Abstract
Insulin resistance has a broad pathogenic impact affecting metabolic, cardio-renal and other disease areas. Extensive studies to dissect the mechanisms of insulin resistance have provided valuable insights to shape current clinical awareness and advance therapeutic practice. However, the development of direct interventions against insulin resistance has been hindered by its complex and highly variable presentations, especially in type 2 diabetes. Among glucose-lowering agents, metformin and thiazolidinediones provide cellular actions that counter some effects of insulin resistance: reduced glucotoxicity and weight-lowering with antidiabetic therapies also improve insulin action, except that endogenously- or exogenously-created hyperinsulinaemia may partially compromise these benefits. Increasing awareness of the pervasiveness and damaging ramifications of insulin resistance heightens the need for more specifically targeted and more effective therapies.
Keywords
Introduction
This short review is written in recognition of the seminal works of Gerald Reaven on the role of insulin resistance in the pathogenesis of type 2 diabetes and cardiovascular (CV) disease. Foregoing reviews in this issue of Diabetes and Vascular Disease Research have eminently recounted these works.1–4 Here, we consider how a growing appreciation of insulin resistance influenced the development of new therapeutics in the field of diabetes.
Multifactorial pathophysiology
Reaven’s Syndrome X (not to be confused with the cardiac syndrome X) describes the clustering of CV risk factors that depend on or associate with insulin resistance. 5 Although Reaven’s6,7 Syndrome X later became subsumed within the so-called Metabolic Syndrome, the two syndromes are not synonymous: insulin resistance can promote CV disease independent of other CV risk factors commonly included in the Metabolic Syndrome such as excess adiposity, and insulin resistance is often associated with compensatory hyperinsulinaemia, at least in its early pathogenesis, which further aggravates metabolic, vascular and haemodynamic disturbances. Studies on the aetiology of insulin resistance and accompanying metabolic and CV abnormalities gave rise to a ‘common soil’ hypothesis of shared origins, and clinical practice recognised that the presence of one feature associated with insulin resistance should prompt suspicion about other CV risk factors. 8 This in turn has promoted a more holistic multifactorial approach to the assessment and management of type 2 diabetes to accommodate metabolic and cardio-renal aspects.9,10
Insulin resistance presents across a wide variety of phenotypes with different responses between tissues, organs and physiological systems. Because this is typically due to multiple ‘bottle-necks’ in the signalling pathways that mediate the biological actions of insulin, it has not been possible to identify a single cure-all molecular target. 2 However, several glucose-lowering agents can ameliorate the effects of insulin resistance with impact on diabetic complications, particularly in type 2 diabetes (Figure 1).
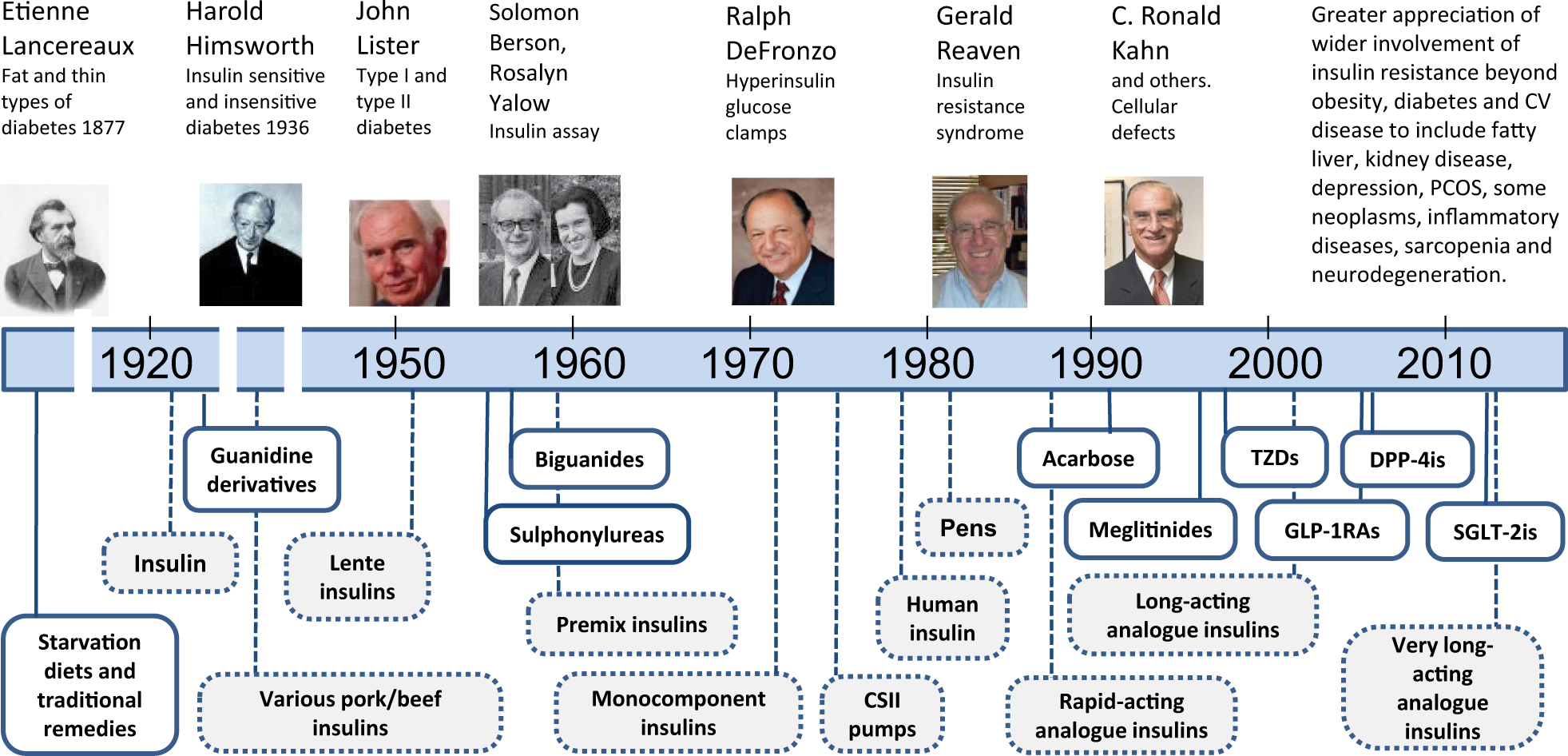
Timeline of some key events regarding insulin resistance and the introduction of diabetes therapies. Metformin was introduced in the United States in 1995.
Insulin
When Harold Himsworth described insulin resistance in the 1930s, he energised debate about different types of diabetes. 11 Studies by Yalow and Berson 12 in the late 1950s noted that insulin concentrations might actually be higher in the early stages of ‘maturity-onset diabetes’, which substantiated the Himsworth premise. Indeed, excess insulin has been mooted as a possible atherogenic factor, 13 and concern about the use of high doses of insulin therapy was highlighted by evidence that raised insulin concentrations do not rectify insulin resistance and may lead to an increasing spiral of insulin demand through further disruption to insulin receptor binding and post-receptor signalling. 14 Thus, an appreciation of Syndrome X helped to redirect attention towards sparing insulin rather than increasing insulin, particularly in the earlier stages of type 2 diabetes.
While this illustrates the rationale for changing the management focus of type 2 diabetes beyond insulin, the main alternative up to the 1990s was the use of sulfonylureas which act mostly by stimulating insulin secretion.
Sulfonylureas
The first sulfonylureas (e.g. carbutamide and tolbutamide) from the mid 1950s and more potent versions from the mid 1960s (e.g. glibenclamide) have well-studied glucose-lowering properties in type 2 diabetes, but incur weight gain and risk of hypoglycaemia. 15 However, the effects of sulfonylureas on insulin resistance have not been consistent, and CV effects are also unclear. The much criticised University Group Diabetes Programme (UGDP) cast doubt on the CV safety of tolbutamide in the late 1960s, 16 but the United Kingdom Prospective Diabetes Study (UKPDS)17,18 and many other trials have shown a better CV prognosis with sulfonylureas than diet/lifestyle but less beneficial than metformin. Similarly, the effects of meglitinides on insulin resistance and CV events remain unclear but appear minimal. Nevertheless, the detrimental impact of hypoglycaemia on CV events and the adverse effects of insulin resistance on islet beta-cell function must be considered in selecting glucose-lowering therapies.19,20
Biguanides
Although several guanidine derivatives were used in the treatment of diabetes in the 1920s and 1930s, their use dwindled as insulin became available, and it was not until the late 1950s that three biguanides (metformin, phenformin and buformin) were introduced in Europe and one (phenformin) was introduced in the United States. 21 Phenformin and buformin were withdrawn in the late 1970s due to high risk of lactic acidosis, and metformin was eventually introduced in the United States in 1995. Since biguanides lower blood glucose in type 2 diabetes without stimulating insulin secretion, it was acknowledged that they counter insulin resistance, and this was initially attributed to increased anaerobic metabolism and an independent reduction of hepatic gluconeogenesis. Lack of weight gain and low risk of hypoglycaemia favoured early use of metformin in type 2 diabetes, supported by mounting evidence for long-term reductions in CV disease.18,22 Through increased appreciation of the pathogenic effects of insulin resistance, Reaven’s studies contributed an important part of the scientific platform for metformin and its present-day position as first-line pharmacological therapy for type 2 diabetes. Reaven’s group also conducted several key studies on the mode of action of metformin, for example, showing the inter-relationship of effects on glucose and lipid homeostasis. 23 The group also noted that metformin prolongs insulin receptor tyrosine kinase activity. 24
Thiazolidinediones
Thiazolidinediones (TZDs) emerged from lipid-lowering clofibrate analogues in the mid 1970s, before peroxisome proliferator–activated receptor (PPAR) molecules had been discovered, but it was not until the late 1990s that the first PPARγ agonist (troglitazone) was introduced and then withdrawn due to unexplained hepatotoxicity. Rosiglitazone and pioglitazone followed promptly: they lowered plasma glucose without raising insulin, mostly through genomic effects that include differentiation of new small insulin-sensitive subcutaneous adipocytes, and improved insulin action in liver and muscle. 25 Pioglitazone also has some PPARα agonism which assists lipid control, but weight gain associated with adipose deposition and renal effects to increase fluid retention and risk of heart failure limited their use. Rosiglitazone was withdrawn in Europe in 2010 amid controversy over possible adverse CV effects, and potential risk of bone fractures has further limited use. Although TZDs provided an antidote to insulin resistance, their limitations illustrate the complexities and ambiguities of increasing insulin action across a breadth of biological functions without modulating effects in different tissues. 26
Incretins
Emanating from studies of the entero-insular axis, the availability of glucagon-like peptide-1 receptor agonists (GLP-1RAs) from 2005 and dipeptidyl peptidase-4 (DPP4) inhibitors from 2006 shifted the therapeutic focus of type 2 diabetes back to the pancreas. These agents do not carry the risk of hypoglycaemia seen with sulfonylureas because they potentiate insulin secretion and suppress glucagon secretion in a glucose-dependent manner (sulfonylureas stimulate insulin secretion independently of the glucose concentration).27,28 Interestingly, GLP-1 RAs and DPP4 inhibitors reduce insulin resistance: this appears to be due, at least in part, to a lowering of glucose concentrations, interrupting the vicious spiral of type 2 diabetes in which insulin resistance generates hyperglycaemia and the ensuing glucotoxicity aggravates insulin resistance. 20 The satiety effect of GLP-1RAs, which is associated with weight loss and decreased adiposity, provides further metabolic and endocrine mechanisms to reduce insulin resistance, and potential additional incretin-based therapies including peptide YY (PYY), oxyntomodulin, derivatives of gastric inhibitory polypeptide (GIP) and antagonists of ghrelin are under investigation. 29
Sodium/glucose co-transporter-2 inhibitors
Sodium/glucose co-transporter-2 (SGLT2) inhibitors introduced in 2012 reduce glucotoxicity and adiposity by eliminating excess glucose in the urine and thereby act indirectly to reduce insulin resistance and spare some of the demand on beta-cell function.30,31 Further evidence that lowering blood glucose will in turn lower insulin resistance is provided by alpha-glucosidase inhibitors which reduce prandial glucose excursions by slowing the rate of carbohydrate digestion.
Adipose and anti-obesity agents to reduce weight
Excess lipids, endocrine factors and pro-inflammatory molecules from adipose tissue are well known to promote the pathogenesis of insulin resistance in obese type 2 diabetes, and several adipokine-based therapies such as adiponectin receptor agonists are receiving consideration as potential approaches to counter insulin resistance. Improvements of insulin action and glycaemic control are consistently reported with caloric restriction and reduced adipose mass (particularly in omental, hepatic and pancreatic locations), whether achieved by dieting, bariatric procedures, SGLT2 inhibitors, GLP-1RAs or other appetite/satiety-modifying therapies.32,33 We may wonder why the age-old energy-reducing approach to treating obese-diabetes has taken so long to regain prominence.
CV and other considerations
It is perhaps an irony that one of the TZDs (rosiglitazone), which improved insulin sensitivity and reduced a range of atherogenic risk markers, should have triggered CV safety concerns and prompted current regulatory requirements for specific and extensive evaluation of CV events with new glucose-lowering agents. 26 Although there are many unanswered questions regarding the subtle interplay of insulin resistance and hyperinsulinaemia on the endothelium and myocardium, it is evident that early intervention to achieve long-term metabolic control and reduce insulin resistance provides an opportunity to improve CV prognosis.18,26 Timing appears to be especially important in this context because late interventions have been less successful against CV disease. The early development of hyperinsulinaemia with insulin resistance may promote hyperfiltration and damage to glomeruli, and insulin resistance is implicated in a wide variety of conditions including polycystic ovary syndrome and dementia indicating the breadth of potential benefits to be gained from effective timely interventions.1–4,34,35
Future
Awareness of insulin resistance as an underlying and modifiable pathogenic factor spanning diabetes, CV and other disease areas makes it an important therapeutic target. However, despite considerable appreciation of insulin–receptor interactions and post-receptor signalling, therapeutic interventions have been unable to rectify or circumvent the complex multi-dimensional defects of insulin resistance. 35 Several current therapies do act, at least in part, to address the metabolic disturbances and provide some protection against adverse CV events associated with type 2 diabetes, but it is unclear how these interventions will impact other disease areas susceptible to insulin resistance. New therapeutic approaches, including small non-peptide molecules that partially mimic insulin effects at the insulin receptor or initiate or potentiate receptor tyrosine kinase activity or target post-receptor pathways, have been identified, but these are still at early stages of investigation. 36 Thus, the therapeutic reversal of insulin resistance seems destined to be an ongoing unmet need for the foreseeable future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
