Abstract
Objective:
Evaluate the effect of sotagliflozin, a dual inhibitor of sodium glucose cotransporter (SGLT) 1 and 2, on arterial stiffness in patients with type 1 diabetes (T1D) treated with sotagliflozin as adjunct to optimized insulin therapy.
Methods:
In this post hoc analysis, indirect markers of arterial stiffness, including pulse pressure, mean arterial pressure (MAP), and double product, were calculated using observed systolic blood pressure (SBP), diastolic blood pressure (DBP), or pulse rate at 24 weeks using data from a pooled patient population from the inTandem1 and inTandem2 randomized controlled trials (n = 1575).
Results:
Baseline characteristics were similar among groups. Relative to placebo at Week 24, sotagliflozin 200 mg and 400 mg reduced SBP by 2.03 mm Hg (95% CI −3.30 to −0.75; p = 0.0019) and 2.85 mm Hg (−4.12 to −1.57; p < 0.0001), respectively. DBP decreased by 1.1 and 0.9 mm Hg, MAP by 1.4 and 1.6 mm Hg, and double product by 202.5 and 221.1 bpm × mm Hg, respectively (p < 0.05 for all). No increases in heart rate were observed.
Conclusion:
In adults with T1D, adding sotagliflozin to insulin significantly reduced blood pressure and other markers of arterial stiffness and vascular resistance.
Introduction
Type 1 diabetes (T1D) is associated with an increased risk of cardiovascular morbidity and mortality, driven by various cardiovascular risk factors including elevated HbA1c and hypertension.1,2 Up to 73% of persons with T1D have hypertension,3,4 which leads to arterial stiffness. Along with associated measures such as pulse pressure (a surrogate for pulse wave velocity), mean arterial pressure (MAP; a measure of cardiac output), systemic vascular resistance, central venous pressure, and double product (a measure of cardiac workload and myocardial oxygen demand), arterial stiffness is considered another independent risk factor for cardiovascular events in T1D.5,6
Sodium glucose cotransporter (SGLT) 2 inhibitors reduce measures of arterial stiffness in adults with type 2 diabetes (T2D),7,8 but these effects have been explored in T1D only in small, short-term studies of empagliflozin.9,10 In the inTandem phase 3 clinical trials, sotagliflozin, an oral dual SGLT1/SGLT2 inhibitor, improved glycemic control, body weight, and systolic blood pressure (SBP) when given as an adjunct to optimized insulin therapy in adults with T1D.11–13 The present analysis evaluated the effects of sotagliflozin on measures of arterial stiffness and vascular resistance in adults with T1D on optimized insulin therapy.
Methods
This post hoc analysis was conducted using pooled data from two 52-week, phase 3, multicentre, randomized, controlled trials—inTandem1 (NCT02384941; conducted in the US and Canada) and inTandem2 (NCT02421510; Europe and Israel)—which assessed the safety and efficacy of sotagliflozin 200 or 400 mg after a 6-week insulin optimization run-in.11,12 The trials had identical study designs wherein optimized insulin therapy continued over the 24-week core treatment period and a 28-week extension period. The trials were conducted in accordance with international standards of good clinical practice; the investigators obtained written informed consent from each participant; and the protocols were reviewed and approved by the respective institutional review boards or independent ethics committees. The trial designs and results have been published previously.11,12
This analysis included all randomized participants from both trials. Eligible patients were ⩾18 years of age with T1D for at least 1 year before trial entry with a screening HbA1c between 7.0% and 11.0% (53–97 mmol/mol). Body mass index (BMI) entry criteria were 20–45 kg/m2 inclusive. Patients were randomized 1:1:1 to placebo, sotagliflozin 200 mg, or sotagliflozin 400 mg, administered once daily as an adjunct to optimized insulin therapy. Patients continued their previous insulin delivery regimen—either continuous subcutaneous insulin infusion (CSII) or multiple daily injections (MDI)—throughout the trial.
This pooled data analysis was conducted using a combined data set from both studies. Endpoints included change from baseline in SBP, diastolic blood pressure (DBP), pulse rate, pulse pressure, MAP, and double product at Week 24 (primary efficacy endpoint). Pulse pressure was calculated as the difference between SBP and DBP. MAP was calculated as the sum of two-thirds of DBP and one-third of SBP. Double product was calculated as pulse rate multiplied by SBP.
Least squares (LS) mean changes from baseline, LS mean differences from placebo, and standard errors (SE) with 95% confidence intervals (CI) were obtained from mixed model repeated measure (MMRM) analysis with treatment, randomization strata of insulin delivery (CSII, MDI), baseline HbA1c category (⩽8.5%, >8.5%), study week, study (either inTandem1 or 2), and treatment-by-time interactions as fixed categorical effects, and baseline value as a covariate.
Results
Baseline demographic characteristics were similar across treatment groups for the 1575 adults included in this pooled analysis (Supplemental Table 1). Most patients were white, and approximately half were women. The mean age was 44 years, and duration of T1D was 21 years. Mean HbA1c was 7.7% (60 mmol/mol); total daily insulin dose was 0.74 IU/kg; and BMI was 29 kg/m2. Mean SBP/DBP was 122/76 mmHg; 27% had a baseline SBP ⩾130 mm Hg and 36% reported having a diagnosis of hypertension. During the treatment period, 73%, 77%, and 65% of patients in the placebo, sotagliflozin 200 mg, and sotagliflozin 400 mg groups, respectively, took a blood pressure lowering agent, the plurality of which were agents acting on the renin angiotensin system (Supplemental Table 1). Mean baseline pulse rate, MAP, and pulse pressure were 74 bpm, 91 mm Hg, and 45 mm Hg, respectively.
Relative to placebo at Week 24, sotagliflozin 200 mg and 400 mg reduced SBP by 2.03 mm Hg (95% CI −3.30 to −0.75; p = 0.0019) and 2.85 mm Hg (−4.12 to −1.57; p < 0.0001), respectively, and DBP by 1.09 mm Hg (−1.92 to −0.27; p = 0.0094) and 0.87 mm Hg (−1.70 to −0.05; p = 0.0384), respectively (Supplemental Table 2). Reductions in SBP observed at 4 weeks were sustained through 24 weeks (Figure 1). DBP reductions followed a similar pattern, and blood pressure changes were not associated with increases in pulse rate. Pulse pressure changed by −1.0 mm Hg (−2.1 to 0.0; p = 0.055) with sotagliflozin 200 mg and −2.0 mm Hg (−3.0 to −1.0; p < 0.001) with sotagliflozin 400 mg relative to placebo, and MAP by −1.4 mm Hg (−2.3 to −0.6; p < 0.001) and −1.6 mm Hg (−2.4 to −0.7; p < 0.001), respectively. Double product also decreased by 202.5 bpm × mm Hg (−356.2 to −48.8; p = 0.01) and 221.1 bpm × mm Hg (−374.8 to −67.2; p = 0.005) with sotagliflozin 200 and 400 mg versus placebo, respectively (Supplemental Table 2). Improvements in these parameters observed with sotagliflozin at 4 weeks persisted through 24 weeks (Figure 1).
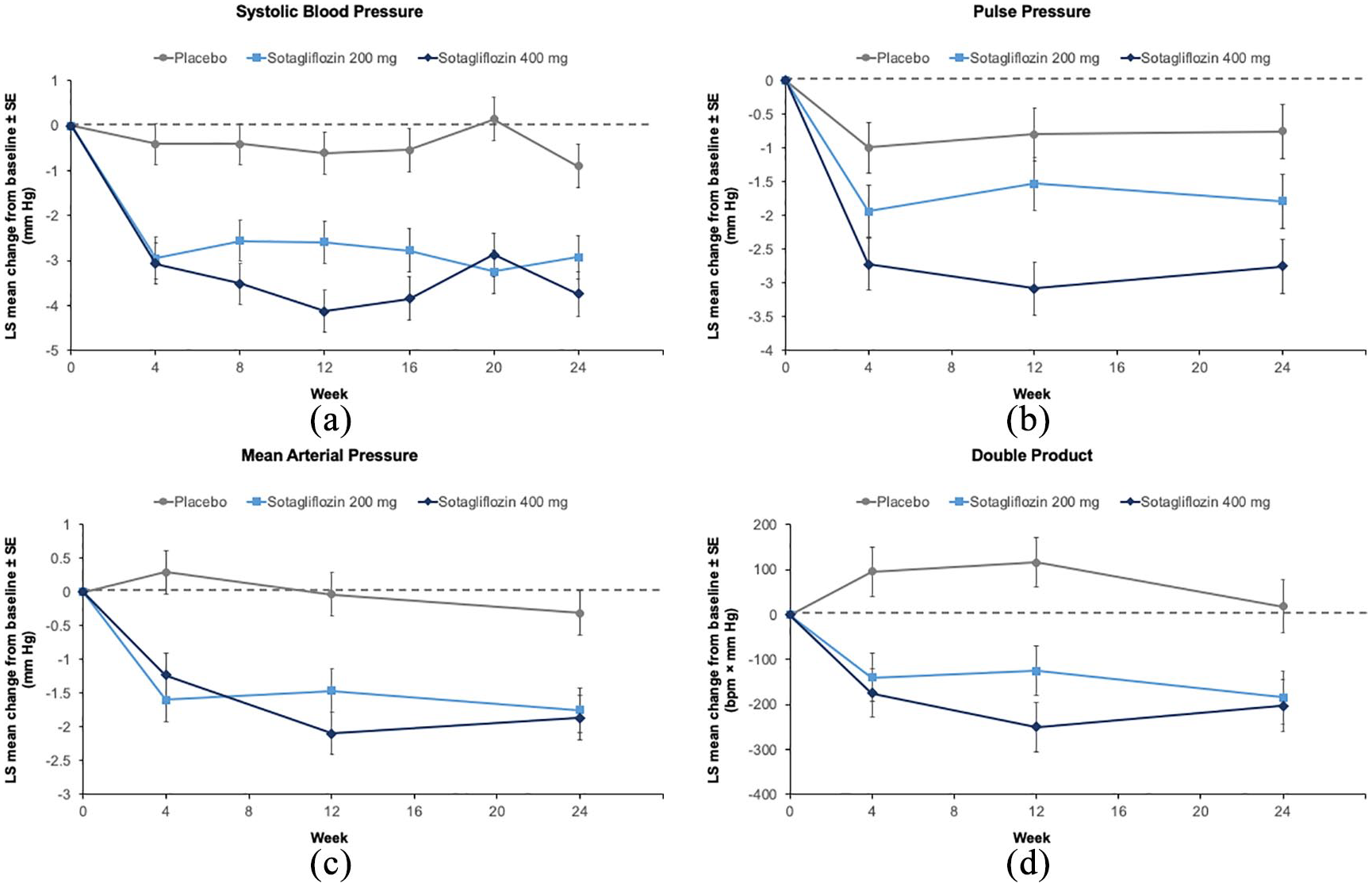
Least squares (LS) mean changes in systolic blood pressure (a), pulse pressure (b), arterial pressure (c), and double product (d) between baseline and week 24 in patients with type 1 diabetes.
Discussion
Among patients with diabetes, each 10-mm Hg increase in SBP, pulse pressure, or MAP increases the risk of CVD by 10%–17%. 6 The observed reductions in these indices in the present study are consistent with findings from studies of selective SGLT2 inhibitors in T2D. 14 In separate post hoc pooled analyses of patients with T2D, empagliflozin and canagliflozin significantly reduced SBP, DPB, pulse pressure, MAP, and double product, with no change in pulse rate.7,8
The present study only assessed blood pressure indices rather than direct measures of arterial stiffness. However, findings from small studies of selective SGLT2 inhibitors in T1D support the benefits of these reductions. During a glucose clamp study involving 40 young (mean age 24 years) patients with T1D, empagliflozin significantly reduced SBP, radial augmentation index, carotid augmentation index, and aortic augmentation index. 10 In another small T1D study, a combination of empagliflozin and metformin improved direct measures of arterial stiffness (pulse wave velocity and common carotid artery stiffness). 9 Collectively these results suggest that SGLT inhibitors improve indirect and direct measures of arterial stiffness in adults with T1D or T2D. These improved measures may contribute to the cardiovascular benefits observed with this class of medicines. 15 In addition, sotagliflozin may further enhance cardiovascular benefits through improvements in postprandial glucose excursions, glycemic variability, and time in range. 16 However, a head-to-head trial would be necessary to determine whether there was any difference between sotagliflozin and other SGLT inhibitors in this regard.
This analysis is limited by its post hoc nature, and the fact that the inTandem trials were not powered to assess arterial stiffness. Furthermore, the current study used estimated and not direct measurements of arterial stiffness. However, the present findings are consistent with those observed with other SGLT inhibitors, suggesting a robust finding across the class.
In conclusion, in this post hoc analysis, sotagliflozin 200 and 400 mg led to significant reductions in SBP and DBP as well as multiple indirect markers of arterial stiffness, including pulse pressure, MAP, and double product, without changes in pulse rates, in adults with T1D on optimized insulin therapy.
Supplemental Material
sj-pdf-1-dvr-10.1177_1479164121995928 – Supplemental material for Effect of sotagliflozin as an adjunct to insulin therapy on blood pressure and arterial stiffness in adults with type 1 diabetes: A post hoc pooled analysis of inTandem1 and inTandem2
Supplemental material, sj-pdf-1-dvr-10.1177_1479164121995928 for Effect of sotagliflozin as an adjunct to insulin therapy on blood pressure and arterial stiffness in adults with type 1 diabetes: A post hoc pooled analysis of inTandem1 and inTandem2 by Helena W Rodbard, Andrea Giaccari, Bertrand Cariou, Satish Garg, Michael J Davies, Kiernan Seth and Sangeeta Sawhney in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
We thank the patients and investigators for participating in the studies. Medical writing support was provided by Amanda M. Justice and funded by Lexicon Pharmaceuticals, Inc.
Author contributions
Authors working for Lexicon Pharmaceuticals, Inc., contributed to the statistical analysis of data, interpretation of data, and/or the writing of this manuscript. All authors had full access to all the data in the studies and had final responsibility for the decision to submit for publication. All authors participated in interpreting the data and critically revising the report, and all authors approved the final version to be published.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.W.R. was a consultant, speaker, and/or received research support from AstraZeneca, Bayer, BI/Lilly, Janssen, Lexicon, Merck, Mylan, Novo Nordisk, Sanofi, and Regeneron. A.G. has undertaken ad hoc consultancy or sponsored lectures for Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, MSD, Mundipharma, Novo-Nordisk, and Sanofi-Aventis and received research grants from AstraZeneca. B.C. has received research funding from Amgen, Pfizer, Sanofi, and Regeneron Pharmaceuticals Inc and has served on scientific advisory boards and received honoraria or consulting fees from Abbott, Akcea, Amgen, AstraZeneca, Genfit, Pierre Fabre, Lexicon Pharmaceuticals Inc., Eli Lilly and Company, MSD Merck & Co., Novo Nordisk, Regeneron, and Sanofi. His institution has received research trial support from Lexicon Pharmaceuticals Inc., and Sanofi. S.G. has received advisory board consulting fees from Medtronic, Roche, Merck, Lexicon, Novo-Nordisk, Sanofi, Mannkind, Senseonics, Zealand, and Eli Lilly and research grants from Eli Lilly, Novo-Nordisk, Merck, Lexicon, Medtronic, Dario, NCI, T1D Exchange, NIDDK, JDRF, Animas, Dexcom, and Sanofi. All research grants, honoraria, and travel expenses are received through the University of Colorado Denver. M.J.D. is employed by Esperion Therapeutics. K.S. is employed by Lexicon Pharmaceuticals, Inc. S.S. is employed by Immuvant, Inc. M.J.D. and S.S. were employed by Lexicon Pharmaceuticals, Inc., at the time the studies were conducted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lexicon Pharmaceuticals, Inc. designed and funded the studies, including the operational execution and medical monitoring of the studies.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
