Abstract
Background
Diabetes type 2 is one of the strongest risk factors affecting coronary artery disease (CAD) and is also a marker of poor short and long-term prognosis in subjects with acute coronary syndrome (ACS) treated with percutaneous coronary intervention (PCI) with subsequent drug-eluting stent (DES) implantation. Chronic local vascular inflammation along with endothelial dysfunction is postulated to be the pathophysiological background of unfavorable results. The second generation of metallic magnesium BRS –Magmaris (Biotronik, Berlin, Germany) had been introduced to clinical practice to overcome these limitations.
Methods
We evaluated 2-years clinical outcomes after Magmaris BRS implantation in NSTE-ACS diabetic (
Results
No significant differences between diabetic and non-diabetes cohorts were noticed in terms of Primary Outcome (cardiac death, myocardial infarction, stent thrombosis) (8.1% vs 3.3%
Conclusions
2-years outcome suggests good safety and efficacy of the magnesium BRS (Magmaris) in NSTE- ACS and concomitant DM. Nevertheless, there is a strong need for large multicenter, randomized, prospective studies for a full assessment of this novel device in diabetic patients with ACS.
Keywords
Introduction
The global number of people diagnosed with diabetes mellitus (DM) is constantly rising, reaching over 600 million cases worldwide. 1 The diabetes pandemic is remarkably affecting the young adult population mainly due to the increased burden of obesity combined with a sedentary lifestyle. This alarming trend set up a new challenge in terms of long-term care particularly the management of cardiovascular complications of DM. Diabetes type 2 is one of the strongest risk factors affecting coronary artery disease (CAD). Furthermore, three out of four patients with DM t.2 will die as a result of cardiovascular complications with specifically CAD as a leading cause of morbidity and mortality. 2 Besides being a risk factor for CAD, DM is also a marker of poor short and long-term prognosis in subjects with acute coronary syndrome (ACS). Furthermore, coronary interventions - the basic therapeutic approach in the ACS subset remain fraught with a high rate of complications in the diabetic population.
Recently several improvements in PCI technique and armamentarium had been introduced to the clinical practice to alleviate this unfavorable trend. Despite undeniable improvement in drug-eluting stent (DES) technology still, DM t.2 is one of the major risk factors of long-term device failure resulting in an increased need for target lesion repeat revascularization.
Although several significant predictors of in-stent restenosis have been identified, the exact causes of DES restenosis remain controversial and not fully understood.3,4 It is likely that multiple factors related to biological, genetic, mechanical, and technical aspects contribute to the etiopathogenesis of this process. In the diabetic population, local vascular inflammation together with endothelial dysfunction5–7 has been postulated to play a crucial role in the pathophysiological background of adverse outcomes. Antiproliferative agents in modern DES suppress smooth muscle cell migration and proliferation by arresting cells in the G1 phase and inducing cell apoptosis.8,9
Furthermore-on the one hand, genetic resistance to the mechanisms of antiproliferative drugs has been well documented. 10 On the other hand, diabetes has been identified as a known risk factor for a systematic proinflammatory status,11,12 which, combined with the presence of the artificial body in the vascular wall, may additionally enhance the local inflammatory process. 3 This was confirmed by elevated levels of systemic inflammatory markers, which have been shown to be early markers of stent failure. Additionally, permanent vessel caging with a metallic stent structure evoke hypersensitivity reactions mainly due to polymer coverage of DES. 13
To counteract these processes, bioresorbable scaffolds (BRS) were introduced to clinical practice. Actually the second generation of metallic magnesium BRS –Magmaris (Biotronik, Berlin, Germany)—had received a CE mark and been introduced to clinical practice and showed favorable short-term clinical outcomes.14–17 This novel clinical concept is based on solving a magnesium backbone scaffold that provides short-term vessel-supporting potency equal to classical DES technology therefore, it helps avoid permanent caging of the vessel and potentially reduce long-term device-related adverse events 18 which might be particularly useful in the diabetic population. Especially since hyperglycemia per se has been shown to disrupt arterial healing and activate chronic local inflammation, promoting vascular architectural abnormalities with concomitant vasomotor dysfunction.19,20 Indeed recently published data support the high efficacy and safety of Magmaris BRS in diabetes subjects in terms of short-term follow-up21,22 still long-term data are missing.
Therefore we designed this study to evaluate the long-term results of magnesium scaffold in the diabetes ACS population.
Materials and methods
Study design and population
The complete trial design along with a full list of inclusion and exclusion was reported previously21,22 Briefly, the study population consisted of patients (
PCI procedures
Treatment of de novo lesions, in patients with ACS subset, was allowed. All PCI procedures had to meet the “4P” criteria, particularly initial successful predilatation of the target lesion along with scaffold high-pressure (not less than 16 atm) post-dilatation with NC balloon (sized 1:1 balloon/scaffold ratio or up to 0.5 mm longer) were mandatory. The use of intravascular imaging was left to the operator`s discretion.
Scaffold characteristic
The Magmaris, previously known as DREAMS 2G, is a bioresorbable magnesium scaffold with CE approval in Europe since June 2016. It is a balloon-expandable, bioresorbable metallic scaffold coated with a poly-L-lactic acid biodegradable polymer (PLLA) eluting sirolimus- (up to 90 days) and with an average time of full scaffold absorption in approximately 1year. The scaffold is available in diameters of 3.0 and 3.5 mm and lengths of 15, 20, and 25 mm.
Follow-up
Patients were evaluated by telephone contact or/and personal scheduled visits at our cardiac center during the post-procedure period at the following time points: post-procedure/in-hospital; 1 and 2 years.
Study endpoint
Clinical component endpoints including death, MI, target lesion revascularization (TLR), target vessel revascularization (TVR), all coronary revascularization, and scaffold thrombosis and restenosis were based on Academic Research Consortium definitions. 23 Myocardial infarction was defined according to the Fourth Universal Definition of Myocardial Infarction. 24
The primary outcome was a safety composite of cardiovascular death, myocardial infarction, and definite or probable in-stent thrombosis at the 1-year, and 2-years of follow-up. The principal secondary outcome was an effective outcome of target-lesion failure (TLF) defined as cardiac death, target vessel myocardial infarction (TV-MI), or target lesion revascularization (TLR).
Statistical analysis
Continuous variables were characterized by mean and standard deviation, categorical variables were characterized by the frequencies. The study cohorts were compared with the nonparametric two-sample Mann–Whitney’s test for continuous variables and Fisher’s Exact Test for categorical variables. Bonferroni correction was applied to adjust for multiple comparisons. P- values ≤ 0.05 were defined as statistical significance. All statistical analyses were performed using the R language by a professional statistician similar to medical analysis.
Results
Baseline clinical characteristic of both groups.

Abbreviations: 1 NSTEMI = non-ST elevation myocardial infraction; PCI = percutaneous coronary intervention; MI = myocardial infraction; LVEF = left ventricular ejection fraction. Bold data-mark statistically significant (
Procedural characteristic of both groups.
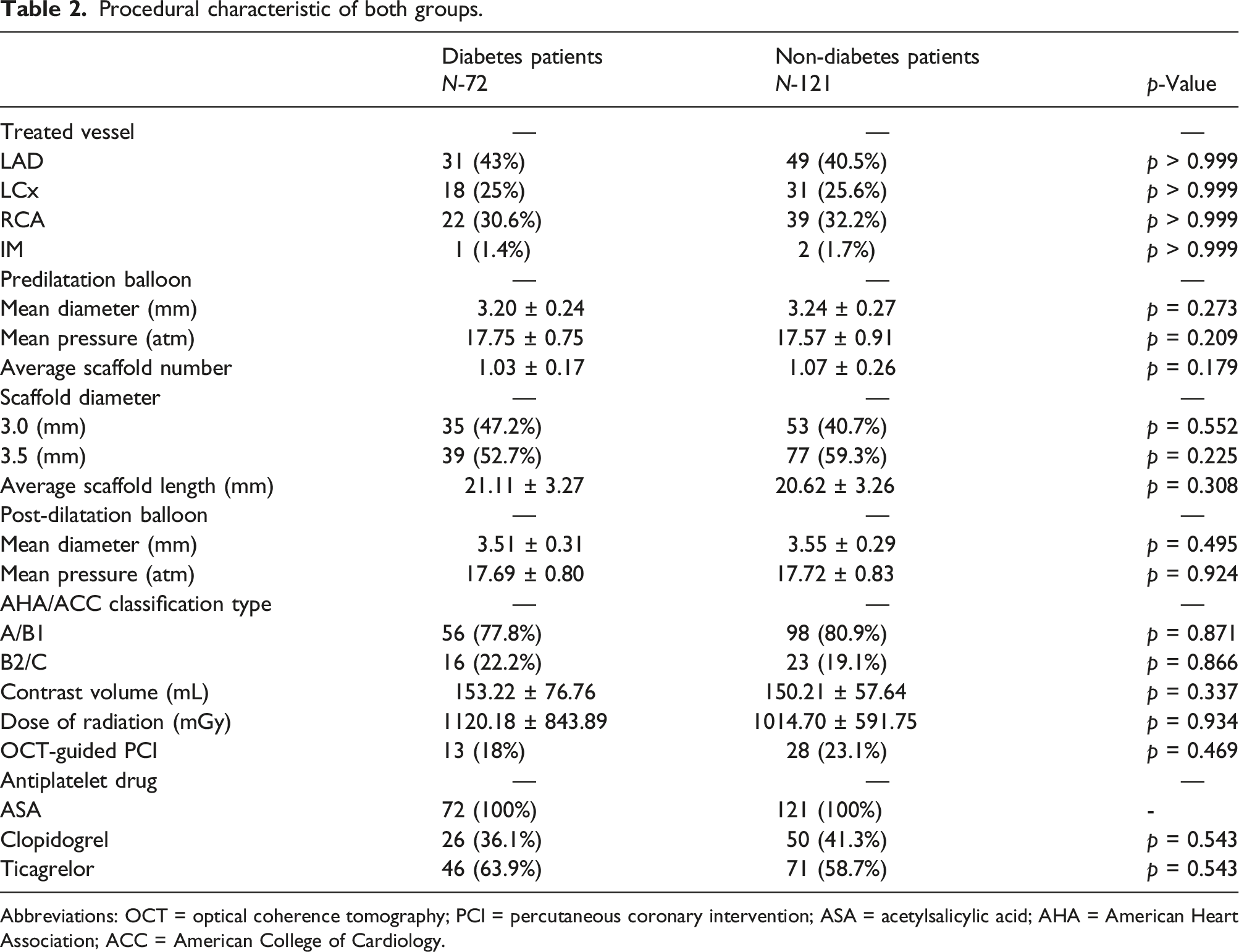
Abbreviations: OCT = optical coherence tomography; PCI = percutaneous coronary intervention; ASA = acetylsalicylic acid; AHA = American Heart Association; ACC = American College of Cardiology.
Clinical outcomes.

Abbreviations: PCI = percutaneous coronary intervention; ASA = acetylsalicylic acid; MI = myocardial infraction.
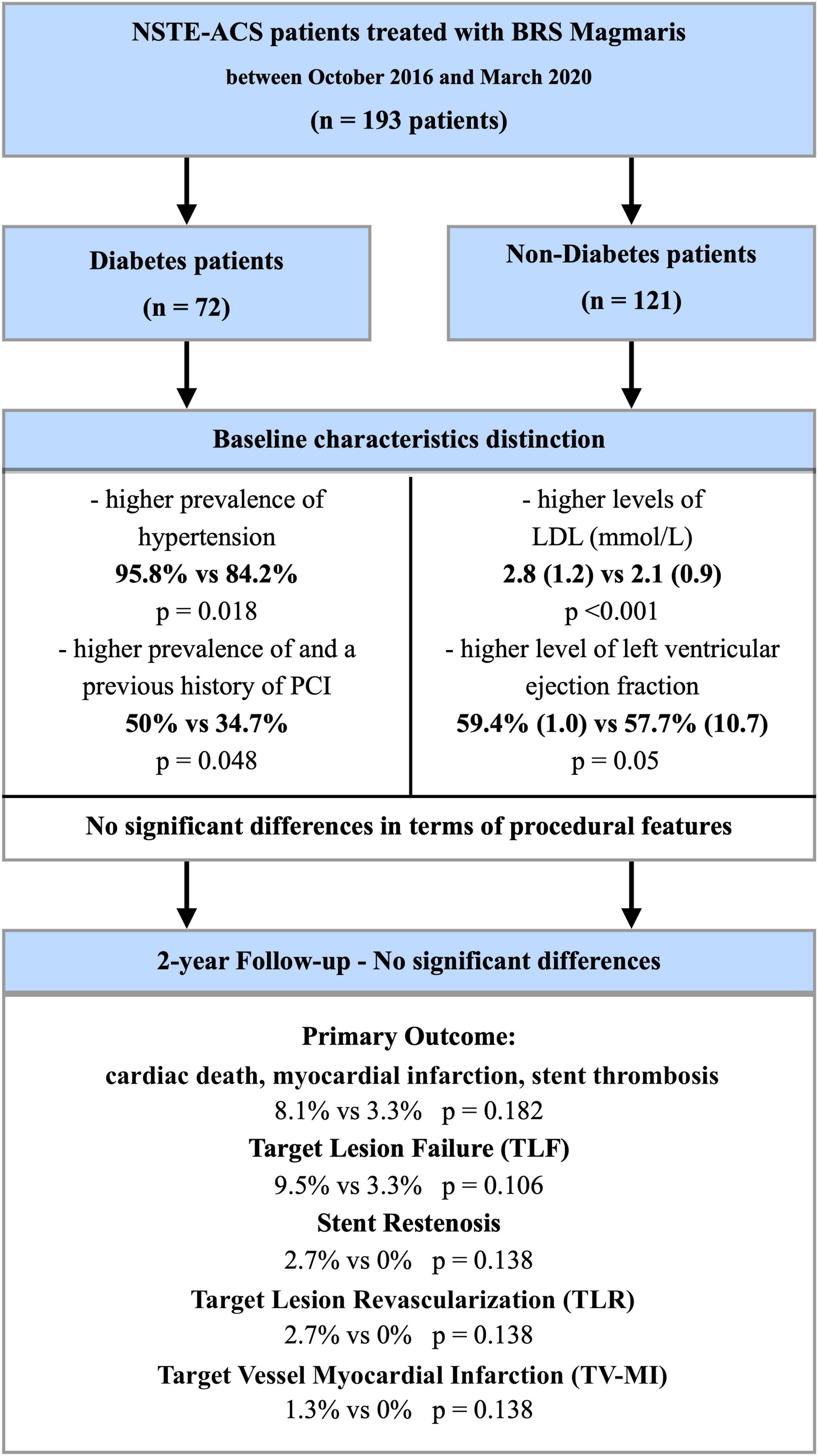
A flow chart of the study design.
Discussion
Despite the undeniable development of stent technology and the undisputed revolution linked with the introduction to the clinical practice of second-generation DES 25 still, the diabetes population seems to have a faint advantage from this novel technology26,27 Several conceptional improvements in stent design and modality had been postulated and introduced to clinical practice to overcome these limitations.28,29 Especially high expectations had been associated with bioresorbable materials30–32 - two different concepts have been proposed to use the advantage of these novel materials. First used combination of modern DES technology with bioresorbable polymers designed for optimal release kinetic of the antiproliferative drugs. Unfortunately, in pooled meta-analysis short-term of the biodegradable polymer showed no superiority compare to classical permanent polymers 33 therefore scar data from long-term follow-up data, suggests a lower risk of stent thrombosis. 34
The second concept is based on the theoretical assumption that the scaffold backbone after providing short-time vessel patency can be fully absorbed. Such an operational model is particularly encouraging in terms of the diabetic ACS subpopulation where vessel scaffolding is necessary to stabilize vulnerable plaque in a short-term period. However, the long-term presence of anartificial material in the vascular wall might enhance the local inflammatory process increasing TLF particularly in-stent restenosis or/and late stent thrombosis.35,36
First-generation BRS, largely represented by the poly-l-lactic acid (PLLA) Absorb (Abbott Vascular, Santa Clara, CA, USA) had shown similar safety and efficacy in diabetic and non-diabetic populations in terms of short-term observation.37–39 However strong correlation between the higher rate of target lesion revascularization and insulin depending diabetes was marked. 40 Interestingly Absorb had shown favorable clinical outcomes in diabetic populations both patency and TLR, even in patients with heavy calcification in terms of subjects who underwent lower limb revascularization due to peripheral artery disease. 40 This fact is probably related to slight differences in terms of the pathogenesis of atherosclerosis in the course of DM where factors such as inflammation, endothelial dysfunction, and vascular smooth muscle cell proliferation seem to be crucial. 5
What needs to be emphasized, the long-term results were far less encouraging Absorb had worse clinical outcomes in diabetic patients as compared to nondiabetic patients. Furthermore, Absorb was associated with higher rates of device thrombosis and TLF compared to Xience at a 3-years follow-up. 41 However, if Absorb BRS were implanted in larger vessels with support to intravascular imagining and in accordance with “4 P” criteria 42 long-term results were more convincing.43,39
Robust evidence related to the long-term outcomes of other non-Absorb BRS in the context of DM are still lacking. Short-term data focused on the diabetes population are available only to magnesium BRS and seem to be favorable
21
also comparing the Magmaris to the second generation of metallic DES.44,22 Data regarding the relationship between DM t2 and other novel BRS are partial and incomplete45–47 making it difficult to analyze and discuss. Observed in our study cohort favorable clinical outcomes (no significant differences in terms of Primary outcome, principally secondary outcome, TLF, with additional lack of stent thrombosis in the study population) after Magmaris implantation in diabetic ACS-NSTE population seems to support data obtained in all-comers Magmaris registries.48–51 Despite no statistically significant difference in terms of study endpoints occur noticeable unfavorable trend in restenosis ratio in DM t.2 subpopulations occurred (8.1% vs 3.3%;
Limitations
In this study, several limitations have occured. The study population is relatively small, all patients were recruited in a single center, however with a good experience in the BRS technology. Even more, the study was non-randomized and all the data had been collected retrospectively. Additionally, the rate of intravascular imaging guidance PCI in this study was comparatively low.
Conclusions
The mid-term outcome observed in our study points at a good safety and efficacy of the magnesium BRS (Magmaris) in NSTE- ACS and concomitant DM. Nevertheless, there is a strong need for large multicenter, randomized, prospective studies aiming at an overall assessment of this novel device in diabetic patients with ACS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Medical Lower Silesian Ethics Board (number 20/07/2016 approval 12.07.2016).
