Abstract
Obstructive sleep apnoea (OSA) is very common in patients with Type 2 diabetes (T2D), which is not surprising considering that obesity is a common risk factor for both conditions. In general population studies, OSA has been shown to be associated with several comorbidities including increased risk of road traffic accidents, T2D, hypertension and lack of nocturnal dipping of blood pressure, hyperlipidaemia, increased inflammation, increased risk of cardiovascular disease and mortality, increased risk of atrial fibrillation, worse quality of life, and erectile dysfunction. However, the impact of OSA on diabetes-related vascular and metabolic outcomes remains unclear. Furthermore, the impact of continuous positive airway pressure (CPAP) treatment in patients with T2D is also unclear. This unclarity regarding the impact of OSA and CPAP in patients with T2D has possibly contributed to the lack of screening for OSA in patients with T2D in the UK despite the high prevalence of OSA in patients with T2D. In this commentary, I provide an overview about OSA with a particular focus on its role and impact in patients with T2D.
Keywords
Introduction
Obstructive sleep apnoea (OSA) is a common medical disorder. While earlier estimates suggested a 4% prevalence in men and 2% prevalence in women, more recent studies indicate a prevalence of 17%–26% of men and 9%–28% of women. 1 This increase in OSA prevalence is likely to reflect the global increase in obesity, which is a major risk factor of OSA,2,3 and/or may reflect higher awareness of this condition leading to increased diagnosis.
As obesity is a common risk factor for OSA and type 2 diabetes (T2D), it is not surprising that these conditions commonly co-exist.2,4 The prevalence of OSA is high in people with T2D (8.5%–86%, with 23.8%–70% for moderate-to-severe OSA);3–10 the variation in OSA prevalence between studies is due to multiple factors including differences in studies populations (e.g. primary care vs secondary care), the methods of diagnosing OSA (polysomnography vs portable devices) and the criteria used to diagnose OSA [apnoea hypopnoea index (AHI) of 5 vs 10 vs 15 or using the oxygen desaturations index rather than AHI]. Similarly, the prevalence of T2D is high patients with OSA (15%–30%). 11 As a result, the International Diabetes Federation (IDF) recommended in 2008 routine screening for OSA in patients with T2D, 12 but despite the IDF statement, a recent survey in the United Kingdom showed that two-thirds of diabetes healthcare professionals were unaware of these recommendations and only 19% had the local diabetes guidelines incorporated assessment for OSA in those at risk. 13
This lack of OSA screening in patients with T2D might be related to the reduced awareness of the associations between OSA and T2D and might also reflect the lack of clear screening strategies and clinical pathways that inform practicing clinicians who to screen, how to screen, what to do after screening and how often to screen. Screening for OSA is further complicated by the fact that many of the symptoms that can be attributed to OSA can also be attributed to diabetes (see below), which makes identifying patients purely on clinical grounds more difficult. Furthermore, there is lack of data regarding the clinical, metabolic and economic impacts of OSA in patients with T2D, the cost-effectiveness of OSA screening and treatment in diabetes and the impact of OSA treatment on diabetes-related outcomes; these issues can make it difficult to justify routine screening for OSA. 14
OSA overview
OSA is characterised by upper airway instability during sleep which results in recurrent episodes of complete or partial upper airway obstruction that leads to recurrent episodes of either complete (apnoea) or partial (hypopnoea) cessation of airflow. 15 These apnoea and hypopnoea events usually result in altered intrathoracic pressure and increased breathing efforts as the patient attempts to breath against a blocked upper airway; this leads to a brief awakening (microarousal) and restoration of upper airway patency with termination of the apnoea/hypopnoea event.2,4 Following restoration of upper airway patency, the same cycle of events will repeat itself resulting in recurrent oxygen desaturations. These cyclical changes in intrathoracic pressure and recurrent microarousals result in sleep fragmentation and disruption of sleep architecture [loss of deep sleep and rapid eye movement (REM) sleep], which in part contribute to the excessive daytime sleepiness (EDS) observed in patients with OSA. 15 In addition to the above-mentioned changes, the apnoea/hypopnoea events are frequently associated with cyclical changes in blood pressure (BP), heart rate and sympathetic activity. 15
Polysomnography (that typically records 12–16 channels) remains the gold standard for diagnosing OSA. 15 However, as polysomnography is time-consuming, resourceful, expensive and requires a hospital stay overnight, other alternatives have been developed. Portable home-based cardio-respiratory Type III devices (typically recording 4–7 channels) are widely used in clinical practice as well as research, and their use is supported by the latest statement from the American Academy of Sleep Medicine (AASM). 16 Type III portable devices have a sensitivity and specificity of 87%–96% and 60%–76%, respectively, to diagnose OSA defined AHI ⩾5 events/hour and 49%–92% and 79%–95%, respectively, for OSA diagnosed as AHI ⩾15 events/hour. 17
Clinically, OSA is associated with a variety of non-specific symptoms. Snoring is the most common symptom, occurring in 95% of patients with only 6% of OSA patients (or their partners) not reporting any snoring. However, it has a poor positive predictive value due to the high prevalence of snorers who don’t have OSA. 15 Witnessed apnoeas can be reported by the partner but they do not correlate with disease severity and can occur in up to 6% of the ‘normal’ population without OSA. 15 EDS is another important feature of OSA, but many OSA patients do not suffer from EDS, at least if measured using the Epworth Sleepiness Scale (ESS). Other OSA symptoms include choking, insomnia, nocturia, sweating, fatigue, morning headaches, erectile dysfunction and autonomic symptoms.15,18 However, many of the above-mentioned OSA-related symptoms are either common in patients without OSA or can be attributed to a diabetes-related cause such as hyperglycaemia, hypoglycaemia, diabetic neuropathy, cardiovascular disease (CVD) or drug-related adverse events, which makes it difficult to suspect OSA on clinical grounds only in patients with T2D.
OSA is associated with several comorbidities including increased risk of road traffic accidents, incipient hypertension and lack of nocturnal dipping of BP, hyperlipidaemia, increased inflammation, increased risk of CVD and mortality, increased risk of atrial fibrillation, worse quality of life and erectile dysfunction.1,10,17,19–28
Continuous positive airway pressure (CPAP) is the mainstay treatment for moderate to severe OSA. CPAP usage for ⩾4 h/night resulted in reduction of the risk of road traffic accidents. 29 In a recent meta-analysis of randomised controlled trials (RCTs), CPAP (compared to sham) was associated with reduction of AHI [weighted mean difference (WMD), −33.8, 95% confidence interval (CI): −42.0 to −25.6]; ESS (−2.0, 95% CI: −2.6 to −1.4); diurnal systolic BP (−2.4, 95% CI: −3.9 to −0.9) and diurnal diastolic BP (−1.3, 95% CI: −2.2 to −0.4) and improved quality of life. 17 One RCT showed that 4 weeks of CPAP improved cognitive function following a stroke compared rehabilitation treatment. 30 Lifestyle and dietary interventions and the use of mandibular advancement devices were also associated with significant improvements in AHI and ESS, but the effect sizes were smaller than CPAP.17,31 Bariatric surgery has also been shown to result in a significant reduction in AHI (weighted decrease 29/h; 95% CI: 22.41 to 36.74). 32 Despite the favourable impact of CPAP on BP and erectile dysfunction when compared to placebo, it was less efficacious in treating erectile dysfunction and hypertension than sildenafil33–36 and valsartan, 37 respectively.
It must be emphasised that while CPAP is the ‘gold-standard’ treatment for patients with OSA in terms of the impact on AHI and EDS, weight loss also has a major role, particularly as it reduces CVD while the evidence for CPAP in altering vascular events is based on observational studies rather than RCTs. In addition, the combination of CPAP and weight loss has been shown to have greater impact on some cardiovascular risk factors compared to CPAP alone.38,39
In the United Kingdom, National Institute for Health and Care Excellence (NICE) recommended the use of CPAP in patients with moderate to severe OSA with EDS (https://www.nice.org.uk/guidance/ta139/chapter/1-Guidance); but despite NICE guidance, the availability and delivery of OSA services vary considerably across the United Kingdom (https://www.blf.org.uk/sites/default/files/OSA_Toolkit_2015_BLF_0.pdf).
OSA and T2D: a chicken and egg?
As highlighted above, OSA and T2D commonly co-exist, but is there a causative relationship between the two conditions and, if so, which one comes first? Most of the literature is focussed on examining the impact of OSA on incident T2D. Indeed, several longitudinal studies and meta-analyses have shown that OSA is a risk factor for the development of T2D independent of obesity and other traditional risk factors [relative risk (RR), 1.4–1.63 depending on the study].2,10,40,41 It is plausible that OSA can lead to the development of T2D as OSA is associated with insulin resistance and β-cell dysfunction; this relationship is mediated by several neurohormonal and inflammatory mechanisms and increased oxidative stress.2,10,42 However, whether CPAP treatment might reduce the incidence of T2D is awaiting the results of ongoing RCTs, but early evidence suggests potential benefit. In one recent RCT, 8 h of CPAP reduced the area under the curve for the 2-h overall glucose response during oral glucose tolerance test (OGTT) compared to placebo and improved insulin sensitivity, 24-h BP and norepinephrine levels. 43 Nonetheless, this study was small (n = 39), of short duration (2 weeks) and was conducted in laboratory environment to ensure high compliance with CPAP under direct supervision which might prove difficult to achieve in real life. In another RCT in patients with severe OSA but without diabetes, CPAP for 12 weeks reversed impaired glucose tolerance in 9/42 patients versus 5/38 patients in the life style counselling group (p = 0.039) without any changes in weight or Homeostatic model assessment- insulin resistance (HOMA-IR). 44 In another observational study, the incidence of T2D over a median of 5 months was lower in patients who had outstanding (⩾90% of nights and 8 h/night), excellent (⩾80% nights and ⩾6 h/night) and good (⩾70% nights and ⩾4 h/night) compliance with CPAP compared to those who were non-adherent to CPAP (0%, 0%, 0.8% and 4%, respectively). 45 The impact of OSA on dysglycaemia, insulin resistance and beta-cell dysfunction is mediated by multiple mechanisms including increased oxidative stress, sympathetic overactivity, activation of the hypothalamic pituitary adrenal access, increased inflammation and adipokine changes; most of which is improved by CPAP. 42
On the other hand, it is also plausible that T2D might also be a risk factor for OSA or worsening of pre-existing OSA, but this has not been explored widely in the literature. One longitudinal study showed that baseline HOMA-IR was an independent predictor of incident witnessed apnoeas [odds ratio (OR): 1.31, 95% CI: 1.13–1.51] after adjustment for age, sex and waist circumference over a period of 6 years. 46 Other independent predictors of witnessed sleep apnoeas included waist circumference, triglycerides levels and smoking. 46 All these factors are common in patients with T2D and hence might increase the risk of developing apnoeas and OSA in patients with T2D. In addition, the weight gain that occurs in patients with T2D with treatment intensification might result in the development of, or worsening of pre-existing, OSA,47–49 while weight loss in patients with T2D improved AHI in a longitudinal study. 50 Furthermore, the development of autonomic neuropathy in patients with T2D might have an impact on upper airway innervation and collapsibility, ventilatory drive and central respiratory centre responses to hypercapnic stimulus, all of which contribute to the pathogenesis of OSA.51,52
It is likely that the relationship between OSA and T2D is bidirectional and that each of these conditions can contribute to the development or worsening of the other. However, understanding this complex relationship is important to develop the appropriate screening and treatment strategies. While both conditions (OSA and T2D) are associated with similar comorbidities [such as hypertension, CVD, hyperlipidaemia and chronic kidney disease (CKD)], whether the impact of having both conditions (i.e. OSA and T2D) on CVD and other comorbidities is greater than having either OSA or T2D alone is the focus of several ongoing studies. In addition, the impact of OSA on the above-mentioned comorbidities in patients with T2D might differ whether OSA was present before or developed after the onset of T2D. In OSA patients who subsequently develop T2D, it is possible that the combination of OSA and T2D results in greater impact on diabetes-related outcomes and intervention targeting OSA might produce larger benefits as the diabetes-related comorbidities are still at earlier stages of development with the potential of reversibility. On the other hand, if OSA developed after the onset of diabetes, then targeting OSA might produce less effects as many complications have already been established and therefore less likely to reverse. However, these hypotheses remain to be examined and explored in ongoing studies and trials.
OSA and glycaemic control in T2D
OSA has been linked to worse glycaemic measures (such as HbA1c, fasting plasma glucose or glucose variability) following adjustment for a wide range of confounders in several cross-sectional studies.2,4,53 However, though biologically plausible, causality between OSA and worse glycaemic control has not been proven as yet due to the lack of longitudinal studies and the conflicting results of clinical trials. The impact of OSA on HbA1c ranged between 0.7% and 3.69%; this variation between studies is likely to be due to methodological differences and variability in OSA severity between studies.2,4,53
Overall, uncontrolled studies of short duration (⩾4 months) showed that CPAP improved glycaemic variability, postprandial glucose levels and HbA1c in patients with T2D, but RCTs did not overall show such benefits.2,4,53 In a small RCT of 42 men with T2D and OSA who were randomised to CPAP versus sham CPAP, 3 months of treatment did not show any impact of CPAP on HbA1c but CPAP compliance was poor, preventing definitive conclusions. 54 In a more recent study of patients with T2D and moderate to severe OSA in which 298 patients were randomised to CPAP versus usual care, CPAP again failed to show an impact on HbA1c but glucose control was fairly good at baseline (HbA1c 7.3%), which may have contributed to the negative findings. 55 In contrast, another RCT in which 50 patients with T2D and OSA were randomised to CPAP versus no CPAP for 6 months, CPAP resulted in greater HbA1c reductions compared to no CPAP (between group adjusted difference, −0.4%; 95% CI: −0.7 to −0.04, p = 0.029). Of note, the compliance in this study was good with average CPAP usage of 5.2 h per night. 56 The conflicting results of these studies might be related to multiple factors including differences in CPAP compliance, baseline glycaemic control and the reserve in β-cell function. Other factors related to OSA might also play an important part; for example, some studies showed that the impact of CPAP might be greater in patients with OSA and EDS versus OSA but not EDS, 57 whereas others showed that the relation between OSA and glycaemic measures mainly relate to apnoea/hypopnoea events that occur in REM sleep compared to non-REM sleep. 58 Further large RCTs that will include patients in the early stages of T2D and that consider factors such as EDS and REM-related AHI and in which CPAP compliance is high are still needed to answer the question whether CPAP treatment improves glycaemic control in patients with T2D.
OSA and hypertension
Epidemiological studies and RCTs have shown a causal link between OSA and incident hypertension and non-dipping BP and that CPAP lowered BP compared to no CPAP or sham CPAP but the impact of CPAP was less than that of valsartan in one RCT (Table 1).21,37,59 However, data in patients with T2D are limited. In an observational retrospective cohort study in patients with OSA and T2D, CPAP was associated with improvements in systolic (−6.81, 95% CI: −9.94 to −3.67 mmHg) and diastolic (−3.69, 95% CI: −5.53 to −1.85 mmHg) BP over 9–12 months period. 60 Similar reductions in BP levels were found after 3 months of CPAP in a RCT in which patients were randomised to early (<1 week) or late (1–2 months) 61 intervention.
Summary of the evidence regarding the relationships between OSA and diabetes-related macro- and microvascular complications and the impact of CPAP.
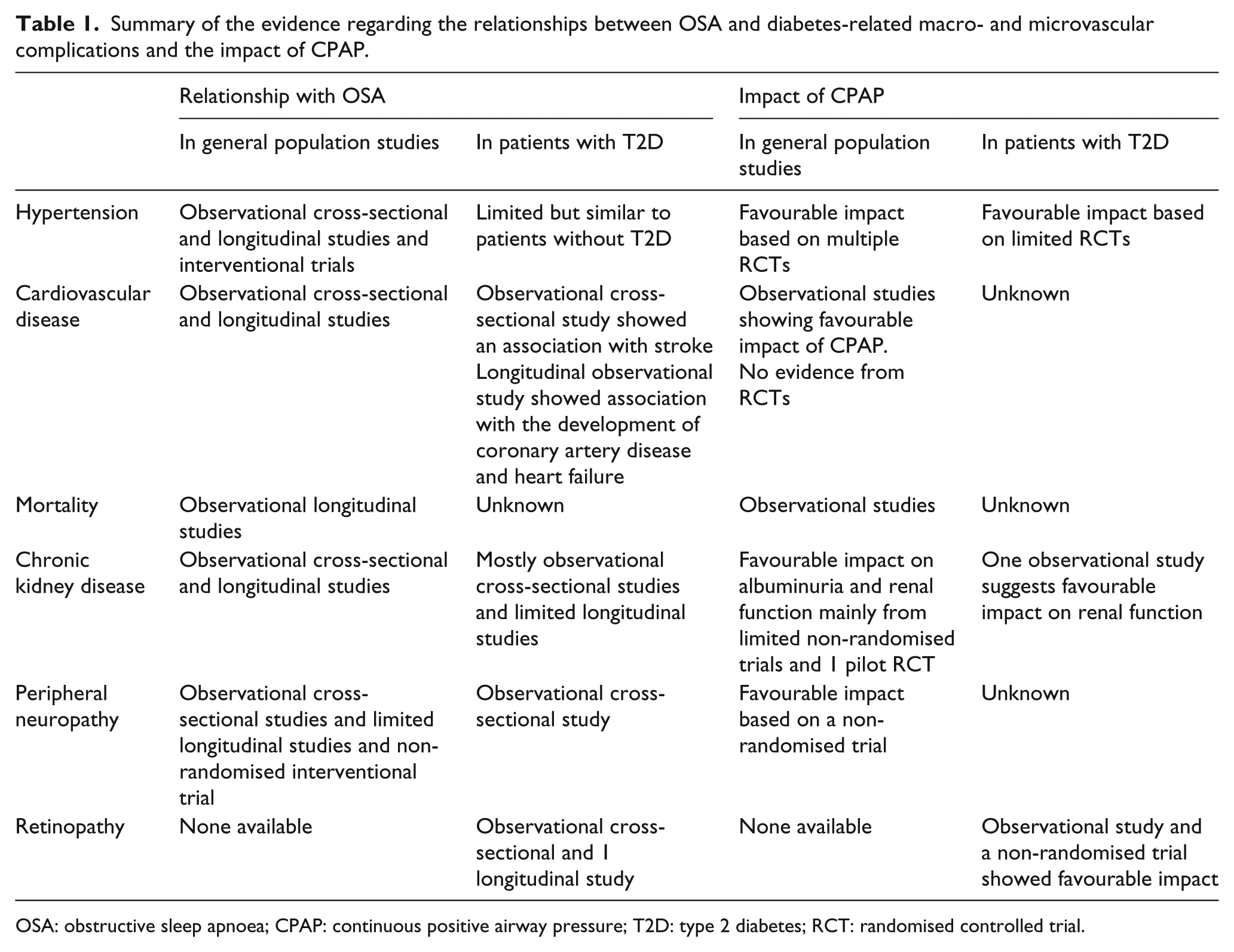
OSA: obstructive sleep apnoea; CPAP: continuous positive airway pressure; T2D: type 2 diabetes; RCT: randomised controlled trial.
OSA and CVD
In general population studies, several cross-sectional and longitudinal studies showed a link between OSA and CVD (Table 1). Based on intravascular ultrasound and CT coronary angiogram, OSA and AHI were found to be associated with a larger atherosclerotic plaque volume.62,63 In addition, a study found a diurnal variation in the occurrence of acute myocardial infarction in that patients with OSA were at increased risk of acute myocardial infarction overnight (midnight to 6 am) compared to those without OSA, suggesting a possible role for the nocturnal OSA-associated events in the development of CVD. 64 Longitudinal observational studies have shown increased risk of incident CVD and mortality in patients with OSA compared to those without OSA over 3–10 years [adjusted OR/hazard ratio (HR) for incident CVD of 1.97–4.9] and that CPAP treatment was associated with lower vascular events and mortality.22–24,65–69 While this looks encouraging, there is no evidence from RCTs that CPAP lowered mortality or CVD.17,70,71 However, most of these RCTs were ⩾12 weeks duration and only one showed a follow-up of more than 1 year; therefore, the number of vascular events and mortality was small.
Data regarding the impact of OSA on CVD in diabetes are rather limited. In the SleepAhead study, AHI was associated with history of stroke (adjusted OR, 2.57; 95% CI: 1.03–6.42), but not coronary artery disease. 72 OSA was found to predict incident coronary artery disease (adjusted HR, 2.2; 95% CI: 1.2–3.9; p = 0.01) and heart failure (adjusted HR, 3.5; 95% CI: 1.4–9.0; p < 0.01) in 132 patients with T2D and a normal baseline exercise echocardiography test over a median follow-up of 4.9 years. 73 It remains unclear, however, whether treating OSA in these patients alters vascular outcome or heart failure in this high-risk population.
OSA and microvascular complications in T2D
OSA shares many of its molecular consequences with hyperglycaemia which can lead to microvascular complications in patients with T2D (Table 1). OSA can result in increased oxidative stress and nitrosative stress, poly (ADP-ribose) polymerase (PARP) activation, increased advanced glycation end-products (AGE), protein kinase C (PKC) activation, inflammation and endothelial dysfunction in patients with and without T2D,2,53,74,75 all of which are involved in the pathogenesis of microvascular complications in T2D. 76
OSA and retinopathy
During darkness, retinal oxygen demand increases and in the context of diabetes the retina cannot meet the added metabolic load imposed by the dark-adapted rod photoreceptors. This exacerbates retinal hypoxia and results in vascular endothelial growth factor (VEGF) secretion, with the consequent development of diabetic retinopathy (DR). 77 Hence, it is plausible that the presence of OSA, with recurrent nocturnal oxygen desaturations, might increase the burden of nocturnal hypoxia on the retina and might result in the development or progression of DR.
OSA is not known to be associated with retinal changes similar to DR in patients without diabetes. However, OSA has been associated with several retinal and ocular manifestations. In one study, an increase of respiratory disturbance index (RDI) from 0 to 10 was associated with a decrease in arteriole-to-venule ratio of 0.01. 78 Other retinal changes described in OSA included retinal arteriolar narrowing and venular widening or branch retinal vein occlusion.79,80
In patients with T2D, the relationship between OSA and DR is complex and the results of multiple studies were inconsistent; this is, in part, due to the poor methodology of some of the available literature. A recent systematic review found that out of 16 studies that examined the relationship between OSA and DR, 15 were cross-sectional in nature and 1 cohort study, 81 which highlights the need for more robust studies in this area. The above-mentioned systematic review showed that the cross-sectional associations between OSA and retinal microvascular changes were inconsistent, but there seems to be an association between OSA and more advanced DR such as pre-proliferative DR, proliferative DR and maculopathy. 81
Our own data (currently undergoing peer review) showed that OSA was associated with sight-threatening DR and maculopathy, but not with background DR in a cross-sectional analysis. However, longitudinally, we found that OSA was an independent predictor of the development of pre-/proliferative DR in patients with T2D after adjustment for confounders.
So overall, the development of DR in its earlier stages seems to be dependent on the presence of diabetes, and OSA appears to enhance progression of DR into sight-threatening retinopathy through the development of pre-proliferative and proliferative changes.
Whether CPAP has an impact on DR treatment and/or progression remains to be seen with several ongoing RCTs in this area. Our unpublished data showed that the progression to pre-proliferative or proliferative DR was less in OSA patients who were CPAP compliant compared with those who were not compliant. In an uncontrolled, hypothesis generating study, CPAP for 6 months was associated with improvement in visual acuity without an impact on macular oedema/thickness, suggesting improved functionality rather than actual change in the oedema. 82 Interestingly, the presence of OSA might affect the response to DR treatment as was shown by one study in which poor response to anti-VEGF therapy with bevacizumab (Avastin) was associated with higher prevalence of OSA in patients with diabetic macular oedema and age-related macular degeneration; 83 whether CPAP can improve the response to DR treatment requires future studies.
OSA and diabetes CKD
In patients without diabetes, OSA has been associated with CKD in cross-sectional and longitudinal studies.2,53,84,85 Small studies of short duration suggested potential beneficial effects of CPAP on albuminuria and renal function.86–89 In patients with T2D, a cross-sectional study of Japanese patients, Oxygen desaturation Index (ODI) ⩾5 was independently associated with microalbuminuria in women but not in men after adjustment for confounders. 90 In another cross-sectional study from the United Kingdom, OSA was associated with CKD in patients with T2D (adjusted OR, 2.64; 95% CI: 1.13–6.16; p = 0.02). 91 Longitudinally, OSA was associated with greater decline in estimated glomerular filtration rate (eGFR) compared to patients without OSA. Moreover, OSA was an independent predictor of study-end eGFR and eGFR decline after adjustment for potential confunders. 91 The impact of CPAP on CKD in patients with diabetes is unclear, but in a longitudinal study, eGFR decline was lowered in patients with OSA who were compliant with CPAP; 91 ongoing RCTs will help to answer this point.
OSA and peripheral neuropathy
In patients without diabetes, OSA and nocturnal hypoxia were associated with peripheral neuropathy (based on nerve conduction studies) independent of age and obesity.92,93 Moreover, one small study demonstrated that OSA was associated with axonal dysfunction and that 6 months of CPAP improved the amplitude of action potentials. 94 Data in patients with T2D are limited with one cross-sectional study showing that OSA was associated with foot insensitivity and clinically evident diabetic peripheral neuropathy, independent of potential confounders. 95 In addition, OSA was independently associated with lower intra-epidermal nerve fibre density (based on skin biopsies) in patients with T2D compared to those without OSA. 75 Furthermore, patients with OSA were more likely to have a history of foot ulceration 75 and one case series suggested that CPAP might aid the healing of foot ulceration in patients with diabetes. 96
OSA and other diabetes-related outcomes
OSA has been associated with worse quality of life and erectile dysfunction which can be improved by CPAP, but data in patients with T2D are lacking. 10
Summary and conclusion
OSA is a common condition and is associated with EDS, road traffic accidents, hypertension, worse glycaemic control, hypertension, CVD, diabetes-related microvascular complications, erectile dysfunction, impaired quality of life and increased mortality. CPAP treatment improves EDS and other OSA-related symptoms, has favourable impact on BP and lowers the risk of road traffic accidents, but there is currently no evidence from RCTs that CPAP can lower CVD or mortality.
OSA is very common in patients with T2D. The impact of OSA and CPAP in patients with T2D is still unclear with most studies having a cross-sectional nature. The limited number of longitudinal studies suggests that OSA is associated with hypertension, CVD, advanced DR and CKD in patients with T2D. Most RCTs of OSA in the context of T2D were focussed on glycaemic control and the results are conflicting, but ongoing RCTs are assessing the impact of CPAP on other diabetes-related outcomes.
While the impact of OSA and its treatment in patients with T2D remains to be fully explored, it is important to consider OSA in patients who have OSA-related symptoms such as EDS as CPAP treatment can improve symptoms. In addition, considering the increased risk of road traffic accidents in some patients with T2D, it is important to consider the contribution of OSA to this risk as CPAP might have a beneficial impact in this context. How to screen, the frequency of screening and the cost-effectiveness for screening for OSA in patients with T2D are questions that remain to be answered.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
A.A.T. is a clinician scientist supported by the National Institute for Health Research in the United Kingdom. The views expressed in this publication are those of the author(s) and not necessarily those of the National Health Service, the National Institute for Health Research or the Department of Health.
