Abstract
While hemoglobin A1c (HbA1c) is commonly used to monitor therapy response in type 2 diabetes (T2D), GV is emerging as an essential additional metric for optimizing glycemic control. Our goal was to learn more about the impact of hypoglycemic agents on HbA1c levels and GV in patients with T2D. A systematic review and network meta-analysis (NMA) of randomized controlled trials were performed to assess the effects of glucagon-like peptide 1 receptor agonists (GLP-1 RAs), sodium-glucose cotransporter (SGLT)-2 inhibitors, dipeptidyl peptidase (DPP)-4 inhibitors, sulfonylurea and thiazolidinediones on Mean Amplitude of Glycemic Excursions (MAGE) and HbA1c. Searches were performed using PubMed and EMBASE. A random-effect model was used in the NMA, and the surface under the cumulative ranking was used to rank comparisons. All studies were checked for quality according to their design and also for heterogeneity before inclusion in this NMA. The highest reduction in MAGE was achieved by GLP-1 RAs (SUCRA 0.83), followed by DPP-4 inhibitors (SUCRA: 0.72), and thiazolidinediones (SUCRA: 0.69). In terms of HbA1c reduction, GLP-1 RAs were the most effective (SUCRA 0.81), followed by DPP-4 inhibitors (SUCRA 0.72) and sulfonylurea (SUCRA 0.65). Our findings indicated that GLP-1 RAs have relatively high efficacy in terms of HbA1c and MAGE reduction when compared with other hypoglycemic agents and can thus have clinical application. Future studies with a larger sample size and appropriate subgroup analyses are warranted to completely understand the glycemic effects of these agents in various patients with T2D. The protocol for this systematic review was registered with the International Prospective Register of Systematic Reviews (CRD42021256363).
Keywords
Introduction
Diabetes is a serious and chronic disease that affects individuals and families worldwide. In 2019, 537 million adults aged 20–79 years have diabetes, and diabetes reached 10% of total health spending. 1 To date, the main goal of diabetes management is hemoglobin A1c (HbA1c) control, which has been the standard parameter for determining the risk of complications in diabetic patients. 2 However, HbA1c fails to capture the time spent within the glucose target range (TIR) of 70–180 mg/dL and glucose variability (GV), 3 both of which are related to vascular outcomes.4–6 Therefore, TIR and GV should also be considered as additional measures for glycemic control in addition to HbA1c.7,8 Currently, the International Consensus on the Use of Continuous Glucose Monitoring recommends employing the coefficient of variation (CV) of <36% as a key indicator of GV, despite the fact that at least 20 GV metrics have been reported in the literature, each with its own set of advantages and disadvantages. 9 Among these metrics, the mean amplitude of glycemic excursion (MAGE) is one of the most widely used in the literature, and it has a higher potential of capturing glucose swings than CV. 10 Thus, MAGE may be able to better represent glucose fluctuation in this context.7,11 Hence, in the current study, we used MAGE as a primary GV metric due to its extensibility.
There is emerging evidence that patients use glucagon-like peptide 1 receptor agonists (GLP-1 RA), dipeptidyl-peptidase 4 (DPP-4) inhibitors, and sodium-glucose cotransporter 2 (SGLT-2) inhibitors can effectively achieve glycemic control.12–14 Subin Lee et al. reported that DPP-4 inhibitors were effective in GV regulation in a direct analysis comparing DPP-4 inhibitors and oral antihyperglycemic drugs (OAD). 14 In addition, Heeyoung Lee et al. compared and analyzed GLP-1 RAs and SGLT-2 inhibitors. 11 According to published studies, SGLT-2 inhibitors, GLP-1 RAs, and DPP-4 inhibitors show pharmacological differences in their mechanisms of action, but effectively reduce glucose levels, complications, and GV in diabetic patients.11,14 However, in existing studies, it is difficult to ascertain the differences between individual drugs because direct comparison drugs for each drug pair exist in part. Although there have been indirect studies regarding HbA1c reduction between antidiabetic treatment pairs, 15 there have been no studies on MAGE, which is another parameter that indicates differences between GV fluctuations. Therefore, the purpose of this study was to compare the effect of each drug pair on GV based on HbA1c and MAGE reduction in type 2 diabetes patients.
Methods
Search strategy and selection criteria
For this systematic review and network meta-analysis, we searched MEDLINE, Embase from database inception through to 28 June 2021. Database search was performed using medical subject heading (MeSH) terms “glycemic variability,” “glycemic fluctuations,” “mean amplitude of glycemic excursions,” in combination with “Diabetes Mellitus, Type 2” “dipeptidyl-peptidase 4 inhibitors”. Terms related to DPP-4 inhibitor, GLP-1 RA and SGLT-2 inhibitor are shown in Supplementary Table S2.
Only randomized control trials (RCTs) that evaluated HbA1c and MAGE of type 2 diabetes in participants aged ≥18 years were included. Five anti-diabetes treatments were intervention compared GLP-1 receptor agonists (RAs), DPP-4 inhibitor, SGLT-2 inhibitors, sulfonylurea, or thiazolidinediones. Our study assessed HbA1c and MAGE of the anti-diabetes treatment pairs. The intervention was monotherapy or add-on therapy, which included GLP-1 RA, SGLT-2 inhibitors, or DPP-4 inhibitors, and the control was either a placebo or other antidiabetic treatment. We limited the language of the publications to English and excluded publications without MAGE data.
This study was prepared according to the PRISMA extension statement for network meta-analyses guidelines (Supplementary Table S1). The protocol for this systematic review was registered with the International Prospective Register of Systematic Reviews (CRD42021256363).
Outcome and data analysis
The primary outcome was MAGE in type 2 diabetes mellitus participants, while the secondary outcome was HbA1c. Uncertain information was decided after a discussion between two or more authors. All outcomes were calculated as the difference between pre- and post-treatment values.
The evaluation of MAGE and HbA1c outcomes were extracted from the included trials. In this network meta-analysis, we used the random-effects model in Bayesian frameworks. Analyses were only conducted when there were two or more trials available for the studied outcome. We then performed Markov Chain Monte Carlo simulations using vague priors 16 and a generalized linear model with Gaussian family distribution and an identity link function.17,18 The analysis was conducted using 10 000 burn-ins, 100 000 iterations, and 1000 adaptations. The deviance information criterion and individual datapoint posterior mean deviance contribution were used to compare goodness-of-fit between the consistency and inconsistency models. The deviance information criterion was also used to select between a fixed-effects or random-effects model.17,18 The outcomes were analyzed using the mean difference (MD) and 95% confidence intervals (CIs). All analyses were generated by R Software with GeMTC package.19–21
The methodological quality assessment was performed by using Cochrane Collaboration’s tool to assess the risk of bias in each trial. 22 The evaluation items included in the present study were random sequence generation, allocation concealment, blinding, incomplete outcome data, selective outcome reporting, and potential bias. Each item was presented as “low risk,” “some concern,” or “high risk.” The graphs were synthesized by Review Manager version 5.4. The quality of evidence of collective outcomes was estimated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) framework. 23 Publication bias was also assessed using funnel plots. 24
The consistency of network meta-analysis was assessed using the node-splitting models to detect whether the results of the direct and indirect comparisons were in agreement within treatment loops. 25 Also, we used the analysis of heterogeneity to quantify the degree of heterogeneity using the I2 calculation. Values of I2 >50% were considered heterogeneous across the trials. 26 We ranked the interventions according to their surface under the cumulative ranking (SUCRA), which ranged from 0 to 1. 27 The SUCRAs is based solely on the point estimates and standard errors of the network estimates and measure the mean extent of the network estimates and the mean extent of certainty that one intervention is superior to another averaged over all competing interventions.
Results
Systematic searching through 28 June 2021, identified 139 studies, of which 17 were included in the final analysis. The flow diagram for the results of the electronic search is described in Figure 1. Flow chart of articles search and screening process.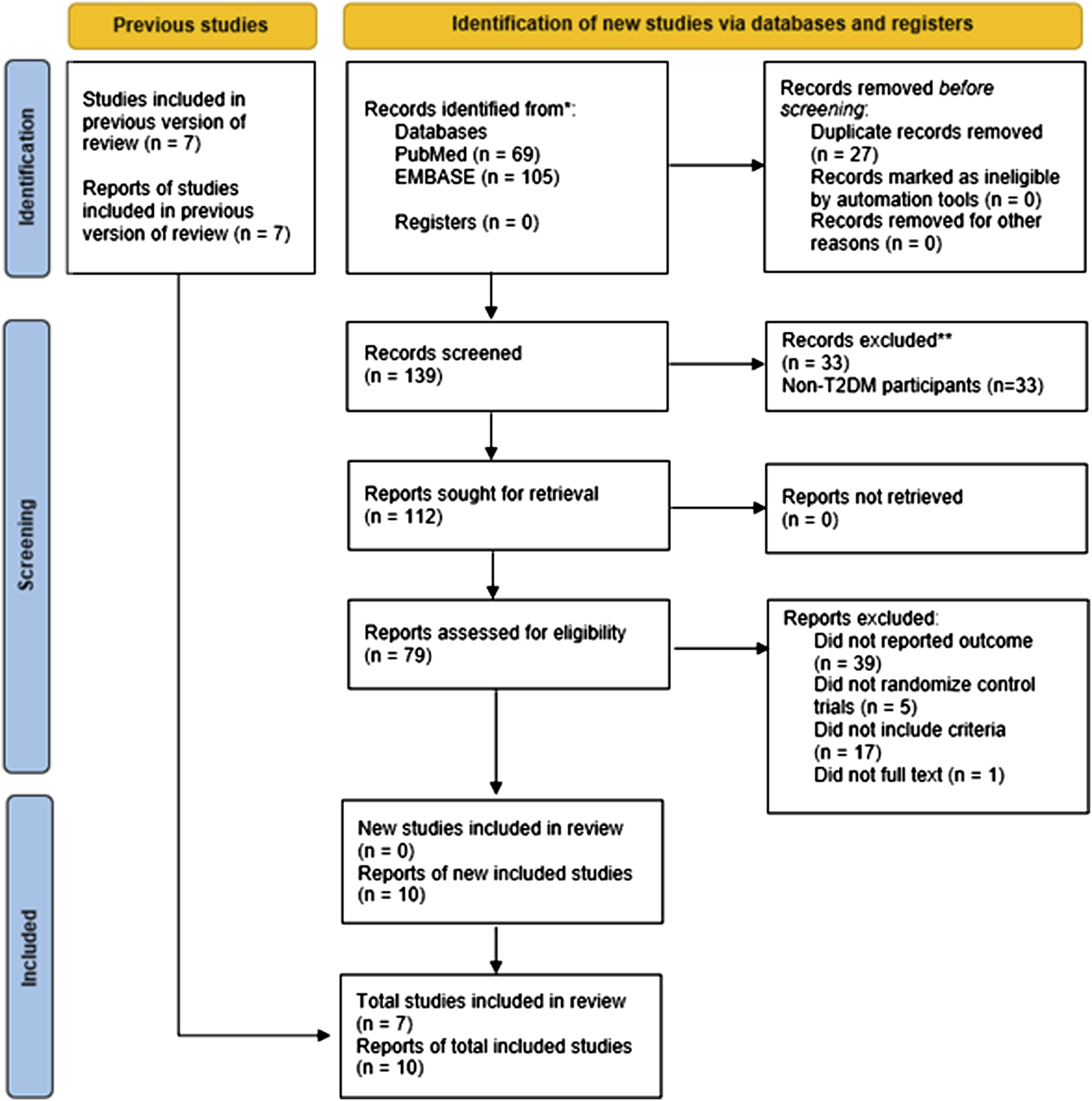
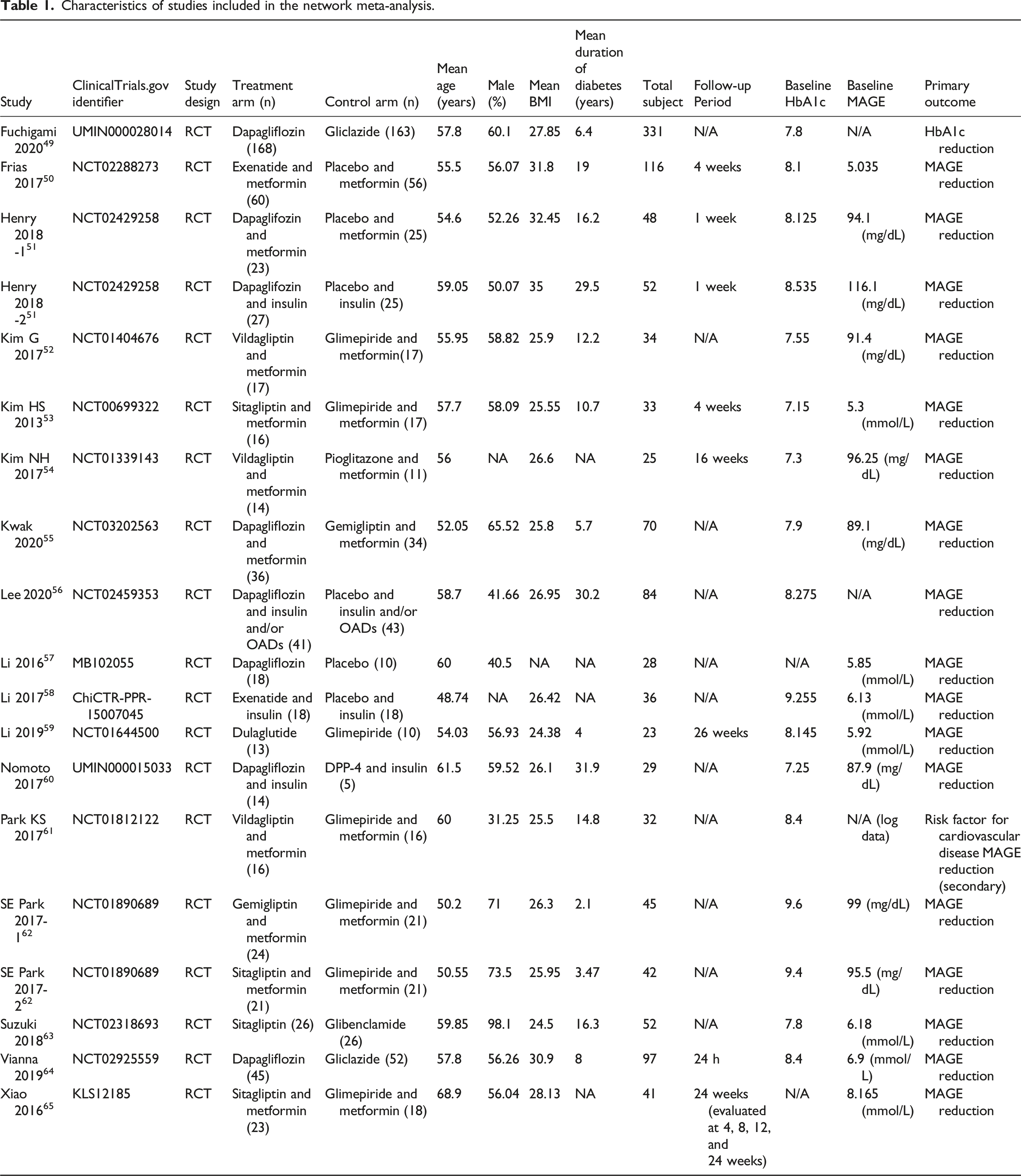
Characteristics of studies included in the network meta-analysis.
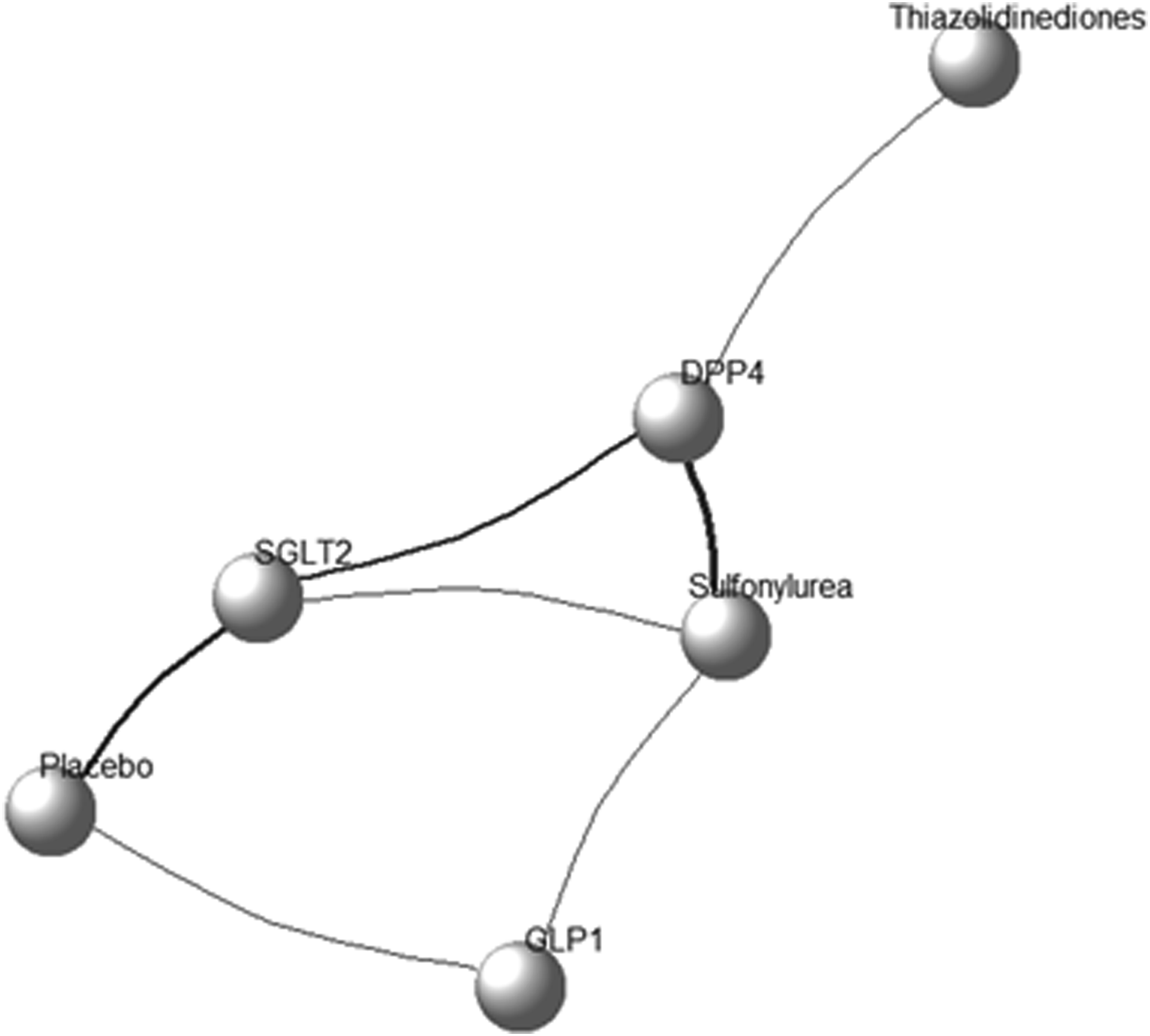
Network plot of HbA1c and MAGE. The size of the nodes is proportional to the number of subjects (sample size) randomized to receive the therapy. The width of the lines is proportional to the number of trials comparing each pair of treatments. GLP-1 = Glucagon-like-peptide-1 receptor agonists; DPP-4 = dipeptidyl-peptidase 4 inhibitor; SGLT-2 = Sodium-glucose cotransporter 2 inhibitors.
Mean amplitude of glycemic excursions (the mean amplitude of blood glucose fluctuations)
Seventeen RCTs were included in MAGE analysis, and 3 pairs were significantly more effective than placebo (DPP-4 [MD: −21.98, 95% CI: −35.91 to −8.04], GLP-1 [MD: −25.51, 95% CI: −37.86 to −13.15], and SGLT-2 [MD: −16.42, 95% CI: −27.36 to −5.48]) (Figure 3(a) and Supplementary Table S4). The network-estimated effect size between antidiabetic classes is shown in Table S7. There was no inconsistency between the direct and indirect comparisons of node-splitting analysis (Supplementary Table S5-6). In the heterogeneity analysis, the global I2 did not identify any heterogeneity across the studies (Supplementary Table S5, global I2 = 46.45%). Figure 4 shows the cumulative rank probability plot. GLP-1 RAs ranked highest (SUCRA 0.83), followed by DPP-4 inhibitors (SUCRA 0.72), thiazolidinediones (SUCRA 0.69), SGLT-2 inhibitors (SUCRA 0.51), and sulfonylurea (SUCRA 0.20). Table S9 details the results. Forest plot between antidiabetic treatment for (a) MAGE, (b) HbA1c in type 2 diabetes patients. HbA1c = hemoglobin A1c; MAGE = timing of mean amplitude of glycemic excursion; MD = mean difference; CI = confidence interval; GLP-1 = Glucagon-like-peptide-1 receptor agonists; DPP-4 = dipeptidyl-peptidase 4 inhibitor; SGLT-2 = Sodium-glucose cotransporter 2 inhibitors. Cumulative rank probabilities for MAGE between antidiabetic treatment in type 2 diabetes patients. Changes in the rank of treatments across different MAGE scores. Cumulative rank probabilities for each treatment were estimated using surface under the cumulative rank curve (SUCRA). SUCRA provided a single summary estimate of relative treatment efficacy by taking the average of cumulative rank probabilities of treatment GLP-1 being ranked 1st best among diabetes treatments. MAGE is best almost surely when the SUCRA index equals 1 and the worst if equals 0. GLP-1 = Glucagon-like-peptide-1 receptor agonists; DPP-4 = dipeptidyl-peptidase 4 inhibitor; SGLT-2 = Sodium-glucose cotransporter 2 inhibitors.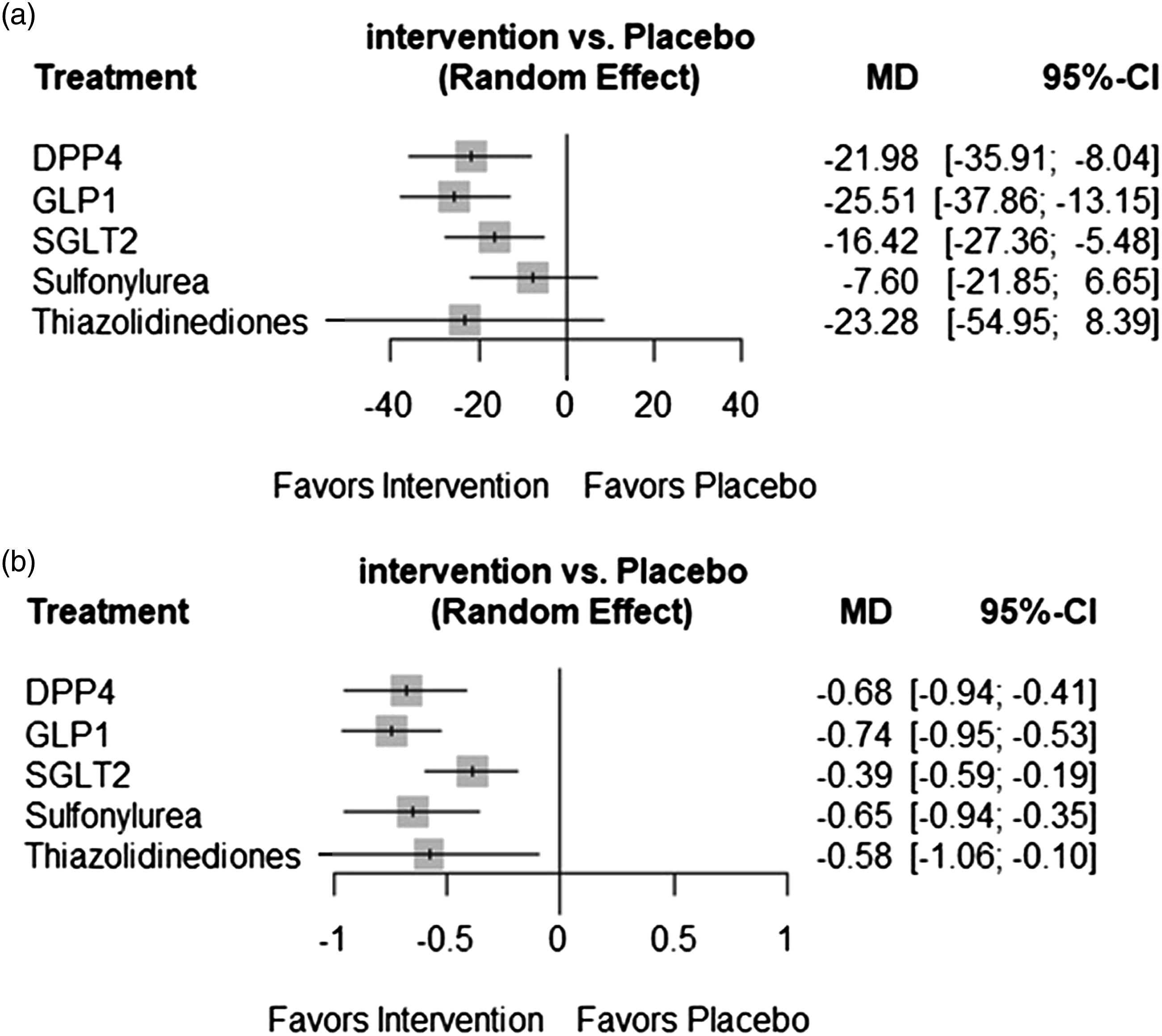
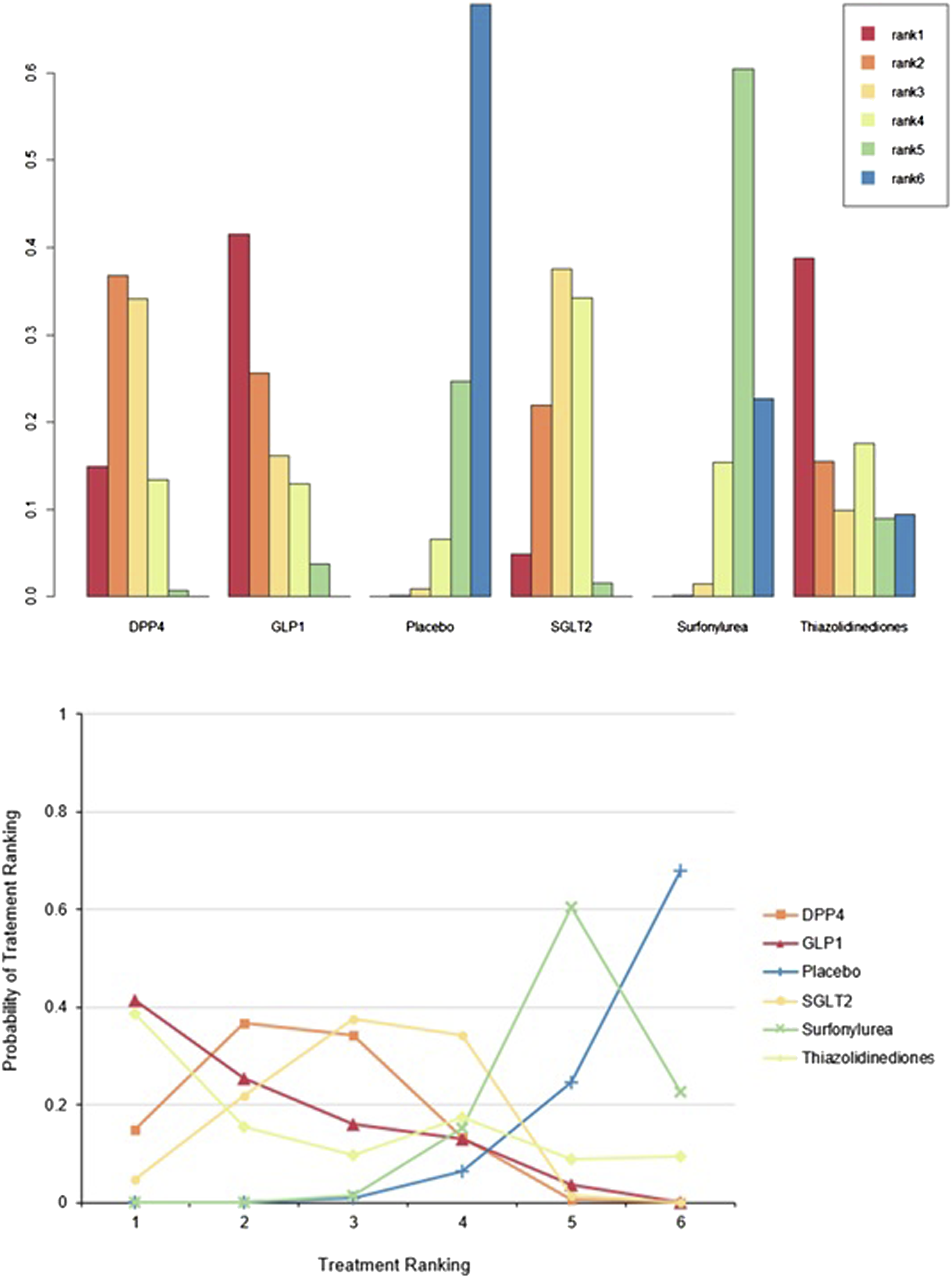
Hemoglobin A1c
Hemoglobin A1c was assessed in 12 RCTs. All interventions significantly decreased HbA1c more than when a placebo was used. (DPP-4 [MD: −0.68, 95% CI:−0.94 to −0.41]; GLP-1 [MD: −0.74, 95% CI: −0.95 to −0.53]; sulfonylurea [MD: −0.65, 95% CI: −0.94 to −0.35]; thiazolidinediones [MD: −0.58, 95% CI: −1.06 to −0.10]; and SGLT-2 [MD: −0.39, 95% CI: −0.59 to −0.19]) (Figure 3(a) and Table S4). HbA1c fluctuation was also conducted between anti-diabetes treatments (Supplementary Table S7). There was no inconsistency between the form of direct and indirect comparisons of node-splitting analysis (Supplementary Tables 5-6, p-value = 0.72). In the heterogeneity analysis, the global I2 did not identify any heterogeneity across the studies (Table S5, global I2 = 28.00%). Figure 5 shows the cumulative rank probability plot. GLP-1 RAs ranked highest (SUCRA 0.81), followed by DPP-4 inhibitors (SUCRA 0.72), sulfonylurea (SUCRA 0.65), thiazolidinediones (SUCRA 0.55), and SGLT-2 inhibitors (SUCRA 0.26). More detail is reported in Supplementary Table S9. Cumulative rank probabilities for HbA1c between antidiabetic treatment in type 2 diabetes patients. Changes in the rank of treatments across different HbA1c scores. Cumulative rank probabilities for each treatment were estimated using surface under the cumulative rank curve (SUCRA). SUCRA provided a single summary estimate of relative treatment efficacy by taking the average of cumulative rank probabilities of treatment GLP-1 being ranked 1st best among diabetes treatments. HbA1c is best almost surely when the SUCRA index equals 1 and the worst if equals 0. GLP-1 = Glucagon-like-peptide-1 receptor agonists; DPP-4 = dipeptidyl-peptidase 4 inhibitor; SGLT-2 = Sodium-glucose cotransporter 2 inhibitors.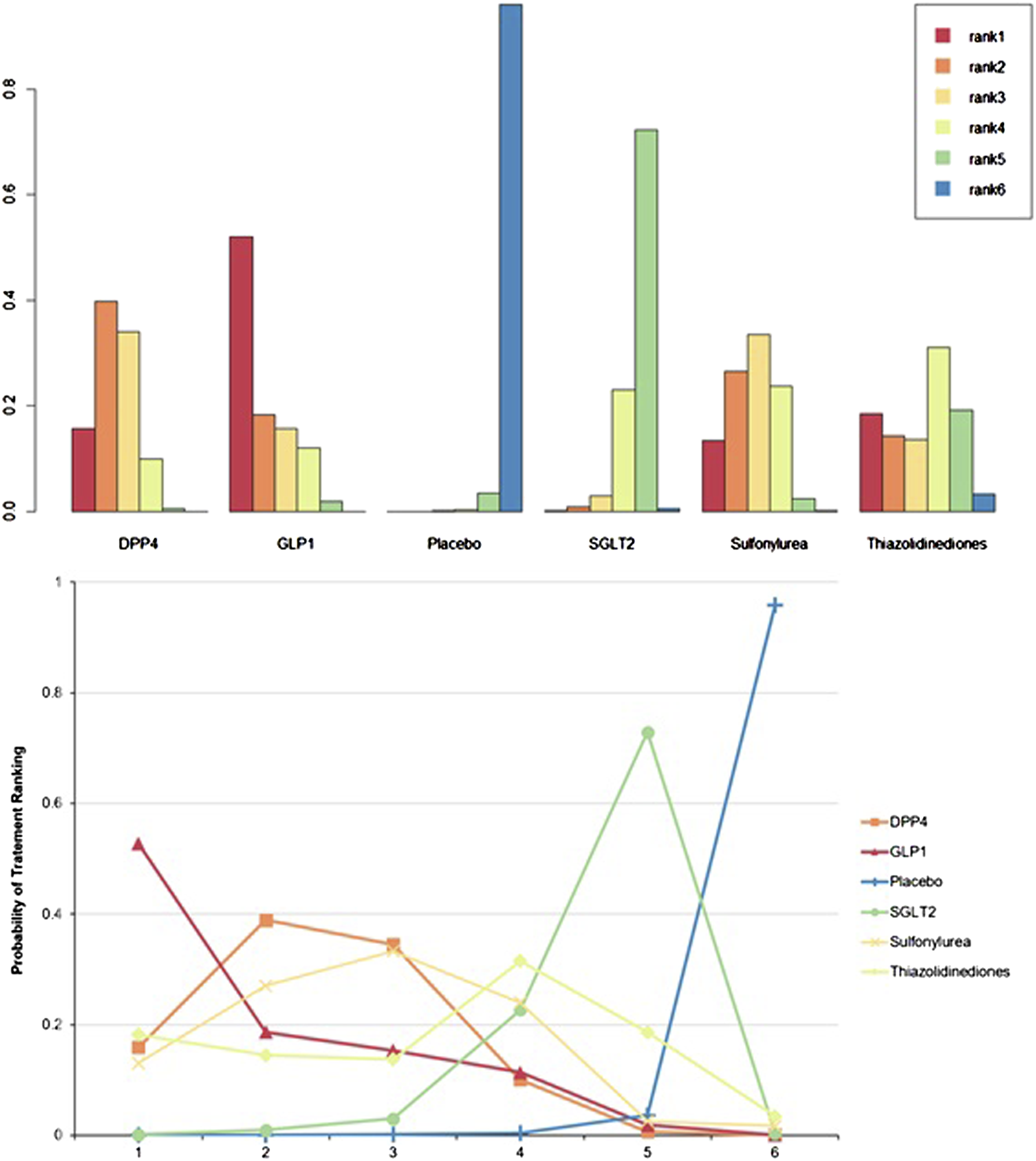
Certainty of evidence evaluated with GRADE framework.

aDowngraded by one when unable to evaluate inconsistency/heterogeneity due to lack of sufficient data (a single study).
Study design: If randomized trials form the evidence base, the quality rating starts at “high.” If observational studies form the evidence, base the quality rating starts at “low.”
Risk of bias: Downgraded for failure to conceal random allocation or blind participants in randomized controlled trials or failure to adequately control for confounding in observational studies.
Inconsistency: Downgraded if heterogeneity represented by I2 statistics or global inconsistency (Q statistic to assess consistency under the assumption of a full design-by-treatment interaction random-effects model) was high.
Indirectness. Downgraded when the assumption of transitivity is challenged, or the result is solely derived from indirect comparisons.
Imprecision: Downgraded when confidential interval (CI) is relatively too large compared to other active drugs.
Publication bias: Downgraded when substantial asymmetry is observed in funnel plot or p < 0.05 in the Egger test.
GRADE Definition (suggested by Puhan et al. in “A GRADE Working Group approach for rating the quality of treatment effect estimates from network meta-analysis”).
High quality: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate quality: We are moderately confident in the effect estimate, i.e., the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low quality: Our confidence in the effect estimate is limited, i.e., the true effect may be substantially different from the estimate of the effect.
Very low quality: We have very little confidence in the effect estimate, i.e., the true effect is likely to be substantially different from the estimate of effect.
Discussion
In this systematic review and network meta-analysis, we compared and evaluated the relative effects of each treatment on GV reduction among patients with type 2 diabetes. Maintaining a normal glycemic level is crucial for preventing or minimizing complications in the treatment of type 2 diabetes. 7 The results of our study revealed that GLP-1 RA was relatively associated with decreased MAGE and HbA1c levels compared with other treatments.
High GV is associated with an increased risk of diabetic macrovascular and microvascular complications, hypoglycemia, mortality, and other adverse clinical outcomes.6,28–30 The most known GV parameters, such as SD, CV, MAGE, are the most relevant metrics to assess glycemic control. 6 Although the clinical significance of intra-day and inter-day blood glucose variability for chronic complications is still controversial, 6 it is expected that the development of CGMS and the development of drugs to improve blood glucose variability will reveal the clinical significance and mechanism in the future. In this NMA, GLP-1 RAs had a relatively greater effect on MAGE reduction than other therapeutic agents, followed by DPP-4 inhibitors and SGLT-2 inhibitors. When compared with SU, both incretin-based therapy (GLP-1 RAs and DPP-4 inhibitors) and SGLT-2 inhibitors have high efficacies in GV reduction since their glucose-lowering mechanisms do not contribute to an increased risk of hypoglycemia.31,32 Incretin is a hormone released by the intestines in response to food consumption. Incretin medication, such as GLP-1 RA or DPP-4 inhibitor, controls meal-related glycemic excursions through augmentation of insulin and inhibition of glucagon secretion. 33 DPP-4 inhibitors rely on endogenous GLP-1 synthesis by increasing it’s half-life, but GLP-1RAs are resistant to DPP-4 degradation and provides super physiologically stimulate GLP-1 receptors.34,35 SGLT-2 inhibitors act by reducing the rate of renal tubular glucose reabsorption, which reduces blood glucose levels without stimulating insulin release. 36 However, because the majority of the studies included in this NMA are short-term, the long-term effects of each treatment on MAGE reduction remain unknown. Therefore, a prospective, multicenter study is warranted to investigate whether a drug or a class of drugs could provide better long-term GV control and whether treatment-mediated GV reduction can minimize the risk of hypoglycemia or vascular complications.
Regarding HbA1c reduction, all antidiabetic drugs exhibited a similar effect, with the exception of SGLT2-inhibitors, which were comparatively less effective. The relatively poor efficacy of SGLT2-inhibitors is likely due to the fact that this NMA included only a few small studies with heterogenous baseline characteristics and background therapy. Consequently, our study’s results may differ from those of other studies with larger sample sizes and adjustment for these confounders.15,37 Intriguingly, the reducing effect of SU on HbA1c observed in the current study is relatively weak, especially when compared with earlier studies.32,38 This is likely due to the fact that all the included studies that used SU as a comparator focused primarily on MAGE as a primary outcome rather than HbA1c reduction. Therefore, the results of our study regarding the HbA1c reduction efficacy should be interpreted with caution.
In terms of GV and HbA1c reduction, our study indicated that incretin-based therapy, including GLP-1RAs and DPP-4 inhibitors, is more beneficial than SGLT-2 inhibitors, SU, and TZD. However, before prescribing antidiabetic medications to a patient, several factors must be considered, including cardiovascular and renal comorbidities, body weight, hypoglycemia risk, cost and accessibility, and patient preference. 39 The latest diabetes guidelines recommend that patients who are unable to achieve their glycemic goal with oral antihyperglycemic drugs and require more aggressive glycemic control to consider combining GLP-1 RAs with basal insulin. 39 They also specify that while selecting additional medications for patients with type2 diabetes, the risk of cardiovascular disease and the possibility of related diseases must be considered.40,41 Based on this study’s findings, patients with diabetes and acute coronary syndrome as well as a high GV may benefit from GLP-1 RAs for preventing midterm MACE or the addition GLP-1 RAs to basal insulin could help patients achieve optimal glycemic control while minimizing the potential for hypoglycemia.42,43 In addition, incretin mimetics (GLP-1 RAs or DPP-4i) treatment would benefit patients dependent on insulin who are experiencing weight gain and hypoglycemia with a high GV profile. 44 However, SGLT-2 inhibitors are recommended for patients with diabetes with or at high risk of HF, diabetic kidney disease, clinically evident atherosclerotic cardiovascular disease, or any combination of these conditions. 45 Importantly, a decreased GV not only affects clinical outcomes. It has the potential to lead to improved treatment continuity, because it is associated with improved treatment satisfaction and quality of life. 6 Therefore, a suitable treatment for diabetes should not rely primarily on MAGE or HbA1c reduction, but should also consider the above mentioned factors.
Direct meta-analysis is limited by the few studies evaluating specific treatment pairs. 46 However, network meta-analysis provides useful evidence, including direct and indirect comparisons to carefully select the best treatment. 47 However, our analysis has several limitations. First not all studies reported HbA1c with MAGE, and only 12 studies reported HbA1c in this analysis. Therefore, HbA1c reduction should be carefully interpreted in this NMA. Second, GLP-1 RAs, which were the most effective in reducing HbA1c in this analysis, were not divided into short- and long-acting subclasses; hence, no analysis was conducted between these subclasses. Since previous studies have confirmed that short-acting GLP-1 RAs are more effective for GV than long-acting, a detailed classification of GLP-1 RAs is necessary. 48 Third, this systematic review and NMA included 17 small studies, and subgroup analysis could not be performed due to differences in the baseline characteristics and background treatment among the studies. Therefore, the power of the mean difference between pairwise comparisons is lacking, and heterogeneity may exist. Therefore, caution is needed in the interpretation of the pairwise indirect comparison, and additional research is needed according to the patient’s condition in accordance with the revised guidelines in future studies.
In conclusion, this systematic review and network meta-analysis comprehensively compared various therapeutics for HbA1c reduction as well as GV markers in patients with type 2 diabetes. This study demonstrates that GLP-1 RAs have relatively high efficacy in terms of both HbA1c and MAGE reduction compared to other antidiabetic treatments. To draw clear conclusions about the major clinical benefits and risks of these therapies, additional studies using a larger sample size and presence of classifications according to patient status are recommended.
Supplemental Material
Supplemental Material - Hypoglycemic agents and glycemic variability in individuals with type 2 diabetes: A systematic review and network meta-analysis
Supplemental Material for Hypoglycemic agents and glycemic variability in individuals with type 2 diabetes: A systematic review and network meta-analysis by SuA Oh, Purja Sujata, Hocheol Shin, Minji Kim and EunYoung Kim in Diabetes and Vascular Disease Research
Footnotes
Author Contributions
E.Y designed the study; S.A collected the data; S.A conducted the data analysis; S.A and E.Y interpreted results and drafted the manuscript; S.P, M.K, and H.C supported quality assessment; all authors finalized the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Research Foundation of Korea (NRF) grant funded by the Korean government (Ministry of Science and ICT, MICT) (NRF2021R1F1A1062044), and by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (Grant Number 2021R1A6A1A03044296), which had no further role in the study design, data collection, analysis, and interpretation, the writing of the report, or in the decision to submit the paper for publication.
Data availability statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
