Abstract
Objective:
Advanced glycation end products and their receptor – RAGE – in the adipose tissues contribute to metabolic derangements in fructose-fed rats. However, it remains unclear whether fructose could cause endothelial cell damage via the activation of AGE-RAGE.
Methods:
Intracellular advanced glycation end products were evaluated by dot blot analysis. Fructose-derived advanced glycation end products (Fruc-AGEs) were prepared by incubating bovine serum albumin with fructose for 8 weeks. Reactive oxygen species generation was measured using a fluorescent probe. Vascular cell adhesion molecule-1 gene expression was analysed by reverse transcription-polymerase chain reaction. Binding affinities of Fruc-AGEs to DNA-aptamer raised against Fruc-AGEs (Fruc-AGE-aptamer) or RAGE were measured with a quartz crystal microbalance.
Results:
Fructose increased the advanced glycation end product–specific fluorescence intensity in assay medium, while it stimulated intracellular formation of advanced glycation end products in human umbilical vein endothelial cells. Furthermore, 0.3 mM fructose for 4 days significantly increased reactive oxygen species generation and vascular cell adhesion molecule-1 gene expression in human umbilical vein endothelial cells. Fruc-AGE-aptamer, but not Control-aptamer, bound to Fruc-AGEs with Kd value of 5.60 × 10−6 M and dose-dependently inhibited the binding of Fruc-AGEs to RAGE. Moreover, Fruc-AGE-aptamer prevented the Fruc-AGE- and fructose-induced reactive oxygen species generation and vascular cell adhesion molecule-1 gene expression in human umbilical vein endothelial cells.
Conclusion:
This study suggests that fructose may elicit endothelial cell damage partly via the activation of AGE-RAGE axis.
Introduction
Consumption of large amounts of food and beverages containing added sugar has been associated with the prevalence of obesity and metabolic syndrome in humans.1,2 Sucrose and high-fructose corn syrup are two common types of added sugar, which are composed of glucose and fructose.1,2 Accumulating evidence has shown that fructose-rich diet evokes insulin resistance and hypertension in rodents, whereas consumption of fructose-sweetened, but not glucose-sweetened, beverages has induced metabolic derangements in overweight humans.1–5 These observations suggest that fructose may contribute to the development of metabolic syndrome independent of excess calorie intake.
Monosaccharides, such as glucose and fructose, can react non-enzymatically with amino-groups of proteins and lipids to form senescent macromolecular derivatives termed advanced glycation end products (AGEs).6–10 The formation and accumulation of AGEs have progressed under diabetic condition, which could cause the structural and functional alternation of proteins and lipids, thereby contributing to the development and progression of aging-related disorders, including atherosclerotic cardiovascular disease.6–10 Moreover, engagement of receptor for AGEs (RAGE) with AGEs elicits oxidative stress and inflammatory reactions in numerous kinds of cells, which could further potentiate the formation and accumulation of AGEs.6–10
We have previously found that activation of AGE-RAGE axis in the adipose tissues is critically involved in metabolic derangements in fructose-fed rats. 11 Indeed, compared with control rats, adipose tissue AGEs, RAGE and oxidative stress levels were increased in fructose-fed rats in association with elevated average fasting blood glucose, while adipocyte size grew larger with decreased gene expression of adiponectin, all of which were inhibited by the treatment with high-affinity DNA-aptamer directed against AGEs. 11 However, it remains unclear whether fructose could cause endothelial cell damage via the activation of AGE-RAGE axis. In this study, we have addressed the issue.
Methods
Materials
Bovine serum albumin (BSA; essentially fatty acid free) was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Preparation of fructose-derived AGEs (Fruc-AGEs)
Fruc-AGE-BSA was prepared as described previously.
12
In brief, BSA (25 mg/mL) was incubated under sterile conditions with 0.5 M fructose in 0.2 M NaPO4 buffer (pH 7.4) at 37°C for 8 weeks. Then, unincorporated fructose was removed by dialysis with phosphate-buffered saline. Control non-glycated BSA was incubated in the same conditions except for the absence of
Preparation of DNA-aptamers raised against Fruc-AGEs (Fruc-AGE-aptamers)
A random combinatorial single-stranded DNA library with normal phosphate ester backbone oligonucleotides (80-mer) was synthesized, and selection of Fruc-AGE-aptamers was performed using systemic evolution of ligands by exponential enrichment (SELEX) as described previously. 13
Cells
Human umbilical vein endothelial cells (HUVECs) obtained from Lonza Group Ltd. (Basel, Switzerland) were maintained in endothelial basal medium supplemented with 2% foetal bovine serum, 0.4% human fibroblast growth factor-B, 0.1% heparin, 0.1% human epidermal growth factor and 0.04% hydrocortisone according to the manufacturer’s recommendation. Experiments were carried out in a medium lacking epidermal growth factor and hydrocortisone (assay medium).
Measurement of AGE-specific fluorescence intensity
Cell culture assay medium was incubated with 0.3 or 0.6 mM fructose at 37°C for 4 days. Then, the fluorescence intensity at 460 nm, with an excitation wavelength of 355 nm, was measured using a plate reader (PerkinElmer Inc., Waltham, MA, USA).
Dot blot analysis
HUVECs were treated with 0.3 mM fructose or mannitol for the indicated time periods. Then, proteins were extracted from HUVECs, and AGE levels were evaluated by dot blot analysis using monoclonal antibody raised against AGEs as described previously.11,14 Colour intensity was analysed by microcomputer-assisted image J.
Binding affinities of Fruc-AGEs to Fruc-AGE-aptamer to RAGE
The binding affinities of Fruc-AGEs to Control-aptamer, Fruc-AGE-aptamer or extracellular AGE-binding v-domain of RAGE (vRAGE) were measured using sensitive 27-MHz quartz crystal microbalance (QCM) (Affinix Q; Initium Inc., Tokyo, Japan) as described previously. 13
Measurement of reactive oxygen species generation
Reactive oxygen species (ROS) generation was measured using a fluorescent probe – carboxy-H2DFFDA (Thermo Fisher Scientific, Waltham, MA, USA) – as described previously. 13
Real-time reverse transcription-polymerase chain reactions
HUVECs were treated with 0.3 mM fructose, 0.3 mM mannitol, BSA or Fruc-AGEs in the presence or absence of Fruc-AGE-aptamer or Control-aptamer for the indicated time periods. Then, total RNA was extracted with NucleoSpin RNA Plus (Takara Bio, Shiga, Japan). Quantitative real-time reverse transcription-polymerase chain reaction (RT-PCR) was performed using Assay-on-Demand and TaqMan 5 fluorogenic nuclease chemistry (Applied Biosystems, Foster city, CA, USA) according to the manufacturer’s recommendation. IDs of primers for human vascular cell adhesion molecule-1 (VCAM-1), GAPDH and β-actin gene were Hs01003372_m1 (Figures 1(d) and 2(d))/Hs00365486_g1 (Figure 3(b)), Hs99999905_m1 and Hs01060665_g1, respectively.
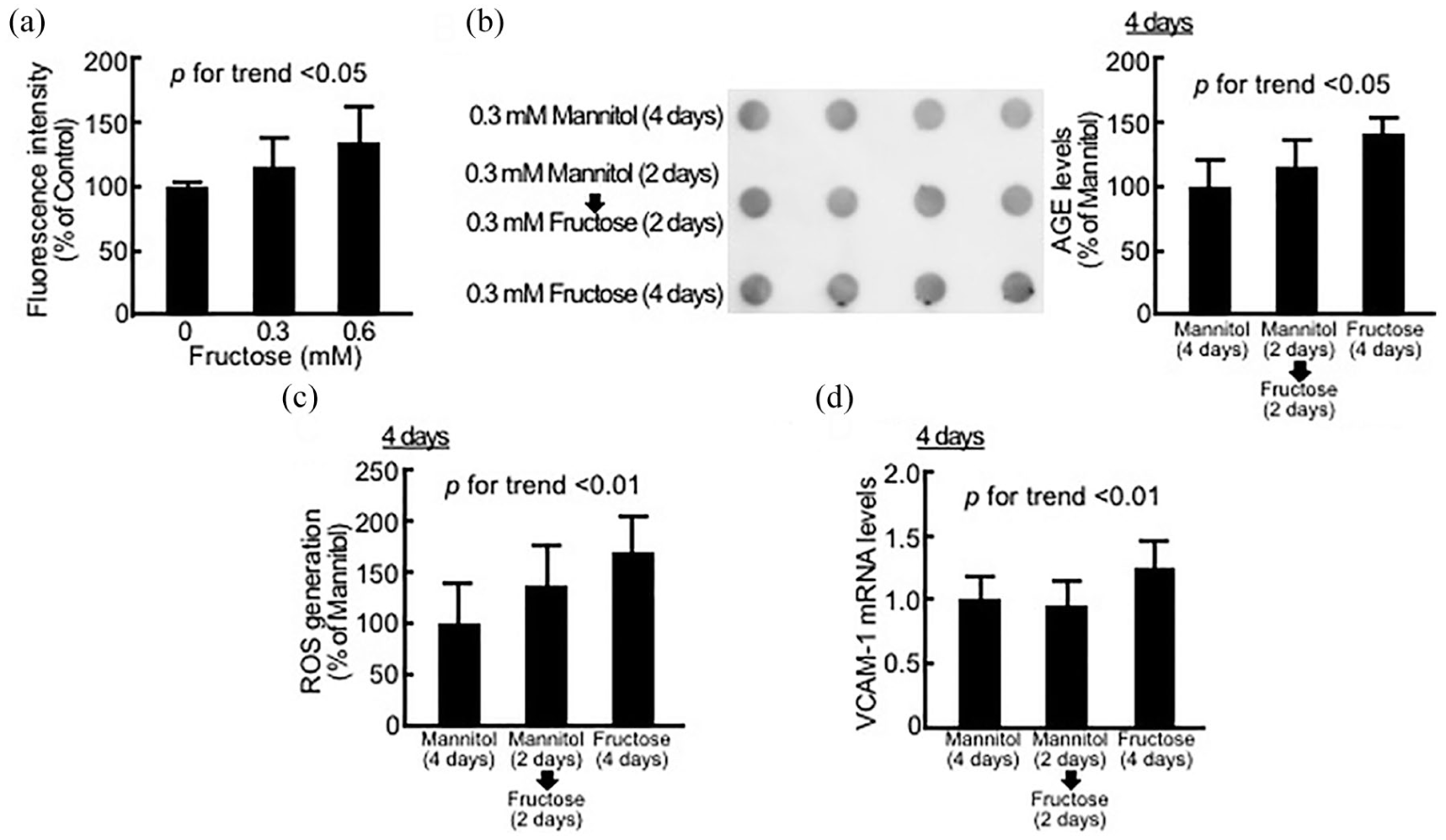
Effects of fructose on fluorescence intensity in assay medium (a), AGE formation (b), superoxide generation (c) and VCAM-1 gene expression (d) in HUVECs. (a) Fluorescence intensity was measured, N = 6 per group. (b) AGE levels in HUVECs were evaluated by dot blot analysis – right panel shows the dot blot membrane and the left panel shows the quantitative data, N = 4 per group. (c) HUVECs were incubated with 10-µM carboxy-H2DFFDA for 30 min and then fluorescence intensity was measured, N = 6 per group. (d) Total RNAs were transcribed and amplified by RT-PCR. Data were normalized by the intensity of GAPDH-derived signals and then related to the values with mannitol, N = 9 per group.
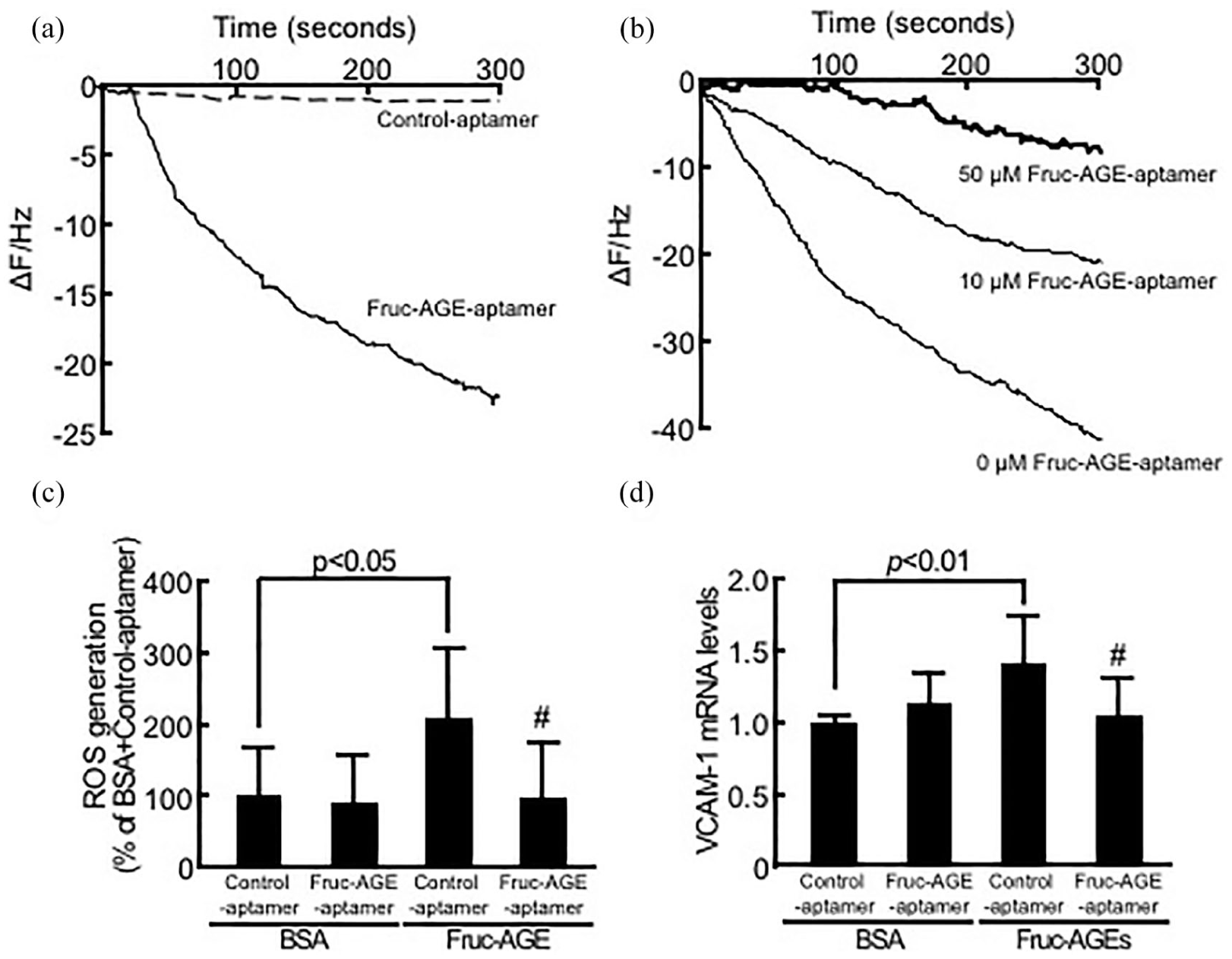
Binding affinities of Fruc-AGEs to Fruc-AGE-aptamer (a) or vRAGE (b) and effects of Fruc-AGE-aptamer on ROS generation (c) and VCAM-1 gene expression (d) in Fruc-AGE-exposed HUVECs. (a) Binding affinities of Fruc-AGE-aptamer or Control-aptamer to Fruc-AGEs immobilized on a QCM surface. (b) Binding affinities of Fruc-AGEs to vRAGE in the presence or absence of Fruc-AGE-aptamer. Representative time courses of the frequency decreases of bound Fruc-AGE-aptamer and Control-aptamer (a) or Fruc-AGEs (b) on the QCM surface, N = 3 per group. (c) HUVECs were incubated with 10-µM carboxy-H2DFFDA for 1 h and then treated with or without Fruc-AGEs for 45 min. ROS generation was evaluated by fluorescence intensity, N = 3 per group. (d) HUVECs were treated with or without Fruc-AGEs for 4 h. Then, RT-PCR was performed. Data were normalized by the intensity of β-actin-derived signals and then related to the values with non-glycated BSA plus Control-aptamer, N = 4 per group.
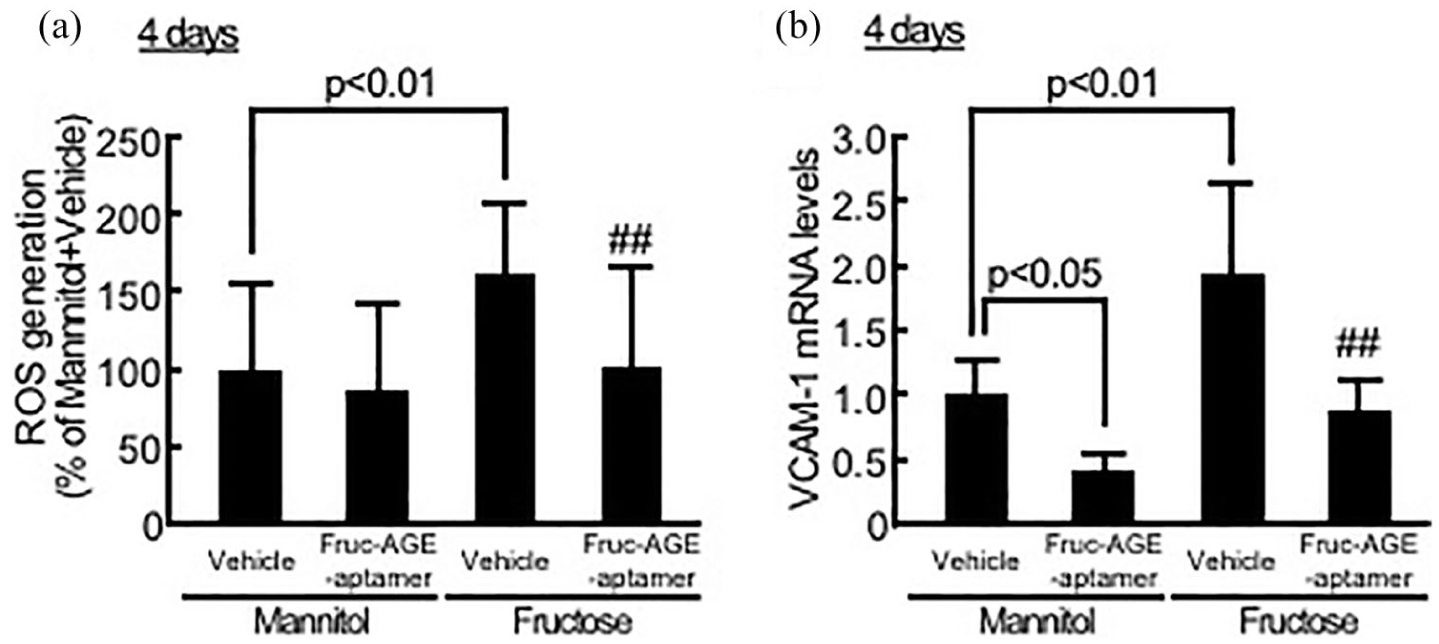
Effects of Fruc-AGE-aptamer on ROS generation (a) and VCAM-1 gene expression (b) in fructose-exposed HUVECs. (a) HUVECs were treated with 0.6 mM fructose or mannitol for 2 days. Then, medium was changed, and cells were treated under the same conditions for further 2 days. Assay medium used for medium change was pre-incubated with 0.6 mM fructose or mannitol for 2 days. After incubating the cells with 40-µM carboxy-H2DFFDA for 15 min, ROS generation was evaluated by fluorescence intensity, N = 6 per group. (b) HUVECs were treated with 0.6 mM fructose or mannitol. Medium was changed every day, and cells were treated under the same conditions for further 3 days. Assay medium used for medium change at days 1, 2 and 3 was pre-incubated with 0.6 mM fructose or mannitol for 1, 2 and 3 days, respectively. Then, RT-PCR was performed. Data were normalized by the intensity of GAPDH-derived signals and then related to the values with mannitol plus vehicle, N = 3 per group.
Statistical analysis
All values were presented as mean ± standard deviation. One-way analysis of variance (ANOVA) or Tukey’s honestly significant difference (HSD) test was performed for multiple statistical comparisons; p < 0.05 was considered significant.
Results
As shown in Figure 1(a) and (b), 4-day treatment with fructose dose-dependently increased the AGE-specific fluorescence intensity in assay medium, while 0.3 mM fructose time-dependently increased the formation of intracellular AGEs in HUVECs compared with 0.3 mM mannitol, an osmotic control. Furthermore, 0.3 mM fructose for 4 days significantly increased ROS generation and VCAM-1 gene expression in HUVECs (Figure 1(c) and (d)).
Sequences of Fruc-AGE-aptamer and Control-aptamer were 5′-tTTggcgggAggcgAgcggggAATggcgggTgTgggggAA-3′ and 5′-aTcgAccTggAggcgAgcAgcTcggATccAgTcgcgTgAg-3′, respectively, where 5′ side of phosphorothioate nucleotides are indicated as capital letters. As shown in Figure 2(a) and (b), Fruc-AGE-aptamer, but not Control-aptamer, bound to Fruc-AGEs with Kd value of 5.60 ± 2.35 × 10−6 M and dose-dependently inhibited the binding of Fruc-AGEs to vRAGE. Moreover, Fruc-AGE-aptamer completely prevented the Fruc-AGE-induced ROS generation and VCAM-1 gene expression in HUVECs (Figure 2(c) and (d)). Fruc-AGE-aptamer alone did not affect ROS generation or VCAM-1 mRNA levels in HUVECs exposed to non-glycated BSA.
As shown in Figure 3(a) and (b), Fruc-AGE-aptamer also significantly inhibited the fructose-evoked ROS generation and VCAM-1 mRNA up-regulation in HUVECs. Fruc-AGE-aptamer decreased VCAM-1 mRNA levels in mannitol-exposed HUVECs as well (Figure 3(b)).
Discussion and conclusion
As far as we know, there were two papers to show that fructose evoked inflammatory and thrombotic reactions in cultured endothelial cells.15,16 However, there is some controversy about the mechanism of action of fructose in endothelial cells. Indeed, one paper showed that 0.25–2.5 mM fructose for 24 h increased intercellular cell adhesion molecule-1 expression in human aortic endothelial cells via inhibition of nitric oxide generation, which was independent of NF-κB activation, 15 whereas the other paper reported that 1 mM, but not 0.25 mM, fructose for 6 h induced thrombotic reactions in HUVECs through the activation of NF-κB via ROS. 16 In this study, we examined the effects of fructose on HUVECs by focusing on the role of AGE-RAGE axis in fructose-induced endothelial cell damage because (1) fructose reacts 10 times more rapidly with proteins than glucose to form AGEs; 17 (2) Fruc-AGEs exist in human serum, and the levels are increased in diabetic patients; 12 (3) Fruc-AGEs induce cellular senescence in HUVECs; 18 and (4) fructose-rich diet increases the formation of AGEs, RAGE expression and oxidative stress generation in the adipose tissues of rats and subsequently induces metabolic derangements, all of which are attenuated by the treatment with DNA-aptamer raised against AGEs. 11
We found here that fructose treatment for 4 days not only enhanced the formation of AGEs in culture medium but also increased the intracellular AGE levels in HUVECs. Furthermore, Fruc-AGEs mimicked the effects of fructose on ROS generation and VCAM-1 gene expression in HUVECs. Since Fruc-AGE-aptamer blocked the binding of Fruc-AGEs to vRAGE and inhibited the deleterious effects of Fruc-AGEs and fructose on HUVECs, this study suggests that relatively long-term exposure to fructose may elicit endothelial cell injury partly via the activation of fructose-derived AGEs and their receptor RAGE axis.
Basal plasma concentration of fructose in healthy humans is about 0.05–0.1 mM, and its levels are increased to 0.25–0.8 mM after oral administration of 18–100 g fructose.15,19 So, consumption of 500 mL soft drink, which contains 27.5 g fructose, is estimated to increase plasma levels of fructose to 0.38 mM. Moreover, Fruc-AGEs are present in appreciable levels in beverages containing high-fructose corn syrup. 20 These observations suggest that regular consumption of large amounts of beverages containing fructose may cause endothelial cell damage partly via the activation of AGE-RAGE system. However, extrapolation of in vitro results involving heavily glycated proteins to the in vivo clinical situation should be undertaken with caution. Therefore, further longitudinal study is needed to examine whether fructose-rich diet increases plasma levels of Fruc-AGEs in humans and whether Fruc-AGE-aptamer could ameliorate endothelial cell damage in animals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported, in part, by Grants-in-Aid for Scientific Research (grant number: 17K08968) (S.i.Y.) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
