Abstract
Aim:
We examined eligibility and preventable cardiovascular disease events in US adults with diabetes mellitus from the Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME).
Methods:
We identified adults with diabetes mellitus eligible for EMPA-REG OUTCOME based on trial eligibility criteria available from the National Health and Nutrition Examination Surveys, 2007–2016. We estimated composite cardiovascular disease endpoints, as well as all-cause deaths, death from cardiovascular disease and hospitalizations for heart failure from trial treatment and placebo event rates, the difference indicating the preventable events.
Results:
Among 29,629 US adults aged ⩾18 years (representing 231.9 million), 4672 (27.3 million) had diabetes mellitus, with 342 (1.86 million) meeting eligibility criteria of EMPA-REG OUTCOME. We estimated from trial primary endpoint event rates of 10.5% and 12.1% in the empagliflozin and placebo groups, respectively, that based on the ‘treatment’ of our 1.86 million estimated EMPA-REG OUTCOME eligible subjects, 12,066 (95% confidence interval: 10,352–13,780) cardiovascular disease events could be prevented annually. Estimated annual preventable deaths from any cause, cardiovascular causes and hospitalizations from heart failure were 17,078 (95% confidence interval: 14,652–19,504), 14,479 (95% confidence interval: 12,422–16,536) and 9467 (95% confidence interval: 8122–10,812), respectively.
Conclusion:
Empagliflozin, if provided to EMPA-REG OUTCOME eligible US adults, may prevent many cardiovascular disease events, cardiovascular and total deaths, as well as heart failure hospitalizations.
The EMPA-REG OUTCOME trial has demonstrated patients with diabetes with known cardiovascular disease (CVD) benefitted from empagliflozin treatment over standard of care alone, with a significant 14% [95% confidence interval (CI): 1%–26%] reduction in the primary composite endpoint of death from CVD causes, non-fatal myocardial infarction or non-fatal stroke. Death from CVD was reduced by 38% (95% CI: 23%–51%), hospitalization for heart failure 35% (95% CI: 15%–50%) and death from any cause 32%. 1
Empagliflozin is a selective inhibitor of sodium glucose cotransporter 2 (SGLT-2) that decreases renal glucose reabsorption to alleviate hyperglycaemia in patients with type 2 diabetes mellitus (T2DM). In brief, eligible subjects for EMPA-REG OUTCOME trial included those who had T2DM and were aged 18 years and older with established CVD. Other inclusion criteria have been previously provided. 1 Table 1 shows the baseline characteristics of the EMPA-REG sample (placebo group data shown).
Descriptive statistics for EMPA-REG eligible participants in NHANES 2007–2016 (with comparison to EMPA-REG OUTCOME eligible participants).
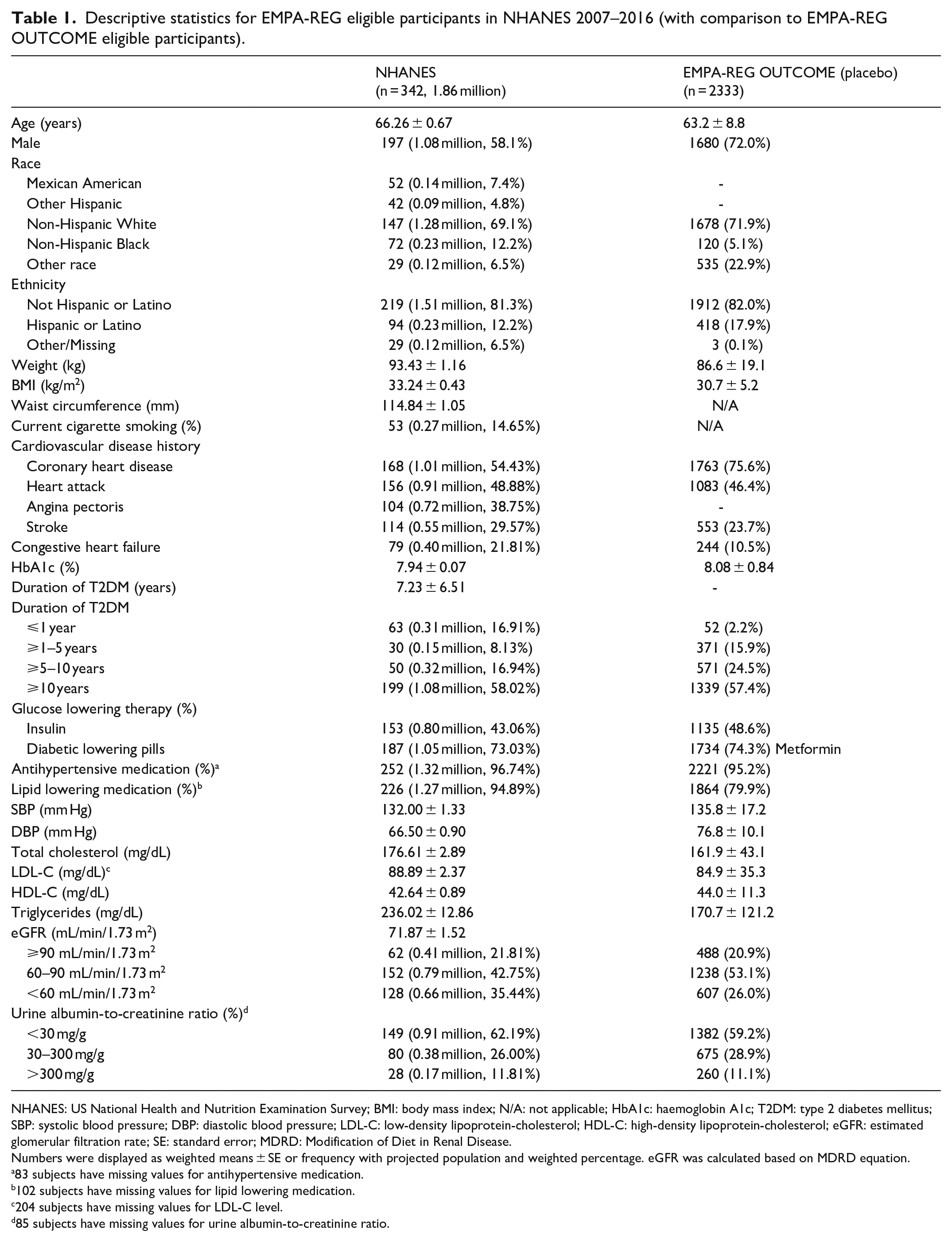
NHANES: US National Health and Nutrition Examination Survey; BMI: body mass index; N/A: not applicable; HbA1c: haemoglobin A1c; T2DM: type 2 diabetes mellitus; SBP: systolic blood pressure; DBP: diastolic blood pressure; LDL-C: low-density lipoprotein-cholesterol; HDL-C: high-density lipoprotein-cholesterol; eGFR: estimated glomerular filtration rate; SE: standard error; MDRD: Modification of Diet in Renal Disease.
Numbers were displayed as weighted means ± SE or frequency with projected population and weighted percentage. eGFR was calculated based on MDRD equation.
83 subjects have missing values for antihypertensive medication.
102 subjects have missing values for lipid lowering medication.
204 subjects have missing values for LDL-C level.
85 subjects have missing values for urine albumin-to-creatinine ratio.
To better understand the potential nationwide applicability and impact of the EMPA-REG OUTCOME study, it is of interest to understand the proportion and estimated number of eligible US adults with diabetes who would fit EMPA-REG OUTCOME criteria from which we are able to project the number of CVD events that could be prevented nationwide. In addition, an understanding of the current cardiovascular risk profile and use of diabetes and other medications among the general US population eligible for EMPA-REG OUTCOME would be of interest.
The US National Health and Nutrition Examination Survey (NHANES) offers an opportunity to examine these issues in non-institutionalized non-selected US adults with diabetes. We have shown barely 20% of US adults with diabetes and CVD are at recommended target levels of low-density lipoprotein cholesterol (LDL-C), blood pressure and haemoglobin A1c (HbA1c), 2 and that aggressive management of major risk factors could prevent over 50% of coronary heart disease events in US adults with diabetes, 3 identifying significant opportunities for these unmet needs.
In this project, we estimated the number of preventable CVD events in the US adult diabetic population from empagliflozin among EMPA-REG OUTCOME eligible persons, based on the number of US adults with T2DM we estimated would fit the EMPA-REG OUTCOME study criteria.
Methods
Sample inclusions
NHANES is a US population-representative cross-sectional evaluation of non-institutionalized US adults containing information on medical history, measured laboratory parameters and prescription medication information. We included US adults from five 2-year NHANES surveys from 2007 to 2016 with T2DM (based on age of onset of 30 years or greater using NHANES criteria), and of these, we utilized the following EMPA-REG OUTCOME eligibility criteria available in NHANES: (a) those who have a known history of CVD, including coronary heart disease, myocardial infarction, stroke or angina; (b) HbA1c of between 7% and 9%, or 7% and 10% if on hypoglycaemic therapy (insulin or oral hypoglycaemic medication); (c) estimated glomerular filtration rate (eGFR) of ⩾30 mL/min/1.73 m2; and (d) body mass index (BMI) of ⩽45 kg/m2. The total number of adults with T2DM fitting these criteria among these five surveys was then projected to the US population using NHANES 10-year sample weighting. This provided the estimated number of millions of persons with T2DM who were empagliflozin eligible based on the above criteria (other more specific eligibility criteria such as revascularization, angiographic stenosis and peripheral arterial disease are not available in NHANES). Sensitivity analyses were done comparing eligible estimated number of subjects across each 2-year survey using 2-year sample weighting each. Since this study involved the use of publicly available de-identified data, it was exempt from the institutional review board review and did not qualify for human subjects research at the University of California, Irvine.
Statistical analysis
We initially examined demographic (age groups, sex, ethnicity), clinical and risk factor distributions [mean/standard deviation (SD) of age, total cholesterol, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) cholesterol, triglycerides, systolic and diastolic blood pressure, HbA1c, BMI, duration of T2DM, eGFR based on creatinine measures, proportion with hypertension, cigarette smoking, family history of CVD and in different T2DM duration categories, eGFR categories, and albumin to creatinine ratio categories] among our empagliflozin eligible subjects, as well as proportions on self-reported insulin or oral hypoglycaemic medication, antihypertensive medication and lipid lowering medication. We then described how these demographic, clinical and risk factor characteristics compared to the actual EMPA-REG OUTCOME trial enrolled participants. Comparisons are made to the EMPA-REG OUTCOME placebo group patients, who were similar in baseline characteristics, including other background diabetes therapy, to the empagliflozin treated participants.
Finally, we estimated the number (in thousands) of preventable CVD events (primary and secondary composite endpoints), heart failure hospitalizations and cardiovascular and total deaths using sample weighting and applying the EMPA-REG OUTCOME published placebo and treatment group event rates for each of these endpoints to the weighted population of empagliflozin eligible US adults with T2DM as estimated in Wong et al. 2 The difference between the estimated number of events based on the placebo and treatment group event rates for each endpoint was designed as the number of preventable events. Analyses were done based on the entire 10-year sample using SAS (version 9.3; SAS Institute, Cary, NC, USA). Population weighted estimates were provided by PROC SURVEY FREQ with the CLWT option providing 95% CIs for these weighted estimates. These were multiplied by the corresponding event rates to get the 95% CIs for the estimated number of events.
Results
From 29,629 US adults we identified (representing 231.9 million), 4672 (27.3 million) were identified with T2DM of which 1067 (6.0 million) had known CVD. After applying other exclusion criteria as available in NHANES, including those without known CVD, HbA1c not between 7% and 10%, BMI >45 kg/m2, eGFR <30 mL/min/1.73 m2, and those with liver disease, we identified 342 subjects representing 1.86 million (95% CI: 1,592,599–2,120,022) US adults who would fit EMPA-REG OUTCOME trial eligibility criteria (Figure 1).
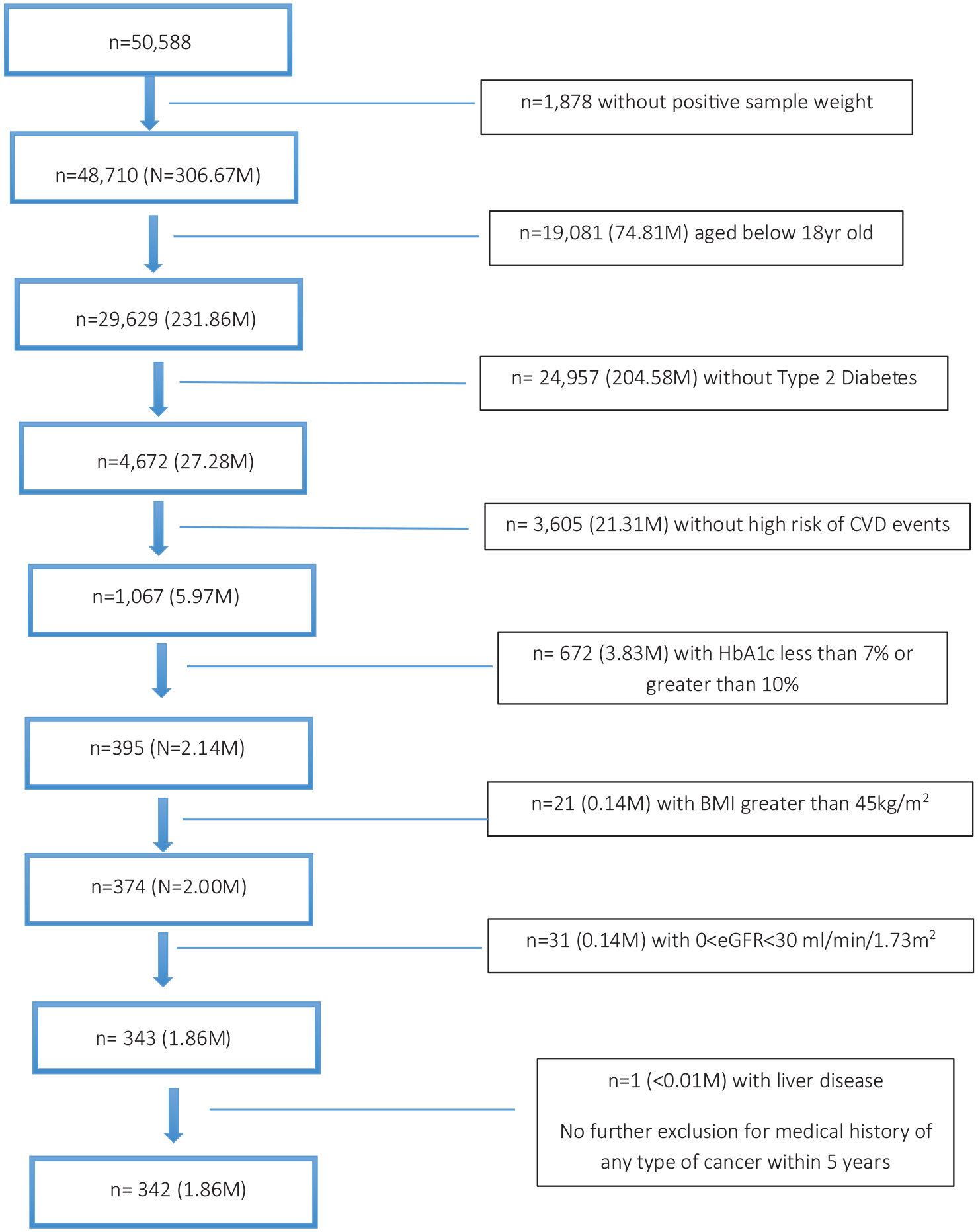
EMPA-REG OUTCOME eligible sample selection from the National Health and Nutrition Examination Surveys, 2007–2016.
Table 1 compared demographic and clinical factors between those EMPA-REG OUTCOME eligible US adults in NHANES with the EMPA-REG OUTCOME placebo group reported values. Our NHANES EMPA-REG OUTCOME eligible US adults compared to the EMPA-REG OUTCOME placebo group subjects tended to be on average 3 years older, less likely male, more likely non-Hispanic Black, with greater BMI, higher total- and LDL-cholesterol, triglycerides and lower systolic and diastolic blood pressure. Our NHANES eligible subjects were also less likely to have coronary heart disease but more likely to have chronic kidney disease (CKD; eGFR <60 mL/min/1.73 m2), prior stroke and heart failure. Patients in our NHANES sample tended to have more recently diagnosed T2DM and more likely to be on lipid lowering medication. Importantly, however, use of insulin and metformin was similar in our EMPA-REG OUTCOME eligible NHANES sample compared to actual EMPA-REG OUTCOME placebo subjects; 15% of our sample were current cigarette smokers and the mean waist circumference of 115 mm indicated most had abdominal obesity. Across survey year, there was a modest variation in the proportion of US adults eligible for empagliflozin based on EMPA-REG OUTCOME criteria, ranging from as low as 0.68% (5.5% of those with T2DM) in 2013–2014 to 0.99% (7.6% of those with T2DM) in 2015–2016, without any significant trend across survey years, with overall eligibility being 0.80% of all adults and 6.8% of those with T2DM (data not shown in the table).
Table 2 shows the estimated number of primary and secondary outcomes and individual endpoints (among those significant in EMPA-REG OUTCOME trial) resulting from empagliflozin ‘treatment’ based on our estimated 1.86 million US adults fitting EMPA-REG OUTCOME eligibility. The estimated number of events if not on empagliflozin (based on the placebo event rates) or on empagliflozin (based on the empagliflozin group event rates) was displayed, with the difference between the two being the estimated preventable CVD events. Using the respective EMPA-REG OUTCOME annual event rates, we also estimated the respective annual events that would occur in each group, with the difference between the preventable events per year. We found approximately 29,700 (95% CI: 25,482–33,920) preventable CVD event rates based on the duration of the EMPA-REG OUTCOME trial (median follow-up time of 3.1 years), translating to 12,066 (95% CI: 10,352–13,780) estimated annual preventable CVD events from empagliflozin treatment. Secondary outcome events prevented were 27,844 (95% CI: 23,889–31,800) or 11,323 (95% CI: 9715–12,932) per year, respectively. Of note, deaths from any cause prevented were 48,263 (95% CI: 41,408–55,121) or 17,078 (95% CI: 14,652–19,504) per year, death from cardiovascular causes 40,838 (95% CI: 35,037–46,640) or 14,479 (95% CI: 12,422–16,536) per year, hospitalizations for heart failure prevented were 25,988 (95% CI: 22,296–29,680) or 9467 (95% CI: 8122–10,812) per year and combined hospitalizations for heart failure or death from cardiovascular causes prevented were 51,976 (95% CI: 44,593–59,361) or 19,306 (95% CI: 16,563–22,048) per year.
Number and absolute event rate of expected preventable cardiovascular outcomes if on EMPA-REG OUTCOME, NHANES 2007–2016.
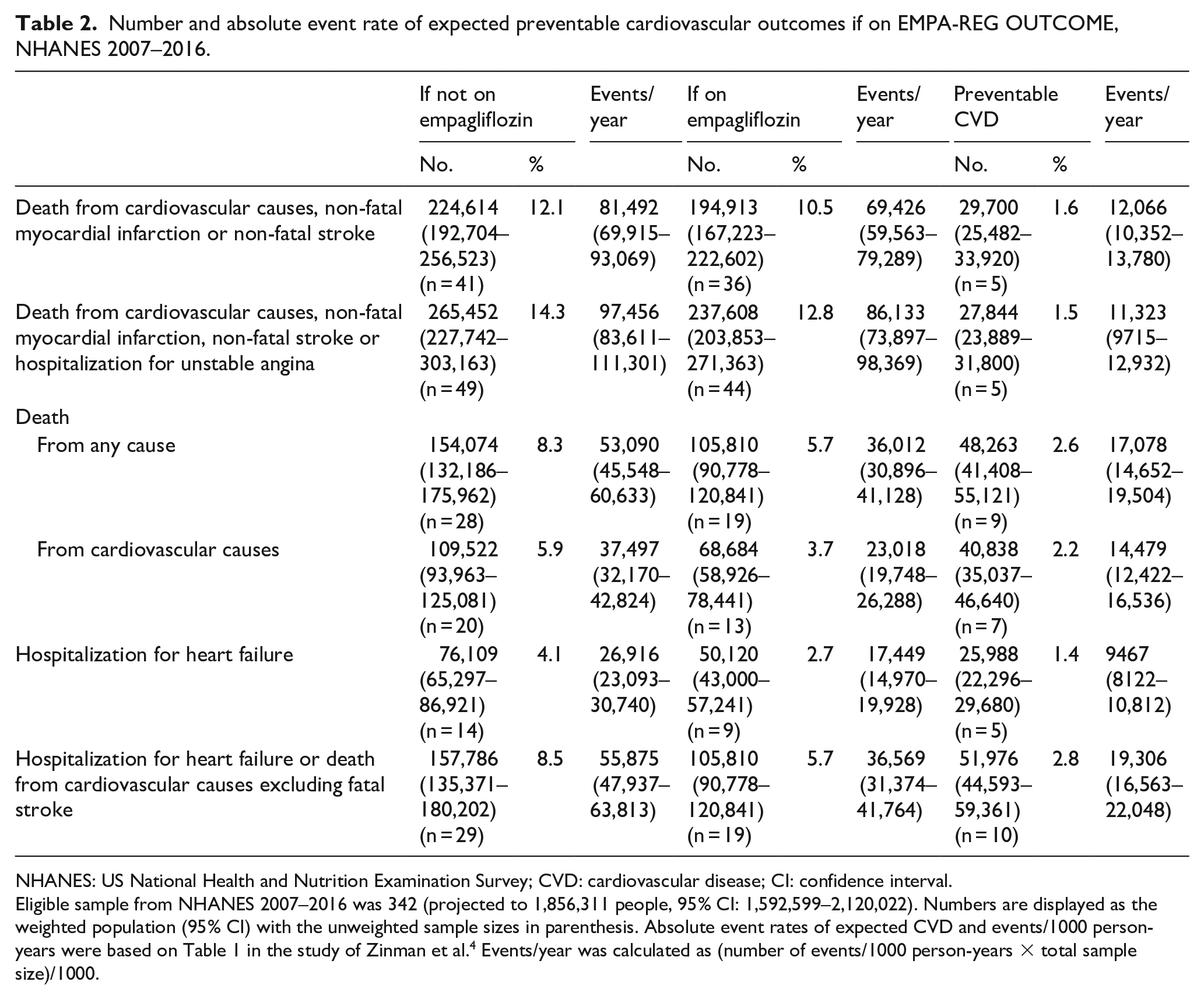
NHANES: US National Health and Nutrition Examination Survey; CVD: cardiovascular disease; CI: confidence interval.
Eligible sample from NHANES 2007–2016 was 342 (projected to 1,856,311 people, 95% CI: 1,592,599–2,120,022). Numbers are displayed as the weighted population (95% CI) with the unweighted sample sizes in parenthesis. Absolute event rates of expected CVD and events/1000 person-years were based on Table 1 in the study of Zinman et al. 4 Events/year was calculated as (number of events/1000 person-years × total sample size)/1000.
Discussion
This study is the first to demonstrate the US population-wide applicability of a major trial of SGLT-2 inhibitor therapy in persons with T2DM with respect to estimating the burden of CVD events that may be prevented. We show roughly a third of the 6 million US adults with both T2DM and CVD meet EMPA-REG OUTCOME eligibility criteria, of which untreated approximately 81,492 primary CVD events would occur annually, but that empagliflozin could annually prevent about 12,066 primary CVD outcome events, as well as 17,078 total deaths, 14,479 cardiovascular deaths and 9467 heart failure hospitalizations over that of placebo.
The SGLT-2 inhibitor class of diabetes medications5,6 are landmark in being the first such class of agents to demonstrate in persons with T2DM, not only cardiovascular event reduction, with particularly consistent reductions in heart failure hospitalizations, but also reductions in blood pressure, weight and other cardiovascular risk factors. Most recently, beneficial impacts on progression of CKD in patients with CKD have been demonstrated, 7 as well as reduced heart failure hospitalization and cardiovascular deaths in those with heart failure. 8 The transformative role of these therapies as cardioprotective agents establishes their key role in the field of preventive cardiology.
The EMPA-REG OUTCOME trial was a landmark in being the first of the SGLT-2 inhibitor trials to demonstrate cardiovascular benefit, showing a 14% (95% CI: 1%–26%) relative risk reduction in the primary composite endpoint of death from cardiovascular causes, non-fatal myocardial infarction or non-fatal stroke, and a more impressive 38% reduction in death from cardiovascular causes (95% CI: 23%–51%), 35% (95% CI: 15%–50%) reduction in hospitalization for heart failure and 32% (95% CI: 18%–43%) reduction in death from all causes. 1 Following this key trial was the CANVAS (CANagliflozin cardioVascular Assessment Study) programme studying canagliflozin and also involving patients with T2DM at high cardiovascular risk, which showed a 14% (95% CI: 3%–25%) relative risk reduction in the same primary composite endpoint and a 33% (95% CI: 13%–48%) reduction in heart failure hospitalization, but no significant reduction in cardiovascular or all-cause mortality. 9 More recently, the DECLARE trial involving dapagliflozin and a broader patient population for which over half were without CVD showed dapagliflozin did not result in a lower rate of major adverse cardiovascular events. There was a significant 17% (95% CI: 5%–27%) relative risk reduction in the co-primary endpoint of cardiovascular death or hospitalization for heart failure, but no significant reduction in cardiovascular or all-cause mortality. 10
While no real-world studies estimating the population-wide impact of these trials in terms of preventable CVD events, others have examined eligibility for these and trials of other glucose lowering agents. In the US Diabetes Collaborative Registry (DCR), Arnold and colleagues showed 26% of over 180,000 patients in the registry to meet EMPA-REG OUTCOME criteria, although only a minority (5.2%) were actually being prescribed an SGLT-2 inhibitor. 11 It was estimated that 354 cardiovascular deaths and 231 heart failure hospitalizations could have been prevented among those patients. The patient population included in the DCR at the time was highly enriched from cardiology practices, thus with an overrepresentation of patients with known CVD, and importantly the DCR does not have sample weighting allowing for extrapolation to the US population. Wittbrodt et al. 12 utilized earlier NHANES surveys from 2009 to 2012 to estimate eligibility in several T2DM outcomes trials, including EMPA-REG OUTCOME. EMPA-REG OUTCOME had the lowest proportion eligible subjects, with 4.1% of T2DM subjects eligible for the trial (this can be compared with our larger proportion eligible of 7% over a wider range of NHANES cohorts up to year 2016) and 39.8% showing eligibility for DECLARE. In addition, among over 1.2 million patients identified from the Royal College of General Practitioners Research and Surveillance Centre database in England, 15.7% of all T2DM patients had the same cardiovascular risk as those in the EMPA-REG OUTCOME trial, of which only 11% had been initiated on an SGLT-2 inhibitor. 13 The Comparative Effectiveness of Cardiovascular Outcomes in New Users of SGLT-2 Inhibitors (CVD-REAL) study was the first large real-world study of patients with T2DM involving more than 300,000 patients across six countries, 87% without a history of CVD. This study propensity matched those on SGLT-2 inhibitors and other glucose lowering drugs and found 39% lower risks of heart failure (HF) hospitalizations and a 51% lower risk of all-cause death in those on SGLT-2 inhibitors compared to other glucose lowering drugs. 14 Most recently, the Empagliflozin and the Risk of Heart Failure Hospitalization in Routine Clinical Care: A First Analysis from the Empagliflozin Comparative Effectiveness and Safety (EMPRISE) study 15 showed from a propensity-score matching analysis of two commercial and one federal claims databases of patients initiating empagliflozin or sitagliptin that the former was associated with a 50% reduction in heart failure hospitalizations, even greater than that observed in EMPA-REG OUTCOME. Future real-world studies of health claims data will provide further validation of the findings regarding the impact of SGLT-2 inhibitors and other therapies from these initial reports. While such studies provide information on the applicability of clinical trial findings to actual clinical practice, such analyses while mimicking clinical trials using propensity-score matching and other techniques cannot replace the results of clinical trials. Residual confounding and other issues, such as immortal time bias, 16 can exaggerate actual results.
Our study has important strengths and limitations. The NHANES surveys are the only US population-representative surveys containing sufficient laboratory and clinical information to provide estimates of eligibility from major clinical trials such as EMPA-REG OUTCOME. NHANES also contains standardized data collection as well as evaluation of medical history. Importantly, however, information on history of CVD is all based on self-reported questions on coronary heart disease, myocardial infarction, stroke and heart failure, without any greater detail. One limitation is that information on prior bypass surgery or percutaneous interventions, angiographic stenosis from coronary angiography, as well as ankle-brachial index was not captured in the NHANES surveys we utilized; such persons fitting EMPA-REG OUTCOME eligibility according to any of these criteria alone (without otherwise reporting as ‘yes’ on one of the self-reported questions for cardiovascular component above) would have not been captured in our analysis, resulting an underestimation of those actually eligible for EMPA-REG OUTCOME. Therefore, our estimates of EMPA-REG OUTCOME eligibility (and therefore projected preventive cardiovascular events) are likely to be conservative. Importantly, our estimates of primary and secondary events in placebo and empagliflozin treated US population depend on our sample being representative of participants in EMPA-REG OUTCOME trial. It has been previously noted by others 17 that large trials of glycaemic control often have limited external validity when applied to a large population-based cohorts such as our NHANES sample; therefore our results must be treated with caution, especially since the baseline risk of our sample is likely to be different than that of the EMPA-REG OUTCOME trial sample. In addition, while it would have been desirable to examine the effects of other SGLT-2 inhibitors studied in other outcomes trials like EMPA-REG OUTCOME, each trial had different inclusion/exclusion criteria, and thus different samples of patients for which the results of a given trial could be applied to. Our NHANES sample had a higher proportion of Black but similar proportion of White participants as EMPA-REG OUTCOME; however, neither had information on certain ethnic groups with high cardiometabolic risk (e.g. South Asians). Also, as the differences in terms of absolute number of events between treated and placebo groups in the sample studied were fairly small, the extrapolated preventable events can only be considered an approximation. We have described several other differences in demographic characteristics and CVD risk factors as well as concomitant treatment between our cohort and the EMPA-REG OUTCOME trial cohort, which of course would impact actual event risk. Our NHANES sample also tended to have more persons who were very recently diagnosed with T2DM and on lipid lowering medication.
In conclusion, our report gives some insight into the burden of CVD events, cardiovascular and total deaths, and heart failure hospitalizations that could potentially be prevented in the US population who would fit empagliflozin eligibility criteria. Recent guidelines have recommended the use of SGLT-2 inhibitor or GLP-1 agents, shown to have cardiovascular benefit, in a broader range of T2DM persons with CVD (e.g. without specific EMPA-REG OUTCOME restrictions such as range of HbA1c included)18,19 and even recently in those with T2DM and multiple risk factors alone. 20 It is therefore possible that many more CVD events, heart failure hospitalizations and deaths could be preventable than what we estimate using the limited EMPA-REG OUTCOME eligibility criteria, especially as these products become more widely used beyond those T2DM patients who already have established CVD. However, ultimately the costs of these therapies must be weighed against the cost savings from hospitalizations and other costs saved from events prevented when deciding which patient subgroups will be most cost-effective for their use.
Footnotes
Acknowledgements
This project was presented in part at the American College of Cardiology Scientific Sessions, March 2019, New Orleans, LA, USA.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Wong receives research support through the University of California, Irvine from Amarin, Amgen, Boehringer Ingelheim and Novo Nordisk. Dr Pak is an employee of Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was made possible by an independent grant to the University of California, Irvine from Boehringer Ingelheim Pharmaceuticals, Inc., who provided financial support for the study.
