Abstract
Advanced glycation end-products, especially toxic advanced glycation end-products derived from glyceraldehyde (advanced glycation end-product-2) and glycolaldehyde (advanced glycation end-product-3), are biologically reactive compounds associated with diabetic complications. We previously demonstrated that toxic advanced glycation end-products were internalised into macrophage-like RAW264.7 cells through scavenger receptor-1 class A (CD204). Toxic advanced glycation end-product uptake was inhibited by fucoidan, a sulphated polysaccharide and antagonistic ligand for scavenger receptors, suggesting that sulphated polysaccharides are emerging candidates for treatment of advanced glycation end-product–related diseases. In this study, we compared the effects of six types of sulphated and non-sulphated polysaccharides on toxic advanced glycation end-product uptake in RAW264.7 cells. Fucoidan, carrageenan and dextran sulphate attenuated toxic advanced glycation end-product uptake. Fucoidan and carrageenan inhibited advanced glycation end-product-2–induced upregulation of SR-A, while advanced glycation end-product-3–induced upregulation of scavenger receptor-1 class A was only suppressed by fucoidan. Dextran sulphate did not affect scavenger receptor-1 class A levels in toxic advanced glycation end-product–treated cells. Chondroitin sulphate, heparin and hyaluronic acid failed to attenuate toxic advanced glycation end-product uptake. Heparin and hyaluronic acid had no effect on scavenger receptor-1 class A levels, while chondroitin sulphate inhibited advanced glycation end-product-3–induced upregulation of scavenger receptor-1 class A. Taken together, fucoidan and carrageenan, but not the other sulphated polysaccharides examined, had inhibitory activities on toxic advanced glycation end-product uptake and toxic advanced glycation end-product–induced upregulation of scavenger receptor-1 class A, possibly because of structural differences among sulphated polysaccharides.
Keywords
Introduction
Accumulated evidence suggests that advanced glycation end-products (AGEs), which result from prolonged exposure of proteins to sugars, are associated with both microvascular and macrovascular complications in diabetic mellitus.1,2 AGEs are also associated with the development of age-related diseases such as cardiovascular disease, Alzheimer’s disease and osteoporosis.3,4 According to their sugar types, AGEs in serum of diabetic patients were classified into six groups. 5 Among the AGEs, those derived from glyceraldehyde (AGE-2) and glycolaldehyde (AGE-3) in particular are known as toxic AGEs and have potent activity compared with other AGEs, such as those derived from methylglyoxal (AGE-4) and glyoxal (AGE-5). For example, our group previously demonstrated that toxic AGEs, but not AGE-4 and AGE-5, led to enhanced inflammatory cytokine production by human blood mononuclear cells. 6 Furthermore, AGE-2 and AGE-3 had high affinity for receptor for AGEs (RAGE) compared with AGE-4 and AGE-5. 7 Therefore, toxic AGEs may play an important role in the pathogenesis of diabetic complications and age-related diseases.
Scavenger receptor-1 class A (SR-A/CD204) was initially discovered as a candidate molecule associated with the recognition and uptake of modified low-density lipoprotein (LDL) and the development of atherosclerosis. 8 Recently, we demonstrated that SR-A was involved in toxic AGE uptake and increased after exposure to toxic AGEs in RAW264.7 cells. 9 Furthermore, SR-A–deficient mice showed resistance to diabetic nephropathy. 10 Taken together, these findings raise the possibility that SR-A is associated with the physiological dysfunctions induced by toxic AGEs.
The development and improvement of technologies such as mass spectrometry have advanced our understanding about glycobiology. Polysaccharides/glycans play roles not only as energy sources but also in diverse physiological functions mediated by their interactions with proteins and lipids. Among the polysaccharides, sulphated polysaccharides easily interact with positively charged molecules because of their high negative charge at neutral pH. Sulphated polysaccharides are widely present in the natural world. Glycosaminoglycans, which include chondroitin sulphate, heparin and hyaluronic acid, are major sulphated polysaccharides in mammalian tissues, while fucoidan and carrageenan are derived from brown and red algae, respectively. Meanwhile, dextran sulphate, which is artificially produced by sulphation of dextran derived from sucrose fermentation by microbes, is a representative sulphated polysaccharide. These sulphated polysaccharides not only have many beneficial biological activities such as anticoagulant, antioxidant, angiogenic and immunomodulatory effects but also possess unique physical properties such as gelation or water retention capacities.11–13 Therefore, sulphated polysaccharides are applied in a wide range of areas, including cosmeceutical, nutraceutical and pharmaceutical products. In addition, several sulphated polysaccharides appear to have anti-diabetic effects, 14 and glycosaminoglycans are useful for the treatment and prevention of diabetic complications. 15 However, the effects of sulphated polysaccharides on the functions of toxic AGEs remain unclear. In a previous study, we demonstrated that fucoidan suppressed toxic AGE uptake and increased SR-A levels induced by toxic AGEs. 9 In this study, we compared the effects of algae-derived sulphated polysaccharides, glycosaminoglycans and artificial sulphated polysaccharides on toxic AGE uptake and SR-A expression in RAW264.7 cells.
Methods
Reagents
Bovine serum albumin (BSA; FUJIFILM Wako, Osaka, Japan) was incubated under sterile conditions with D-glyceraldehyde (AGE-2) (Sigma–Aldrich, St. Louis, MO, USA) or glycolaldehyde dimer (AGE-3) (Sigma–Aldrich) in 0.2-M phosphate buffer (pH 7.4) at 37°C for 7 days. As a control, BSA was incubated under the same conditions without additional compounds. After the incubation, AGE–BSA and BSA were dialysed for 2 days at 4°C. The endotoxin concentration of AGEs at 100 µg/mL was measured by SRL (Okayama, Japan) as 1.2 pg/mL. Fucoidan derived from Fucus vesiculosus (Sigma–Aldrich), dextran 500,000 (FUJIFILM Wako), low-molecular-weight (LMW) dextran sulphate (average MW, 5000; FUJIFILM Wako), high-molecular-weight (HMW) dextran sulphate (average MW, 500,000; FUJIFILM Wako), chondroitin sulphate C sodium salt (FUJIFILM Wako), heparin (Nacalai Tesque, Kyoto, Japan), hyaluronic acid (FUJIFILM Wako) and neocarrahexaose-24,41,3,5-tetra-O-sulphate (Dextra Laboratories, Reading, UK) were dissolved in ultrapure water. λ-carrageenan (FUJIFILM Wako) was dissolved in ultrapure water and incubated at 60°C for 30 min. All reagents were prepared under sterile conditions.
Cell culture
The mouse macrophage cell line RAW264.7 was obtained from DS Pharma Biomedical (Osaka, Japan). RAW264.7 cells were cultured in Dulbecco’s modified eagle medium (DMEM) containing 2 mM glutamine and 10% heat-inactivated fetal bovine serum (FBS) at 37°C under 5% CO2.
Fluorescent labelling of AGEs
Each protein was incubated with a 20× amount of Alexa Fluor 488 C5 maleimide (Thermo Fisher Scientific, Waltham, MA, USA) at room temperature for 2 h in phosphate-buffered saline (PBS) and then dialysed against PBS at 4°C for 2 days. Total protein concentrations were quantified by the Bradford method using a Bradford protein assay kit (Bio-Rad Laboratories, Kidlington, UK). The fluorescence intensity of Alexa Fluor 488–labelled compounds was measured using an ARVO MX 1420 (PerkinElmer Japan, Yokohama, Japan) with excitation at 485 nm and emission at 535 nm. The fluorescence intensities of Alexa Fluor 488–conjugated BSA, AGE-2 or AGE-3 per unit dosage were adjusted by adding the respective unlabelled proteins.
Flow cytometric analysis for AGE uptake in RAW264.7 cells
RAW264.7 cells were seeded in 24-well plates at 1 × 105 cells/well. After adhesion to the plates, the cells were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 500 µg/mL (fucoidan, carrageenan, hyaluronic acid) or 1 to 1000 µg/mL (dextran sulphate, chondroitin sulphate, heparin) in the presence or absence of Alexa Fluor 488–labelled BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. Subsequently, the cells were harvested and processed twice by rinsing with fluorescence-activated cell sorter (FACS) wash buffer (PBS supplemented with 2.5% normal horse serum, 0.1% sodium azide, 10 mM 2-[4-(2-Hydroxyethyl)-1-piperazinyl]ethanesulfonic acid (HEPES)) and centrifugation (200 × g, 5 min, 4°C). The cells were then resuspended in 200 µL of PBS (−) and stained with propidium iodide (PI) (2 µg/mL; Dojindo Laboratories, Kumamoto, Japan) to exclude PI-positive dead cells. Thereafter, analysis was performed using a FACS CantoII (BD Biosciences, San Jose, CA, USA), and the data were processed with BD FACSDiva software (BD Biosciences) to determine the mean fluorescence intensity (MFI) of Alexa Fluor 488–labelled BSA, AGE-2 and AGE-3.
Flow cytometric analysis for SR-A on RAW264.7 cells
RAW264.7 cells were seeded in 24-well plates at 1.0 × 105 cells/well and concomitantly incubated with each sulphated polysaccharide and BSA or AGEs (200 µg/mL) for 1 h. Subsequently, the cells were harvested and rinsed with FACS wash buffer followed by centrifugation (200 × g, 5 min, 4°C) and incubated with phycoerythrin-conjugated mouse anti-CD204 antibody (4 ng, 130-102-328; Miltenyi Biotec, Bergisch Gladbach, Germany) at 4°C for 30 min. After rinsing with wash buffer and centrifugation (200 × g, 5 min, 4°C), 200 µL of PBS (−) was added to the cell pellet followed by staining with PI (2 µg/mL), FACS CantoII analysis and data processing using BD FACSDiva software to determine the MFI of CD204 on RAW264.7 cells.
Statistical analysis
All data are expressed as mean ± standard error of mean (SEM). Significant differences were determined by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for comparisons among more than three groups using GraphPad Prism version 7 software (GraphPad Software, San Diego, CA, USA). Values of p < 0.05 were regarded as significant.
Results
Effects of sulphated polysaccharides on toxic AGE uptake by RAW264.7 cells
Accumulation of AGEs was observed in macrophage-derived foam cells in early human atherosclerotic lesions, 16 suggesting that inhibition of AGE uptake in macrophages may be important for the prevention and treatment of AGE-related diseases. In this study, to compare the effects of individual sulphated polysaccharides on toxic AGE uptake, cells were concomitantly treated with each sulphated polysaccharide and fluorescently labelled BSA, AGE-2 or AGE-3 (200 µg/mL) and measured for their fluorescence intensity by flow cytometric analysis. For these experiments, fluorescently labelled BSA, AGE-2 and AGE-3 were prepared using Alexa Fluor 488–conjugated maleimide, which binds to the thiol groups of proteins, under identical experimental conditions. We found that glycolaldehyde-derived AGE (AGE-3) exhibited the lowest fluorescence intensity (data not shown), indicating that AGE-3 was more greatly modified with BSA than AGE–2. The concentration ranges of the sulphated polysaccharides were determined based on our previous report and other reports on the physiological effects of sulphated polysaccharides.9,17,18 The maximum concentration of carrageenan and hyaluronic acid was 500 µg/mL because of their limited solubility in sterile water. For all polysaccharides examined, no cytotoxic effect was observed even when the concentration was increased to 500 µg/mL (fucoidan, carrageenan, hyaluronic acid) and 1000 µg/mL (HMW dextran sulphate, chondroitin sulphate, heparin) (Supplemental Figure and Supplemental Table).
Consistent with our previous findings, AGE-2 and AGE-3 at 200 µg/mL showed enhanced uptake in RAW264.7 cells (Figure 1(a)). Uptake of both AGE-2 and AGE-3 was dose dependently suppressed by the algae-derived sulphated polysaccharides fucoidan at 10–500 µg/mL (IC50: AGE-2, 9.72 µg/mL; AGE-3, 127.9 µg/mL) and carrageenan at 100 and 500 µg/mL (IC50: AGE-2, 94.4 µg/mL; AGE-3, 105.8 µg/mL) (Figure 1(b) and (c)). In addition to algae-derived sulphated polysaccharide, HMW dextran sulphate at 100–1000 µg/mL inhibited toxic AGE uptake (IC50: AGE-2, 15.0 µg/mL; AGE-3, 6.19 µg/mL) (Figure 2(a)).
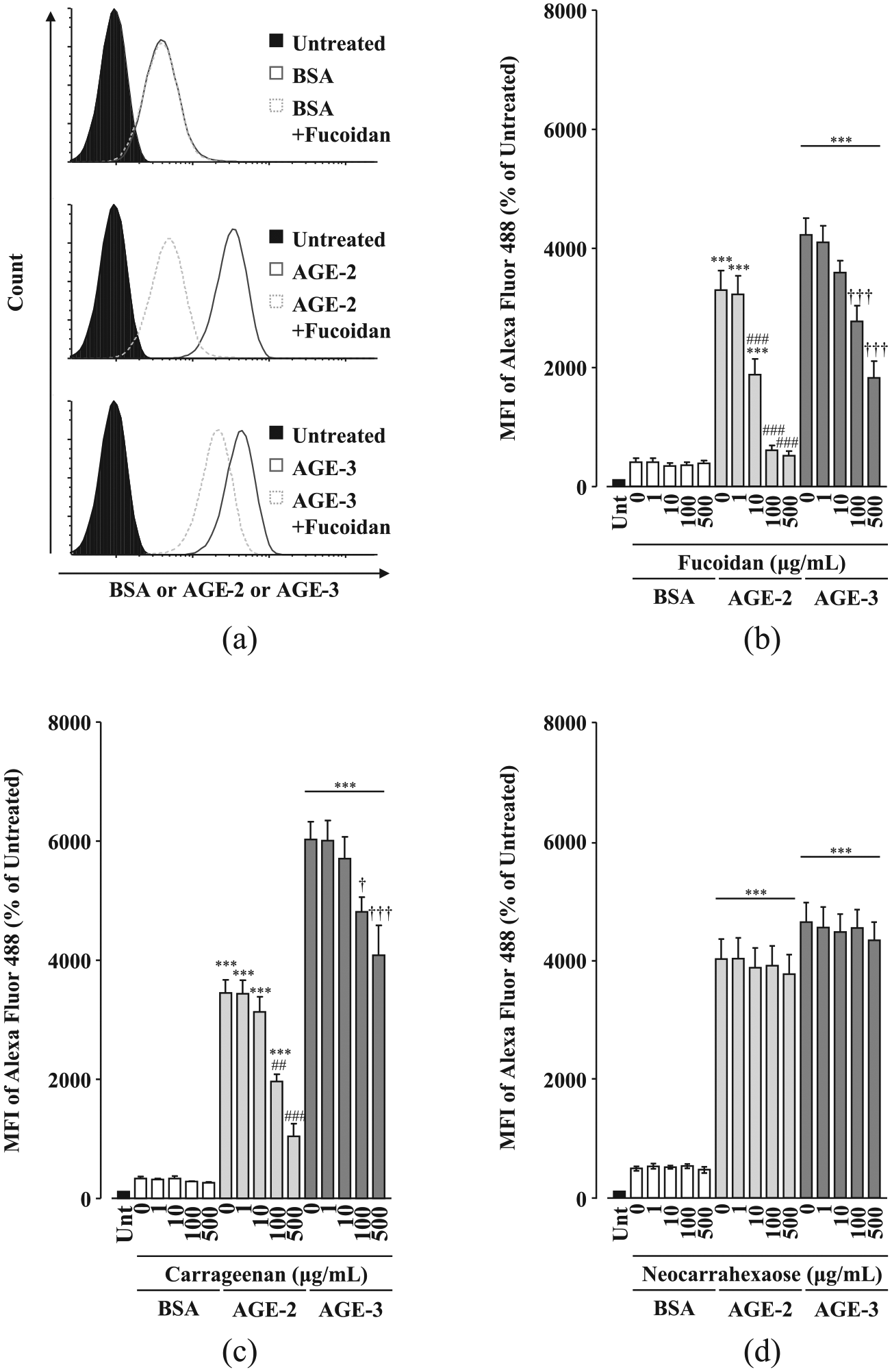
Effects of algae-derived sulphated polysaccharides on toxic AGE uptake by RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 500 µg/mL in the presence and absence of fluorescence-labelled BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. Cellular uptakes of fluorescently labelled BSA, AGE-2 or AGE-3 were determined by flow cytometry. (a) Representative flow cytometric analysis of BSA, AGE-2 or AGE-3 endocytosed by RAW264.7 cells treated with fucoidan (500 µg/mL). (b) Fucoidan (n = 5). (c) Carrageenan (n = 5). (d) Neocarrahexaose-24,41,3,5-tetra-O-sulphate (n = 3). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
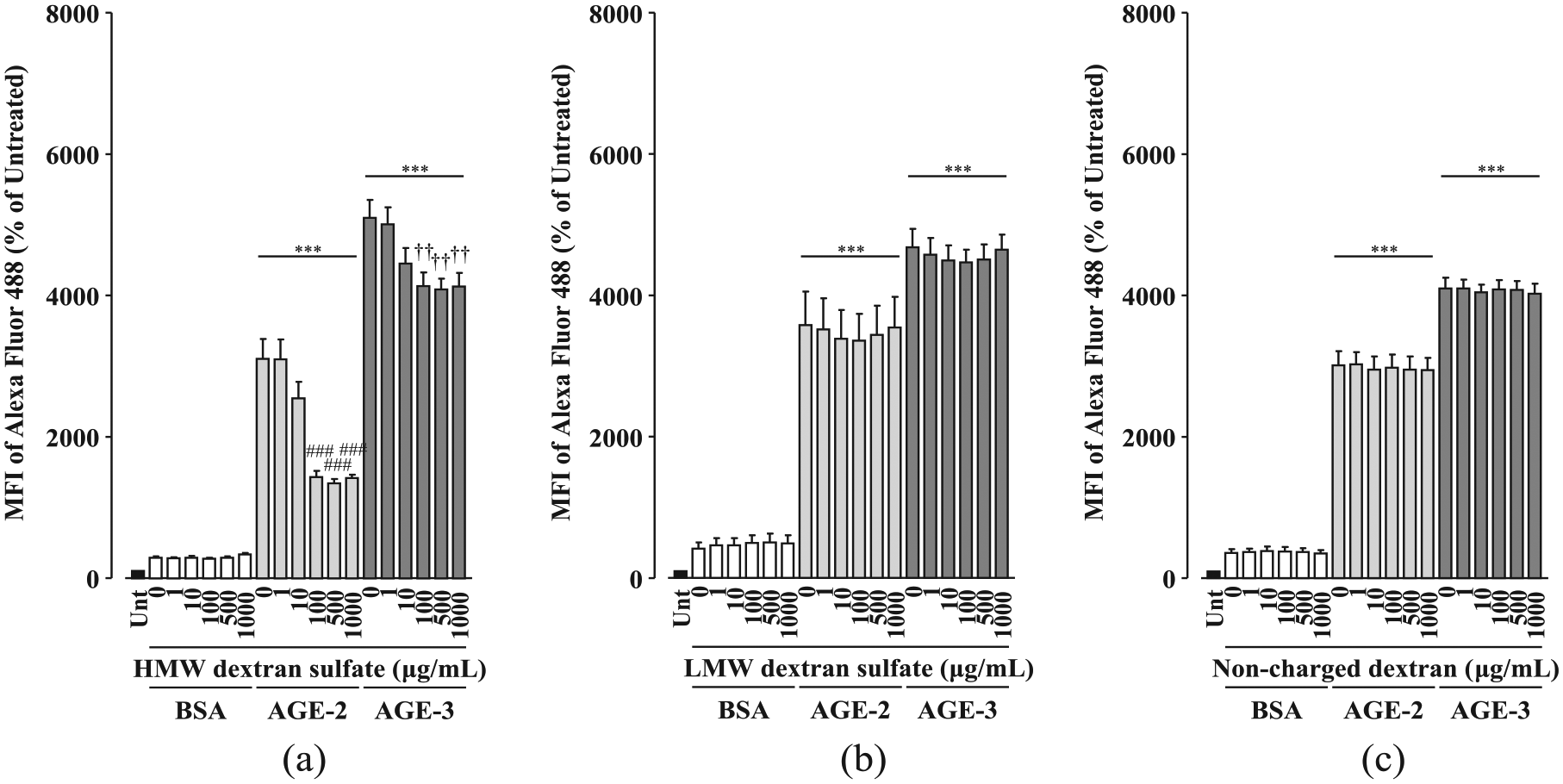
Effects of artificial sulphated polysaccharides on toxic AGE uptake by RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 1000 µg/mL in the presence and absence of fluorescently labelled BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. Cellular uptakes of fluorescently labelled BSA, AGE-2 or AGE-3 were determined by flow cytometry. (a) HMW dextran sulphate (n = 5). (b) LMW dextran sulphate (n = 5). (c) Non-charged dextran (n = 5). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
In contrast, glycosaminoglycans including chondroitin sulphate, heparin and hyaluronic acid had no effect on toxic AGE uptake within the concentration range of 1.0 to 1000 µg/mL (Figure 3(a) to (c)).
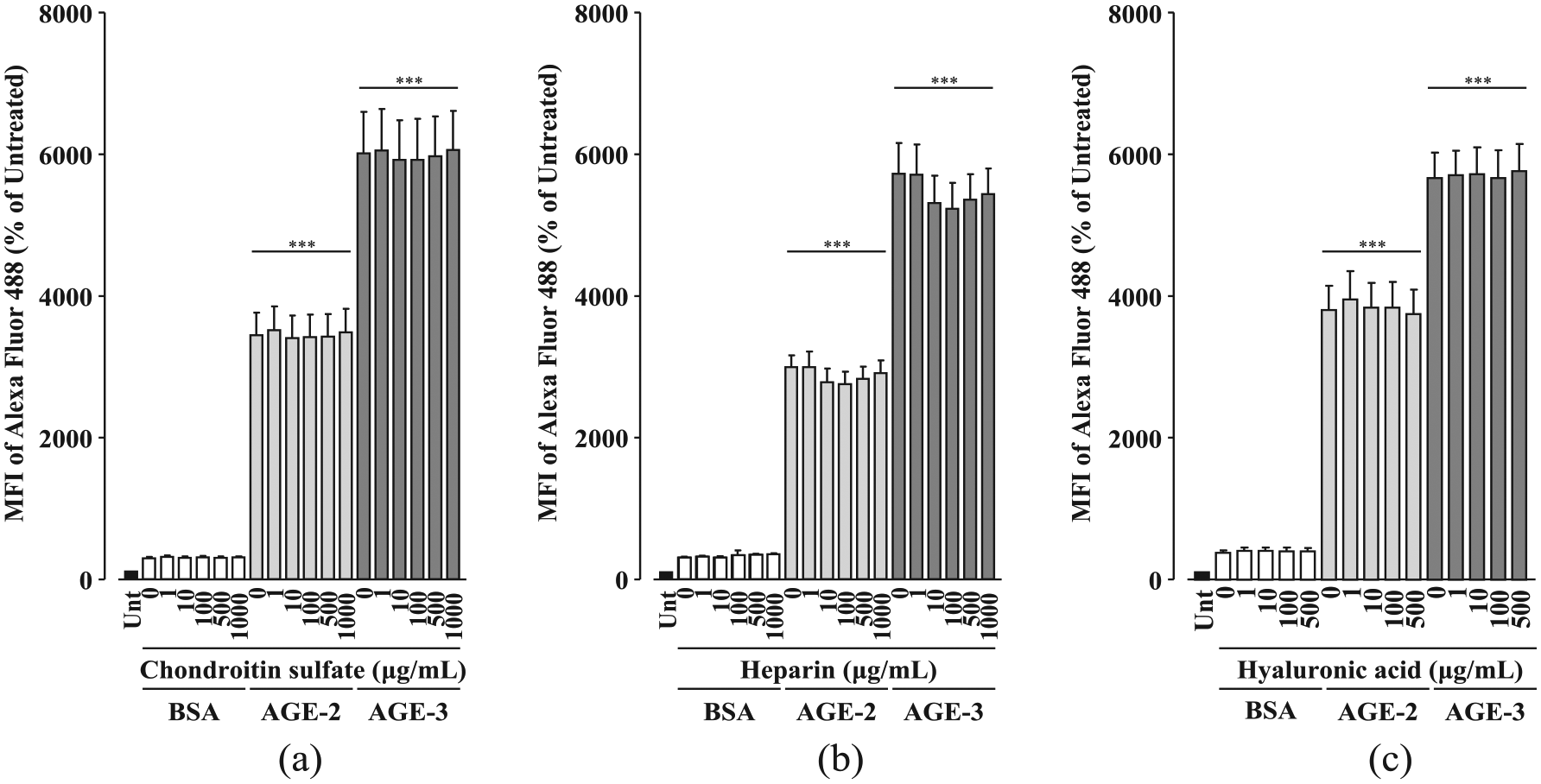
Effects of glycosaminoglycans on toxic AGE uptake by RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 500 µg/mL (hyaluronic acid) or 1 to 1000 µg/mL (chondroitin sulphate and heparin) in the presence and absence of fluorescence-labelled BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. Cellular uptakes of fluorescently labelled BSA, AGE-2 or AGE-3 were determined by flow cytometry. (a) Chondroitin sulphate (n = 5). (b) Heparin (n = 5). (c) Hyaluronic acid (n = 5). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
It is well known that fucoidan, carrageenan and dextran sulphate, but not chondroitin sulphate, heparin or hyaluronic acid, are ligands for SR-A.19,20 Accumulating structural evidence has indicated that SR-A ligands are essentially macromolecular and polyanionic compounds.20,21 Consistent with these findings, in contrast to HMW dextran sulphate, LMW dextran sulphate and non-charged dextran had no significant effect on toxic AGE uptake (Figure 2(b) and (c)). In addition, we investigated the activity of neocarrahexaose-24,41,3,5-tetra-O-sulphate, a degradation product of carrageenan with several biological activities including immunomodulatory and anti-tumour effects. 22 The molecular mass of neocarrahexaose-24,41,3,5-tetra-O-sulphate was low (MW: 1344.99) compared with that of carrageenan. neocarrahexaose-24,41,3,5-tetra-O-sulphate had no activity on toxic AGE uptake at increasing concentrations from 1 to 500 µg/mL (Figure 1(d)). Taken together, these results suggest that sulphated polysaccharides may be potential candidates for inhibitors of toxic AGE uptake via SR-A in macrophages.
Effects of sulphated polysaccharides on toxic AGE-induced upregulation of SR-A in RAW264.7 cells
Enhanced SR-A expression in human monocyte-derived macrophages was reported to be induced by exposure to high glucose. 23 Moreover, macrophages expressing high levels of SR-A were likely to form foam cells. 24 Clinically, increased expression of SR-A was observed in foam cells in atherosclerotic lesions. 25 Therefore, we investigated the effects of sulphated polysaccharides on AGE-induced upregulation of SR-A.
Fucoidan at 100 and 500 µg/mL and carrageenan at 500 µg/mL completely abolished AGE-2–induced upregulation of SR-A, while AGE-3–induced upregulation of SR-A was only suppressed by fucoidan at 500 µg/mL (Figure 4(a) to (c)). HMW dextran sulphate did not affect toxic AGE-induced upregulation of SR-A, whereas HMW dextran sulphate at 100–1000 µg/mL increased the SR-A levels in BSA-treated cells (Figure 5), indicating that dextran sulphate as well as toxic AGE induces upregulation of SR-A.
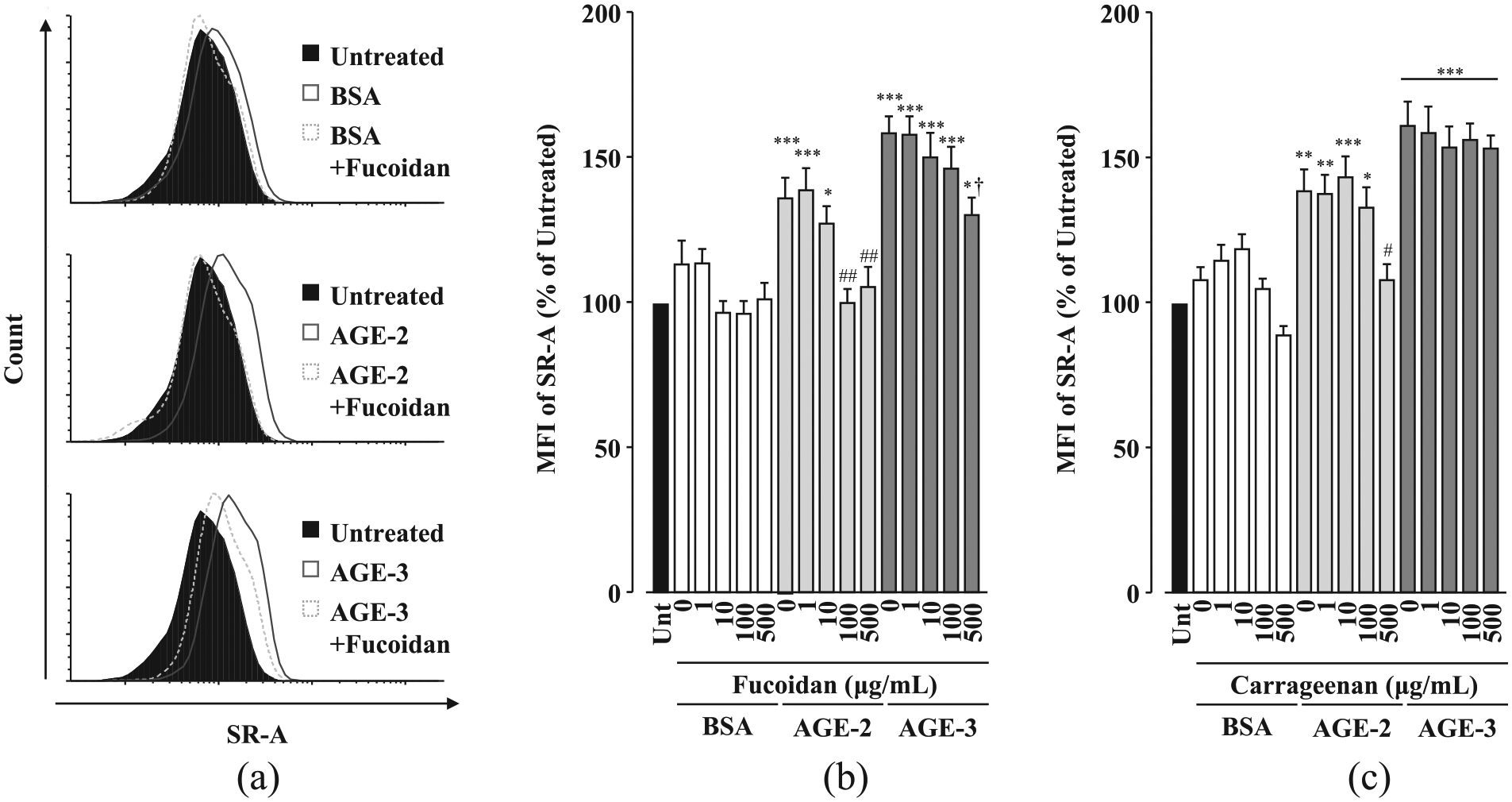
Effects of algae-derived sulphated polysaccharides on the toxic AGE-induced upregulation of SR-A in RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 500 µg/mL in the presence and absence of BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. The SR-A levels on the cell surface membrane were determined by flow cytometry. (a) Representative flow cytometric analysis of SR-A expression on RAW264.7 cells concomitantly treated with BSA, AGE-2 or AGE-3 and fucoidan (500 µg/mL). (b) Fucoidan (n = 8). (c) Carrageenan (n = 8). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
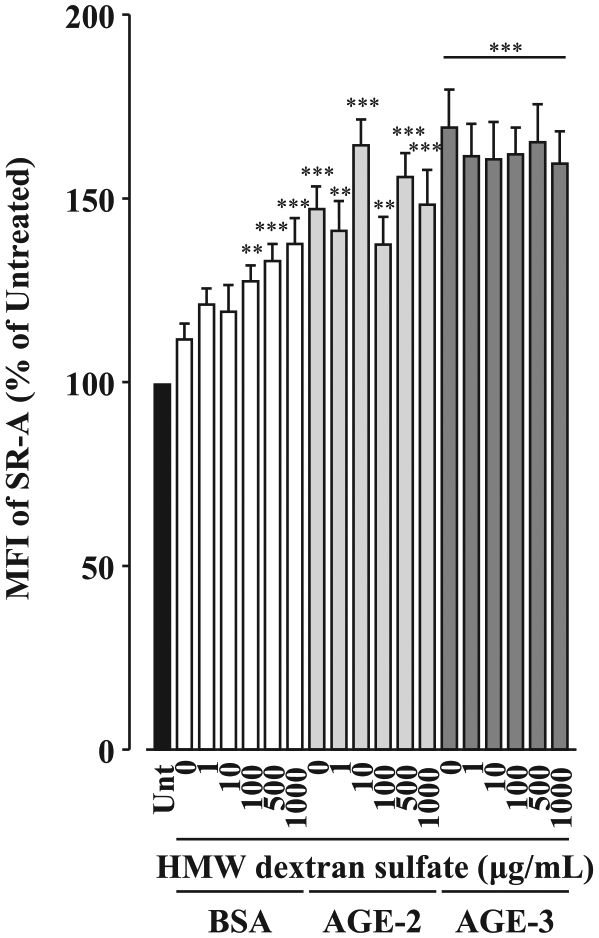
Effects of HMW dextran sulphate on toxic AGE-induced upregulation of SR-A in RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with HMW dextran sulphate at increasing concentrations from 1 to 1000 µg/mL in the presence and absence of BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. The SR-A levels on the cell surface membrane were determined by flow cytometry. (n = 7). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
Among the glycosaminoglycans that did not inhibit toxic AGE uptake in RAW264.7 cells, chondroitin sulphate at 1000 µg/mL decreased AGE-3–induced, but not AGE-2–induced, upregulation of SR-A (Figure 6(a)). Conversely, both heparin and hyaluronic acid had no effect on toxic AGE-induced upregulation of SR-A (Figure 6(b) and (c)).
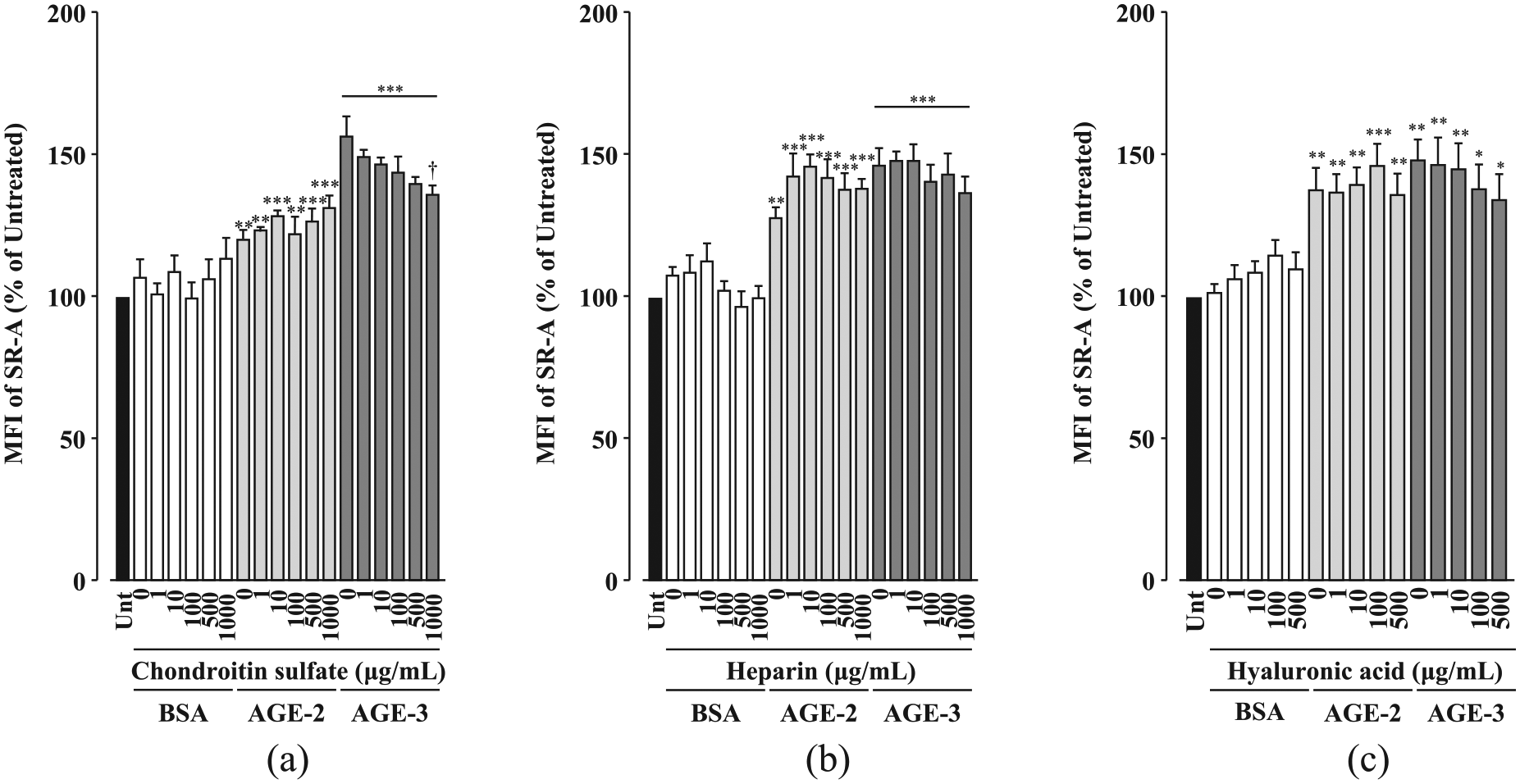
Effects of glycosaminoglycans on toxic AGE-induced upregulation of SR-A in RAW264.7 cells. RAW264.7 cells seeded at 1 × 105 cells/well were concomitantly treated with each sulphated polysaccharide at increasing concentrations from 1 to 500 µg/mL (hyaluronic acid) or 1 to 1000 µg/mL (chondroitin sulphate and heparin) in the presence and absence of BSA, AGE-2 or AGE-3 at 200 µg/mL for 1 h. The SR-A levels on the cell surface membrane were determined by flow cytometry. (a) Chondroitin sulphate (n = 5). (b) Heparin (n = 5). (c) Hyaluronic acid (n = 7). Data are expressed as means ± SEM and were analysed by one-way ANOVA followed by Tukey’s test.
Taken together, these results indicate that only fucoidan lead to inhibition of toxic AGE uptake mediated by the suppression of toxic AGE-induced upregulation of SR-A, while inhibition of toxic AGE uptake by carrageenan and dextran sulphate occur independently of SR-A expression. In addition, chondroitin sulphate attenuates AGE-3–induced upregulation of SR-A without inhibition of AGE-3 uptake.
Discussion
Previously, we investigated the uptake of toxic AGEs in RAW264.7 cells 9 with the following findings: AGE-2 and AGE-3 at concentrations ranging from 0.2 to 200 µg/mL enhanced their own uptake in a concentration-dependent manner. AGE-2 and AGE-3 increased the expression of several scavenger receptors including lectin-like oxidised LDL receptor 1 (LOX-1), haemoglobin scavenger receptor (CD163), SR-A and mannose receptor-1 (CD206), but not RAGE and class B scavenger receptor (CD36). Neutralising antibodies against SR-A, but not against RAGE, LOX-1, CD36, CD163 and CD206, were able to inhibit AGE-2 or AGE-3 uptake by almost 60% or 70%, respectively. Neutralising antibodies against SR-A also suppressed toxic AGE-induced increased expression of scavenger receptors. Similar to the anti-SR-A antibody, fucoidan attenuated the action of toxic AGEs. These findings suggest that SR-A may play a crucial role in toxic AGE uptake in RAW264.7 cells. On the other hand, several scavenger receptors are expressed in RAW264.7 cells and involved in the regulation of signal transduction pathways.26,27 As neutralising antibody against SR-A partially inhibit toxic AGEs uptake in RAW264.7 cells, other scavenger receptors may contribute to the toxic AGEs uptake in RAW264.7 cells.
In this study, we found that the effect on toxic AGE uptake and SR-A expression was differ among sulphated polysaccharides. Although the underlying mechanisms of effects by individual sulphated polysaccharides remain unclear, we discuss two possibility based on structural and chemical characteristics of sulphated polysaccharide.
First, we focus on sulphated polysaccharides used can be divided into the exogenous and endogenous sulphated polysaccharide. Exogenous sulphated polysaccharides including fucoidan, carrageenan and dextran sulphate is never present in mammalian tissues, while endogenous sulphated polysaccharides including chondroitin sulphate, heparin and hyaluronic acid are contained in mammalian tissues as glycosaminoglycan. Algae-derived fucoidan and carrageenan are essential components of the algal cell wall. The precise structure of fucoidan remains unclear, because it has a branching and heterogeneous backbone structure based on sulphated L-fucose and other sugars such as glucose, galactose, mannose, xylose and uronic acid (Figure 7). 28 The structure and components of fucoidan appear to depend on the species as well as environmental factors, suggesting that they may be associated with adaptation to environmental changes such as osmotic and mechanical stress. 29 Carrageenan is broadly divided into three classes, κ-, λ- and ι-, according to their gelation and position of sulphation. 30 The λ-carrageenan used in this study was composed of repeated disaccharides of galactose 2-sulphate and galactose 2, 6-disulphate (Figure 7). The artificially produced dextran sulphate used was composed of sulphated glucose with almost entirely α-(1 → 6) glycosidic linkages (Figure 7). Thus, the exogenous sulphated polysaccharides appeared to be mainly composed of single sugar components. In contrast, the endogenous sulphated polysaccharides including mammalian glycosaminoglycans were composed of regularly repeating disaccharide units with combinations of amino sugar, N-acetylglucosamine or N-acetylgalactosamine and uronic acid, glucuronic or iduronic acid (Figure 7). At this point, it remains unclear whether these structural differences between the exogenous and endogenous sulphated polysaccharides contributed to their inhibitory activity for uptake of toxic AGEs. Further study is needed.
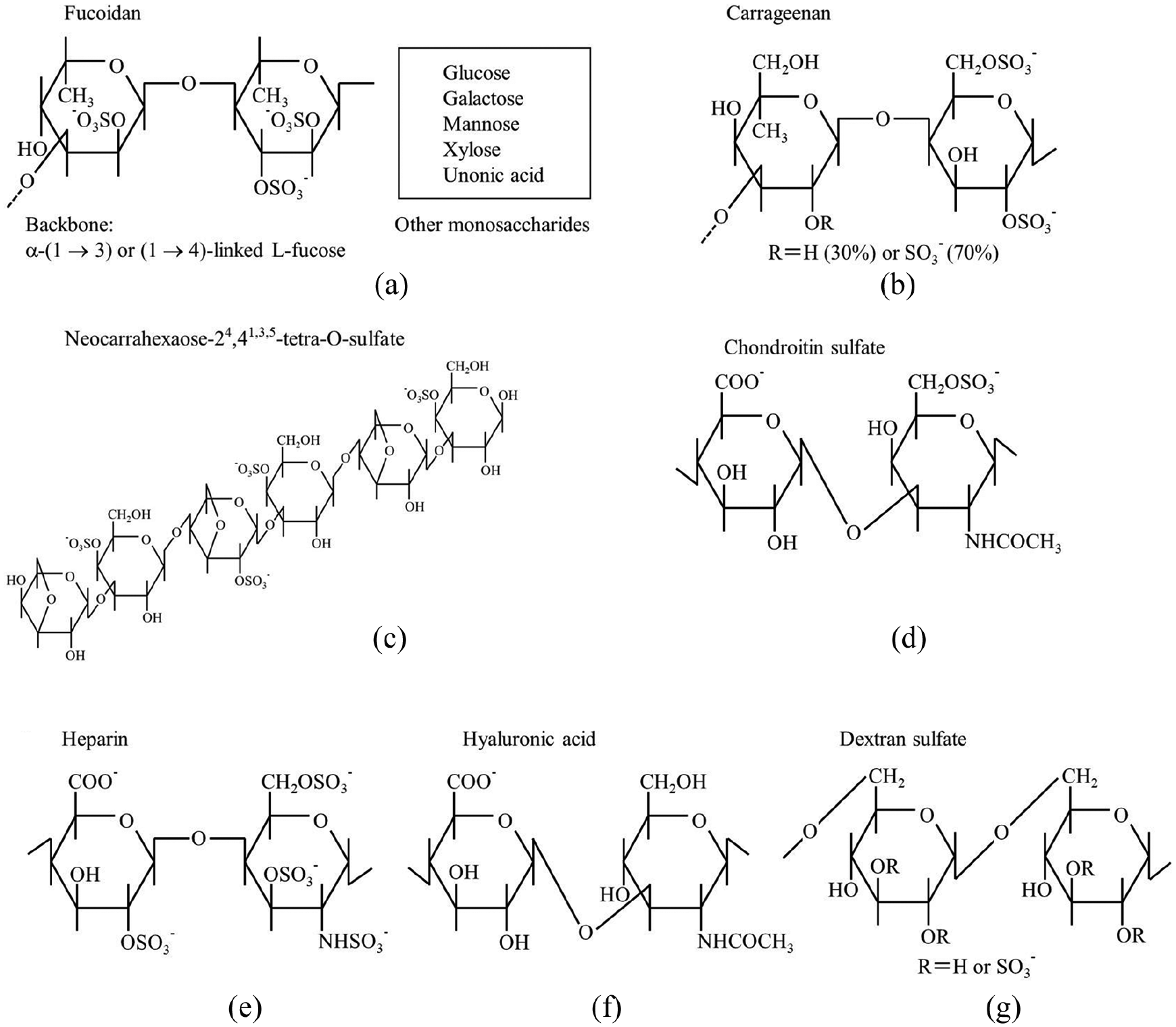
Primary structure motifs of sulphated and non-sulphated polysaccharides. (a) Fucoidan derived from F. vesiculosus. (b) Carrageenan. (c) Neocarrahexaose-24,41,3,5-tetra-O-sulphate. (d) Chondroitin sulphate. (e) Heparin. (f) Hyaluronic acid. (g) Dextran sulphate. Fucoidan derived from F. vesiculosus largely consists of L-fucopyranose units with α-(1 → 3) or (1 → 4) glycosidic linkages. λ-carrageenan is composed of galactose 2-sulphate and galactose 2,6-disulphate. Chondroitin sulphate C sodium salt is composed of repeating disaccharide units, glucuronic acid and N-acetylgalactosamine, consisting of α-(1 → 3) and β-(1 → 6) glycosidic linkages. Heparin is composed of repeating disaccharide units, uronic acid (glucuronic acid or iduronic acid) and glucosamine (N-acetylglucosamine or glucosamine), consisting of α-(1 → 4) glycosidic linkages. Hyaluronic acid is composed of repeating disaccharide units, glucuronic acid and N-acetylgalactosamine, alternately consisting of β-(1 → 3) and β-(1 → 4) glycosidic linkages. Dextran sulphate is only composed of sulphated glucose with almost entirely α-(1 → 6) glycosidic linkages.
Second, in polysaccharides, the degree of sulphation (DS; average number of sulphate groups per monosaccharide or disaccharide repeating unit) is an important parameter that determines their physiological activity. In general, desulphation of polysaccharides decreases their physiological activity. Exogenous sulphated polysaccharides tend to have more sulphate groups than endogenous sulphated polysaccharides, because of their need to interact with the extracellular matrix under high salt conditions. 31 The DS per disaccharide in exogenous sulphated polysaccharides fucoidan, carrageenan and dextran sulphate was reported to be 1.18, 1–3 and 1–4, respectively. 32 Meanwhile, the DS in endogenous sulphated polysaccharides chondroitin sulphate and heparin was 0.9 and 2.5, respectively, and hyaluronic acid had carboxyl groups instead of sulphate groups.33–35 Furthermore, exogenous sulphated polysaccharides had slightly higher molecular masses than endogenous sulphated polysaccharides excepted for hyaluronic acid. The molecular masses of fucoidan and carrageenan were reported to be 38.2 and 600–2000 kDa, respectively.32,36,37 For endogenous sulphated polysaccharides, the molecular masses of chondroitin sulphate, heparin and hyaluronic acid were reported to be 22, 15 and 200–2000 kDa, respectively.33–35 Taken together, sulphated polysaccharides such as fucoidan which have a certain level of molecular masses and sulphate may attenuate toxic AGE uptake. It still remains unclear how individual sulphated polysaccharide affect SR-A expression.
We have shown that algae-derived fucoidan, but not the other sulphated polysaccharides examined, had inhibitory activities on toxic AGE uptake and toxic AGE-induced upregulation of SR-A. In contrast, algae-derived carrageenan exerts inhibition of toxic AGE uptake without affecting the upregulation of SR-A by AGE-3. It has been reported that immunoreactivity in macrophages were different between fucoidan and carrageenan.17,38,39 Moreover, fucoidan exerted an anti-diabetic effect mediated by improving glucose intolerance and prevention of diabetes complications in animal model.40,41 Taken together, among the exogenous sulphated polysaccharides, fucoidan may be a beneficial compound for the treatment of diabetic complications.
Our study has a number of limitations. The first limitation is that we have investigated the effect of sulphated polysaccharides on toxic AGE uptake and SR-A expression using only flow cytometry. Cell surface expression of SR-A is crucially important to recognise and endocytose toxic AGE in RAW264.7 cells. We believe that cell surface expression of SR-A is suitable for analysis by flow cytometry. The second limitation is that our findings are obtained by a single murine cell line. It has been reported that inflammatory responses differ between RAW264.7 cells and human blood cell. 42 Further studies are needed to confirm the effect of sulphated polysaccharides using human blood cell.
Anti-AGE therapies involving suppression of AGE formation and injection of antagonists for AGE-binding receptors have recently attracted attention. Although their underlying mechanisms for impairment of physiological functions remain unclear, AGEs are considered to be toxic compounds. Macrophage is one of the target cells for toxicity induced by AGE and contributes to the pathogenesis of the diabetes and its complications.43,44 Sulphated polysaccharides are widely used as cosmeceutical, nutraceutical and pharmaceutical products and are easy to apply as medicine. Further studies are needed to determine whether sulphated polysaccharides can ameliorate toxic AGE-induced biological impairments such as vascular dysfunctions in vivo.
Key messages
Toxic advanced glycation end-products (AGEs) are endocytosed into RAW264.7 cells
Algae and artificial sulphated polysaccharides inhibit toxic AGE uptake
Glycosaminoglycans fail to inhibit toxic AGE uptake
Algae-derived fucoidan only inhibit toxic AGE-induced upregulation of SR-A
Fucoidan may be useful for treatment and prevention for AGE-related diseases
Supplemental Material
Supplementary_Figure – Supplemental material for A comparative study of sulphated polysaccharide effects on advanced glycation end-product uptake and scavenger receptor class A level in macrophages
Supplemental material, Supplementary_Figure for A comparative study of sulphated polysaccharide effects on advanced glycation end-product uptake and scavenger receptor class A level in macrophages by Takashi Nishinaka, Shuji Mori, Yui Yamazaki, Atsuko Niwa, Hidenori Wake, Tadashi Yoshino, Masahiro Nishibori and Hideo Takahashi in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors would like to thank the staff at the Central Research Facilities, Kindai University Faculty of Medicine, Center for Instrumental Analyses and Center for Morphological Analyses for their technical assistance. The authors thank Alison Sherwin, PhD, from Edanz Group (![]() ) for editing a draft of this manuscript.
) for editing a draft of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (C) Grant Numbers 18K06905 to H.T., 17K01881 to A.N., Early-Career Scientists Grant Numbers 18K15035 to T.N., Grants-in-Aid for Young Scientists (Start-up) Grant Numbers 17H07272 to Y.Y. and Japan Agency for Medical Research and Development (AMED) Grant Number 15LK0201014h0003 to M.N. Additional funding was received from Kindai University Grant-in Aid for Encouragement of Young Scientists Grant Number SR01 to T.N. H.T. was also supported by the Ministry of Education, Culture, Sports, Science and Technology (MEXT)-Supported Programme for the Strategic Research Foundation at Private Universities (S1411037).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
