Abstract
Objective:
The primary objective of the study was to estimate the prevalence of sub-clinical left ventricular dysfunction among asymptomatic diabetic patients, while the secondary objectives were to determine its association with microvascular complications and to find correlation with the baseline clinical and demographic parameters.
Material and methodology:
This was a cross-sectional study conducted on 226 type 2 diabetic patients who did not have any diagnosed cardiac disease, baseline ECG abnormality or cardiac symptoms. Two-dimensional strain echocardiography was performed to estimate the prevalence of left ventricular systolic dysfunction by measuring global longitudinal strain rate (cutoff < 18). Its association with microvascular complications was analysed with SPSS 23 software. Other baseline clinical parameters and demographic profile were also analysed.
Result:
Among 226 patients (151 males, 75 females), cardiac abnormality was found in 29.2% patients. Diabetic microvascular complications (e.g. neuropathy, retinopathy and nephropathy) were strongly associated with it (each with
Conclusion:
It was found that sub-clinical left ventricular dysfunction was found in significantly high proportion among patients with microvascular complications. Hence, routine screening of all diabetics for such complications and subsequently high-risk patients undergoing strain echocardiography can be a very cost-effective diagnostic, therapeutic and prognostic modality.
Keywords
Introduction
Diabetes mellitus has become the most prevalent non-communicable disease and its incidence is globally increasing. According to the World Health Organization (WHO) statement, the number of people with diabetes has increased from 108 million in 1980 to 422 million in 2014. The global prevalence of diabetes among adults over 18 years of age has risen from 4.7% in 1980 to 8.5% in 2014. 1 Data from Southeast Asia are even worse with 88 million adults (age group of 20–79 years) living with diabetes in 2019 and is estimated to increase to 153 million by 2045. 2 Left ventricular systolic dysfunction has been identified in diabetic patients even with preserved left ventricular ejection fraction (LVEF) without overt coronary artery disease or heart failure (HF). 3 In addition to glycaemic parameters and traditional risk factors, diabetes-related vascular complications have also been found to be associated with cardiac dysfunction.4–6 Microvascular complications (nephropathy, neuropathy, retinopathy) can be detected in any outpatient department without undergoing any time-consuming or expensive investigation. These are part of a routine evaluation for any diabetic patients. These microvascular complications can play an important role as a screening marker for cardiac evaluation. Because most of the cardiac dysfunctions in diabetics remain asymptomatic for a long time during initial period. 7 We do not have adequate data about complex relation between these two entities. Considering the huge diabetes burden in India and Southeast Asia, this information can be a policy changing factor. Hence, this study was conducted in an attempt to understand the correlation and determine the strength of association between microvascular complications and cardiac dysfunction
Methods
This was a cross-sectional study conducted in the department of diabetes and metabolism (Internal Medicine) together with department of cardiology in our institution over a period of 15 months. After obtaining institutional ethical committee clearance, a total of 400 type 2 diabetes mellitus (T2DM) patients were screened after taking written and informed consent. Based on their clinical symptoms, past medical records, baseline ECG and routine blood investigation, 226 patients were included in the study. All the patients with diagnosed cardiac conditions or symptoms suggestive of cardiac diseases, uncontrolled hypertension (BP > 140/90), abnormal ECG and comorbidities like chronic smoking, alcoholism, chronic kidney disease, cerebrovascular events, thyroid disorder, anaemia and obesity were excluded. All the included participants were evaluated for nephropathy (by 24-h urinary protein), neuropathy [by nerve conduction study (NCS)] and retinopathy (by ophthalmoscopy examination by experienced ophthalmologist). They underwent conventional two-dimensional (2D) echocardiography with commercially available PHILLIPS EPIQ 7C echocardiography machine and speckle trekking strain imaging (Figure 1) with the ‘QLAB software’ provided in-built by the manufacturer. Data were recorded on MS office Excel sheet and various clinical and demographic parameters were analysed with SPSS version 23. LV systolic dysfunction was defined by global longitudinal strain (GLS) rate <18. The value of
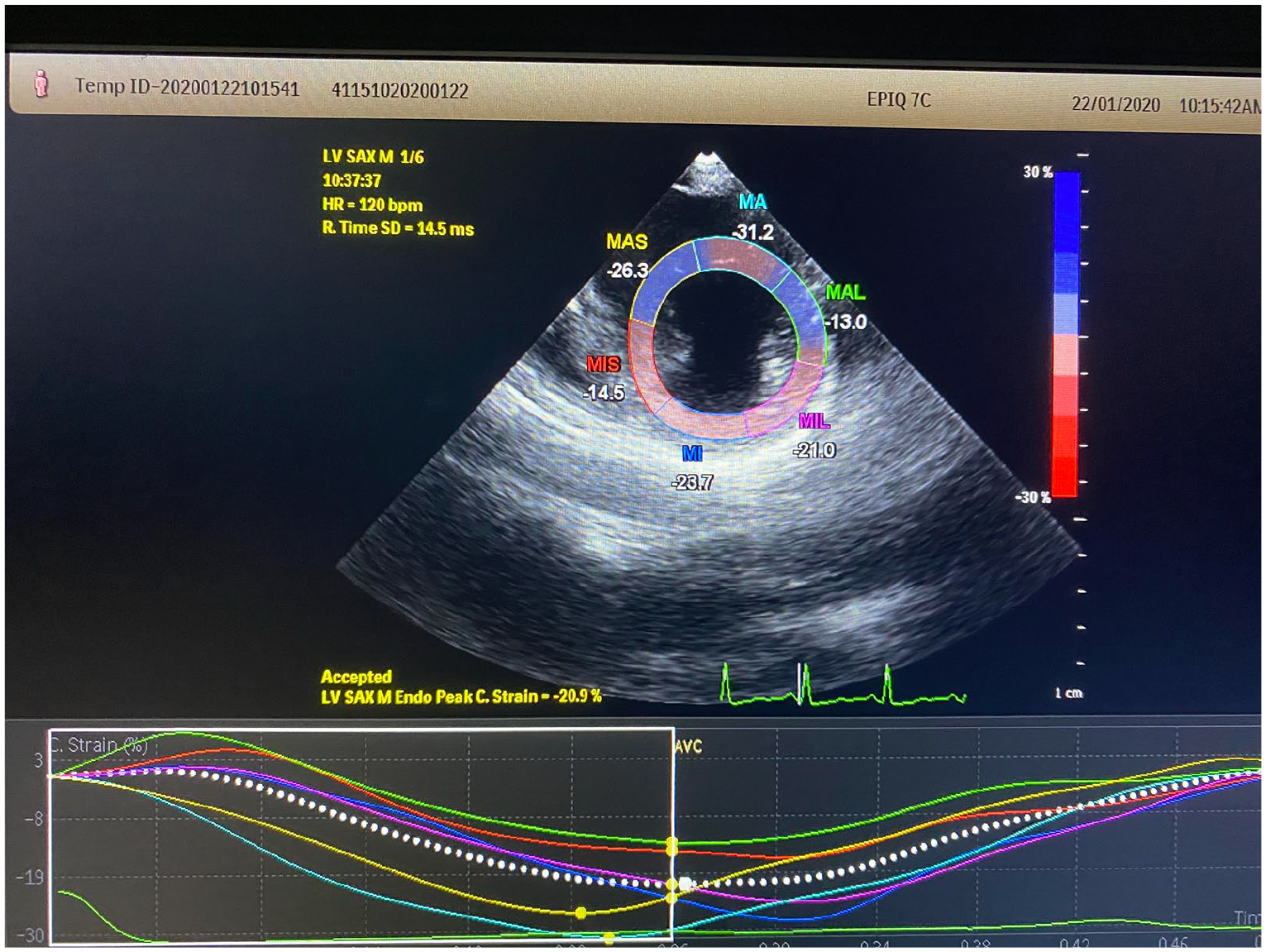
Acquisition of strain imaging in para-sternal short axis.
Results
A total of 226 patients with a mean age of 52.66 ± 8.43 years underwent transthoracic 2D echocardiography along with strain imaging. Baseline demographic parameters are shown in Table 1. Of 226 participants, 66 (29.2%) were found to have sub-clinical LV dysfunction as assessed by GLS <18%. Among the baseline characteristics, older age (
Baseline parameters of the study population.
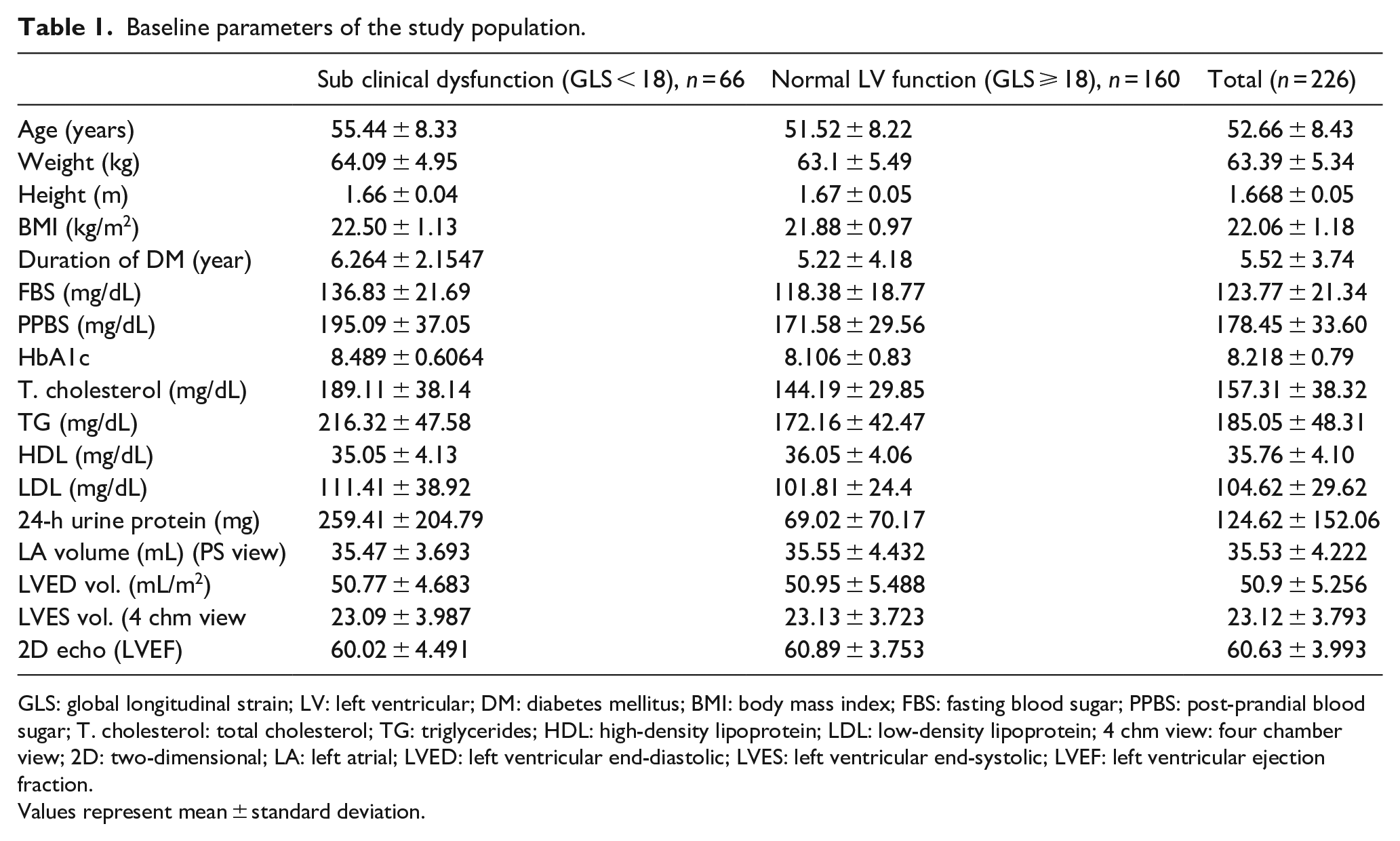
GLS: global longitudinal strain; LV: left ventricular; DM: diabetes mellitus; BMI: body mass index; FBS: fasting blood sugar; PPBS: post-prandial blood sugar; T. cholesterol: total cholesterol; TG: triglycerides; HDL: high-density lipoprotein; LDL: low-density lipoprotein; 4 chm view: four chamber view; 2D: two-dimensional; LA: left atrial; LVED: left ventricular end-diastolic; LVES: left ventricular end-systolic; LVEF: left ventricular ejection fraction.
Values represent mean ± standard deviation.
Association between the various parameters with global longitudinal strain (GLS) rate by univariate and multivariate analysis.
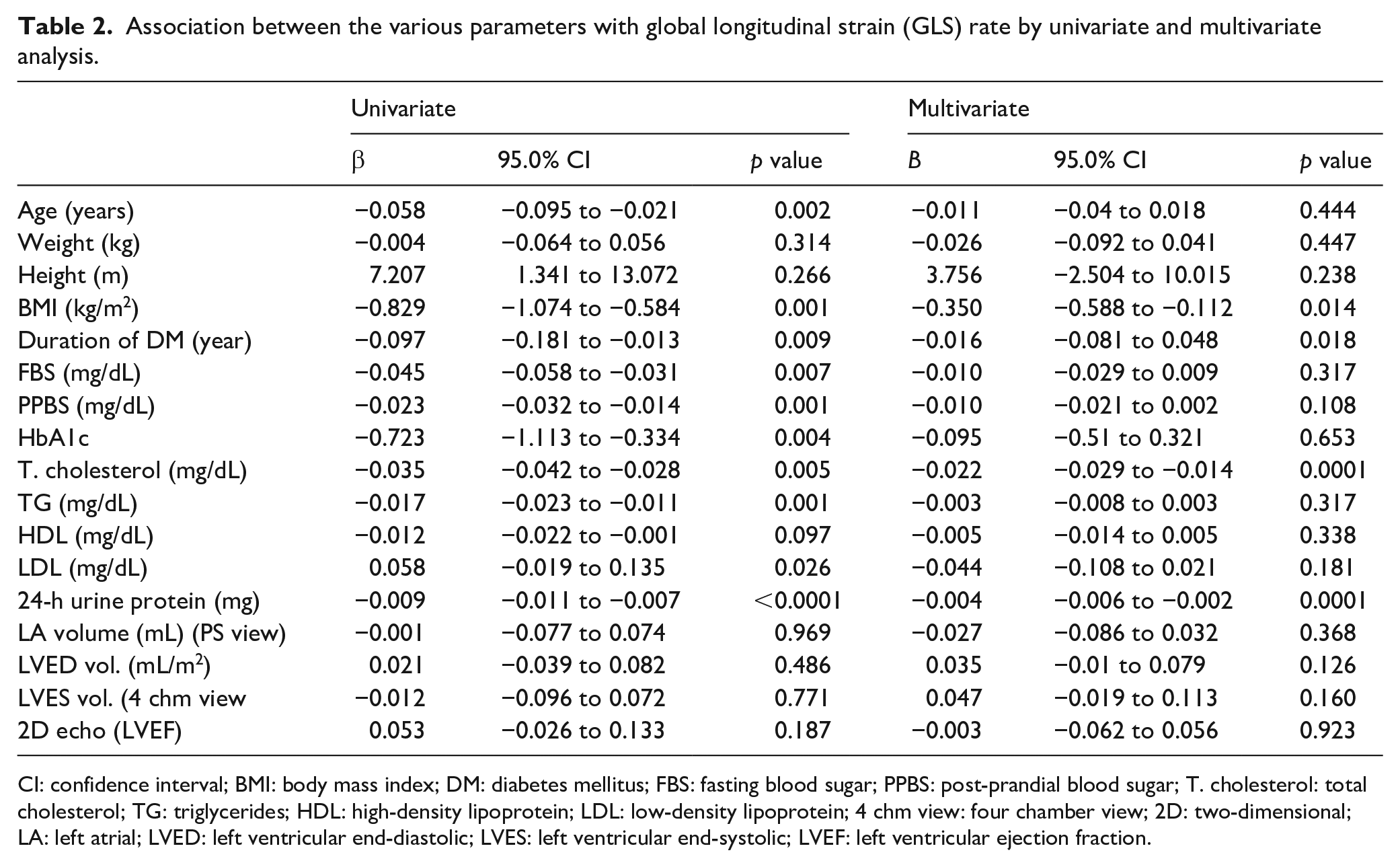
CI: confidence interval; BMI: body mass index; DM: diabetes mellitus; FBS: fasting blood sugar; PPBS: post-prandial blood sugar; T. cholesterol: total cholesterol; TG: triglycerides; HDL: high-density lipoprotein; LDL: low-density lipoprotein; 4 chm view: four chamber view; 2D: two-dimensional; LA: left atrial; LVED: left ventricular end-diastolic; LVES: left ventricular end-systolic; LVEF: left ventricular ejection fraction.
In terms of microvascular complications, out of 65 neuropathy patients, 40 (61.5%) were found to have sub-clinical LV dysfunction [95% CI = 1.910–6.350, odds ratio (OR) = 3.483,
Association between sub-clinical systolic dysfunction and diabetic neuropathy.
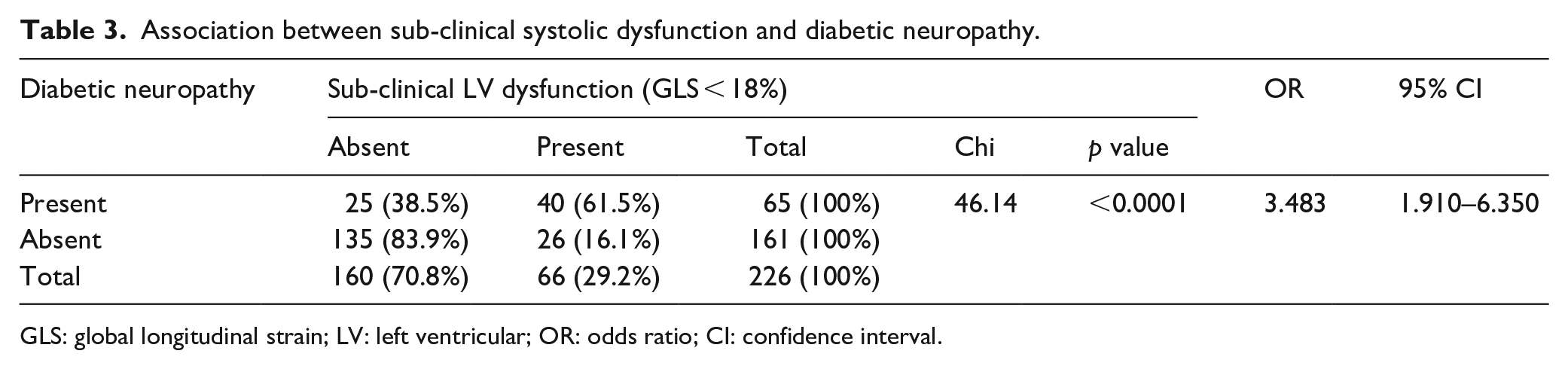
GLS: global longitudinal strain; LV: left ventricular; OR: odds ratio; CI: confidence interval.
Association between diabetic retinopathy and sub-clinical systolic dysfunction.
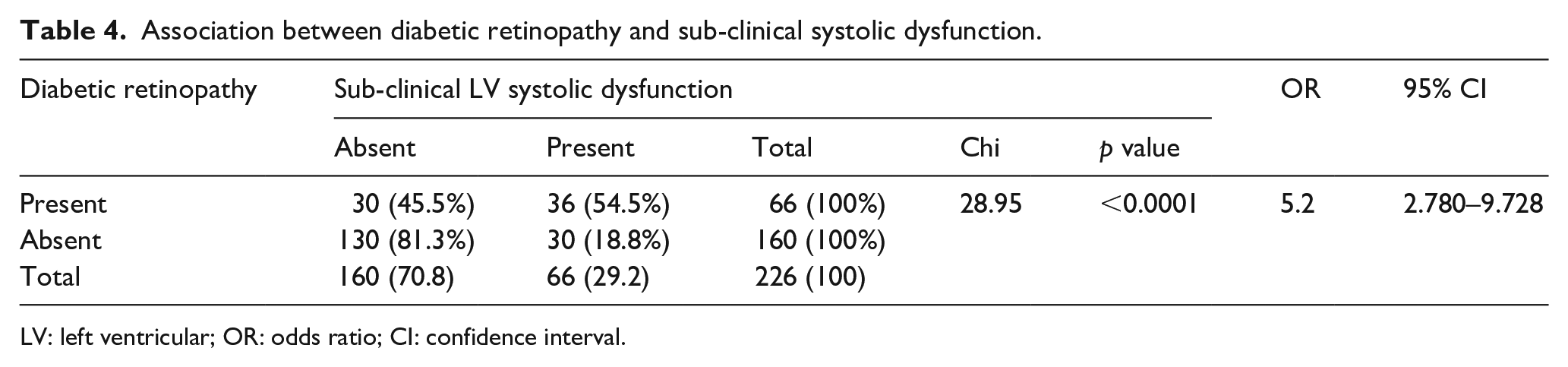
LV: left ventricular; OR: odds ratio; CI: confidence interval.
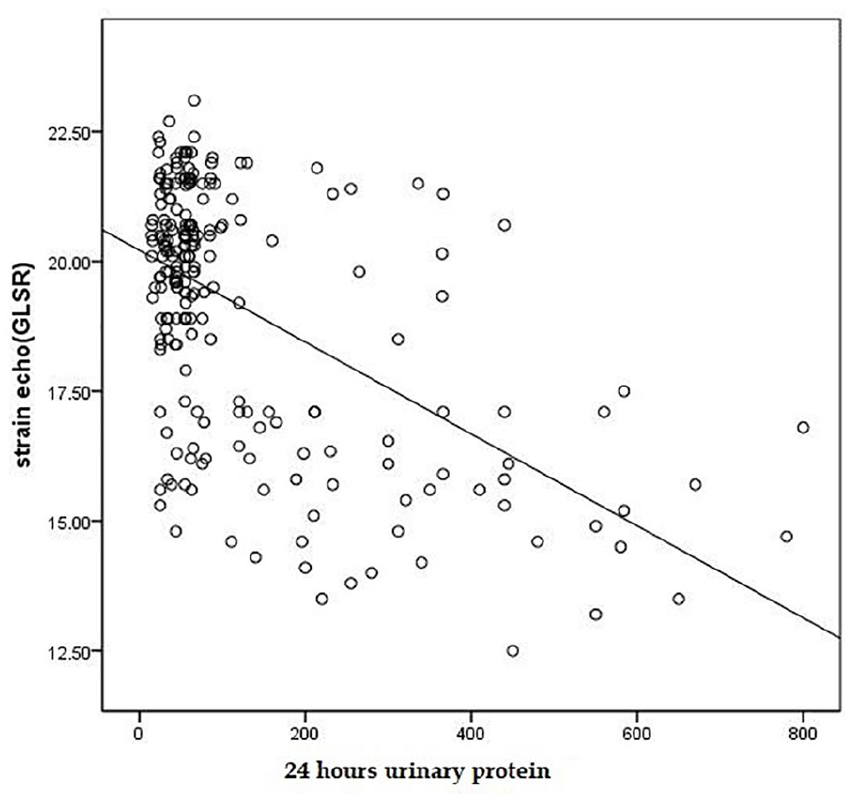
Scatter diagram showing relation between GLS and 24-h urinary protein.
Discussion
The major contributor of morbidity and mortality in both type 1 and type 2 diabetes is related to direct or indirect effects of hyperglycaemia on vascular system. 8 Diabetic patients generally have two- to fivefold increased risk of cardiac disease when compared to non-diabetics. It comprises both systolic and diastolic heart failure with worse outcome once symptomatic heart failure develops. The term diabetic cardiomyopathy (DCM) was coined almost 40 years ago and was initially used to describe ventricular dysfunction in the absence of coronary artery disease and hypertension in diabetic patients. 9 Insulin resistance and hyperglycaemia are the major etiological factors as they increase the levels of free fatty acids, growth factors and causes imbalance in demand and supply in cardiomyocyte. Furthermore, they also promote excessive production and release of reactive oxygen species, which induces oxidative stress leading to abnormal gene expression and cardiomyocyte apoptosis. 10
The overall prevalence of diabetes-related cardiac dysfunction is found to be 16.9%, whereas in the hospital setting it is found to be around 18%. 11 With the advancement of diagnostic modalities, many sub-clinical cardiac dysfunctions are getting identified among diabetic patients. 12 Our study revealed 29.2% of patients having sub-clinical dysfunctions as assessed by measuring GLS rate in otherwise asymptomatic patients with a normal 2D echocardiography. Considering the fact that most of the confounding factors that affect heart were excluded, it implies that hyperglycaemia with insulin resistant can be an independent factor in its pathogenesis. This is consistent with various previous studies. 13
Diabetic complications are divided as either acute or chronic with sub-classification of chronic into microvascular, macrovascular and non-vascular complications, and diabetic nephropathy, retinopathy and neuropathy are the classical microvascular complications. While macrovascular complications include coronary artery disease, peripheral vascular disease (PVD) and cerebrovascular disease, 14 cardiovascular complications fall under macrovascular complication as it is believed to be due to underlying compromised coronary perfusion. 15 Because of similarity of pathogenesis between cardiac and microvascular complications at the molecular level, researchers have been trying for a long time to establish a clinically useful relationship between the two entities.
The current study has further strengthened the belief of mutual existence of cardiac and microvascular complications. In addition, the study also leaves a question to ponder whether microvascular complications can be considered as sine quo non for cardiovascular complications, as we could see that 40 (61%) of those with diabetic neuropathy were also found to have sub-clinical cardiac dysfunction. Being asymptomatic, they would have been missed otherwise. Diabetic neuropathy has been found to have variable occurrence among different studies. One of the largest population-based studies done by Zeigler et al. 16 showed prevalence of neuropathy to be approximately 8.54% in type 1 and 13.46% in type 2 diabetic patients. On the other hand, a study done 4 years back by Popescu et al. 17 found that 66% in type 1 and 59% in type 2 were having diabetic neuropathy in western population. Probably the rapidly rising number of diabetic patients and inclusion of better diagnostic tools for neuropathy might explain differences in these numbers. Nerve conduction studies (NCSs) are recommended as the gold standard for objectively diagnosing diabetic peripheral neuropathy (DPN). Abnormal NCS together with signs and symptoms are the best diagnostic approach for DPN as advised by the Toronto Consensus committee. The characteristics of electrophysiological findings are reduced amplitude, slowing of sensory and motor nerve signal conduction velocity and prolonged F-wave latency along with absent Hoffman reflex. 18 Our results support previous studies.
A meta-analysis of 12 studies showed that cardiac autonomic neuropathy (CAN) is associated with silent myocardial infarction (MI) detected by the exercise test with prevalence ratios of 0.85 to 15.53, 6 while in another multicentre randomized study, patients with established cardiac diseases were found to have increased risk of developing neuropathy over 10 years of follow-up. 19 So, from the review of literature, the relationship between cardiovascular disease (CVD)-related mortality and CAN has been almost well demonstrated. 20 But the evidence on the relationship between DPN and CVD seems to be erratic and scanty. Probably our study can serve as an evidence to further emphasize the relation between the two. Considering the disease burden of diabetes, it is important to screen for neuropathy by simple clinical examination and questionnaire so that their CVD-related mortality and morbidity can be prevented or delayed. A more holistic approach by diabetologist, cardiologist and neurologist is needed in this scenario.
A similar correlation (
The current study also clearly demonstrates a positive correlation between diabetic retinopathy and LV systolic dysfunction. We can see in our result that 36 (54.5%) patients had sub-clinical LV systolic dysfunction out of 66 patients with diabetic retinopathy (all had non-proliferating retinopathy). Probably inclusion of only asymptomatic patients yielded this milder version of retinopathy. However as per the evidence, severity of retinopathy is associated directly with the severity of CVD. Shoeibi et al. in their study showed that after adjustment for traditional risk factors for CVD, the risk of CVD remained markedly increased in the presence of diabetic retinopathy. Risk of CVD was significantly higher in PDR [proliferative diabetic retinopathy vs non-proliferative diabetic retinopathy (NDR)] (18.7 ± 10.0% vs 11.3 ± 8.4%,
Along with these three microvascular complications, other traditional risk factors like older age, history of hypertension, dyslipidaemia and poor glycaemic parameters (e.g. FBS, PPBS, HbA1c) were associated with LV dysfunction. However, hyperglycaemia remains as an important contributor.
Conclusion
The strong association between microvascular complications and CVD risk is proven in our study. Our study is unique as it included all the three parameters of microvascular complication in a single study group of population and is probably the first of its kind. Hence, a risk stratification for early CVD evaluation based on these readily detectable microvascular parameters should be given a priority in any diabetologists’ or general physician’s outpatient clinic.
Key messages
India together with Southeast Asia carries the highest burden of diabetes mellitus and CVD being the most common cause of mortality, it is always important to detect this condition early. Hence, being able to identify those high-risk patients in their asymptomatic stage based on the presence of these microvascular complications is the key learning point from our study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
