Abstract
Diabetes mellitus is a salient risk factor for the development of cardiovascular diseases (CVDs). Part of this risk is associated with the presence of advanced glycation end products (AGEs), which have been shown to up-regulate platelet or endothelial cell inflammatory and thrombogenic responses that are associated with CVDs. However, platelets perform mechanisms that alter endothelial cell inflammatory and thrombogenic responses, and endothelial cells perform similar mechanisms on platelets. Thus, our goal was to evaluate platelet and endothelial cell inflammatory and thrombogenic reactions that AGEs elicit during concurrent exposure. Endothelial cells were incubated with AGEs for 5 days, after which platelets were added. A time course for CVD inflammatory and thrombogenic responses was quantified as a function of extent of glycation. In general, the presence of platelets reduced AGE-induced endothelial cell responses associated with CVD progression and the presence of endothelial cells reduced platelet adhesion and activation responses, as compared with individual exposures. In general, the presence of irreversibly glycated albumin promoted CVD development to a greater extent than reversibly glycated albumin. This suggests that under diabetic conditions, platelets and endothelial cells can negatively feedback on each other, likely via enhanced adhesion, to elicit a reduced response associated with CVD progression.
Introduction
Cardiovascular diseases (CVDs) remain to be the leading cause of death in the Western world. Coronary heart disease, which is the most common CVD, leads to nearly 400,000 deaths per year and over US$100 billion in health-care costs.1,2 Diabetes is considered a major risk factor for the development of CVDs, and it is thought to instigate disease progression through the presence of advanced glycation end products (AGEs) in the vasculature. AGEs are proteins (or other molecules) that become glycated after the long-term exposure to sugars. However, the role of glycation extent on CVD development remains unclear. Early glycation (up to 2-week exposure), which has not passed through the Amadori rearrangement, is generally thought to be reversible, whereas late glycation (glycation time of more than 6 weeks), which passes through the Amadori rearrangement, is generally thought to be irreversible. We aimed here to determine the role of glycation extent on modulating endothelial cell and platelet functions that are common during CVDs and how the interaction of these two cell types may alter disease progression.
Numerous reports document the role of AGEs on endothelial cell functions observed during CVDs. AGEs can interact with endothelial cells via the receptor for advanced glycation end products (RAGE), which induces the generation of reactive oxygen species and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). 3 The up-regulation of these products has been shown to enhance the expression of adhesion molecules [such as intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1)] and pro-inflammatory cytokines [interleukin-1 (IL-1), interleukin-6 (IL-6)] and reduce endothelial cell anti-thrombotic properties and the bioavailability of endothelium-derived nitric oxide; each of which is associated with endothelial dysfunction. It is well accepted that an increasing concentration of AGEs has a more deleterious effect on endothelial cells; however, the role of glycation extent has not been well characterized. In the past, we have aimed to determine the role of glycation extent on endothelial cell functions associated with CVDs and have in general shown that reversibly glycated AGEs do not elicit a significant effect on endothelial cell responses associated with CVDs as compared with paired non-glycated samples, but irreversibly glycated AGEs are consistently detrimental to endothelial cell functions. This is especially prevalent for endothelial cell metabolic activity, viability, expression of ICAM-1, tissue factor and thrombomodulin and shear-induced cytoskeletal dynamics.4–6 Here, we aimed to extend this work by including possible cross-talk between platelets and endothelial cells.
Platelets are a salient cell for the progression and initiation of CVDs. Platelets from diabetic patients and healthy platelets exposed to AGEs have been shown to have altered functions. For instance, platelets from patients with diabetes mellitus aggregate to a greater extent when antagonized with serotonin. 7 Healthy platelets exposed to AGEs are more susceptible to aggregation induced by multiple agonists.8,9 Platelet shear-induced activation and the expression of CD41a and CD62P were all enhanced after exposure to AGEs. Importantly, our work again showed that the glycation extent plays an important role in the AGE-induced platelet functional changes. 9 Here, we aimed to determine how the presence of endothelial cells can alter platelet functional changes induced by AGEs.
As discussed, there has been a significant amount of work that aimed to elucidate the effects of AGEs on endothelial cells and platelets. However, in diabetics, the AGE population is a heterogeneous mixture of reversibly glycated and irreversibly glycated proteins, and thus, it is important to elucidate the role of each of these AGEs on cellular functions. In previous work, we have examined the role of glycation extent on platelet or endothelial cell function and in general observed an increased thrombotic/inflammatory response after irreversibly glycated albumin exposure but not for reversibly glycated albumin.4–6,9 Here, we examine the inflammatory and thrombogenic potential of endothelial cells and platelets concurrently exposed to AGEs glycated to various extents. We hypothesized that irreversibly glycated AGEs would elicit a more robust response than paired non-glycated proteins and reversibly glycated AGEs. Additionally, the presence of platelets would accelerate the endothelial cell response, whereas the presence of endothelial cells would accelerate the platelet response. We also hypothesized that these responses would be a function of time; with a greater incubation duration, the extent of endothelial cell or platelet activation would be greater.
Methods
Synthesis of AGEs
Bovine serum albumin (BSA) was glycated as previously reported by us without modifications 9 from the same batch as previously reported. 10 The albumin was chemically modified with an average of 6 glucose molecules after 2 weeks of glycation, 25 glucose molecules after 6 weeks of glycation and 30 glucose molecules after 8 weeks of glycation. To cell samples, 2 mg /mL glycated (termed AGE) or non-glycated albumin (termed BSA) was added. All samples were compared to a paired sample with no added albumin (termed No Added BSA).
Endothelial cell culture and platelets
Human umbilical vein endothelial cells (HUVECs) were purchased from ScienCell Research Laboratories (Carlsbad, CA, USA) as passage-one cells and were used between passages three and six for all experiments. Cells were maintained in endothelial cell growth media with added endothelial cell growth supplement (1×), foetal bovine serum (5%), penicillin (10 U/mL) and streptomycin (10 µg/mL, all from ScienCell Research Laboratories). HUVECs were passaged with trypsin at confluence, at which time either non-glycated or glycated albumin was added to the cells for 5 days, at a final concentration of 2 mg /mL. 10 Fresh platelet-rich plasma (PRP) pheresis packs (anti-coagulated with 0.32% sodium citrate) were obtained from Oklahoma Blood Institute (Oklahoma City, OK, USA). Washed platelets were obtained via centrifugation of PRP at 1100 × g for 9 min. The platelet pellet was resuspended in a 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) platelet buffer as previously reported 9 and added to the culture immediately after counting for up to 4 h. For statistical purposes, endothelial cell seeding density was maintained at ~1000 cells/cm2 per experiment and the platelet concentration was maintained at 200,000 platelets/µL.
Endothelial cell metabolic activity
A standard 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) assay was conducted to document the metabolic activity via mitochondrial dehydrogenase of endothelial cells, as previously reported without modifications 6 (Sigma–Aldrich). The resulting solution, after formazan crystals were dissolved, was gently mixed, and absorbance at 630 nm was quantified using a microplate reader (BioTek ELx800). For statistical analysis, all metabolic activity data were normalized by the metabolic activity of endothelial cells exposed to No Added BSA and without exogenous platelets. Additionally, a standard live/dead cell cytotoxicity assay was used to confirm the MTT data.
Endothelial cell inflammatory and thrombotic markers
Enzyme-linked immunosorbent assay (ELISA) was used to quantify the surface expression of ICAM-1, thrombomodulin and tissue factor. HUVECs were washed with phosphate buffered saline (PBS) and fixed in 0.5% glutaraldehyde for 15 min. Glutaraldehyde was removed with two PBS washes. A volume of 100 mM glycine + 1% BSA (30 min, room temperature) was used for blocking/neutralizing. Cells were washed and then incubated with 5 µg/mL of the specific primary antibodies (Ancell Corporation, Bayport, MN, USA) for 60 min. Cells were washed and then incubated with 10 µg/mL of an alkaline phosphatase–conjugated secondary antibody for 60 min. Binding was detected with p-nitrophenyl phosphate (pNPP) treatment, which detects alkaline phosphatase activity. Colour development was read at 405 nm in a microplate reader. For statistical analysis, all ELISA data were normalized by the surface expression of the particular marker of endothelial cells exposed to No Added BSA and without exogenous platelets.
Endothelial cell connexin-43/caveolin-1 expression
Immunofluorescence microscopy was used to quantify the expression and localization of connexin-43 and caveolin-1. For connexin-43 and caveolin-1 dual staining, after exposure conditions, HUVECs were washed in PBS and fixed in 1.5% glutaraldehyde for 15 min. After washing, PBS + 1% BSA was added for blocking/neutralizing for 30 min. Cells were washed and incubated with 0.6 µg/mL anti-human connexin-43 (Abcam, Cambridge, MA, USA) and 2 µg/mL anti-human caveolin-1 (Invitrogen, Carlsbad, CA, USA) for 60 min (room temperature). Cells were washed and incubated with two fluorescent secondary antibodies, each at a concentration of 5 µg/mL for 60 min (room temperature). Finally, cells were stored in PBS + 1% BSA for imaging. For statistical analysis, all data were normalized by the surface expression of endothelial cells exposed to No Added BSA and without exogenous platelets.
Platelet adhesion, aggregation and activation
Timed samples of washed platelets were assayed for the surface expression of CD41a [glycoprotein IIb (GPIIb), aggregation], CD31 [platelet–endothelial cell adhesion molecule (PECAM), adhesion], phosphatidylserine (activation) and CD62P (P-selectin, activation) to determine a possible mechanism for endothelial cell/platelet functional changes and the extent of platelet functional changes. In previous work, our group and others have correlated the expression of CD41a to platelet aggregation responses and CD62P to platelet activation responses.9,11 PE-anti-CD41 (1:50, Ancell Corporation), PE-anti-CD31 (1:50, Ancell Corporation), annexin V (1:50, Invitrogen) and fluorescein isothiocyanate (FITC)-anti-CD62P (1:50, Ancell Corporation) were incubated with platelet samples for 30 min at 37 °C in the dark. Platelets were then diluted 1:5 in platelet buffer and immediately analysed on a flow cytometer (Accuri C6) for receptor expression data. All mean fluorescence were normalized to IgG1 Isotype Control from Murine Myeloma (MOPC) (negative control) and sonicated (30 s) cells (positive control), for statistical purposes.
Statistical analysis
All data were normalized as described above. Normalized data were averaged within one trial, and the averaged independent data were used for statistical analyses. For all experiments, a total of at least four to five independent experiments were conducted. Data were analysed for significance using a two-way analysis of variance (ANOVA) (grouped by duration and additive) with the least-squares means (LSMEANS) post hoc test to determine differences between pre-determined comparisons. All statistical analyses were conducted in SAS (v9.3) with α = 0.05.
Results
Endothelial cell metabolic activity
The metabolic activity of endothelial cells exposed to albumin glycated for various durations in the presence of platelets was quantified as a function of exposure duration to platelets (Figure 1). Interestingly, the metabolic activity, as quantified by the activity of mitochondrial dehydrogenase, was not dependent on the presence of glycated albumin or platelets or exposure time. In parallel, the cell viability and cell density were not altered as a function of time, with the presence of platelets or the presence of glycated albumin (data not shown).
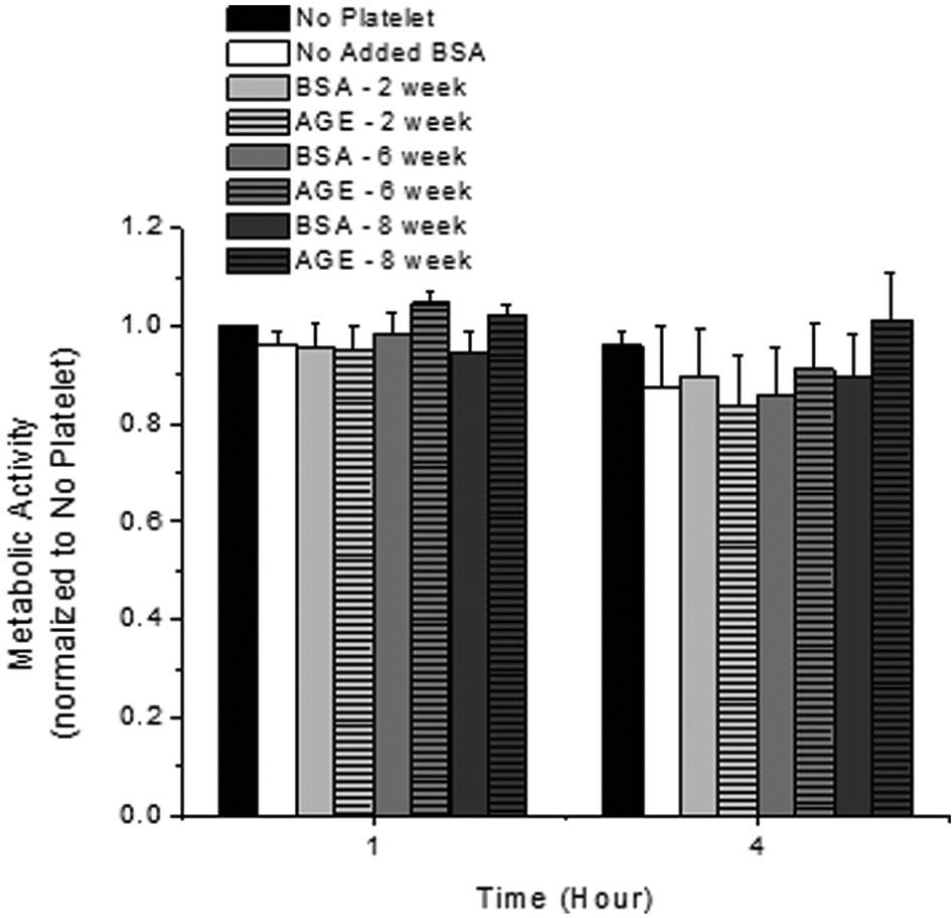
Endothelial cell metabolic activity as a function of incubation duration with platelets and glycated albumin additive. All data are reported as the mean ± standard error of the mean for four independent experiments.
Endothelial cell inflammatory/thrombotic markers
The surface expression of endothelial pro-inflammatory and thrombotic markers was quantified in the presence of platelets as a function of time and presence of glycated albumin glycated to various extents (Figure 2). Decreased thrombomodulin and increased tissue factor expression are associated with an increased thrombotic potential. A decreasing trend for thrombomodulin expression was observed during the exposure to irreversibly glycated albumin (Figure 2(a)), but this was not a function of time. Similarly, an increasing trend for tissue factor expression was observed during the exposure to irreversibly glycated albumin (Figure 2(b)), and this was statistically significant for endothelial cells exposed to albumin glycated for 8 weeks for 1 h. Although there was not a significant change in the thrombotic properties of endothelial cells exposed to glycated albumin as a function of time, the inflammatory potential, as quantified by the surface expression of ICAM-1, was significantly enhanced for endothelial cells that were exposed to irreversibly glycated albumin (Figure 2(c)), as compared with the paired non-glycated samples and the negative control conditions. Interestingly, expression of ICAM-1 was not a function of exposure duration.
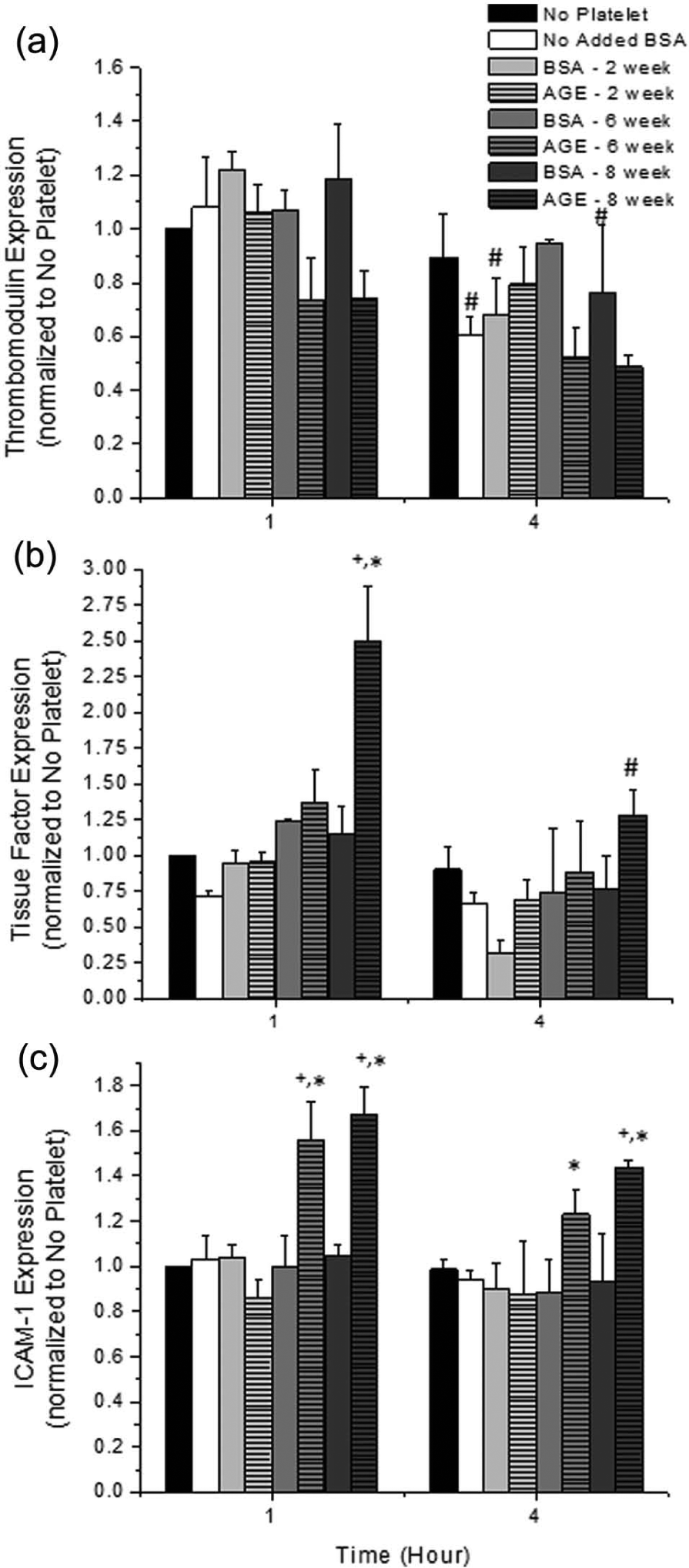
Surface expression of (a) endothelial cell thrombomodulin, (b) tissue factor and (c) ICAM-1 in the presence of platelets as a function of incubation duration with platelets and glycated albumin additive. All data are reported as the mean ± standard error of the mean for at least four independent experiments.
Connexin-43 and caveolin-1 expression
The endothelial cell surface expression of connexin-43 and caveolin-1 was quantified to determine the extent of stable communicative network formation under these conditions (Figure 3). With enhanced communicative network formation, one would expect an increase in CVD progression. We observed that both connexin-43 and caveolin-1 expression increased as a function of glycation extent; with irreversibly glycated albumin, there was a significant up-regulation of both of these markers. This was not a function of time.
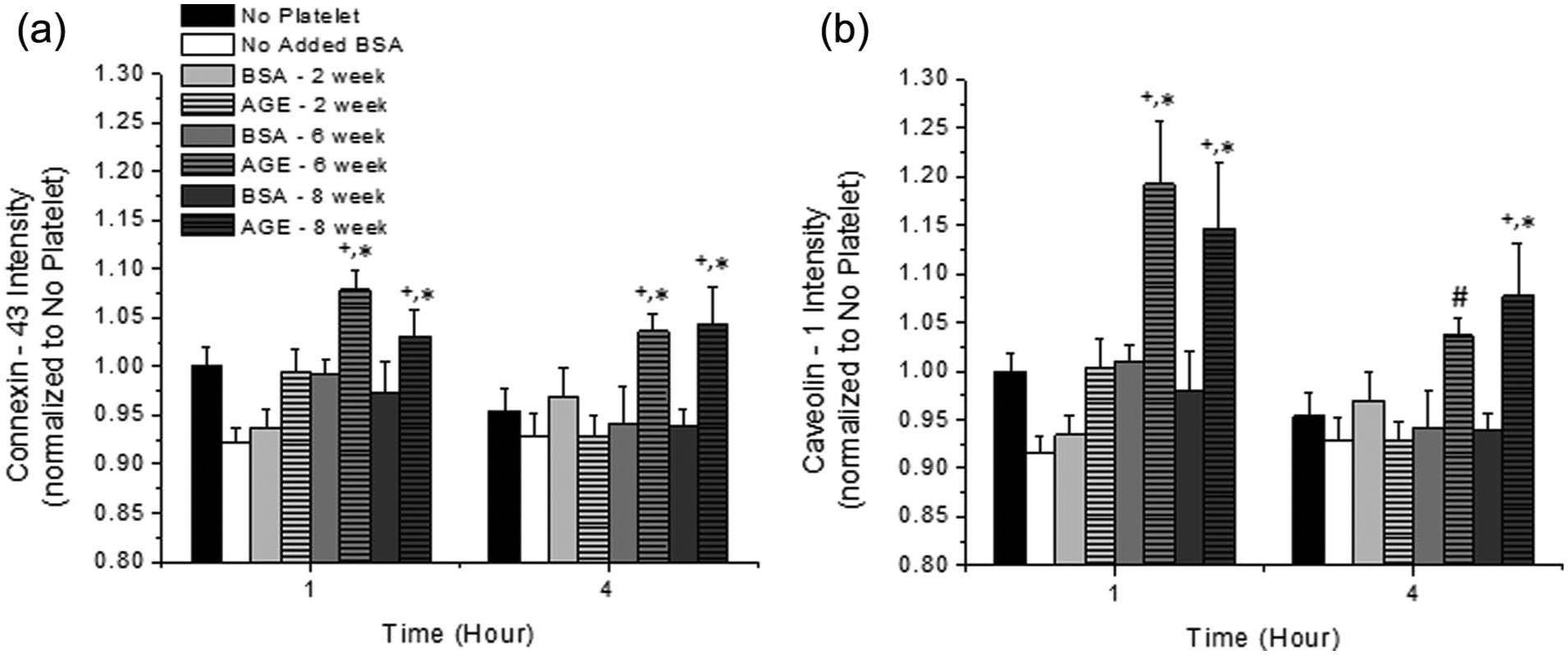
Surface expression of endothelial cell (a) connexin-43 and (b) caveolin-1 in the presence of platelets as a function of incubation duration with platelets and glycated albumin additive. All data are reported as the mean ± standard error of the mean for at least eight independent experiments.
Platelet activation, aggregation and adhesion
Platelet activation, aggregation and adhesion potential were quantified as a function of exposure duration and glycated albumin additive in the presence of endothelial cells (Figure 4). Phosphatidylserine expression is a marker for platelet activation. We observed a time-dependent decrease in phosphatidylserine expression for control conditions and platelets that were exposed to non-glycated albumin (Figure 4(a)). This is typical for quiescent platelets left on a lab bench. However, platelets that were exposed to irreversibly glycated albumin were observed to have a significant increase in phosphatidylserine expression as compared with the No Added BSA control and paired non-glycated albumin samples, which would suggest a heightened activation potential. Platelet adhesion (CD31, PECAM, Figure 4(b)) and platelet aggregation (CD41a, GPIIb, Figure 4(c)) potential were generally not a function of time or glycated albumin additive under these conditions, although there was an increasing trend with time and a marginal increasing trend for irreversibly glycated albumin. Finally, platelet CD62P (P-selectin, Figure 4(d)) expression, which is indicative of platelet activation (via α-granule fusion) and adhesion, showed no statistical significance as a function of exposure time or additive; however, there was an increasing trend for cells that were exposed to all glycated albumin.
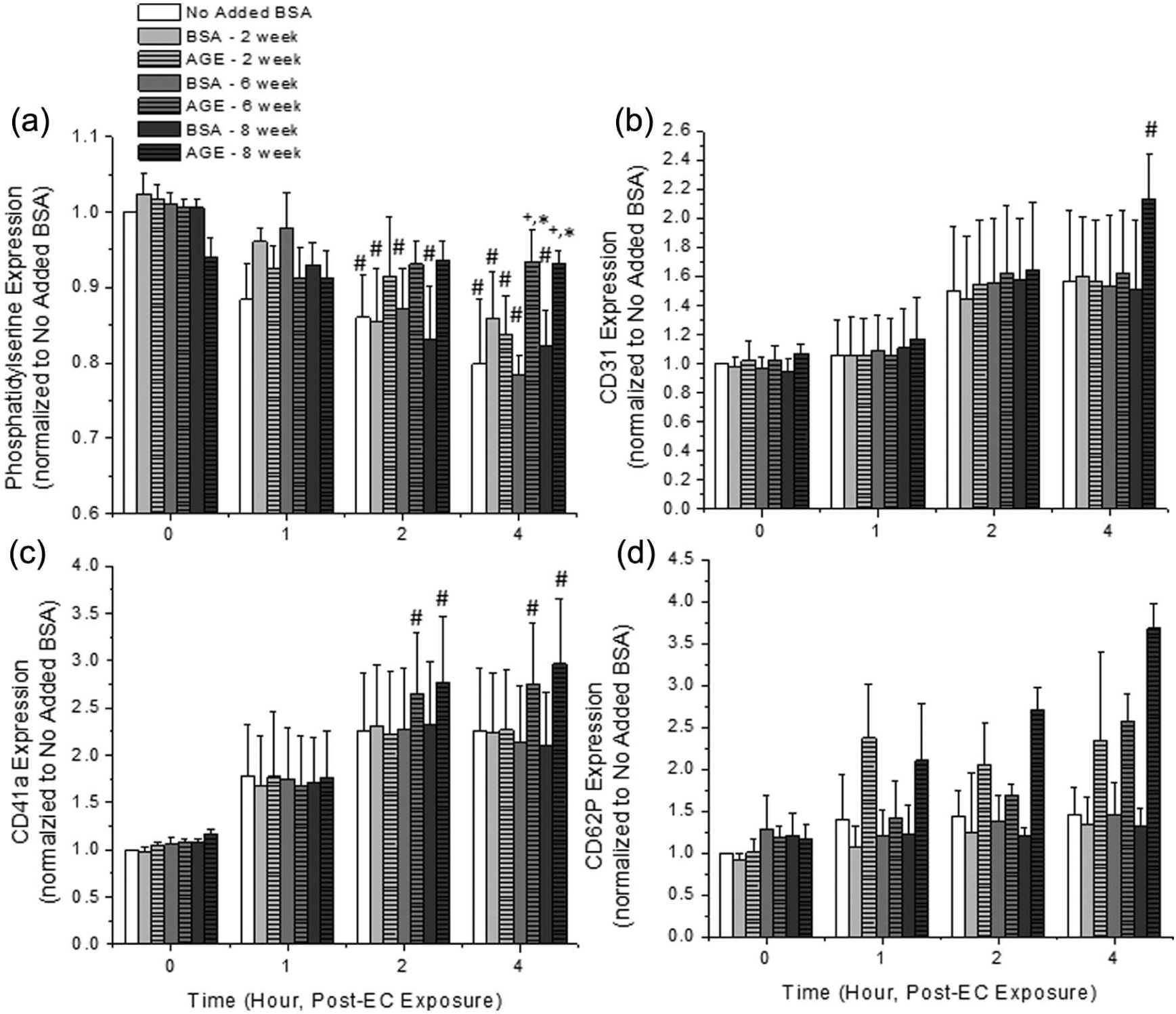
Expression of (a) platelet phosphatidylserine, (b) CD31 (PECAM), (c) CD41a (GPIIIa) and (d) CD62P (P-selectin) in the presence of endothelial cells as a function of incubation duration and glycated albumin additive. All data are reported as the mean ± standard error of the mean for at least four (phosphatidylserine) or five (CD31, CD41a, CD62P) independent experiments.
Discussion
Endothelial cell metabolic activity
We observed no change in endothelial cell metabolic activity in response to the presence of glycated albumin, glycated to any extent, and platelets (Figure 1). In all of our previous works, which investigated the role of glycation reversibility in endothelial cell metabolic activity, the metabolic activity of endothelial cells was altered in response to irreversibly glycated albumin (general decrease of ~50%) and/or shear stress (general increase of ~30%).4–6 Other groups have observed altered endothelial cell metabolic activity, quantified by oxidative indicators, in response to glycated albumin. 12 Additionally, glycated low-density lipoprotein has also been shown to alter endothelial cell metabolic activity in culture. 13 Thus, there is growing evidence that AGEs can alter the metabolic activity of endothelial cells. However, in the presence of platelets, which is a more physiologically relevant in vitro model, this altered metabolism was abolished, suggesting that an inhibitory endothelial cell–platelet interaction occurs under these conditions. Therefore, it is possible that in vivo endothelial cell–mediated CVD development under diabetic conditions may be tempered by the presence of platelets.
Endothelial cell inflammatory/thrombotic markers
Using ELISA techniques, we quantified the surface expression of thrombomodulin, tissue factor and ICAM-1 (Figure 2) to observe endothelial cell inflammatory and thrombotic potential. Platelets were removed prior to ELISA measurements, so there should be minimal effects associated with platelet adhesion to the endothelium. In general, irreversibly glycated albumin enhanced the surface expression of tissue factor and ICAM-1, with marginal changes to the thrombomodulin expression. In our previous work, we typically observed a significantly enhanced inflammatory response, via an increased ICAM-1 expression (average of ~80% increase for static conditions and > 100% increase for shear conditions), coupled with an increased thrombotic response, via an increased tissue factor expression (average of ~60% increase for static conditions and > 100% increase for shear conditions) and a decreased thrombomodulin expression (average of ~30% decrease for static conditions and a ~50% decrease for shear conditions).4–6 In our previous work, this was especially prevalent for irreversibly glycated albumin. In cell culture experiments, others have also reported an enhanced ICAM-1 expression, an enhanced thrombogenicity and a decreased thrombomodulin expression, in response to glycated albumin.14–16 Thus, previous reports suggest that AGEs have a direct effect on enhancing endothelial cell inflammatory and thrombotic responses by modulating ICAM-1, tissue factor and/or thrombomodulin expression. However, our current observations suggest that in a more physiologically relevant model, the effects of AGEs are somewhat tempered by the presence of platelets in regard to ICAM-1, tissue factor and thrombomodulin expression.
Connexin-43 and caveolin-1 expression
Expression of connexin-43 and caveolin-1 was observed to monitor the progression of angiogenesis and the establishment of communicative networks (Figure 3). In general, we observed an increased surface expression of these two markers when endothelial cells were incubated with irreversibly glycated albumin (in the range of 20% for all conditions).4–6 There has been no consensus on the expression of these two markers; some groups have observed an up-regulation, while others have observed a down-regulation of these markers. The expression profile appears to be very highly dependent on the cell type and the conditions studied.4–6,17–19 In the presence of platelets, AGEs appear to restrain the surface expression of connexin-43 and caveolin-1 under control, non-glycated and reversibly glycated conditions and somewhat promote the response of cells when exposed to irreversibly glycated albumin.
Platelet activation, aggregation and adhesion
In order to determine possible mechanisms of platelet–endothelial cell interactions, we quantified the expression of various platelet markers that are associated with activation, aggregation and adhesion after exposure to AGEs (Figure 4). In general, in the presence of endothelial cells and irreversibly glycated albumin, we saw an enhancement of platelet adhesion, via CD41a (GPIIb), and platelet activation, via phosphatidylserine expression. In our previous work, we saw an increased expression of all platelet surface markers investigated, aggregation and thrombogenic potential in response to irreversibly glycated albumin (~40% increased aggregation, ~50% increased activation and ~50% increase in CD41a and CD62P expression). 9 Similar responses have been observed by others, suggesting that AGEs may alter platelet functions.7,20 However, in the presence of endothelial cells, giving platelets the ability to adhere to a different cell type, the response of platelets is somewhat tempered. This seems to be mediated by endothelial cell–platelet adhesion via GPIIb, which is the only marker that is significantly up-regulated as a function of time for irreversibly glycated albumin. Note that the markers that we investigated were up-regulated but this does not necessarily mean that the proteins were active.
Conclusion
In general, all of our previous works and the work of the scientific community have shown that AGEs can alter endothelial cell and platelet functions to promote inflammatory and thrombotic responses that are commonly observed during CVDs. However, it appears that the model systems that were employed, including our own, did not capture the entire story since under more physiologically relevant conditions (e.g. the combined incubation of AGEs, platelets and endothelial cells), the CVD-associated responses are somewhat tempered. This would suggest that there is some communication occurring between platelets and endothelial cells to restrain disease progression. It is well accepted that AGEs can alter endothelial cell and platelet responses, but the extent of these altered responses appears to be somewhat reduced under these more relevant conditions. With that said, there are still limitations to the model system that we employed, including static flow conditions, reduced platelet concentrations and the absence of other cell types (e.g. leukocytes) and plasma proteins.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by The Oklahoma Center for the Advancement of Science and Technology (award number HR09-158).
