Abstract
The sodium-dependent glucose transporter 2 (SGLT2) inhibitors are an important emerging class for the treatment of diabetes. Development of SGLT2 inhibitors has been oriented around a desire for high selectivity for the SGLT2 protein relative to the SGLT1 protein. More recently, genetic and pharmacology research in mice has indicated that gastrointestinal SGLT1 inhibition may also be an appropriate therapeutic target to treat diabetes. Combining SGLT1 and SGLT2 inhibition in a single molecule would provide complementary insulin-independent mechanisms to treat diabetes. Therefore, sotagliflozin (LX4211) has been developed as a dual inhibitor of SGLT1 and SGLT2. The differentiating clinical features of dual inhibitor of SGLT1 and SGLT2 include a large postprandial glucose reduction, elevation of glucagon-like peptide 1 and modest urinary glucose excretion. These features may have clinical implications for the use of sotagliflozin in the treatment of both type 1 and type 2 diabetes.
Keywords
Introduction
Sodium-dependent glucose transporter 2 (SGLT2) plays an important role in the regulation of blood glucose. 1 It is expressed predominantly in the kidney where it reabsorbs approximately 90% of the filtered glucose load. In patients with type 2 diabetes, SGLT2 is overexpressed and glucose re-absorption continues, exacerbating hyperglycaemia. 2 Inhibition of SGLT2 enhances urinary glucose excretion (UGE) and therefore represents a viable treatment strategy for the management of type 2 diabetes and potentially type 1 diabetes as well. At present, several SGLT2 inhibitors are approved in the United States and other countries for the treatment of type 2 diabetes.3 –5
SGLT1 is the primary transporter responsible for absorption of glucose in the gastrointestinal tract. 6 Similar to SGLT2 in the kidney, SGLT1 is overexpressed in the gastrointestinal tract of patients with type 2 diabetes. 7 It has been suggested that SGLT1 inhibition may not be desirable because infants born with homozygous loss of function mutations in the SGLT1 gene have glucose–galactose malabsorption causing diarrhoea that can be life-threatening, and because it is believed that phlorizin, a relatively non-selective dual inhibitor of SGLT1 and SGLT2, was limited in clinical use by gastrointestinal side effects. 8
In contrast, research at Lexicon Pharmaceuticals, Inc., suggested that partial SGLT1 inhibition might provide benefits that could not be achieved with SGLT2 inhibition alone. Lexicon Pharmaceuticals developed several thousand strains of genetically altered mice and performed physiologic tests that provided insight on the potential inhibition of many different proteins, including SGLT1 and SGLT2. 9
Background and conception of sotagliflozin
Mice lacking SGLT1 showed a key physiological pattern defining gastrointestinal SGLT1 inhibition. Postprandial glucose was dramatically reduced. Glucose was delivered more distally into the intestine, where it was metabolized by bacteria into short chain fatty acids. L-cells detected the glucose and short chain fatty acids, and in turn, they released glucagon-like peptide 1 (GLP-1). They also released peptide YY (PYY), a satiety hormone. These effects were not observed in SGLT2 knockout mice, confirming that they were the result of decreased SGLT1 and not SGLT2. 10
The experience with SGLT1 knockout mice also provided a perspective on safety. SGLT1 knockout mice maintained on a diet containing glucose and galactose exhibited unformed or watery stools, decreased food intake and reduced weight gain, which provided additional evidence that complete inhibition of intestinal SGLT1 might not be desirable. However, mice heterozygous for the SGLT1 mutation fed a diet containing glucose had normal stools, food intake and weight gain, but still exhibited increased delivery of glucose to the distal small intestine and caecum and increased GLP-1 release after a meal challenge containing glucose. This suggested that there might be a window for achieving a benefit from partial SGLT1 inhibition while avoiding the gastrointestinal side effects associated with complete inhibition of SGLT1. 10
Preclinical studies of sotagliflozin
Lexicon Pharmaceuticals developed a drug discovery programme that evaluated several drug candidates with differing degrees of SGLT1 and SGLT2 inhibition. Pharmacology studies with a variety of compounds confirmed that compounds such as sotagliflozin, with dual activity against SGLT2 and SGLT1, produced greater improvements in glycemic control and A1C than selective SGLT2 inhibitors, but did not trigger diarrhoea or other apparent gastrointestinal related adverse effects. Sotagliflozin was selected as the optimal drug candidate from this programme (Figure 1). 11
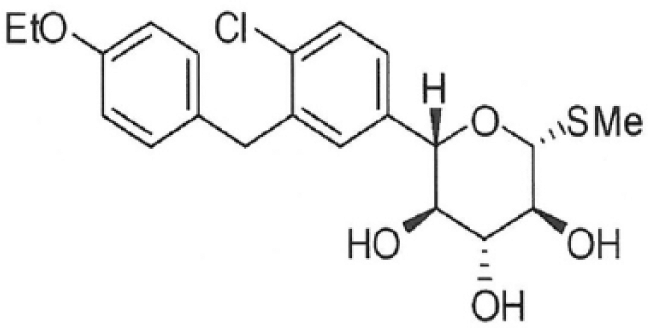
Structure of sotagliflozin.
The ability of sotagliflozin in vitro to inhibit SGLT1 and SGLT2 was established. Sotagliflozin inhibited SGLT2 with an IC50 (concentration causing half of maximal inhibition) of 0.0018 µM, and it inhibited SGLT1 with an IC50 of 0.036 µM. These results indicated that sotagliflozin is a dual inhibitor of SGLT1 and SGLT2 with approximately 20-fold selectivity for SGLT2 over SGLT1. 11 This dual inhibition was confirmed in animal pharmacology studies that demonstrated that sotagliflozin produced increased UGE, delivery of glucose to the caecum, increased postprandial GLP-1 and PYY release. These results were associated with significant reductions in postprandial glucose.12,13
Initial phase 1 studies of sotagliflozin
The initial single-dose and multiple-dose studies of sotagliflozin were performed in healthy subjects to identify its pharmacokinetic and pharmacodynamics properties. 14 A range of doses up to 500 mg in a liquid formulation was evaluated. The maximum circulating concentration on day 7 of sotagliflozin after multiple-dose administration was 165 ng/mL, and the total exposure (AUC0−tau) was 1172 ng h/mL. The pharmacokinetic half-life was 29 h, supporting once-daily dosing.
In these studies, the maximum 24 h UGE was 44 g when sotagliflozin was administered as a single dose. This was achieved at the 300 mg dose, and further increasing the dose did not provide greater UGE despite increased systemic exposure of sotagliflozin. The maximum UGE was 36 g on day 7 after multiple dosing. This UGE reflects a SGLT2 effect in the kidney, but it is less than reported with high doses of selective SGLT2 inhibitors. For example, 24 h UGE of 60–70 g has been reported after administration of canagliflozin in healthy subjects. 15 The apparently lower UGE with sotagliflozin may relate to intestinal SGLT1 inhibition; lower peak postprandial glucose levels mean less glucose is filtered by the kidney than with a selective SGLT2 inhibitor.
Phase 2a study of sotagliflozin and evaluation of GLP-1 response
Sotagliflozin was then evaluated in patients with type 2 diabetes not adequately controlled with metformin. 12 A total of 36 patients had their metformin withdrawn, and they were treated with sotagliflozin (150 mg or 300 mg qd in a liquid formulation) or placebo for 28 days (randomized 1:1:1) in a clinical research unit. Multiple metabolic parameters were followed.
Sotagliflozin reduced fasting plasma glucose by an average of 52 and 60 mg/dL in the 150- and 300-mg qd dosage groups, respectively, as compared with a 9 mg/dL increase in the placebo group. Glucose tolerance, as measured by the 4-h area under the curve (AUC) of a standard 75 g oral glucose tolerance test, was significantly improved with sotagliflozin. At day 27, glucose AUC values were 38% and 39% lower in the 150- and 300-mg qd dosage groups, respectively, as compared with the placebo group. Adjusting for baseline glucose values, glucose excursions during the oral glucose tolerance test at day 27 were 46% lower in both sotagliflozin groups compared with the placebo group (Figure 2).
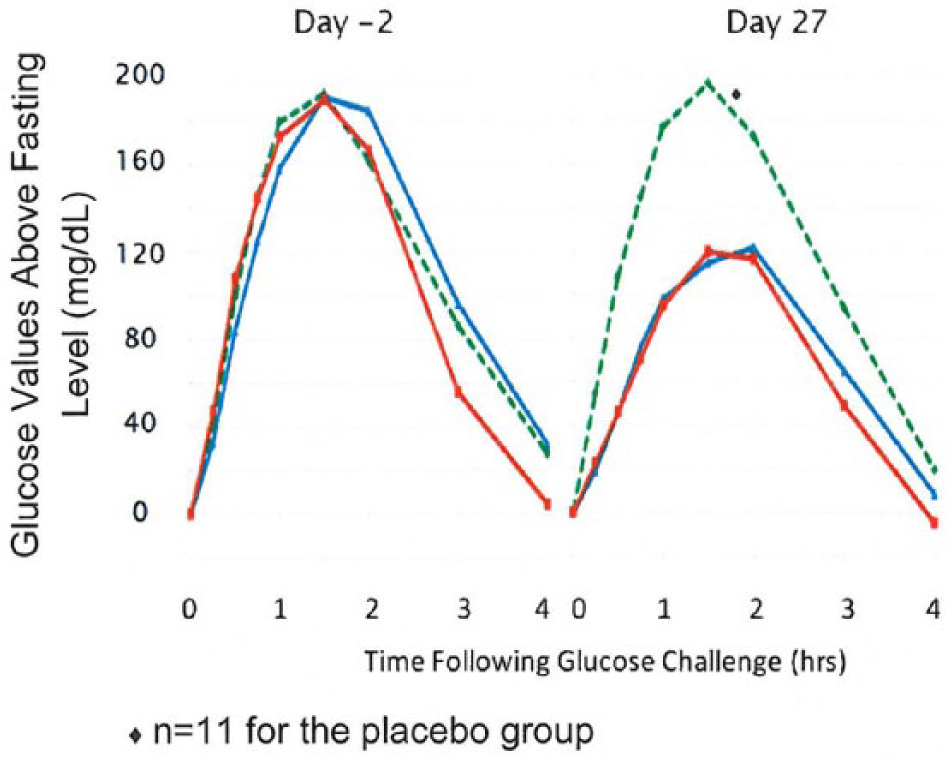
Postprandial glucose excursions with sotagliflozin in patients with type 2 diabetes. Baseline (day −2) and day 27 results are provided for placebo (green dashed line), 150 mg qd sotagliflozin (blue solid line) and 300 mg qd sotagliflozin (red solid line).
These improvements in glucose control were reflected by statistically significant reductions in A1C at 28 days. The A1C reductions were −0.49%, −1.15% and −1.25%, respectively, in the placebo, 150- and 300-mg qd dose groups (p < 0.05 for each group vs placebo). UGE over 24 h approached 50 g on the highest dose at day 28. As in the phase 1 experience, this is modest UGE compared with selective SGLT2 inhibitors. Generally, 80–100 g of 24 h UGE has been reported in patients with type 2 diabetes receiving canagliflozin at doses ⩾ 100 mg, and similar amounts have been reported with empagliflozin.15,16
The high glycemic efficacy observed with only modest UGE suggested that clinically relevant gastrointestinal SGLT1 inhibition was present. To further explore this hypothesis, a prospective test of the GLP-1 response to sotagliflozin was performed in a cohort of 12 additional patients with type 2 diabetes. Significant elevations of GLP-1 and PYY were identified over the course of 13 h (covering three scheduled meals) with sotagliflozin 300 mg qd. These changes were associated with significant reductions in glucose and insulin, without any change in glucagon.
The physiology of gastrointestinal SGLT1 inhibition was explored more thoroughly in a dose-timing study evaluating the effects of sotagliflozin on postprandial glucose, GLP-1, PYY and UGE. 17 A total of 12 healthy subjects received multiple doses of sotagliflozin (at a total daily dosage of 400 mg orally in a solid formulation, which was similar in exposure to 300 mg of the liquid) or placebo. The study included a crossover design where patients were dosed at different times relative to meals. Results demonstrated that dosing immediately before breakfast maximized the metabolic effects of sotagliflozin throughout the day.
The dose-timing study showed that sotagliflozin reduced postprandial glucose in healthy volunteers. The average peak glucose in the placebo group was slightly greater than 130 mg/dL after breakfast, while it was approximately 105 mg/dL with sotagliflozin. Reductions in postprandial glucose excursions were also observed at lunch and dinner. There were significant elevations in GLP-1 and PYY. This was the same pattern of gastrointestinal SGLT1 inhibition seen in preclinical studies.
There was also evidence of postprandial insulin reduction. The average peak insulin after breakfast was above 80 µU/mL on placebo. With sotagliflozin administered before breakfast, it was <40 µU/mL (Figure 3). Since healthy volunteers were absorbing less glucose, they released less insulin. These results strongly suggested that sotagliflozin might have a role in the treatment of type 1 diabetes: just as healthy volunteers released less insulin, patients with type 1 diabetes might require less meal-time (bolus) insulin.
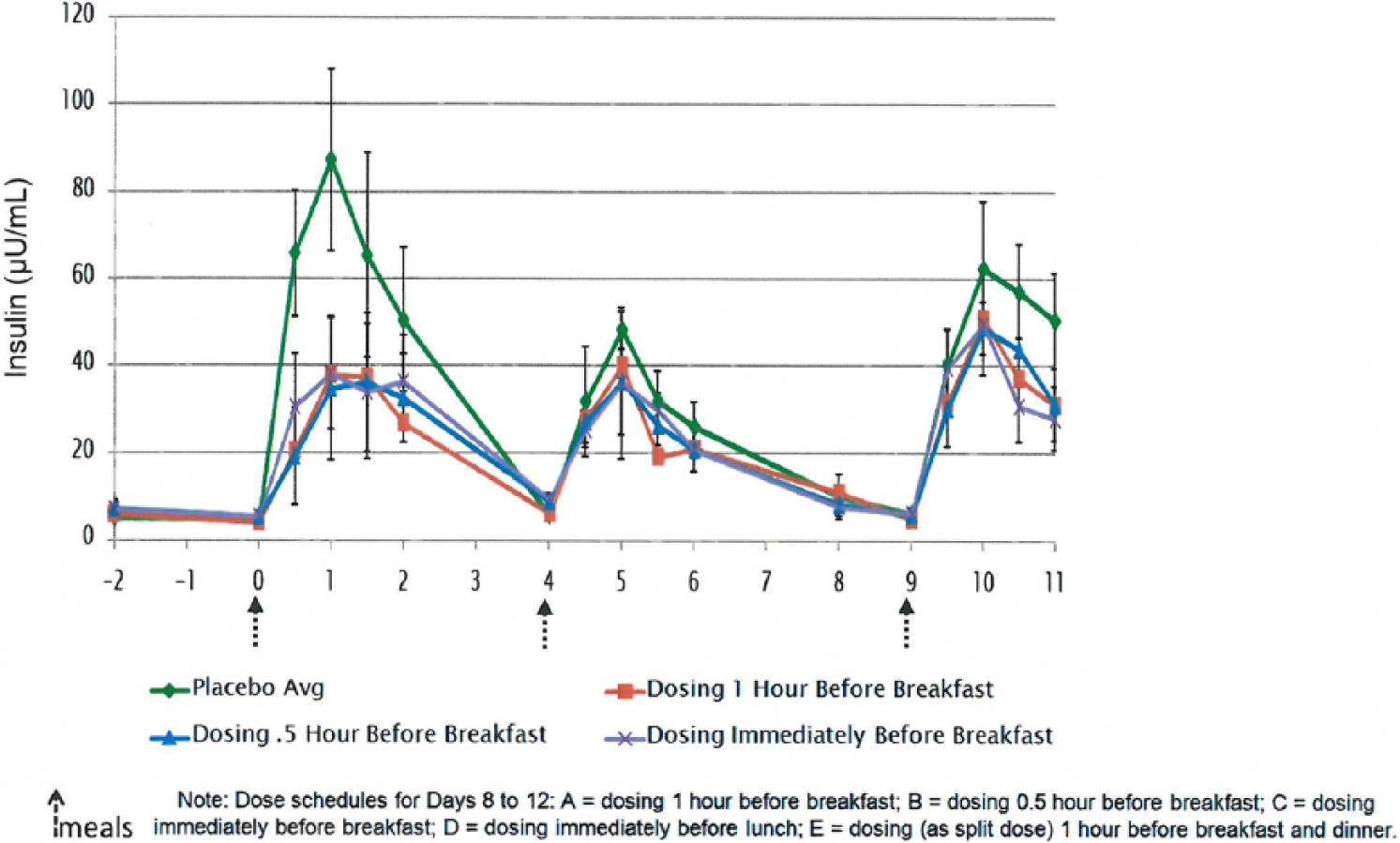
Insulin levels after sotagliflozin dosing in healthy volunteers.
The elevations in GLP-1 also suggested that sotagliflozin could have synergy with DPP4 inhibitors like sitagliptin. While sotagliflozin acts to enhance GLP-1 excretion, DPP4 inhibitors reduce the inactivation of GLP-1. A single-dose study was performed with 400 mg sotagliflozin (in tablet form) and 100 mg sitagliptin, administered as monotherapy or in combination in patients with type 2 diabetes. 18 This was a crossover study, with each participant washing out their metformin and then taking each of the three treatments (sotagliflozin alone, sitagliptin alone and the two in combination) in six different sequences, three patients each; 18 patients were enrolled and treated.
Sotagliflozin as monotherapy reduced postprandial glucose, elevated total GLP-1 and PYY and reduced insulin compared with the baseline assessment. When given with sitagliptin, there was a clear and statistically significant synergy in active GLP1-elevation. When taking the two together, patients with type 2 diabetes had significantly better glucose profiles that were achieved with less insulin. These results suggested that further study of the combination is warranted, and the insulin results further supported evaluating sotagliflozin for the treatment of type 1 diabetes.
To complete phase 2 development of sotagliflozin, three additional studies were designed. A 12-week dose-ranging study of patients with type 2 diabetes was conducted on a background of metformin.19,20 A second study was performed in patients with type 2 diabetes and renal impairment. 21 This is a special population where the gastrointestinal SGLT1 inhibition of sotagliflozin could be especially relevant since selective SGLT2 inhibitors have reduced efficacy in renal impairment. A third study was in patients with type 1 diabetes. 22
Phase 2b dose-ranging study of sotagliflozin
The dose-ranging study of sotagliflozin in patients with type 2 diabetes evaluated placebo and 75 mg qd, 200 mg qd, 200 mg bid and 400 mg qd doses of sotagliflozin administered in tablets. 19 The primary endpoint was A1C reduction. A total of 299 patients were randomly assigned, and the mean A1C at baseline was 8.1%.
Sotagliflozin decreased A1C in a dose-dependent manner throughout the 12-week study. 20 The 400 mg qd dosage reduced A1C by 0.92% (10 mmol/mol) from baseline, while placebo reduced A1C by 0.09%. The 400 mg qd dosage also produced the largest reduction in fasting plasma glucose, 29 mg/dL. Results for A1C and fasting plasma glucose reductions were highly significant, and there were also significant reductions in weight (approximately 2 kg at the 400 mg qd dosage) and blood pressure (approximately 6 mmHg systolic blood pressure for sotagliflozin vs placebo).
In the dose-ranging study, there were several assessments of UGE. Complete, 24-h urine collections were obtained in a sub-study. Spot checks of urine were also performed in the sub-study and in the population overall. All assessments identified a clear plateau in UGE that was reached at the lower 200 mg qd dosage despite a dose proportional increase in systemic exposure to sotagliflozin with the higher dosages.
The dose relationship to UGE was very different than the dose relationship to A1C reduction (Figure 4). The 400 mg qd dosage reduced A1C by 0.92% from baseline, while the 200 mg qd dosage reduced A1C by only 0.52% (Figure 4(a)). However, the added efficacy of 400 mg qd was achieved without any increase in UGE compared with 200 mg qd (Figure 4(b)). These data suggested that SGLT2-mediated glycemic efficacy peaked at 200 mg qd, while SGLT1-mediated effects increased with higher dosage. The UGE on 400 mg qd was <60 g/day, which appeared modest compared with reports of UGE for canagliflozin and empagliflozin.15,16 The amount of UGE may be relevant to safety events such as genitourinary infections and hypovolaemia.
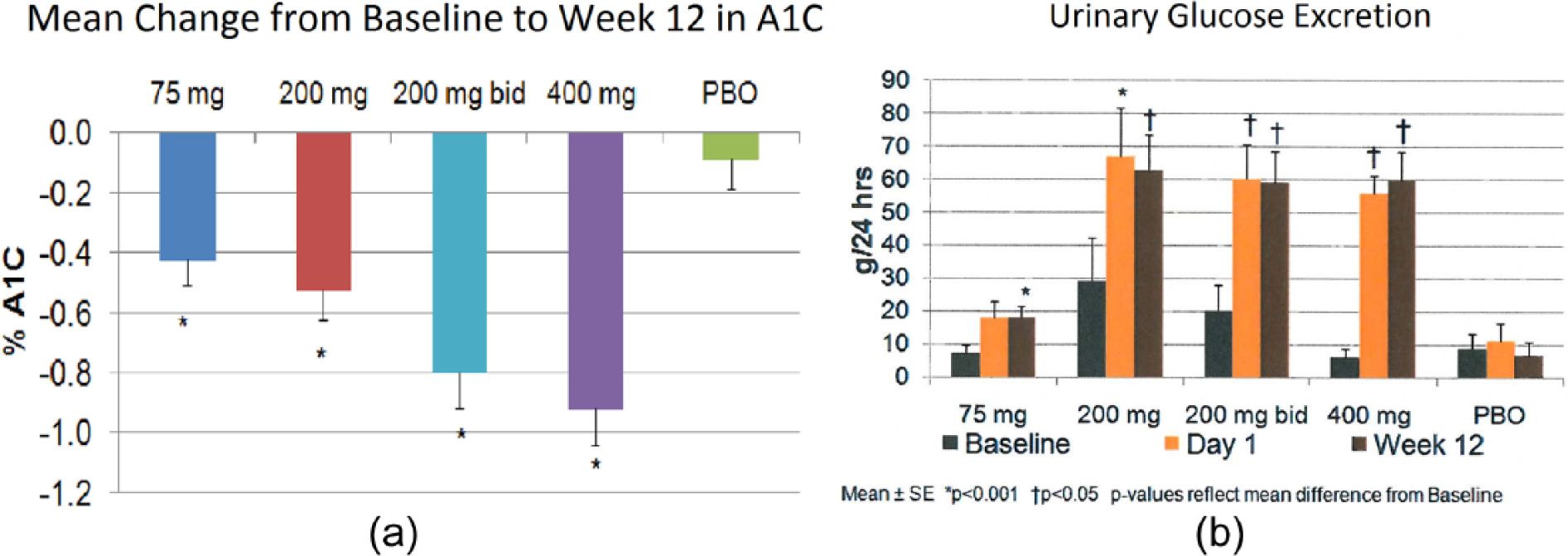
Changes in A1C and urinary glucose excretion with sotagliflozin in a 12-week dose-ranging study of patients with type 2 diabetes. The dose-response for (a) A1C reduction (Figure 4(a)) is different from that for (b) urinary glucose excretion (Figure 4(b)).
The dose-ranging study provided the largest sotagliflozin safety experience to date, involving administration of approximately 20,000 doses. Sotagliflozin exhibited a favourable safety profile and was well tolerated. The proportion of patients with adverse events was 66.7% on placebo and 57.6% on sotagliflozin 400 mg qd. The overall low incidence of adverse events with sotagliflozin may have been related in part to a reduction in metabolic adverse events. There were six cases of hyperglycaemia or worsening diabetes with placebo and only one with sotagliflozin 400 mg qd. There were no reports of hypoglycaemia.
The gastrointestinal safety profile of sotagliflozin was similar to that of placebo. The proportions of patients with gastrointestinal adverse events were 22% on sotagliflozin 400 mg qd and 20% with placebo. There were five reports of diarrhoea with sotagliflozin 400 mg qd and four with placebo. There was one case of constipation with sotagliflozin 400 mg qd and four on placebo. Nausea occurred in six patients with sotagliflozin 400 mg qd and three patients with placebo.
Genitourinary events, including urinary tract infections and vaginal yeast infections, occurred in three patients on sotagliflozin 400 mg qd and one patient on placebo. There were no discontinuations due to any of these events. There was no balanitis. Hypotension was reported in two patients. One received sotagliflozin 75 mg qd, and the other received placebo.
The overall experience of the dose-ranging study was consistent with sotagliflozin 400 mg qd providing clinically relevant SGLT1 inhibition in the gastrointestinal tract and SGLT2 inhibition in the kidney. While GLP-1 and PYY were not assessed in this study, the presence of gastrointestinal SGLT1 inhibition was suggested by the added efficacy achieved with 400 mg qd compared with 200 mg qd without any increase in UGE. The 400 mg qd dosage was selected for the remaining phase 2 studies.
Phase 2 study of sotagliflozin in renal impairment
While renal impairment studies often focus solely on pharmacokinetics, the sotagliflozin renal impairment study was designed to demonstrate significant improvements in glycemic control. 21 It was considered as a proof of concept for dual inhibition of SGLT1 and SGLT2. In a population where selective SGLT2 inhibitors have shown limited or no efficacy (targeting solely the kidney), sotagliflozin was hypothesized to produce clear signs of improved glycemic control with a pattern of gastrointestinal SGLT1 inhibition: large reductions in postprandial glucose, significant elevations in GLP-1 and PYY and modest UGE.
The renal impairment study randomly assigned 31 patients with type 2 diabetes and moderate to severe renal impairment [defined as estimated glomerular filtration rate (eGFR) between 15 and 59 mL/min/1.73 m2] to either sotagliflozin 400 mg qd or placebo. Treatment was given for 7 days. A pre-specified analysis divided the population into those with eGFR 45–59 mL/min/1.73 m2 (16 patients) and those with eGFR < 45 mL/min/1.73 m2 (15 patients). The cut-off of 45 mL/min/1.73 m2 was chosen because the US Food and Drug Administration (FDA) guidelines for selective SGLT2 inhibitors advise against their use in patients with this degree of renal impairment due to lack of efficacy.23 –25
Sotagliflozin 400 mg showed statistically significant reductions in postprandial glucose on day 7 compared with placebo (p = 0.005). These results reflected AUC for glucose measured at multiple time points from a pre-dose assessment to 4 h after a standard meal. Sotagliflozin also reduced fasting plasma glucose and elevated GLP-1 and PYY. Among those with eGFR < 45 mL/min/1.73 m2, the magnitude of effect was not reduced, and the statistical significance of postprandial glucose reduction was maintained (p = 0.002 for sotagliflozin vs placebo). A systolic blood pressure reduction of 10 mmHg versus placebo was seen (p < 0.05), and it was maintained in patients with eGFR < 45 mL/min/1.73 m2. These effects were achieved in the low eGFR group with an increase in UGE of only 20 g per 24 h versus placebo. Results suggest clinically relevant SGLT1 inhibition for this population.
A1C was not measured in this 7-day study, but the magnitude of improvement of glycemic control suggests the potential for significant A1C reduction in longer term studies of patients with type 2 diabetes and moderate to severe renal impairment. The AUC reductions for postprandial glucose reflect approximately 45–65 mg/dL differences in glucose during the assessment period after a standard meal for sotagliflozin compared with placebo.
The safety in the study supported further evaluation of the 400 mg qd dosage in longer term studies of renal impairment. There were no serious adverse events, no discontinuations due to adverse events, and the overall incidences of adverse events were similar with sotagliflozin and placebo.
Phase 2 study of sotagliflozin in type 1 diabetes
A phase 2 study of sotagliflozin in type 1 diabetes was recently completed. 22 It was initiated with a pioneer group of three patients treated in an open-label fashion with sotagliflozin 400 mg. The experience in these three patients was used to determine how best to adjust and evaluate insulin dosing in a subsequent cohort of patients randomized to sotagliflozin versus placebo. While the full results of the randomized cohort have yet to be published, a perspective of the pioneer group experience is provided here to describe the potential profile of dual inhibition of SGLT1 and SGLT2 in type 1 diabetes. The protocol was conducted in accordance with the Declaration of Helsinki.
Each of the pioneer patients entered a run-in period where the baseline levels of insulin use and glucose control were assessed. This period included the initiation of continuous glucose monitoring to ensure adequate safety and to provide a perspective of changes in postprandial glucose. Two nights before the end of the run-in period, patients were admitted to an in-patient unit. On the day before receiving sotagliflozin, a standardized breakfast was administered. On that night, the basal insulin (administered through a pump in each of the pioneer patients) was reduced by 10%.
On the next morning, patients received sotagliflozin 400 mg for the first time, immediately before the same standard breakfast given the day before. However, they received no breakfast bolus insulin. Decisions about insulin dosing at lunch and dinner were individualized based on the glucose response.
Continuous glucose monitoring results for the first patient are provided in Figure 5. The day prior to therapy (Figure 5(a)) had higher glucose excursions at breakfast, lunch and dinner compared with the first dose of sotagliflozin (Figure 5(b)). On the first day of sotagliflozin, the patient received no bolus insulin at breakfast, only 0.7 units at lunch, no bolus at dinner and the basal insulin rate had been reduced 10% the night before. The patient took sotagliflozin for 28 days as an outpatient, reducing basal insulin by 17% and bolus insulin by an average of 56% over that time period. The A1C was 7.1% baseline and 7.1% at the end of the study.
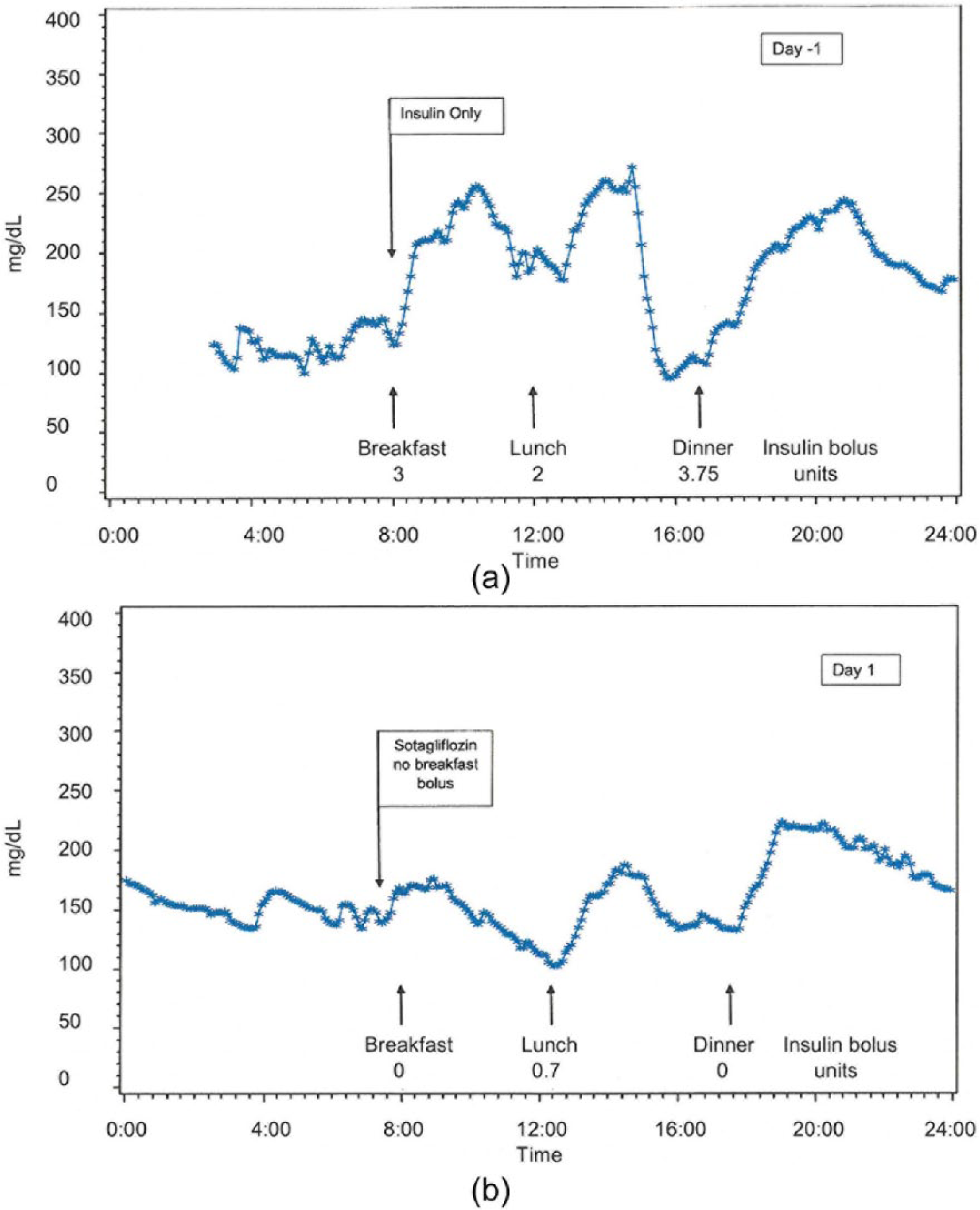
Continuous glucose monitoring of the first patient to receive sotagliflozin for the treatment of type 1 diabetes. Recordings are shown after standardized meals on (a) day −1 (Figure 5(a)) and (b) day 1 (Figure 5(b)).
The second pioneer patient experienced similar changes in glucose and insulin: basal insulin was reduced by 21%, and bolus insulin was reduced by 58%. The A1C was 8.8% at baseline and 6.9% at the end of the study.
Pioneer patient 3 had reductions of 22% and 20%, respectively, in basal and bolus insulin. The patient entered the study with poor glucose control and high insulin doses likely due to insulin resistance associated with obesity. The patient experienced a substantial reduction in the time spent with glucose values > 180 mg/dL, from 29% at baseline to 10% with sotagliflozin. At the same time, the patient reduced the percent time with glucose < 70 mg/dL from 8% to 3%. All three pioneer patients experienced weight reductions of approximately 2% of body weight in 28 days.
There were two episodes of mild hypoglycaemia during the 1-week baseline screening period (prior to administration of sotagliflozin) among the three pioneer patients. There was only one episode of mild hypoglycaemia during a cumulative experience of 12 weeks with administration of sotagliflozin.
The experience of the pioneer patients provides only a qualitative perspective of the mechanism of action of sotagliflozin. However, it suggests that a reduction in glucose absorption from the gastrointestinal tract may reduce glucose variability related to meals. Combining this effect with SGLT2 inhibition in the kidney may reduce the need to adjust insulin dosing decisions so frequently. As an adjunct to insulin, dual inhibition of SGLT1 and SGLT2 may help improve the overall benefit/risk of managing type 1 diabetes, allowing insulin to be used more effectively within a safer dose range.
Other recent studies with sotagliflozin have extended the safety experience and expanded the pharmacokinetic data by evaluating single doses up to 2000 mg and multiple doses up to 800 mg qd, all with sotagliflozin tablets.26,27 The results are consistent with those of the phase 2 studies and support longer term evaluation of sotagliflozin in a broad population.
Discussion
Efficacy of sotagliflozin
The clinical efficacy of sotagliflozin to date has been encouraging, suggesting robust A1C reduction, potential synergy with DPP4 inhibitors, possible efficacy in type 2 diabetes with renal impairment and the opportunity to improve the overall benefit/risk of treating type 1 diabetes. Larger and longer term studies are warranted. These can provide a better understanding of the degree of metabolic change over time with sotagliflozin. In the phase 2 dose-ranging study, A1C improved at each visit and had not yet clearly reached a plateau at 12 weeks. Similarly, weight in the sotagliflozin groups was changing throughout the study, and long-term weight change may contribute to A1C effects.
In the population with type 1 diabetes, dual inhibition of SGLT1 and SGLT2 could provide unique benefits to two distinct groups of patients. For the patient who has reasonably good A1C control, it may serve primarily to reduce bolus insulin dosing, in which case the benefits may be weight loss and a reduction in postprandial ‘overcorrection’ hypoglycaemia. Weight loss is relevant because 50% of patients with type 1 diabetes are either overweight or obese. 28 Hypoglycaemia is the limiting factor in controlling type 1 diabetes and remains a significant cause of death.29–31
For the patient with type 1 diabetes and poor glycemic control, the principal benefit of dual inhibition of SGLT1 and SGLT2 may be A1C reduction. The use of insulin alone has not been sufficient to provide A1C control for the majority of patients with type 1 diabetes. More aggressive use of insulin is not satisfactory for these patients because of the risk of hypoglycaemia. There is a need to make better A1C control a reality for patients with type 1 diabetes.
Another potential topic of long-term efficacy is cardiovascular risk. Several large cardiovascular outcome trials have failed to show clear benefits from oral antidiabetic agents.32,33 However, postprandial glucose has been identified as an important risk factor for cardiovascular disease in several cohorts, including the Honolulu Heart Study and others.34–36 The combination of postprandial glucose reduction, weight loss and blood pressure reduction seen with dual inhibition of SGLT1 and SGLT2 may be relevant to high-risk patients.
In this respect, it will be important to further characterize the blood pressure changes seen with sotagliflozin. In the dose-ranging study of patients with type 2 diabetes, the placebo-subtracted systolic blood pressure reduction with sotagliflozin 400 mg was approximately 6 mmHg (p < 0.001). In the renal impairment study, it was approximately 10 mmHg (p < 0.05), and the systolic blood pressure reductions were maintained even in those with eGFR < 45 mL/min/1.73 m2 and relatively low UGE.
These observations raise the question as to whether the blood pressure effects go beyond the diuresis of renal SGLT2 inhibition, and whether SGLT1 inhibition may contribute to blood pressure lowering. GLP-1 agonists are known to reduce systolic blood pressure, potentially by affecting the secretion of atrial natriuretic peptide.37,38 Therefore, the observed effects of increased circulating GLP-1 with sotagliflozin may contribute to its blood pressure lowering.
Weight loss also should be examined long-term, and its relationship to osmotic diuresis, GLP-1 and PYY release and potentially insulin reduction should be characterized. Weight loss in the range of selective SGLT2 inhibitors has been observed in all phase 2 studies of sotagliflozin, but there have not been yet detailed anthropometric measures or examination of body fat composition.
Safety of sotagliflozin
Longer term studies of sotagliflozin are needed to provide additional information on several aspects of safety. While UGE has been modest and there were few genitourinary infections in the phase 2 dose-ranging study, it is unclear whether the genitourinary safety profile will be different from that of selective SGLT2 inhibitors.
Another safety aspect is hypoglycaemia. The incidence of hypoglycaemia has been low across the clinical development programme to date, and there were no cases at all during the phase 2 dose-ranging study in patients with type 2 diabetes. It is possible that sotagliflozin may have a low incidence of hypoglycaemia due to its reduction of insulin requirements. This could be especially relevant in type 1 diabetes and for patients with type 2 diabetes who are already taking insulin.
The gastrointestinal safety profile of sotagliflozin to date contrasts with the prior focus of the medical and scientific community in avoiding SGLT1 inhibition. It has been suggested that phlorizin, a non-selective SGLT inhibitor, caused gastrointestinal adverse events. 16 However, a first-hand report of very high doses of phlorizin in healthy subjects described no diarrhoea. 39 Furthermore, phlorizin is rapidly metabolized into phloretin, which impacts glucose transporters beyond SGLT1 and SGLT2, so the history of phlorizin use cannot be considered a true test of the tolerability of SGLT1 inhibition. 16 It may be that sotagliflozin dosages above 400 mg qd daily could be less well tolerated, but the results for the 400 mg qd dosage to date reflect a relevant therapeutic window.
One potential aspect of SGLT1 inhibition may be transient nausea. There were six cases with sotagliflozin 400 mg qd compared with three on placebo in the dose-ranging study. The number of cases was too small to draw any conclusions, but nausea is an expected result of GLP-1 elevation and has been reported with GLP-1 analogs. 40
Transient declines in GFR have been reported with SGLT2 inhibitors, likely due to osmotic diuresis.23 –25 The same is expected with sotagliflozin. In patients with hypertension and chronic renal insufficiency, it is not uncommon for the serum creatinine concentration to rise as blood pressure is lowered. 41 A small, early rise followed by a stable plateau in creatinine in this setting has been associated with long-term protection of renal function. 42 However, close follow-up of creatinine may be important with sotagliflozin in the setting of renal impairment, just as it is for antihypertensive agents.
Conclusion
Dual inhibition of SGLT1 and SGLT2 with sotagliflozin has a clinical profile of postprandial glucose reduction, GLP-1 elevation and modest UGE. Phase 1 and phase 2 studies have described this pattern and have identified special opportunities for synergy with DPP4 inhibitors, for treatment of patients with type 2 diabetes and renal impairment and for the treatment of type 1 diabetes. Larger and longer term studies are warranted to fully characterize the efficacy and safety of sotagliflozin.
Footnotes
Declaration of conflicting interests
Dr Lapuerta is an employee of Lexicon Pharmaceuticals, Inc. and owns Lexicon stock. Dr Zambrowicz is an employee of Lexicon Pharmaceuticals, Inc. and owns Lexicon stock. Dr Strumph is an employee of Lexicon Pharmaceuticals, Inc. and owns Lexicon stock. Dr Sands is a former employee of Lexicon Pharmaceuticals, Inc., is a current consultant for Lexicon and owns Lexicon stock.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
