC of basal insulin/GLP-1RA should measure and report more data on self-reported outcomes that PWD perceive and value, such as HRQoL, treatment satisfaction, sexual dysfunction, depressive symptoms or DRD.2,14
Conclusion
The use of the SGLT-2i and GLP-1RA should be weighed on a balance between benefits and harms (cost, side effects, fear of injection, contraindications) within a shared decision-making for an individualized glycemic target, in which PIO, such as cardiorenal protection and improvement of HRQoL, can be achieved independent of HbA1c. Through the attenuation of cardiorenal complications and MM, the SGLT-2i and GLP1-RA could improve the HRQoL of PWD. The transformation of the contemporary management of T2D from glucose- and insulin-centric to person-centered could be facilitated by the pleiotropic effects of the SGLT-2i and GLP-1RA on several non-glycemic, patient-important aspects of living with T2D.
Footnotes
Acknowledgements
The authors wish to thank Mr. Timothy Miller, Project Manager, London, United Kingdom (UK) and Dr. Maria Katsouli, Paediatric Registrar post CCT, Surrey, UK, for the language check of the manuscript and Dr. Eumorfia-Maria Ladogianni, General Practitioner, Aliartos, Boeotia, Greece, for her important remarks.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.