Abstract
Diabetes mellitus, besides disrupting the carbohydrate metabolism process, also induces vascular disease and impacts nearly all the types and sizes of blood vessels. In fact, vascular complications cause majority of the morbidity, hospitalizations and mortality of patients with diabetes mellitus. Retinopathy, nephropathy and neuropathy (microvascular complications) impact hundreds of millions of diabetics and normally target those having long-term or uncontrolled forms of the disease; however, these disorders can also exist at the time of diagnosis or in those yet to be diagnosed. The Kingdom of Saudi Arabia is the biggest country in the Middle East that occupies around four-fifths of the Arabian Peninsula supporting a population of more than 33.3 million people. The prevalence of diabetes mellitus is increasing at an alarming rate in Saudi Arabia. Over 25% of the adult population is suffering and that figure is projected to more than double by 2030. In fact, diabetes mellitus has approximately registered a 10-fold upsurge in the past three decades in Saudi Arabia. However, the prevalence and risk factors of microvascular complications in diabetes mellitus patients have not yet been clearly documented in Saudi Arabia. Hence, in this review, we aim to provide an overview of the microvascular complications among patients with diabetes in Saudi Arabia, utilizing data from the currently available published literature. This is an attempt to facilitate the government and healthcare systems aware of the enormous worth of prevention, early detection and appropriate management of such microvascular complications.
Keywords
Introduction
According to the Diabetes Atlas (8th edition), almost 425 million people globally, or 8.8% of adults in the 20- to 79-year age bracket, are reckoned to suffer from diabetes. The number of diabetics will soar to 451 million if the age category is stretched to include ages of 18–99 years. Assuming that this drift continues, then by 2045, there will be 693 million people between 18 and 99 years, or 629 million individuals from 20 to 79 years of age with diabetes. 1 Moreover, the global annual cost incurred for healthcare by individuals with diabetes is US$727 billion, which translates as US$1 for every US$8 paid for healthcare. 1
The Middle East and North Africa (MENA) region has been projected to register the highest prevalence of diabetes overall in the wake of the meteoric rise in economic development, urbanization and lifestyle pattern changes in these regions.2,3 The International Diabetes Federation (IDF) MENA region (2017) reported that globally, 425 million people have been estimated to have diabetes, among which more than 39 million reside in the MENA region; by 2045, this number is anticipated to escalate to 67 million. 2 The Kingdom of Saudi Arabia (KSA) is not exempt from this worldwide epidemic, and diabetes is clearly recognized as the most daunting health issue it faces. The disturbing rise of diabetes in Saudi Arabia is referred to as an epidemic in many research works.3–6 Recently, the IDF also reported that there were 3,852,000 cases of diabetes in Saudi Arabia. 2 It is well demonstrated that poorly managed diabetes leads to serious diabetes-related vascular complications, reduced quality of life and early death. 7 However, in Saudi Arabia, the prevalence and risk factors of microvascular complications in patients with diabetes mellitus (DM) have not yet been clearly documented. Therefore, the goal of this study is to investigate the various aspects of microvascular complications in those individuals with diabetes, utilizing the data from the published literature available.
Methods
The PubMed, Scopus, Google scholar and Google archives were explored for pertinent information. The titles and abstracts were reviewed, as well as the bibliography of the articles selected. The following keywords were employed to identify the appropriate articles, namely, diabetes complication, vascular diseases, DM, type 2 diabetes (T2D), type 1 diabetes (T1D), paediatric diabetes, microvascular complications diabetic retinopathy (DR), diabetic neuropathy, diabetic peripheral neuropathy (DPN), diabetic nephropathy, awareness and Saudi Arabia. These words were utilized either individually or together to permit a large-scale literature search. The guidelines of the Critical Appraisal Skills Programme (CASP) were followed for the case-control and cohort studies as well as to construct a framework for quality estimation. Studies which included critical limitations either in methodology or reporting were precluded. 8 Data dealing with sample size, duration, and design and study methodology were identified and employed as indicators to assess quality.
Microvascular complications of diabetes
Implementation of changes in lifestyle patterns, like maintaining a robust body weight, staying on a healthy diet, being physically active, avoiding smoking and consuming moderate amounts of alcohol can prevent the onset and complications of diabetes in many instances.9,10 There is sufficient evidence to show that individuals with DM are more susceptible to various types of short- and long-term impediments, which frequently result in early demise.11,12 Diabetes patients also have a characteristic inclination towards greater rates of morbidity and mortality because this type of DM is subtle in onset and slow to be recognized. 13 Diabetes patients face high risk of microvascular complications developing in the small blood vessels. Microvascular complications are normally long-term and distinguishable as DR, nephropathy or neuropathy.14,15 DM has the pathologic warranty of involving the vasculature and causing microvascular complications. 15 The occurrence of microvascular complications in DM patients in Saudi Arabia has not yet been clearly documented. Such data are crucial, as vascular complications are responsible for nearly 50% of all fatalities among individuals with DM in the developed nations. 16
Diabetic retinopathy
Microvascular disease has been reported to be strongly connected to hyperglycaemia. 17 Across the range of chronic hyperglycaemia normally faced in clinical practice, a 11-fold increase in retinopathy is marked when compared with a twofold increase in coronary artery disease.18,19 Although hyperglycaemia is still significant, few patients may show early signs of retinopathy, in fact as early as 7 years prior to the establishment of frank T2D, pointing to the part played by insulin resistance. 20 According to World Health Organization (WHO), 2% of diabetics may become legally blind within roughly 15 years of the onset of the disease, while nearly 10% may end up with severely impaired vision. This has been reported in around 77% of T2D within 10 years of disease onset, and almost in every single T1D. 21 Besides the threat of hyperglycaemia and duration of diabetes mellitus, other retinopathy-related factors include hypertension, smoking and dyslipidaemia. These factors, besides other pathophysiologic mechanisms like insulin resistance and inflammation, may add to the severity of the microvascular disease. 22
From studies conducted in various parts of Saudi Arabia, a high incidence of DR has been reported among diabetics.23,24 In southern part of Saudi Arabia, a study done with 740 diabetes patients revealed that the retina could not be visualized in 4.1% patients, 68.1% showed no signs of retinopathy at all, 18% exhibited mild DR, 5.3% demonstrated observable DR, 3.5% had referable DR and 1.1% expressed proliferative diabetic retinopathy (PDR). 24 Generally speaking, 27.8% patients showed any form of retinopathy. With respect to maculopathy, the macula could not be visualized in 3.9% patients, 83% had no evidence of the disease, 7.8% revealed observable maculopathy and 5.3% showed referable maculopathy; thus, 13.1% of the patients exhibited some detectable form of maculopathy. On the whole, 28.1% of patients suffered from either retinopathy and/or maculopathy of their eyes. Furthermore, in 5.7% of the patients, sight-threatening diabetic retinopathy (STDR) could be detected, induced by either referable retinopathy or maculopathy, or both. 24 From reports on a study performed on 3051 patients in Taif, Saudi Arabia, DR was estimated at 36.8% and STDR was 17.5%. 25 In another study done in Abha on 401 patients, DR showed an overall incidence of 36.4%. In fact, 57.5% of the patients showed mild non-proliferative diabetic retinopathy (NPDR), while 19.9% revealed moderate NPDR and 11.0% suffered from severe NPDR. Finally, 11.6% of diabetes patients showed PDR. Only 7.2% of the DR patients exhibited exudative and focal maculopathy. Significantly, maculopathy was closely linked to severe NPDR and PDR. The retinopathy rate was strongly related to those individuals older in age, with onset at a younger age, with lengthier disease duration and poor blood sugar control, accompanied by hypertension (on treatment with medication), using insulin and having multiple complications 26 (Table 1).
Description of the included studies for diabetic retinopathy.
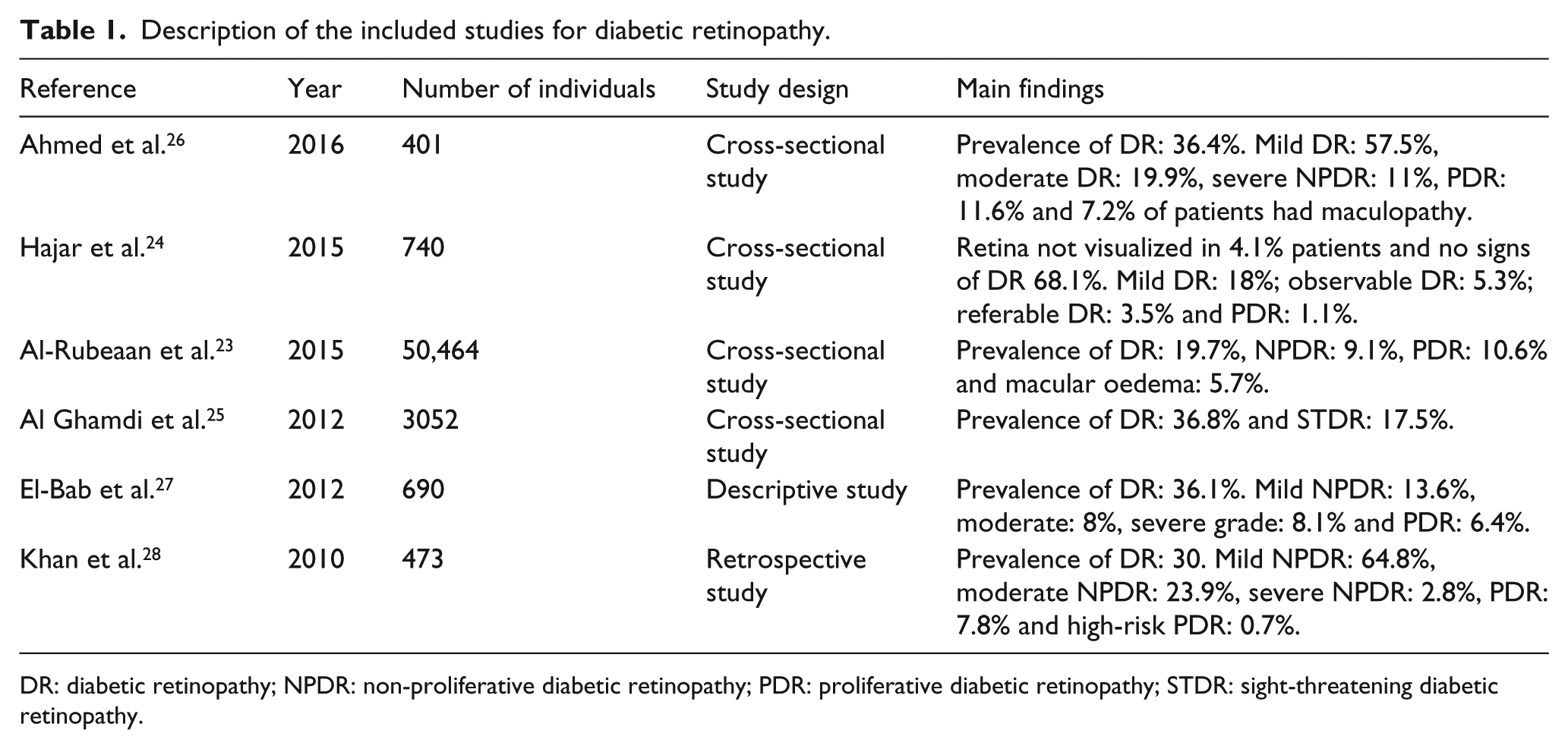
DR: diabetic retinopathy; NPDR: non-proliferative diabetic retinopathy; PDR: proliferative diabetic retinopathy; STDR: sight-threatening diabetic retinopathy.
In an investigation of 690 diabetes patients in Al-Madinah Al-Munawarah, 36.1% were reported to have retinopathy. While 13.6% patients suffered from mild NPDR, 8% had moderate grade and 8.1% experienced severe grade. Another 6.4% showed PDR. 27 In a recent study from Saudi National Diabetes Registry (SNDR) in Saudi Arabia on 50,464 T2D patients aged ⩾25 years, an overall incidence of 19.7% of DR was registered, in which 9.1% exhibited NPDR, 10.6% had PDR and 5.7% showed macular oedema (ME). For DR, diabetes duration and age are the most important risk factors with odds ratio (OR) and 95% confidence interval (95% CI) 8.88 (8.30–9.50) and 5.76 (5.10–6.55), respectively. The risk for DR increased significantly with the presence of nephropathy, neuropathy, insulin use, poor glycaemic control, hypertension and male gender. On the contrary, the T2D Saudi cohort saw a significant drop in the risk for DR with smoking, hyperlipidaemia and obesity. 23 The DR showed a general prevalence of 30% from the findings of a study from the Al Hasa region. Of these patients, 64.8% revealed mild NPDR, with 23.9% showing moderate NPDR, 2.8% experiencing severe NPDR, 7.8% demonstrating PDR and 0.7% revealing high-risk PDR. 28 These variations in the prevalence of DR may result from the differences in the age distribution of the various populations, but may also be caused by the types of study methods used and population sampling. Reeling under the heavy burden of this disease, the Saudi Ministry of Health (MOH) in association with the Saudi National Prevention of Blindness Committee (NPBC) has commissioned more research studies to assess the enormity of the issue in other parts of this huge country. 24
Despite the raising awareness among the patients with diabetes in Saudi Arabia concerning the effect of this disease upon their eyes, there still continues to be negligible or nil knowledge regarding its risk factors and prevention.29,30 The pressing need in Saudi Arabia, therefore, is to raise this level of awareness in order to reduce the number of DR-induced incidence of blindness. A hospital-based, cross-sectional study (n = 357) in Saudi Arabia revealed that only 61% of patients with DM had any awareness of DR. Among these, 38% understood the necessity for annual retinal examinations, while 70% were completely unaware of the treatment for DR. Interestingly, only 50% of all the respondents scheduled eye checkup visits.31,32 Very recently, from a cross-sectional study done among patients with DM who visited the primary healthcare centres at the MOH in Jeddah, it became clear that 82.6% of them appreciated the fact that DM could affect their eyes; they cited physicians, ophthalmologists, television and family members as the common information sources on this subject. Around 36% of these patients indicated that they had not been advised by their doctors regarding this. More than 50% did not feel that DM affected their vision. In fact, those who had never had any diagnosis of DR were more than 58%. Around 35% never attended their scheduled eye checkups, although nearly 59% had some concept that DR could cause blindness. 30 When compared the develop countries, the lack of an effective programme to restrain the meteoric escalation of T2D in Saudi Arabia has resulted in a significant development of this disease, which according to the latest information emphasizes that T2D cannot be excluded in children below 18 years of age. Children with the diagnosis of T2D come from all the Arab countries, and obesity is the characteristic sign in this age category. 33 The finding of Saudi studies comparable with other Arab countries. An investigative study on 513 diabetes patients in the United Arab Emirates (UAE) revealed that while 19% showed retinopathy, most of them (74%) were unaware of their state of health. 34 Further results revealed that patients with hypertension, microalbuminuria, peripheral vascular disease, coronary artery disease and neuropathy had higher incidence of retinopathy. Likely, some other Arab countries also showed higher prevalence of DR; Egypt (42%), Oman (42.4%) and Kuwait (40%).35–37 Another study reported that across MENA countries, retinopathy affected 17%–54% of people with diabetes aged 49–60 years. Men were generally more commonly affected by DR. 38
Diabetic neuropathy
Diabetic neuropathy is a group of nerve disorders, and it is a common troublesome complication of both T1D and T2D, leading to the highest morbidity and mortality and resulting in a vast financial problem for diabetes care. The clinical evaluation of diabetic neuropathy and its management options is multifactorial.39,40 Diabetic neuropathy, particularly polyneuropathy, denotes the most predominant manifestation of diabetes in the nervous system. However, they remain often overlooked, with undiagnosed polyneuropathy in one-third of patients with T1D and in around 60% of those with T2D.41–44
Over the last few decades, only a small number of studies have been performed on diabetic neuropathy in Saudi Arabia. In a research work done on 552 Saudi participants, 110 were diagnosed with DPN, indicating 19.9% prevalence. 45 The participants having DPN were more advanced in age, had diabetes for a longer duration and with greater incidence of abdominal obesity and hypertension when compared with their counterparts lacking DPN. 45 From a multicenter, cross-sectional study in which 242 T2D patients who attended the National Guard primary healthcare clinics in Riyadh were assessed, 35% of them with T2D in this investigation had painful DPN. 46 Findings from another study from the central part of Saudi Arabia revealed that 5.6% had neuropathy. 47 From a study involving a total of 201 T2D patients who were followed in the King Fahad National Guard Hospital Diabetic Clinic, Riyadh, Kingdom of Saudi Arabia, 24% of the patients showed mild forms of neuropathy, 22% exhibited moderate forms and 36% experienced severe neuropathy. 48 Another study done using 237 patients demonstrated that 132 (56%) patients had symptomatic diabetic neuropathy, while 58 (57%) showed subclinical neuropathy in asymptomatic patients. They also reported that old age, T2D over a long time period, poor control and the smoking habit were the risk factors related to symptomatic diabetic neuropathy. 49 In Saudi Arabia, in a nationally representative diabetes population, 65.3% incidence of painful DPN had been reported earlier 50 (Table 2).
Description of the included studies for diabetic neuropathy.
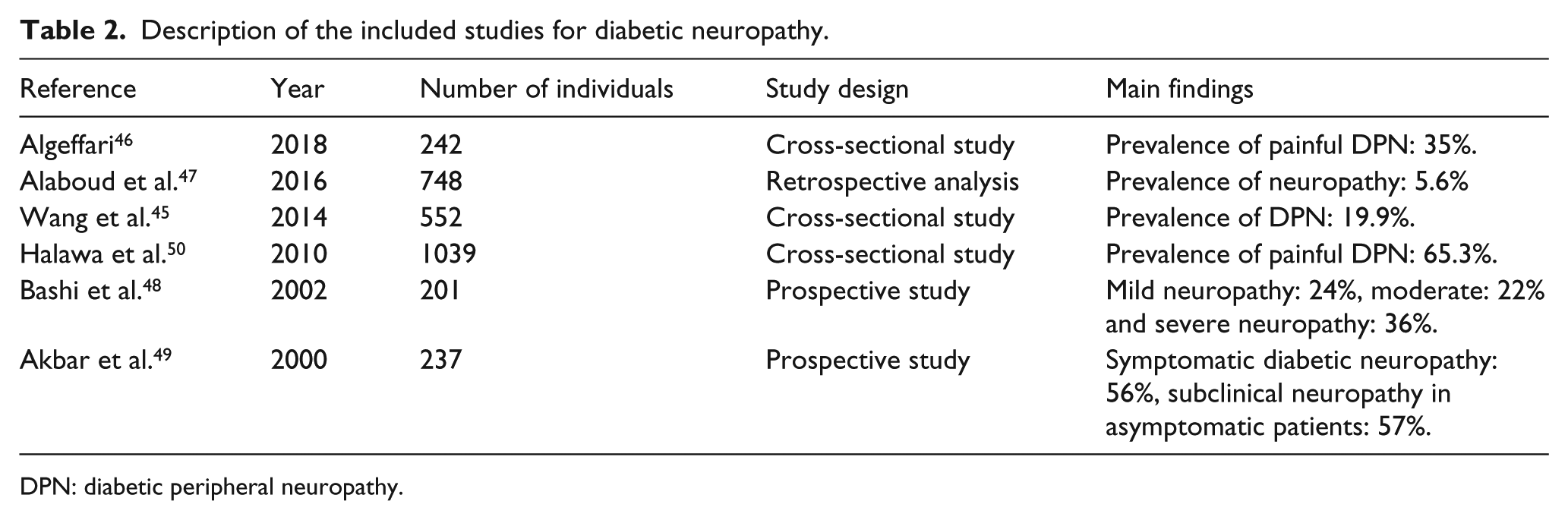
DPN: diabetic peripheral neuropathy.
The prevalence of neuropathy associated with diabetes was very high in the MENA region. 38 The occurrence of diabetic neuropathy among people in communities with diabetes for 10 years was 35% in the UAE and 37% in Bahrain.38,51 Diabetic neuropathy was even more common among the clinic-attending population, affecting 54% of outpatients at clinics across Egypt, Lebanon, Jordan, Kuwait, and the UAE. 52 Importantly, a recent meta-analysis performed on studies representing different Arab countries among the T1D patients concluded that diabetic neuropathy is common in adults and children with T1D, but prevalence differs widely. The risk increases as a person gets older and has had diabetes longer. 53 Unfortunately, there is inadequate awareness about neuropathy among the MENA population. A study also reported that there is a lack of awareness in the MENA among both patients and physicians regarding neuropathy and the associated risks factors, due to the lack of education and specialist centres for its management. 54
Diabetic nephropathy
Diabetic nephropathy occurs in around 7% of T2D patients at the time they received the diagnosis of DM. It is observed in ⩽12% of T1D after about 7 years, while around 25% of T2D patients show evidence of nephropathy after nearly 10 years post diagnosis. 55 Proteinuria is the characteristic indicator for diabetic nephropathy, widely accepted as the principal reason for end-stage renal disease (ESRD). It is also regarded as the first evidence of kidney damage in patients with diabetes. Nearly 10 years ago, evidence of ESRD in T2D patients was cited as a ‘medical catastrophe of worldwide dimension’. 56 Furthermore, recently, it is well recognized that diabetic nephropathy is one of the major reasons of increased morbidity and mortality among diabetes, directly by causing ESRD and indirectly by increasing other risk factors. 57 In fact, a recent study found that diabetic nephropathy was the most common cause of ESRD, accounting for 30.4% in Saudi Arabia. 58 It is reported by many studies that the prevalence of nephropathy associated with diabetes was very high in the MENA region.35,51,59
Saudi Arabia indicated that nearly one-third of T2D population has diabetic nephropathy and it needs to be extensively studied to explore risk factors that might be related to its gradually increasing occurrence in Saudi Arabia. 60 A study was conducted recently on 54,670 T2D patients ⩾25 years of age who were shortlisted from the SNDR and investigated for evidence of diabetic nephropathy. With the prevalence of diabetic nephropathy being 10.8% on the whole, 1.2% accounted for microalbuminuria, 8.1% for macroalbuminuria and 1.5% for ESRD. The factors age and duration of diabetes as vital risk factors powerfully influence the prevalence of diabetic nephropathy, ranging from 3.7% in patients 25–44 years of age and having a duration of diabetes >5 years to 21.8% in patients ⩾65 years in age with a duration of diabetes ⩾15 years. The variables diabetes duration, retinopathy, neuropathy, hypertension, age > 45 years, hyperlipidaemia, male gender, smoking and chronologically poor glycaemic control are significant high-risk factors for diabetic nephropathy. 61 From a hospital-based retrospective analysis of T2D patients, in which 1952 files were reviewed, only 621 (31.8%) fulfilled the criteria for diabetic nephropathy. The glomerular filtration rate (GFR) was observed to decrease from a baseline value of 78.3 (30.3) mL/min/1.73 m2 to 45.1 (24.1) mL/min/1.73 m2 at the final visit, with a mean rate of decline in the GFR of 3.3 mL/min/year. The progression of nephropathy was evident in 455 (73.3%) patients; 250 (40.3%) patients saw the value of their first-hospital-visit serum creatinine level double within a mean of 10.0 (6.0) years. At the culmination of the study, 16.5% of the cohort developed ESRD and was given dialysis. The GFR value was > 90 mL/min/1.73 m2 at the first hospital visit, duration of diabetes >10 years, proteinuria was persistent, systolic blood pressure > 130 mm Hg and retinopathy was present with significant markers related to the progression of nephropathy. 62 Another study was performed using T2D patients between 35 and 70 years of age, with duration of diabetes >10 years, including individuals with microalbuminuria, macroalbuminuria and ESRD in Saudi Arabia. The patients were compared with those having normal albumin excretion. The report showed that, among the 427 patients, 184 (43%) showed microalbuminuria, 83 (19%) possessed macroalbuminuria and 160 (37%) had ESRD. None of the remaining 213 (50%) patients had nephropathy. Patients with nephropathy were characteristically older in age, with a longer duration of diabetes, and a lower monthly income and body mass index than patients without nephropathy 11 (Table 3).
Description of the included studies for diabetic nephropathy.
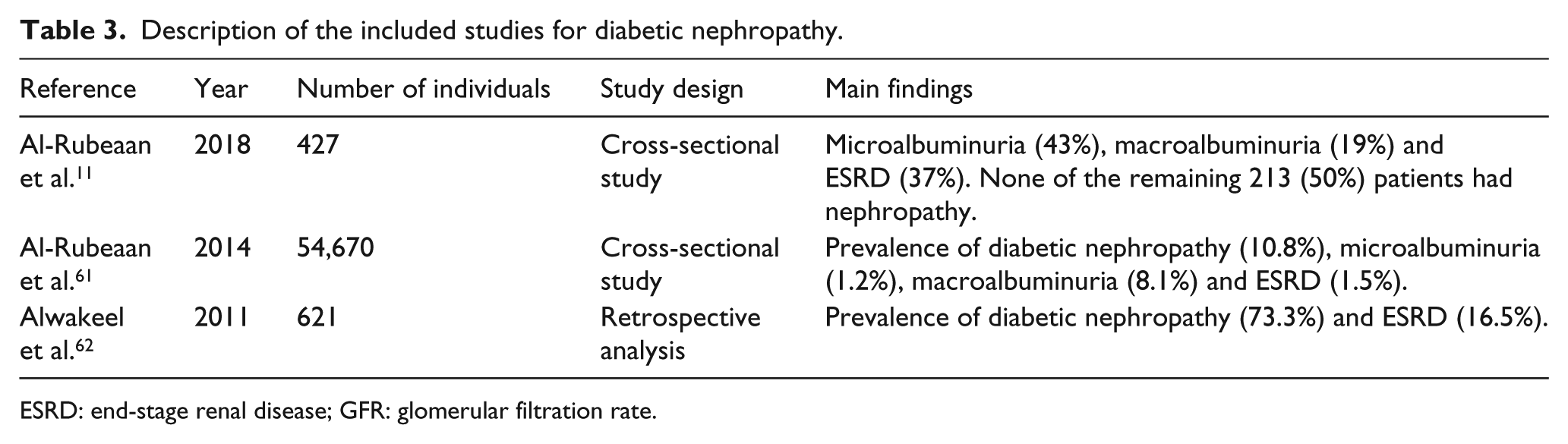
ESRD: end-stage renal disease; GFR: glomerular filtration rate.
It should be noted that similar to Saudi Arabia, Arab Gulf countries also showed a high incident rate of diabetic nephropathy. In the UAE, diabetes was identified the major cause for ESRD, seen in 23.3% of the patients. Therefore, because more than one-third of the patients with diabetes collectively ended up having kidney disease, it is probable that diabetic kidney disease is another significant health problem in the Arab Gulf countries.63,64 In a study from Oman reported that the prevalence of diabetic nephropathy was 42.5%. 65 Importantly, a review reported that the diabetic nephropathy in the Arab Gulf countries was positively related to male gender, body mass index, poor diabetes control and presence of DR and/or neuropathy. 64
Practice recommendations
With the correct diabetes management and suggested lifestyle changes, people with diabetes are able to avoid or delay the onset of complications.66,67 In order to avoid the severe complication, patients who previously identified with diabetes required to follow the routine examination.
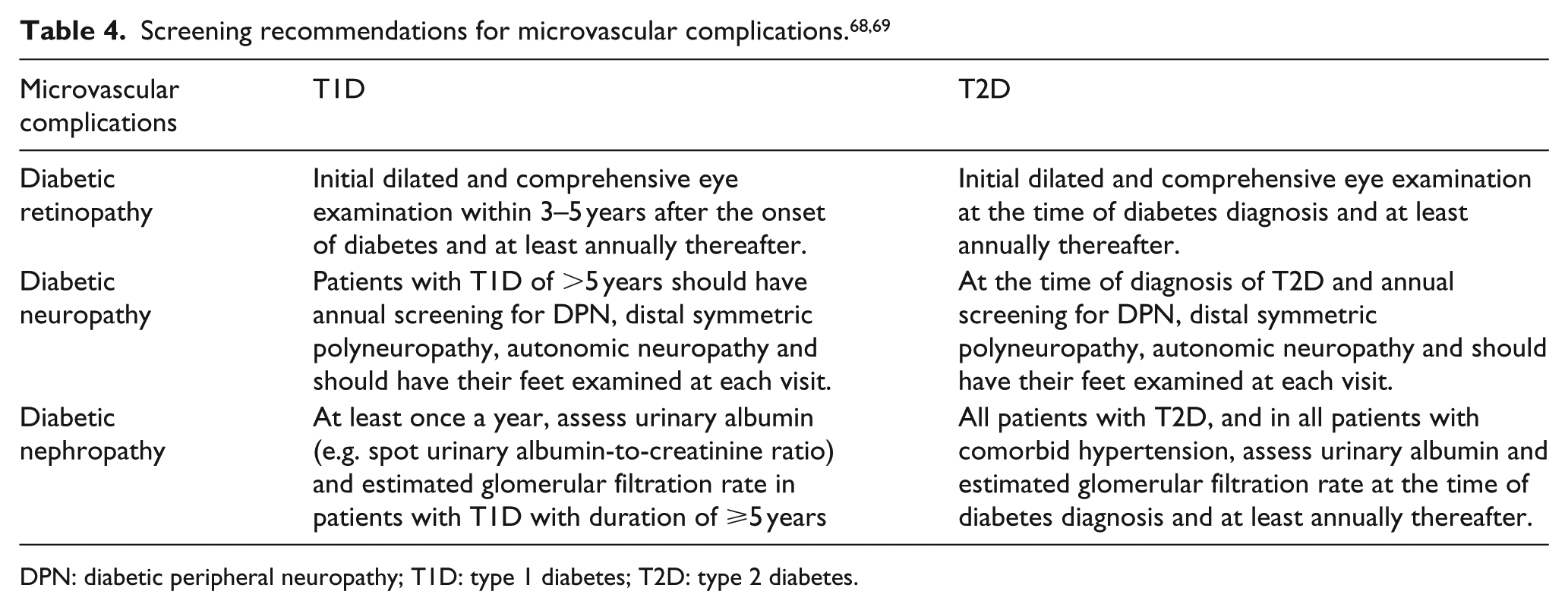
A comprehensive eye examination and dilation within 3–5 years after the onset of diabetes is essential for all patients with T1D, and such screening at the time of diagnosis is mandatory for T2D. 68 All patients should strive for ideal glucose and blood pressure control, in order to decrease the possibility of developing DR or experiencing progression of retinopathy. Patients with T1D of more than 5 years should have annual screening for microalbuminuria, and patients with T2D should undergo such screening at the time of diagnosis and yearly afterwards. Also, diabetes patients should perform serum creatinine measurement annually. 68 Patients who have microalbuminuria or macroalbuminuria should be treated with an angiotensin-converting enzyme inhibitor or angiotensin-receptor blockers. Routine referral to a nephrologist is imperative for patients who have a GFR <60 mL/min or with uncontrolled hypertension or hyperkalemia. 68 All patients with diabetes should undergo screening for distal symmetric polyneuropathy at the time of identification and yearly afterwards. Patients who have peripheral neuropathy should commence with appropriate foot self-care, including wearing special footwear to avoid their risk of ulceration and required referral for podiatric care regularly. Screening for autonomic neuropathy should take place at the time of diagnosis of T2D and beginning 5 years after the diagnosis of T1D. Medication to control the signs of painful peripheral neuropathy may be beneficial in improving the quality of life in patients, but does not appear to revise the natural course of the disease. Hence, patients should continue to strive for the good conceivable glycaemic control (Table 4).
According to the previous studies from Saudi Arabia, it is recommended every Saudi above 30 years of age should be screened for both T2D and pre-diabetes to comprise the diabetes and its complications.4,5 It is important to note here that the American Diabetes Association (ADA) guidelines usually updated every year with recent published data and easily applied in clinical practice. We advocated the application of the ADA 2018 guidelines for all Saudi diabetic clinics to prevent/delay the diabetes-related microvascular complications.29,69
Diabetes education
Education plays a significant role not only in the prevention of diabetes itself but also in preventing its complications. 70 Currently, health education to prevent diabetes microvascular complications in Saudi Arabia is non-existent. Major gaps also exist in education about health and nutrition among the Saudi populations. Furthermore, Saudi diabetes patients have lack of awareness about diabetes risk factors, which leads to the risk for microvascular complications.4,5 From a recent study in Saudi Arabia, it became evident that around 40.3% of patients with diabetes were unaware of DM. 71 Another study reported that the diabetes knowledge score among the Saudi population was 67.4%; however, the scores for general knowledge on the disease was 71.1%; the risk factors was 63.4%; symptoms was 80.8% and complications was 47.7%. 62 This evidently specifies the alarming need for greater efforts in educating the general and diabetes population on diabetes and the complications associated with it.4,5
Conclusion
The high incidence of the diabetes-related complications in the Saudi population poses a crucial problem from the clinical and public health perspectives. The DM and its complications are rapidly snowballing into a huge epidemic, posing a threat to nullify the advantages of modernization and economic rejuvenation. Saudi Arabia has identified several socioeconomic, dietary and lifestyle variables related to DM and its associated complications. Exhaustive studies investigating the part played by these factors and their roles in augmenting the DM associated complications are urgently required. Besides, majority of the research done in Saudi Arabia is cross-sectional and focused on restricted sample sizes, which most often involve only certain parts of the country. More multi-dimensional and multi-sectoral research is urgently required to strengthen the evidence base and to collect greater knowledge as a basis for methods and programmes to tackle the diabetes and its complications. It is recommended that every Saudi citizen, 30 years of age and above, must undergo screening for both T2D, pre-diabetes and its complications to comprehensively study the disease and its associated complications. Also, the need for establishing a national prevention programme as early as possible is a vital need. Through this programme, a check can be maintained on diabetes and its complications and the modifiable risk factors can be dealt with at the community level, with greater concentration being focused on the high-risk groups.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.