Abstract
Objective:
To compare the efficacy and safety of combined treatment with thrombus aspiration and intracoronary tirofiban in patients with acute myocardial infarction (MI), with elective percutaneous coronary intervention (PCI).
Methods:
Patients undergoing elective PCI during recovery from acute MI were randomized into two groups; the intervention group received thrombus aspiration and intracoronary tirofiban; the control group received conventional PCI. Baseline clinical characteristics, postoperative coronary blood flow (thrombolysis in myocardial infarction [TIMI] grade), TIMI myocardial perfusion (TMP) grade, no/slow reflow rate and cardiac function (measured by echocardiography 1 month postoperatively) were evaluated. Major adverse cardiac event rate and bleeding complications during surgery (and at 1 month and 1 year postoperatively) were also evaluated.
Results:
A total of 80 patients were included. Postoperatively, the number of patients with TIMI flow grade 3 and the mean TMP grade were both significantly higher, and the no/slow reflow rate was significantly lower, in the intervention group versus the control group. Echocardiography indicated that cardiac function was significantly improved in the intervention group compared with the control group. There were no major complications in either study group.
Conclusion:
Thrombus aspiration combined with intracoronary tirofiban during recovery from acute MI was effective and relatively well tolerated.
Keywords
Introduction
Percutaneous coronary intervention (PCI) is an effective therapy for patients with acute myocardial infarction (MI), although prognosis following the procedure could be improved. 1 Acute MI leads to a significant thrombi burden in vessels with lesions. 1 During stent placement and balloon dilatation, mobile broken thrombi can cause obstruction and/or spasms in the distal vessels, which result in no or slow reflow. This in turn means that myocardial tissue cannot achieve effective reperfusion, which can negatively influence prognosis. 2 Two methods used to prevent the occurrence of no/slow reflow after PCI are the application of enhanced thrombus removal devices and anticoagulant antiplatelet therapy. 3 A higher incidence of bleeding is associated with methods that use low-molecular weight heparin or triple antiplatelet therapy, both of which can increase mortality rates during hospitalization. 4 Thrombus aspiration during emergency PCI can improve the myocardial reperfusion index and prognosis in patients with acute ST-segment elevation during MI.5,6 Although coronary thrombus aspiration can effectively remove the thrombus blocking the coronary vessel and improve coronary blood flow, residual thrombus debris can lead to distal microvascular embolization. 7 The application of platelet glycoprotein IIb/IIIa receptor antagonists (in combination with antiplatelet agents, anticoagulants and anti-inflammatory drugs) can decrease infarct size, reduce the degree of microvascular damage and improve myocardial perfusion. 8
Research has shown that, with a severe thrombus burden, application of thrombus aspiration and tirofiban during an emergency PCI procedure can improve blood no/slow reflow and lead to improved outcomes. 9 There remains, however, a substantial thrombus load in patients with acute MI who have exceeded the emergency PCI time window (12 h). 10 Clinical studies on how to reduce thrombus burden, decrease no/slow reflow and improve target vessel blood flow and myocardial perfusion are lacking.
The present study evaluated the efficacy and safety of combined treatment with coronary thrombus aspiration and tirofiban in patients with acute MI who had a high coronary thrombus load during elective PCI.
Patients and methods
Study population
Acute MI patients who had exceeded the emergency PCI time window (12 h), who were being treated at the Department of Cardiology, The First Affiliated Hospital of Henan Science and Technology University, Luoyang, Henan, China, between July 2008 and December 2009, were enrolled in the study. Eligible subjects were evaluated according to international standards (namely, the 2009 American College of Cardiology Foundation/American Heart Association [ACCF/AHA] guidelines for the management of ST-elevation MI 11 ) and met the following inclusion criteria: acute ST-segment elevation myocardial infarction (STEMI); onset > 12 h prior to hospital admission; received elective PCI 10–30 days after the acute MI; coronary angiography showed infarct-related artery (IRA) tips or thrombus burden lesions (defined as a coronary thrombosis coronary strip, oval- or irregular-shaped filling defect with retention of contrast medium or dye); infarct-related vessel diameter ≥ 2.5 mm; no contraindications for anticoagulant and antiplatelet therapy. Patients who had been taking anticoagulants were excluded from the study.
Patients were randomized into one of two groups: the intervention group, which received administration of thrombosis aspiration combined with intracoronary injection of tirofiban during PCI; the control group, which underwent conventional PCI. Patients were encoded using a random coding table and were assigned a single number for the study intervention (thrombus aspiration) group or a double number for the control (no thrombus aspiration) group.
The study was approved by the local Ethics Committee of The First Affiliated Hospital of Henan Science and Technology University. All patients provided written informed consent prior to participating in this study.
Study treatment and procedures
All patients underwent routine antiplatelet and anticoagulant therapy before the operation, which included 300 mg/day aspirin orally and 300 mg/day clopidogrel orally, from hospital admission until operation. Routine elective coronary angiography was performed, with study or control interventional therapy, 10–30 days after acute MI in all acute MI recovery patients.
In the intervention group, thrombus aspiration was carried out, followed by an intracoronary injection of 10 ml of 5 mg/100 ml tirofiban (Wuhan Grand Pharmaceutical Group, Wuhan, China; batch number: H20041165). This was followed by stenting, or stenting after balloon dilatation, and a further intravenous infusion of 0.1 µg/kg/min tirofiban was maintained for 48 h. To carry out thrombus aspiration using visual coronary angiography, a guiding catheter was inserted: the guide wire was inserted as far as the IRA lesions. The catheter for thrombosis aspiration – a 3.8 F ZEEK manual aspiration catheter (Zeon Medical Inc., Tokyo, Japan) – was then passed along the guide wire to the IRA (being careful not to touch and cross the blood clots), and the tail was connected with a screw injector for aspiration. The thrombus aspiration catheter was then used to carry out deep aspiration to the point that it exited the body due to continuous negative pressure. The catheter was drained of blood and thrombi, and flushed repeatedly with a heparin-saline solution (heparin sodium 12500 IU, dissolved in 250 ml sodium chloride). The above steps were repeated until the thrombus shadow on visual coronary angiography disappeared, or was considerably reduced, and the thrombolysis in myocardial infarction (TIMI) 12 grade-flow was improved. TIMI blood flow was monitored throughout the procedure and graded as follows: 0, no prorsad blood flow perfusion in the vessel at the distal end; 1, prorsad blood flow perfusion in the vessel but no engorgement at the distal end; 2, vessel fully engorged after more than three cardiac cycles; 3, vessel fully engorged after three cardiac cycles. Satisfactory improvement in the condition of infarct-related arterial blood flow was assessed by the investigator according to TIMI flow grade immediately after the procedure. The assessment of TIMI flow grade and TIMI myocardial perfusion (TMP) grade were necessarily unblinded due to the radically different nature of the study interventions; however, for the purposes of statistical analyses of these data, the statistician was blinded to the study group allocation.
The control group underwent conventional PCI without thrombosis aspiration or administration of tirofiban in the coronary artery; PCI was conducted according to the guidelines of ACCF/AHA-STEMI. 11
After surgery, low-molecular weight heparin (10000 IU/ml; 40 mg every 12 h) was administered subcutaneously for 1 week; 100 mg/day aspirin and 75 mg/day clopidogrel were both administered orally for ≥ 12 months. All patients were followed-up by telephone for 1 year (at 1, 3, 6 and 12 months postoperatively). All follow-up data were collected by full-time medical staff who were unaware of the study protocol or study groups.
Study assessments
Efficacy of was assessed by evaluating the following. The condition of TIMI flow recovery after coronary stenting. The no/slow reflow rate; slow flow was defined as TIMI 2 level flow; no reflow was defined as TIMI 0–1. The TIMI myocardial perfusion grade (TMP) was recorded after stent implantation using Ultravist® Solution 370 (Bayer Vital GmbH, Leverkusen Germany) and graded as follows; grade 0, no significant tissue level perfusion (no contrast agent through the tissue or staining); grade 1, a blush of contrast agent was seen in the myocardium, but it was not empty (there was still contrast agent in the myocardium at the next cardiac cycle); grade 2, slow evacuation of contrast agent in the myocardial tissue (contrast agent was still stranded in the myocardial tissue within three cardiac cycles); grade 3, normal emptying of contrast agent in myocardial tissue (contrast agent was emptied after three cardiac cycles).
Left ventricular ejection fraction (LVEF), early diastolic velocity and late diastolic flow velocity ratio (E/A) at the mitral annulus and left ventricular end-diastolic diameter (LVEDD) were assessed by echocardiography, 1 month after surgery.
Major adverse cardiac events (MACE), including cardiac death, target vessel revascularization and recurrent MI, were assessed within 1 year following surgery. The incidence of bleeding complications was assessed during the operation and at 1 month and 1 year postoperatively. The need for restenosis was also recorded for all patients throughout the study period.
Statistical analyses
Data were expressed as mean ± SD or number and percentage of patients. Differences between the two groups were assessed by an independent-samples
Results
Baseline clinical characteristics of patients undergoing elective percutaneous coronary intervention (PCI) during recovery from acute myocardial infarction (MI), who received combined treatment with thrombus aspiration and intracoronary injection of 10 ml of 5 mg/100 ml tirofiban (intervention group) or conventional PCI (control group; no thrombosis aspiration or tirofiban administration).

Data presented as mean ± SD values or
There were no statistically significant between-group differences (
Angiography and cardiac-function results in patients undergoing elective percutaneous coronary intervention (PCI) during recovery from acute myocardial infarction, who received combined treatment with thrombus aspiration and intracoronary injection of 10 ml of 5 mg/100 ml tirofiban (intervention group) or conventional PCI (control group; no thrombosis aspiration or tirofiban administration).
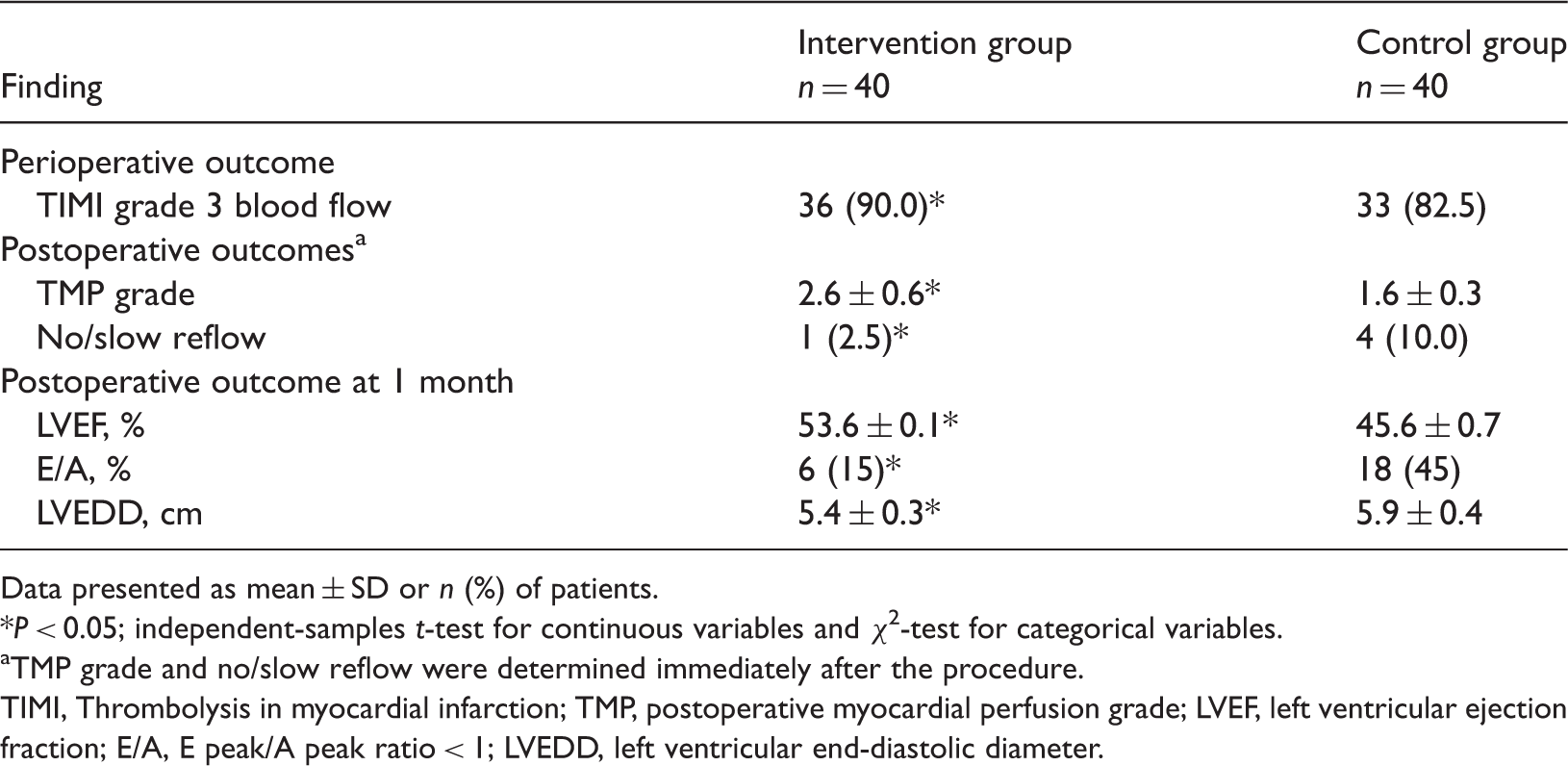
Data presented as mean ± SD or
TMP grade and no/slow reflow were determined immediately after the procedure.
TIMI, Thrombolysis in myocardial infarction; TMP, postoperative myocardial perfusion grade; LVEF, left ventricular ejection fraction; E/A, E peak/A peak ratio < 1; LVEDD, left ventricular end-diastolic diameter.
Comparison of bleeding complications and major adverse cardiovascular events (MACE) over 1 year following surgery in patients undergoing elective percutaneous coronary intervention (PCI) during recovery from acute myocardial infarction, who received combined treatment with thrombus aspiration and intracoronary injection of 10 ml of 5 mg/100 ml tirofiban (intervention group) or conventional PCI (control group).

Data presented as
There were no statistically significant between group differences (
Discussion
Evidence-based medicine has confirmed that unstable plaque rupture is the main initiating factor in the majority of cases of acute MI due to plaque rupture in coronary atherosclerosis, platelet aggregation, thrombosis, or complete or incomplete coronary occlusion. 13 The structure of unstable plaque fibrous caps is weak, as these plaques are formed by a large irregular plaque with a soft lipid core, and a large number of inflammatory cells and macrophages. 14 Haemodynamic changes or increased vascular stress, especially inflammatory cell infiltration within the local plaque and the systemic inflammatory response, will lead to plaque rupture and induce thrombosis. 15 Beyond antiplatelet and antithrombotic treatment and reopening the infarction-related artery by emergency PCI, thrombus aspiration devices and distal protection devices are designed for coronary thrombosis intervention.16,17
It has been confirmed that thrombus aspiration with emergency PCI can significantly reduce no/slow reflow and the incidence of distal-end thrombosis, and improve target vessel flow and clinical outcomes.18,19 For patients not treated by emergency PCI in the acute phase of MI, thrombi remain within the infarct-related artery during the recovery stage. 10 During elective coronary angiography, if patients are treated with routine PCI, the broken thrombus can easily block small blood vessels distal to the site of balloon dilatation and the implantation stent; this can lead to no/slow reflow and impact the immediate and long-term prognosis for the individual. 20 If patients with acute MI treated by emergency PCI show an overloading thrombus indicated by coronary angiography, this strongly suggests a high risk of no/slow reflow, which may decrease the efficacy of PCI. 21
Reduction or elimination of the thrombus burden is an urgent problem. In studies of emergency PCI for acute MI, attention is being paid to enhancing antithrombotic and anticoagulant therapy and/or to applying thrombus aspiration, in order to reduce the catheter thrombus load. 9 There is, however, still a significant thrombotic lesion load after elective PCI in patients with aMI. 10 Clinical research on improving blood flow, myocardial perfusion and clinical outcomes in these patients are lacking.
The present blinded, randomized, controlled study showed that thrombus aspiration during elective PCI, in combination with intracoronary injection of tirofiban, improved postoperative TIMI grade 3 flow and mean postoperative TMP, and reduced the rate of no/slow reflow compared with conventional PCI. Moreover, LVEF was significantly higher, and the number of patients with E/A ratio < 1 and the mean LVEDD were both significantly lower, 1 month postoperation. These findings suggest that thrombus aspiration, combined with intracoronary injection of tirofiban, may help to reduce distal microvascular embolization, improve myocardial microcirculation and decrease no/slow reflow rate, such that myocardial perfusion is complete and cardiac function is improved.
Because the thrombus aspiration device is easy to use, prevents injury and can be reused in surgery, thrombus aspiration is an effective coronary intervention; it reduces the thrombus burden, coronary thrombosis and distal embolization, and decreases the incidence of no/slow reflow, thereby improving the patient's prognosis. 22 The VAMPIRE study showed that the application of thrombus aspiration during emergency PCI did not increase surgical complexity or extend the operation time, and that thrombus aspiration devices could improve the microcirculation of patients with STEMI myocardial reperfusion compared with conventional PCI. 23 The JETSTENT study suggested that, in cases of STEMI with obvious thrombosis in coronary angiography, thrombus aspiration in the infarct-related artery before routine stent implantation would significantly improve myocardial perfusion and reduce the 6-month follow-up MACE. 24 Among patients with a relatively long lag time between infarction and PCI, to prevent distal embolization and no/slow reflow, active thrombus aspiration should be used to clear thrombi or plaque material (especially within the molecular constituents of plaque) if the patient has a TIMI flow grade is 0–1 or 2–3 and severe stenosis. 25 The present study showed that combined thrombus aspiration and intracoronary injection of tirofiban improved coronary flow, myocardial perfusion and cardiac function. There was no occurrence of cardiac death, target vessel revascularization, recurrent MI or other MACE, demonstrating that this method was had a relatively good safety profile, as well as showing favourable efficacy.
The key to treatment of the no/slow reflow phenomenon is to inhibit the induction of microtreatment thrombosis. Tirofiban is a highly-specific nonpeptide platelet GP IIb/IIIa receptor antagonist that directly, rapidly and reversibly blocks platelet aggregation; tirofiban also improves endothelial function. 26 An investigation of intravenous tirofiban before PCI, compared with conventional PCI alone, showed that injection of tirofiban before PCI in patients with acute STEMI significantly reduced blood clots and the no/slow reflow phenomenon, and increased myocardial perfusion. 3 Intracoronary injection of tirofiban was shown to increase the coronary drug concentration, 8 such that no/slow reflow coronary blood flow improved immediately. The main side-effects of tirofiban are bleeding and thrombocytopenia. 7 Among the 40 patients in the intervention group, five reported epistaxis, gum bleeding, haematuria, skin ecchymosis or bleeding at puncture sites, all of which improved after treatment. There was no significant difference between the two groups in the incidence of adverse reactions. This suggests that intracoronary injection of tirofiban did not increase the risk of bleeding and other complications, and is worthy of clinical application.
In summary, the application of intracoronary thrombus aspiration catheter injection combined with tirofiban has a relatively good safety and efficacy profile in elective PCI after acute MI. Further studies with larger sample sizes are needed, to evaluate the clinical value and long-term prognosis following this procedure.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
