Abstract
Asymmetric dimethylarginine (ADMA), an endogenous nitric oxide synthase inhibitor, plays a role in endothelial dysfunction, an initial step of atherosclerosis. Advanced glycation end products (AGEs) also contribute to accelerated atherosclerosis. However, a pathophysiological crosstalk between ADMA and AGEs remains unclear. In this study, we investigated the relationship between ADMA and AGE level in patients with end-stage renal disease (ESRD) due to diabetic nephropathy. We also examined whether and how AGEs increased ADMA generation by cultured endothelial cells (ECs). Plasma ADMA levels were positively associated with serum AGE level and were inversely correlated with endothelial function determined by flow-mediated vasodilatation. AGEs dose dependently increased reactive oxygen species (ROS) generation in ECs, which was blocked by antisense DNA raised against receptor for AGEs (RAGE). Furthermore, AGEs decreased messenger RNA (mRNA) level of dimethylarginine dimethylaminohydrolase (DDAH)-II, an enzyme for ADMA degradation, reduced its total enzymatic activity and resultantly increased ADMA, all of which were completely blocked by an antioxidant, N-acetylcysteine. These results suggest that the AGE-RAGE-mediated ROS generation could be involved in endothelial dysfunction in diabetic ESRD patients partly by increasing the ADMA generation via suppression of DDAH activity in ECs.
Introduction
Reducing sugars such as glucose can react non-enzymatically with amino groups of proteins to form Amadori products. 1 Over the course of days to weeks, these Amadori products undergo further rearrangement reactions to form irreversibly cross-linked senescent macroprotein derivatives called advanced glycation end products (AGEs).1–3 The formation and accumulation of AGEs have been known to progress at an accelerated rate under diabetes, especially in patients with end-stage renal disease (ESRD). 3 Furthermore, there is accumulating evidence that AGEs elicit oxidative stress generation in various cell types through the interaction with a receptor for AGEs (RAGE) and subsequently evoke inflammatory and thrombogenic reactions, thereby contributing to accelerated atherosclerosis in diabetes.4–6 Therefore, AGEs may be one of the causative factors that could explain the increased risk of cardiovascular disease (CVD) in diabetic patients with ESRD.
Nitric oxide (NO) is a pleiotropic molecule critical to a number of physiological and pathological processes in humans. 7 Since NO not only inhibits the inflammatory-proliferative reactions in vascular wall cells but also exerts anti-thrombogenic and endothelial cell (EC) protective properties in vivo,7,8 impaired production and/or bioavailability of NO are also considered to play a role in atherosclerosis in humans.7–9 Indeed, circulating level of asymmetric dimethylarginine (ADMA), an endogenous NO synthase inhibitor, is increased in diabetic patients, especially with ESRD, 10 and is associated with future cardiovascular events in these subjects. 10 However, a pathophysiological crosstalk between ADMA and AGEs remains to be elucidated. In this study, we investigated the relationship between ADMA and AGE levels in 15 patients with ESRD due to diabetic nephropathy. We also examined here whether and how AGEs increased ADMA generation by human cultured aortic ECs.
Design and methods
Materials
Bovine serum albumin (BSA; essentially fatty acid free and essentially globulin free, lyophilized powder) and N-acetylcysteine (NAC) were purchased from Sigma (St. Louis, MO, USA),
Subjects
Fifteen type 2 diabetic patients with ESRD due to nephropathy (13 males and 2 females; mean age = 61 years; 24-h creatinine clearance = 5.0 ± 1.0 mL/min, range = 2.3–9.8 mL/min) who did not undergo haemodialysis therapy were enrolled in the present study. All patients received insulin injection for the treatment of blood glucose. All patients received anti-hypertensive drugs such as angiotensin-II type 1 receptor blockers (n = 12), angiotensin-converting enzyme inhibitors (n = 2) and calcium channel blockers (n = 14). Four patients were treated with statins. The protocol was approved by the ethical committee of our institution and informed consent was obtained from all patients.
Measurements of endothelial function
Endothelial function was determined by flow-mediated vasodilatation (FMD) of brachial artery as described previously. 11 Two blinded investigators evaluated the FMD data. The mean value of two measurements was calculated.
Measurement of ADMA and AGEs
ADMA level was measured with a high-performance liquid chromatography as described previously. 12 Serum level of carboxymethyllysine, one of the representative circulating AGEs, was determined by an enzyme-linked immunosorbent assay at a commercially available laboratory (SRL, Inc., Tokyo, Japan).
Preparations of AGEs
AGE-BSA was prepared as described previously.
13
In brief, BSA was incubated under sterile conditions with
Cells
Human immortalized aortic ECs with SV40T antigen 14 were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Nissui Pharmaceutical Co., Tokyo, Japan) containing 10% fetal bovine serum (FBS; Filtron Pty Ltd, Brooklyn, Australia), 100 U/mL penicillin (Life Technologies, Grand Island, NY, USA) and 100 µg/mL of streptomycin (Life Technologies) at 37°C in a humidified 5% CO2–95% air atmosphere. AGE treatments were carried out in a medium containing 0.5% FBS.
Intracellular reactive oxygen species generation
Intracellular reactive oxygen species (ROS) generation was measured by using a fluorescent probe CM-H2DCFDA (Molecular Probes, Inc., Eugene, OR, USA) as described previously. 13
Assay with antisense oligodeoxyribonucleotides
A phosphorothioate antisense complement of human RAGE messenger RNA (mRNA) and the corresponding sense oligodeoxyribonucleotide were synthesized and purified as described previously. 13 Sequences of antisense and sense oligonucleotides were 5′-CAACTGCTGTTCCGGCT-3′ and 5′-AGCCGGAACAGCAGTTG-3′, respectively, which corresponded to nucleotides 6–22 of human complementary DNA (cDNA). 15 An amount of 10 µM oligonucleotides was added to the medium, with 100 µg/mL AGE-BSA or non-glycated control BSA, in which ECs were grown. After 24 h, intracellular ROS generation was measured. We confirmed that 10 µM antisense but not sense oligomers raised against RAGE mRNA reduced the RAGE protein level to 1/10 of that of control cells (data not shown).
Semi-quantative reverse transcription–polymerase chain reactions
Poly(A)+RNAs were isolated from ECs and were analysed by reverse transcription–polymerase chain reactions (RT-PCR) as described previously. 16 The amounts of poly(A)+RNA templates (30 ng) and cycle numbers (30 cycles for dimethylarginine dimethylaminohydrolase (DDAH)-I gene; 26 cycles for DDAH-II gene; 22 cycles for β-actin gene) for amplification were chosen in quantitative ranges, where reactions proceeded linearly, which had been determined by plotting signal intensities as functions of the template amounts and cycle numbers. Primer sequences used in semi-quantitative RT-PCR were 5′-CGTGGCCGTGGTGTGCGAGGA-3′ and 5′-CAGTTCAGACATGCTCACGGGG-3′ for human DDAH-I mRNAs and 5′-TTTCTTCGTCCTGGGTTGCCTG-3′ and 5′-ATCCTTTTCCCTACACTCTCCCCTC-3′ for human DDAH-II mRNAs.
DDAH enzymatic activity
Total DDAH enzymatic activity was measured as described previously.
12
In brief, the cell lysate was incubated with 4 µM ADMA and 0.1 mM sodium phosphate buffer (pH = 6.5) in a total volume of 0.5 mL for 6 h at 37°C. The reaction was stopped by the addition of equal volume of 10% trichloroacetic acid, and the supernatant was boiled with diacetyl monoxime [0.8% (wt/vol) in 5% acetic acid] and anti-pyrine [0.5% (wt/vol) in 50% sulphuric acid). The amounts of
Statistical analysis
All data were expressed as mean ± standard error (SE). Linear regression analysis was performed to evaluate the correlations among circulating levels of ADMA and AGEs and FMD. Experimental groups were compared by analysis of variance (ANOVA) and, when appropriate, with Scheffe’s test for multiple comparisons. A value of p < 0.05 was considered significant.
Results
Characteristics of the patients
Characteristics of the patients are shown in Table 1. Mean circulating level of AGEs of our patients was 6.7 U/mL. Systolic blood pressure of 129 ± 5.6 mmHg and diastolic blood pressure of 74 ± 2.4 mmHg were well controlled. Fasting plasma glucose was 107 ± 5.1 mg/dL, and HbA1c was 6.4% ± 0.2%.
Clinical characteristics of the subjects.
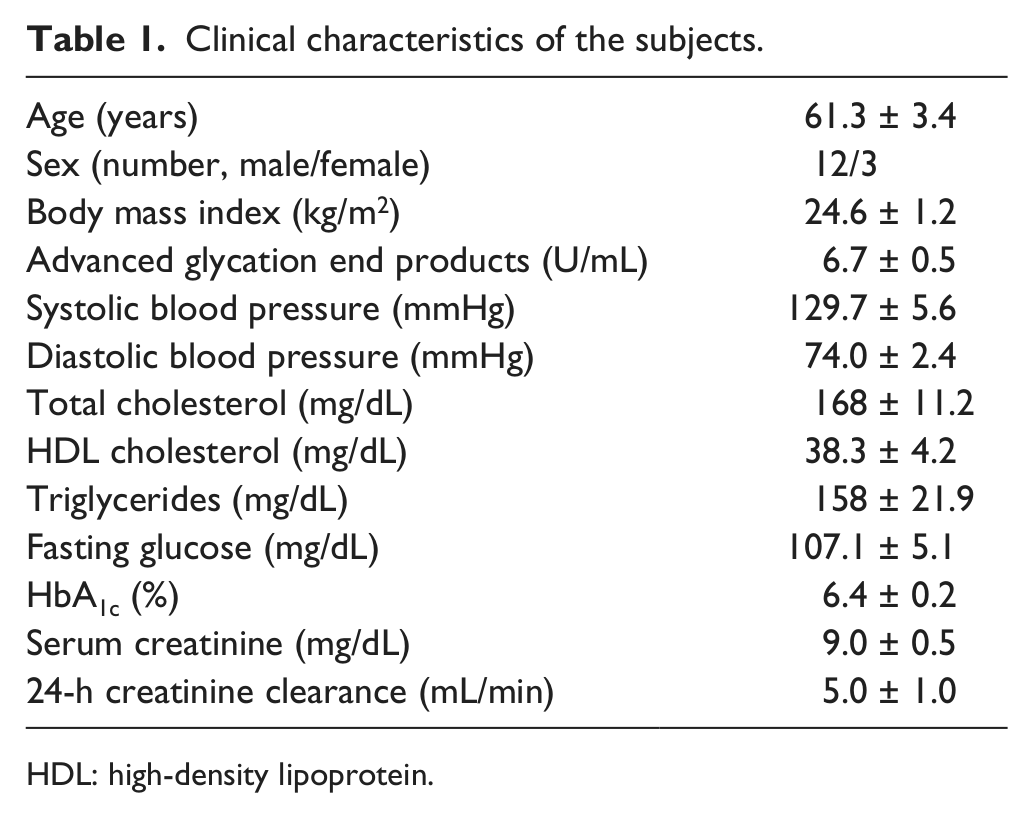
HDL: high-density lipoprotein.
AGEs, ADMA and endothelial function in patients with diabetic nephropathy
As shown in Figure 1(a), plasma level of ADMA was positively associated with circulating AGE level in patients with ESRD due to diabetic nephropathy (r = 0.52, p < 0.05). Furthermore, ADMA level was inversely correlated with FMD in our subjects (r = −0.54, p < 0.05; Figure 1(b)), whereas other cardiometabolic risk factors, including age, blood pressure, lipid parameters, fasting plasma glucose, HbA1c, renal function and body mass index, were not associated with FMD. In addition, plasma level of ADMA was not significantly correlated with endothelial function evaluated by FMD (r = −0.33, p = 0.23).
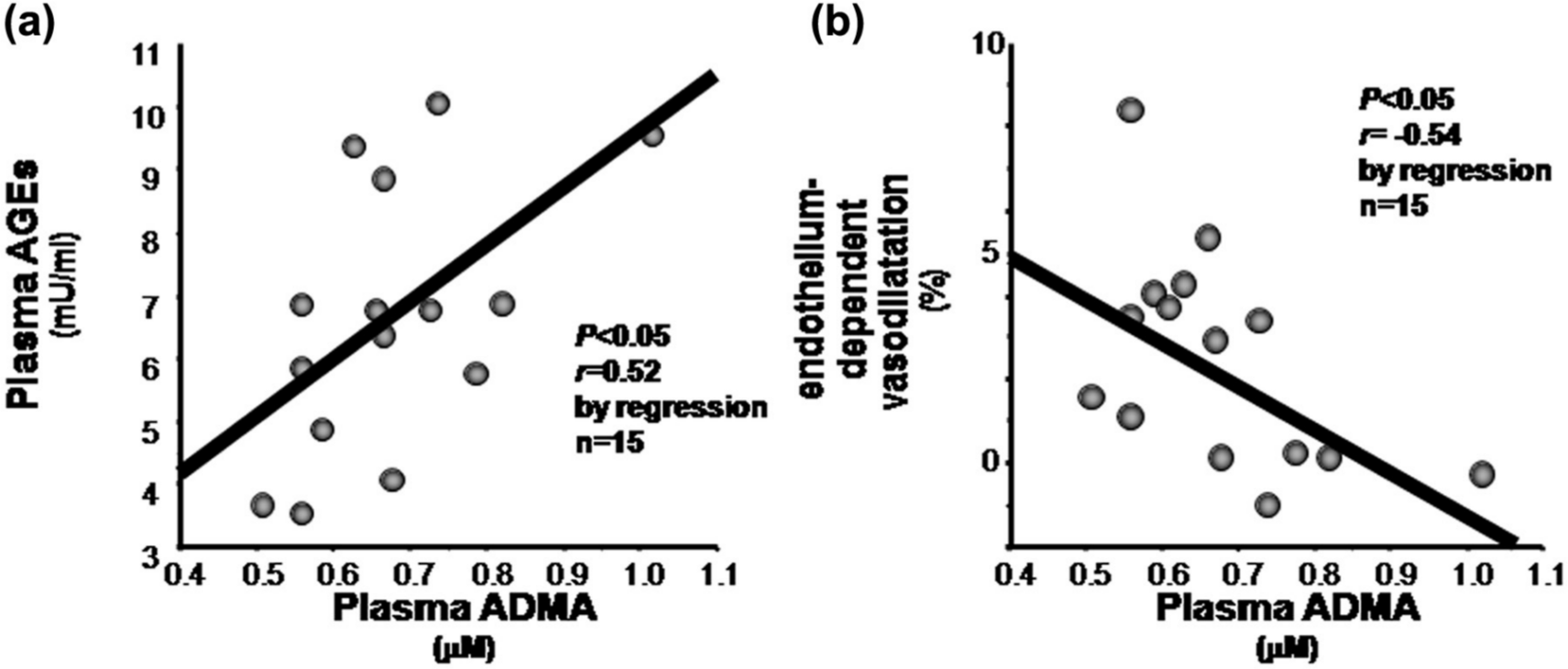
Correlations between plasma level of ADMA and (a) circulating AGE and (b) FMD.
Effects of AGEs on DDAH-ADMA system in cultured human aortic ECs
To clarify the mechanistic link between AGEs and ADMA, we examined whether and how AGEs increased ADMA production by ECs. As shown in Figure 2, AGEs dose dependently increased ROS generation in cultured human aortic ECs, which was blocked by antisense but not sense DNA raised against RAGE mRNA. Furthermore, AGEs decreased mRNA level of DDAH-II, a rate-limiting enzyme for ADMA degradation, but not DDAH-I in ECs (Figure 3). In addition, AGEs reduced its total enzymatic activity and resultantly increased ADMA generation by ECs, all of which were completely blocked by an antioxidant, NAC (Figure 4(a) to (c)).
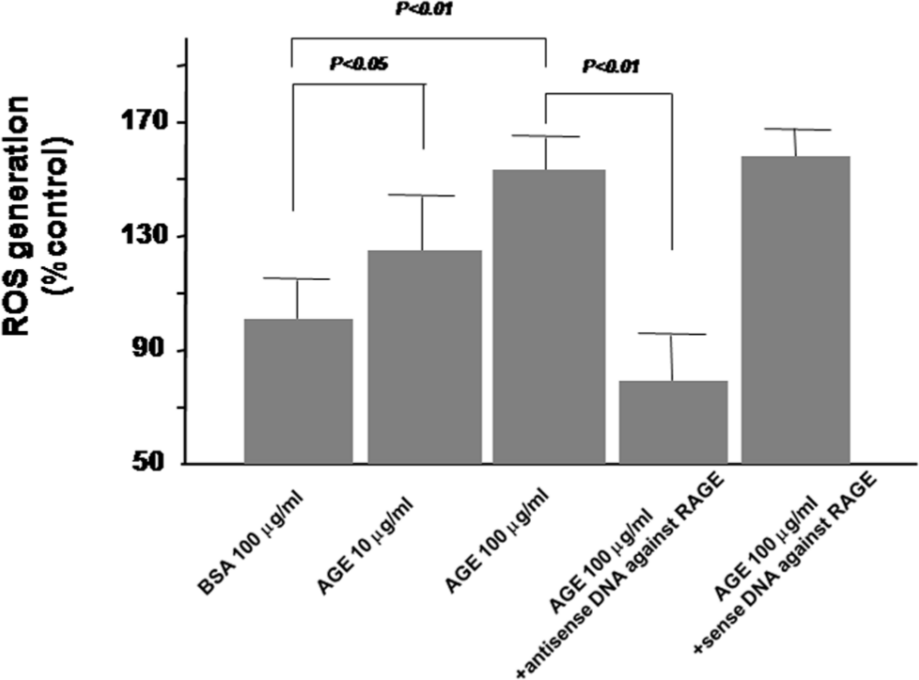
Effects of AGEs on ROS generation. ECs were pre-incubated with or without 10 µM antisense or sense oligodeoxyribonucleotides raised against RAGE mRNA. After 12 h, the cells were treated with 10–100 µg/mL of AGEs or 100 µg/mL of non-glycated BSA for 24 h. Then, ROS generation was measured (N = 14 per group).
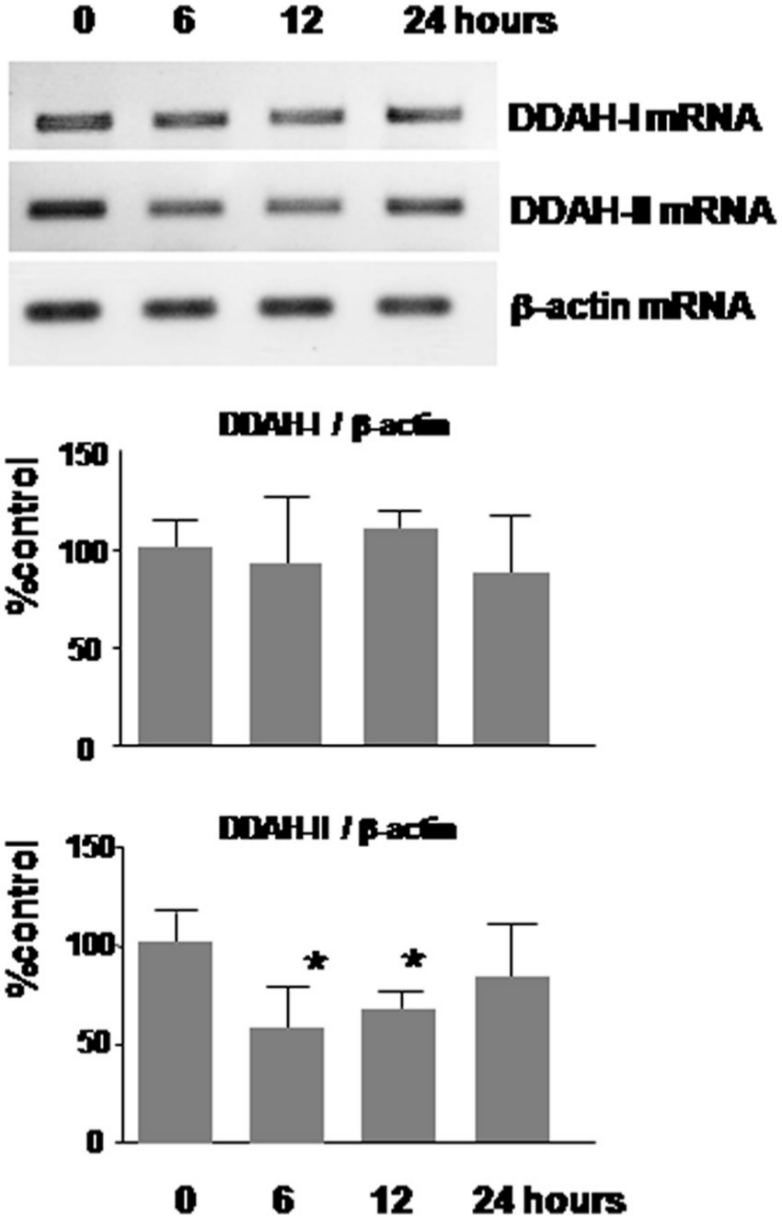
Effects of AGEs on DDAH-I and DDAH-II mRNA levels. ECs were incubated on 6 well plates and then stimulated with 100 µg/mL of AGEs for the indicated periods. Then, DDAH-I and DDAH-II mRNA expressions were determined by RT-PCR. Upper panels show the representative bands of RT-PCR. Lower panels show the quantitative data. Data were normalized by the intensity of β-actin mRNA-derived signals and related to the value obtained with the control (N = 6 per group).
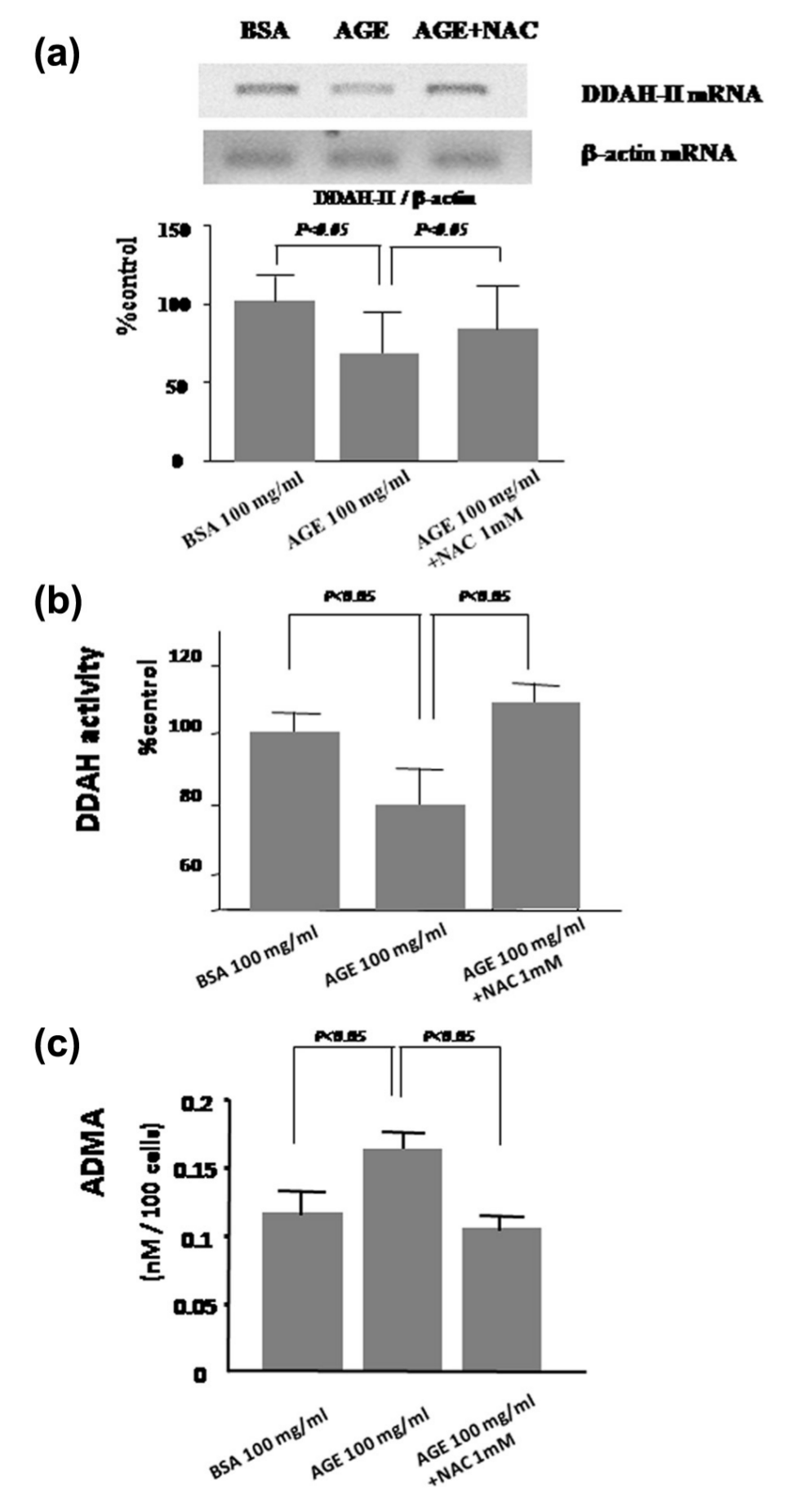
Effects of AGEs and antioxidant on (a) DDAH-II mRNA level, (b) DDAH enzymatic activity and (c) ADMA generation in ECs. (a) ECs were pre-incubated with or without 1 mM NAC. After 1 h, the cells were treated with 100 µg/mL of AGEs or non-glycated BSA for 12 h. Then, DDAH-II mRNA level was analysed by RT-PCR. Upper panels show the representative bands of RT-PCR. Lower panels show the quantitative data. Data were normalized by the intensity of β-actin mRNA-derived signals and were related to the value obtained with the control. N = 6 per group. (b and c) ECs were pre-incubated with or without 1 mM NAC. After 1 h, the cells were treated with 100 µg/mL of AGEs or non-glycated BSA for 48 h. Then, (b) DDAH enzymatic activity and (c) ADMA generation were measured: (b) N = 12 per group and (c) N = 12 per group.
Discussion
In the present study, we found that plasma ADMA level was associated with circulating AGE level and was inversely correlated with endothelial function in type 2 diabetic patients with ESRD due to nephropathy. The findings have extended the previous observations showing that serum AGE level was positively associated with ADMA level in non-diabetic patients with early stage chronic kidney disease (CKD) 17 and that AGE level was correlated with endothelial dysfunction in diabetes.18,19 Moreover, we also found here for the first time that (1) AGEs reduced DDAH-II mRNA level and resultantly suppressed DDAH enzymatic activity and ADMA generation in cultured aortic ECs, all of which were blocked by the treatment of an antioxidant NAC, and (2) AGE-induced ROS generation was reduced by antisense DNAs raised against RAGE mRNA. Therefore, although we did not find here the direct correlation between serum AGE level and endothelial function probably due to small sample size, given that AGEs have exerted pleiotropic actions on a variety of cells via ROS generation,4,20 our present observations suggest that the AGE-RAGE-mediated ROS generation could be involved in endothelial dysfunction in diabetic patients with ESRD partly by increasing the ADMA generation via suppression of DDAH-II expression in ECs.
DDAH-II is a dominant isoform of DDAH in ECs, which could mainly degrade ADMA. 21 DDAH enzymatic activity in ECs has been shown to be suppressed under oxidative stress conditions. 22 Furthermore, overexpression of DDAH-II gene is reported to inhibit the elevation of ADMA level and, subsequently, to improve endothelial dysfunction in diabetic rats. 23 Since there is accumulating evidence that increased plasma level of ADMA is associated with endothelial dysfunction, carotid intima media thickness and CVD in high-risk subjects such as patients with diabetes and CKD,24–26 the present findings suggest that oxidative stress generation-mediated DDAH-II suppression, which is evoked by the AGE-RAGE system, could contribute to ADMA accumulation and accelerated atherosclerosis in diabetic patients with ESRD. Small interference RNAs raised against DDAH-II significantly inhibited endothelium-dependent vasodilation of rat mesenteric arterioles, whereas small interfering RNA (siRNA) against DDAH-I had lesser effects, 27 thus supporting our speculation. However, vascular endothelial-specific DDAH-I-deficient mice have also been shown to exhibit an increase in ADMA concentration associated with attenuated endothelial function. 28 Therefore, although AGEs did not affect DDAH-I mRNA level in the present study (Figure 3), we cannot totally exclude the possibility that AGEs could affect DDAH-Ienzymatic activity in ECs because there is no reliable method to measure the activity of DDAH-I and DDAH-II separately.
Although we did not have the data of circulating levels of AGEs in diabetic patients with almost normal kidney function, several articles have already shown that circulating level of AGEs is significantly increased as the renal function deteriorated in patients with diabetes.29,30 These observations suggest that the crosstalk between the AGE-RAGE axis and ADMA-DDAH system observed in cell culture system could apply to ‘diabetic patients with ESRD’.
In conclusion, the present results suggest that the AGE-RAGE-mediated ROS generation could be involved in endothelial dysfunction in diabetic patients with ESRD by increasing the ADMA generation via suppression of DDAH-II expression and its activity in ECs. AGE-ADMA-DDAH axis may be a therapeutic target for preventing accelerated atherosclerosis in these patients.
Footnotes
Conflict of interest
None declared.
Funding
This work was supported by Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture, Tokyo; a grant from Japan Foundation of Cardiovascular Research, Tokyo; and a grant from Diabetes Masters Conference, Tokyo, Japan.
