Abstract
Background:
Adipose tissue produces pro-inflammatory mediators involved in the atherosclerotic process. We investigated whether 12-month exercise training in patients with type 2 diabetes mellitus and coronary artery disease would reduce circulating levels and genetic expression of mediators in the interleukin-18, Caspase-1 and NLR pyrin domain containing 3 pathways. Correlations to glucometabolic variables; fasting glucose, HbA1c, duration of diabetes, insulin, C-peptide, insulin resistance (measured by homeostatic model assessment indexes – insulin resistance) and body mass index at baseline were further assessed.
Methods:
137 patients (aged 41–81 years, 17.2% female participants) were included and randomized to a 12-month exercise programme or to a control group. Fasting blood and adipose tissue samples were taken at inclusion and after 12 months.
Results:
No statistically significant difference in changes of any variable between the intervention and the control group was found. At baseline, a positive correlation between insulin and homeostatic model assessment indexes – insulin resistance, interleukin-18 expression in adipose tissue and an inverse correlation between some glucometabolic variables and leukocyte expression of NLR pyrin domain containing 3 and Caspase-1 were observed.
Conclusion:
No significant effects of long-term exercise training were observed on the inflammasome-related mediators in our patients with combined coronary artery disease and type 2 diabetes mellitus. The observed correlations may indicate a pro-inflammatory state in adipose tissue by overweight and a compensatory downregulation of these mediators in circulating leucocytes.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is considered as one of the major risk factors for developing cardiovascular disease (CVD), 1 in which the atherosclerotic process is the main underlying condition. Atherosclerosis was previously considered to be a lipid disorder; however, in recent years, the prominent role of inflammation and immune response in the formation of the atherosclerotic plaque has become well recognized. 2 This involves the recruitment and activation of many different cell types, including endothelial cells, smooth muscle cells, monocyte-derived macrophages, and T-cells, all contributing to a pro-inflammatory environment. 3
Interleukin (IL)-18 is a pro-inflammatory cytokine involved in the development and progression of both atherosclerosis and T2DM. 4 It is also known as an interferon-gamma (IFN-γ)-inducing factor and is biologically active after being cleaved by Caspase-1. 5 Caspase-1 is, in turn, activated by the NLR pyrin domain containing 3 (NLRP3). Circulating IL-18 has been identified as an independent predictor of CVD, 6 particularly related to hyperglycemia. 7 Also, patients with T2DM and multi-vessel coronary artery disease (CAD) seem to be characterized by higher serum IL-18 levels, which may explain their vulnerability to secondary cardiovascular events.4,6 Increased gene expression of Caspase-1 in aortic tissue has been demonstrated to be correlated to the severity of CAD, 8 and the expression of NLRP3-inflammasome in subcutaneous adipose tissue (AT) and peripheral blood monocytes has also been shown to be significantly higher in patients with CAD compared with healthy individuals.9,10
The AT has been recognized as an important source of pro-inflammatory cytokines, secreted from both the adipocytes and the infiltrated immune cells, particularly in overweight patients.11,12 As AT is vascularized, cytokines from AT will give rise to a systemic inflammation, which may influence the development of atherosclerosis, insulin resistance, T2DM and metabolic syndrome. 12 Despite some reported links between glucose and the IL-18 pathway, the importance of glycaemic control for the inflammasome-related mediators in T2DM is not fully understood.
Exercise training has evolved into an established evidence-based therapeutic strategy with prognostic benefits in CAD, mainly through the beneficial influence on CVD risk factors. 13 Improved glycaemic control and beneficial effects on systolic blood pressure, triglycerides and waist circumference by exercise training are also evident,14,15 whereas the effect on pro-inflammatory mediators is not clarified, especially in patients with combined CAD and T2DM.
We aimed to investigate whether exercise training would reduce the levels of circulating IL-18, as well as gene expression of IL-18, Caspase-1 and NLRP3 in circulating leukocytes and in AT in patients with CAD and T2DM. Any association between these inflammatory mediators and glucometabolic variables, as well as any relation to CAD sub-groups, was further assessed.
We believe that exercise training might reduce the inflammatory burden, here investigated through IL-18 and its upstream regulators, and thereby decrease the risk of future cardiovascular events.
Methods
Study population
This is a sub-study of the EXCADI (exercise training in patients with T2DM and CAD) trial, in which patients with T2DM and CAD (n = 137) were included at the Department of Cardiology, Oslo University Hospital, Oslo, Norway, between August 2010 and March 2012. The study design has previously been described in detail. 16 In brief, the subjects were randomized 1:1 to a combination of endurance and strength training or to a control group with conventional follow-up by their general practitioner. All patients had known T2DM and stable CAD verified by coronary angiography. Exclusion criteria were presence of proliferative retinopathy, end-stage renal disease, cancer, stroke or acute myocardial infarction (MI) within the last 3 months, unstable angina, decompensated heart failure, serious arrhythmia, severe valvular disease, severe rheumatologic disease, chronic obstructive pulmonary disease stadium GOLD IV, thromboembolic disease, ongoing infections, severe musculoskeletal disorders and other disabilities limiting the ability for physical activity.
Patients with previous MI and/or diabetic microvascular complications [defined as history of nephropathy, neuropathy or retinopathy, and/or abnormal monofilament test and/or (micro-) albuminuria] were defined to have advanced vascular disease. Hypertension (HT) was defined as those on antihypertensive medication. To estimate insulin resistance, the updated homeostatic model assessment indexes – insulin resistance (HOMA2-IR) was applied.
The Regional Committee of Medical Research Ethics in South-Eastern Norway approved the study, and all patients gave their written informed consent to participate. The study was conducted in accordance with the Declaration of Helsinki.
Physical exercise intervention
The physical exercise programme was developed and conducted in collaboration with the Norwegian School of Sport Sciences. Details on the exercise programme have previously been described. 16 Briefly, the exercise group had two group-based sessions per week with qualified instructors, and a third home-based session, for a total volume of at least 150 min per week. About two-thirds were aerobic and one-third resistance training, and each of the supervised sessions included parts with high-intensity interval training. The weekly, unsupervised home-based exercise session was registered in exercise diaries. General activity (outside the training programme) was not accounted for in either group.
A 24-h dietary recall was used for assessment of dietary intake at baseline and after the 1-year intervention. 16
Laboratory methods
Venous blood samples were drawn in fasting condition by standard venipuncture between 08:00 and 10:00 a.m. before intake of morning medication at time of inclusion and after 1 year. Subcutaneous AT taken from the gluteal region was immediately frozen at −80°C until RNA extraction. PAXgene tubes (PreAnalytiX, Hombrechtikon, Switzerland) were collected for RNA extraction from circulating leukocytes. Routine blood samples including HbA1, insulin, and C-peptide were determined by conventional methods. Serum was prepared by centrifugation within 1 h at 2500g for 10 min for the determination of circulating IL-18, measured by enzyme-linked immunosorbent assay (ELISA) (MBL, Medical & Biological Laboratories Co., Ltd, Nagoya, Japan). The inter-assay coefficient of variation (CV) in our laboratory was 7.7%.
Total RNA was extracted using the PAXgene® Blood RNA Kit (PreAnalytiX Qiagen, Hilden, Germany), with an extra cleaning step (RNeasy® MinElute® Cleanup Kit; Qiagen). RNA quality and quantity (ng/µL) were determined by the NanoDrop™ 1000 Spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). Total RNA from the AT was isolated, including disruption and homogenization in Tissuelyser (Qiagen), using a High Pure RNA Tissue Kit (Hoffman-La Roche Ltd, Basel, Switzerland), according to a combination of the kit protocol and previous experience in our laboratory. Extracted RNA from both sources was stored at −80°C until analyses. Complementary DNA (cDNA) was synthesized from equal amount of RNA with qScript™ cDNA SuperMix (Quanta Biosciences Inc., Gaithersburg, MD, USA). Real-time polymerase chain reaction (PCR) was performed on a ViiA™ 7 instrument, using TaqMan® Universal PCR Master Mix (P/N 4324018) and TaqMan® assays for IL-18 (Hs00155517_m1), NLRP3 (Hs00918082_m1) and Caspase-1 (Hs00354836_m1) (Applied Biosystems, Foster City, CA, USA). β2-microglobulin (HS99999907 m1) (Applied Biosystems) was used as endogenous control, and mRNA levels were determined by relative quantification (RQ) using the ΔΔCT method. 17
Statistical analyses
Demographic data are given as proportions, mean (±standard deviation) or median (25th and 75th percentiles) for skewed data. Differences between groups at baseline were analysed by Student’s t-test or Mann–Whitney U test for continuous variables and chi-square test for categorical variables. Within-group changes after intervention were analysed by Wilcoxon Signed Rank test, and differences in change between randomized groups were performed by Mann–Whitney U test. Baseline associations were analysed by the use of Spearman’s rho correlations. Statistical calculations were performed using SPSS version 24 (SPSS Inc., Chicago, IL, USA). p-values < 0.05 were defined as statistically significant.
Results
Baseline characteristics of the total population and according to the randomized groups are shown in Table 1. No significant differences in baseline characteristics between the randomized groups were observed. Of the 137 included patients, 123 completed the study and 9 patients with the lowest adherence to the intervention principle were excluded. 16 Thus, a total of 114 patients were analysed for the intervention effect.
Baseline characteristics of the total study population (n = 137) and according to the randomized groups.
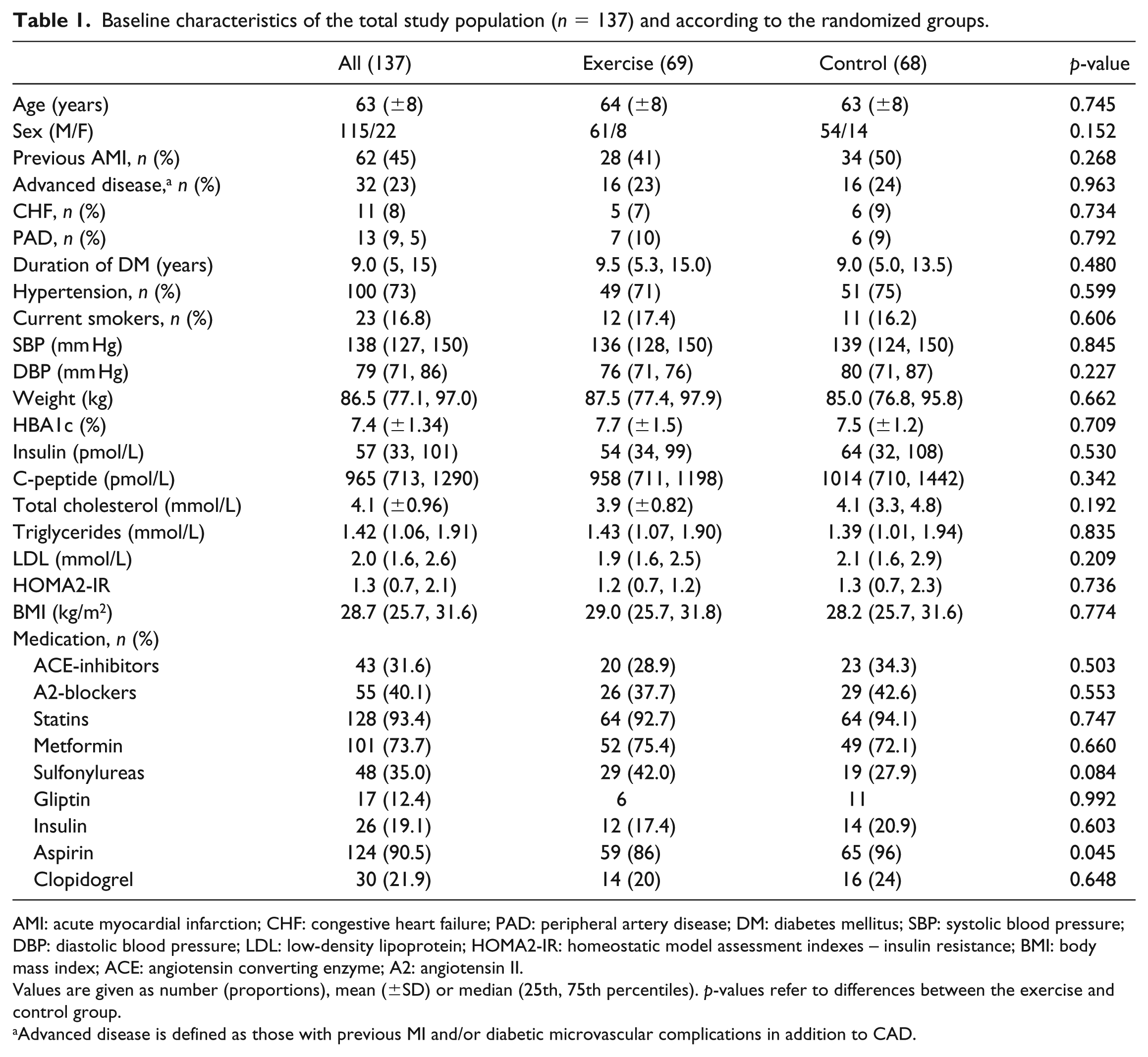
AMI: acute myocardial infarction; CHF: congestive heart failure; PAD: peripheral artery disease; DM: diabetes mellitus; SBP: systolic blood pressure; DBP: diastolic blood pressure; LDL: low-density lipoprotein; HOMA2-IR: homeostatic model assessment indexes – insulin resistance; BMI: body mass index; ACE: angiotensin converting enzyme; A2: angiotensin II.
Values are given as number (proportions), mean (±SD) or median (25th, 75th percentiles). p-values refer to differences between the exercise and control group.
Advanced disease is defined as those with previous MI and/or diabetic microvascular complications in addition to CAD.
As previously reported, there were no significant between-group differences in changes in weight, waist circumference, energy intake nutrients or diabetes medication during the study period. 16
Blood samples at baseline were available in all, whereas after intervention, the numbers of successfully analysed samples were 114 for circulating IL-18 and 107 for leukocyte gene expression of IL-18, NLRP3 and Caspase-1, respectively. The number of successfully analysed AT samples at baseline and after intervention was 102 and 64 (IL-18), 69 and 26 (Caspase-1) and 62 and 17 (NLRP3), respectively. The limited number of AT samples was mainly because of patients’ unwillingness to take fat tissue biopsies, particularly after the intervention period, and inadequate quantity and quality of the samples. Due to limited number of samples with acceptable quality, especially after intervention, results on Caspase-1 and NLRP3 in AT were excluded.
Effects of exercise training
Levels of the inflammatory mediators in the exercise and control groups before and after the intervention period are shown in Table 2 and Figure 1.
Inflammatory mediators at baseline and after 12 months of intervention in both randomized groups.
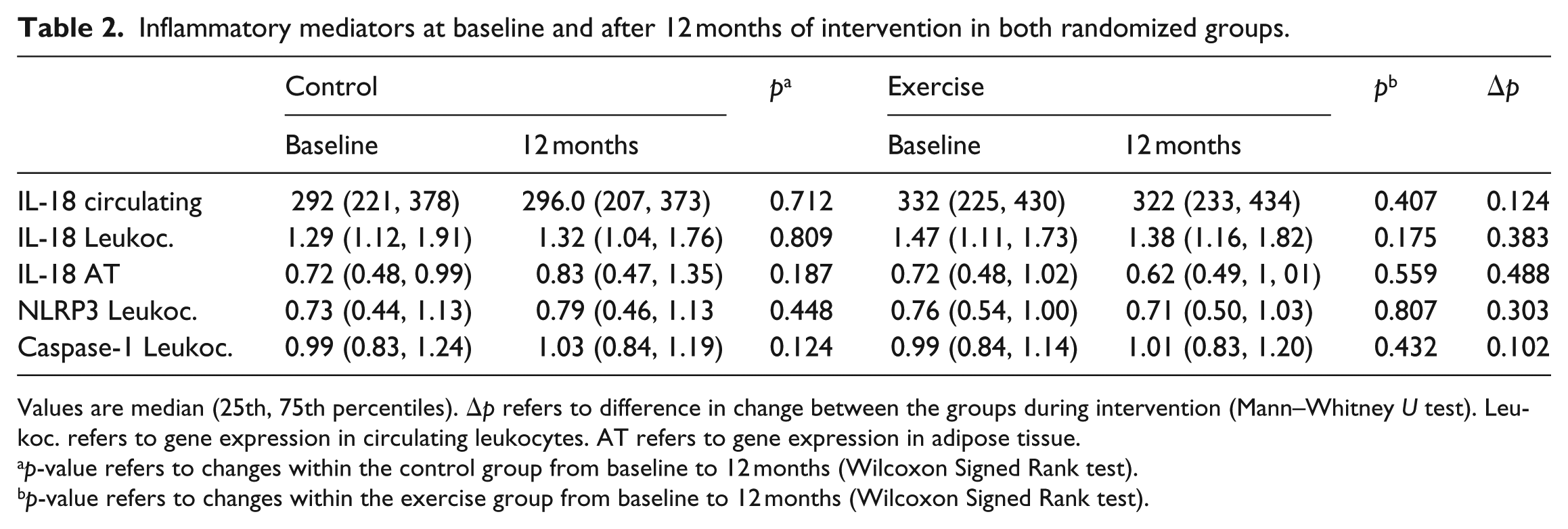
Values are median (25th, 75th percentiles). Δp refers to difference in change between the groups during intervention (Mann–Whitney U test). Leukoc. refers to gene expression in circulating leukocytes. AT refers to gene expression in adipose tissue.
p-value refers to changes within the control group from baseline to 12 months (Wilcoxon Signed Rank test).
p-value refers to changes within the exercise group from baseline to 12 months (Wilcoxon Signed Rank test).
Relative gene expression of the measured markers before and after 12 months in both groups. Relative gene expression of the measured markers in circulating leukocytes and in adipose tissue (AT) before and after 12 months of intervention.
Circulating levels
Circulating levels of IL-18 did not differ between the groups at baseline, and the difference in change between the two randomized groups did not reach statistical significance.
Gene expression in circulating leukocytes
No statistically significant differences in the expression of IL-18, Caspase-1 and NLRP3 were observed between the groups, neither at baseline and after 12 months nor in the change after intervention (Table 2 and Figure 1).
Gene expression in subcutaneous AT
There was also no statistically significant difference in the gene expression of any of the measured markers in AT before and after intervention (Table 2 and Figure 1).
Advanced vascular disease
When analysing the intervention effect according to the sub-groups of patients presenting with advanced vascular disease (n = 79), no significant difference at baseline or in change in either group after 12 months were observed, except for a more pronounced decrease in circulating IL-18 in the exercise group compared to the control group (p = 0.007) (data not shown).
Baseline associations to glucometabolic variables
Our data showed limited significant correlations between circulating IL-18 and the glucometabolic variables, except for a correlation to C-peptide (p < 0.001) (Table 3). IL-18 expression in circulating leukocytes was inversely correlated to C-peptide (p = 0.009) and body mass index (BMI) (p = 0.004), whereas IL-18 expression in AT correlated positively to insulin (p < 0.001), HOMA2-IR (p = 0.004) and BMI (p = 0.002). The expression of NLRP3 and Caspase-1 in circulating leukocytes was inversely correlated to insulin (p = 0.013 and p = 0.011, respectively), C-peptide (p < 0.001 and p = 0.004, respectively), HOMA2-IR (p = 0.002 and p = 0.001, respectively) and BMI (p = 0.002 and p = 0.007). No significant correlations between expression of NLRP3 and Caspase-1 in AT and the glucometabolic variables were observed.
Baseline correlations between glucometabolic variables and inflammatory mediators.
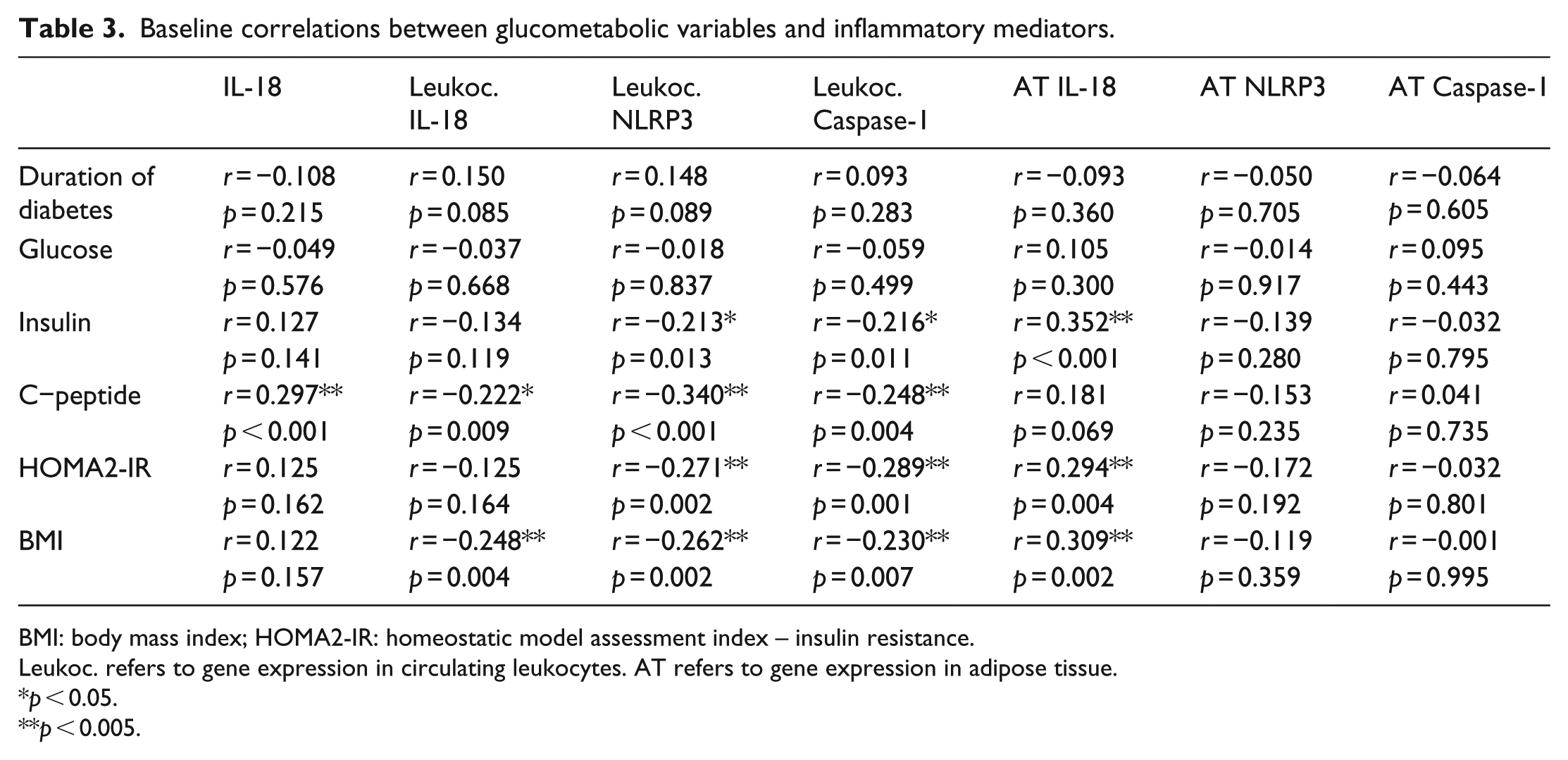
BMI: body mass index; HOMA2-IR: homeostatic model assessment index – insulin resistance.
Leukoc. refers to gene expression in circulating leukocytes. AT refers to gene expression in adipose tissue.
p < 0.05.
p < 0.005.
Inflammatory variables in CAD sub-groups at baseline
The examined sub-groups were sex, HT, smoking history and advanced vascular disease, as defined. The results are presented in Table 4.
Differences between sub-groups in baseline levels of inflammatory mediators.
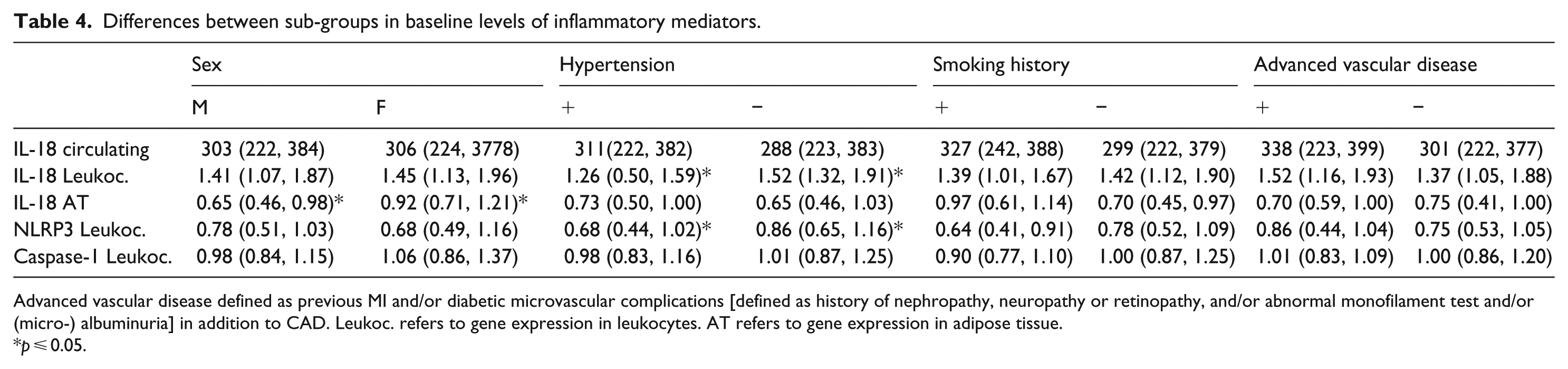
Advanced vascular disease defined as previous MI and/or diabetic microvascular complications [defined as history of nephropathy, neuropathy or retinopathy, and/or abnormal monofilament test and/or (micro-) albuminuria] in addition to CAD. Leukoc. refers to gene expression in leukocytes. AT refers to gene expression in adipose tissue.
p ⩽ 0.05.
In AT, significantly higher IL-18 expression was found in women compared to men (p = 0.03). In circulating leukocytes, lower IL-18 and NLRP3 expression were found in patients with HT compared to those without (p ⩽ 0.03, both). Otherwise, no statistically significant differences were observed, although numerically higher levels of circulating IL-18 were found in patients with HT, in smokers and in patients with advanced vascular disease.
Discussion
The main finding in our study on patients with combined T2DM and CAD was that the levels of inflammatory mediators related to the investigated IL-18 regulatory pathway did not change significantly after the exercise intervention. Some associations between the measured markers and glucometabolic variables were demonstrated, especially in relation to C-peptide and BMI. In AT, IL-18 expression was significantly more pronounced in women, whereas in patients with HT, the expression of IL-18 and NLRP3 in leukocytes was significantly attenuated.
Although significant effects on the pro-inflammatory mediators after intervention were lacking, we observed a tendency towards lower values of circulating IL-18, and lower expression of the IL-18 gene in AT and in leukocytes after 12 months. Looking at the population, especially taken into consideration their age, weight, established CAD and several years with T2DM, it raises the question whether this population had too advanced disease to achieve the hypothesized effect of reduced inflammation after 12-month exercise training. It is also intriguing whether the amount of exercise training was sufficient to detect the expected effects. As previously reported, 16 there was no significant change in weight in this group, again reflecting the possibility of insufficient exercise training, especially when taken into consideration that AT is regarded as an important source for the production of the investigated inflammatory mediators. Previous studies have shown an association between body weight reduction and plasma levels of IL-18 18 and between BMI and expression of IL-18 in AT. 19 This may indicate AT to be an important source of IL-18 production and might reflect a close relationship between a pro-inflammatory state and obesity. Furthermore, a small study with 23 obese subjects that underwent a 15-week lifestyle intervention with an average weight loss of 18 kg, showed a 22% reduction in circulating IL-18. 20 There are, however, studies showing reduction of pro-inflammatory markers after exercise, regardless of weight change. A small study conducted on 36 subjects with metabolic syndrome, showed reduced serum levels of IL-18 in patients that underwent a 12-week training programme or received pravastatin, compared to the control group. 21 Another study showed a significant decrease in IL-18 without significant change in weight after exercise in subjects with T2DM, without vascular complications. 22 This indicates that reduction in the pro-inflammatory markers is achievable without significant weight change, though the subjects in these studies seemed to have less disease progression compared to our study population.
The effects of exercise training on gene expression of IL-18 and its regulators in leukocytes have, to our knowledge, not previously been reported in humans. There is also limited data on exercise effects on NLRP3 and Caspase-1. Ringseis et al. 23 were able to demonstrate that resistant and endurant exercise in obese mice on a high fat diet lead to a reduction in AT expression of NLRP3 after a 10-week exercise programme.
At baseline, we found a significant correlation between expression of IL-18 in AT and BMI, insulin and HOMA2-IR. AT expression of IL-18 has been reported to be positively correlated to insulin resistance, and the observed reduction in AT IL-18 expression after exercise training was thought to partly be due to the beneficial effects on insulin resistance.20,24 This is in accordance with our hypothesis of inflammation being closely linked to not only obesity per se but also a pathological metabolic state.
The baseline findings with the inverse correlations between gene expression of NLRP3 and Caspase-1 in circulating leukocytes and the glucometabolic variables could be indicative of a downregulatory mechanism of their expression in leukocytes with increasing insulin production and insulin resistance. This poses the question of the leukocytes potential protective role in this particular setting, also highlighted by the observed inverse correlation between gene expression of IL-18 in circulating leukocytes and C-peptide and BMI in our study. This is partly consistent with our recently published results from a study conducted on young adult men, demonstrating an inverse correlation between leukocyte expression of NLRP3 and insulin, C-peptide and BMI. In that particular study, we also showed even stronger associations between glucometabolic variables and circulating and AT gene expression of IL-18 compared to this study. 25 This difference between our two studies may, in part, be explained by the comorbid population in this study, heavily medicated on statins and anti-diabetic drugs, hence an altered glucometabolic state and a different composition of the AT is most likely present. However, the clear similarities between our two studies are interesting and supports our claim that glucometabolic variables are associated with an inflammatory state, particularly in AT.
We could further demonstrate a correlation between circulating IL-18 and C-peptide, which is partly in accordance with a study on young female participants showing a possible role of C-peptide in predicting CVD. 26 This might be suggestive of an increased inflammatory activity in patients with increased insulin production and overweight, which is also consistent with other previous findings20,25 and our initial assumptions. Overweight and insulin resistance seems to be closely linked to gene expression of IL-18 in AT as discussed previously.20,24 C-peptide is widely used and accepted in clinical practice as a measure of β-cell function and insulin resistance. 27 It is co-secreted with insulin in equimolar amounts in the islets of Langerhans. The kinetics of its degradation is not exactly the same as insulin, but its concentration in serum does closely reflect the insulin-producing capacity of β-cells in pancreas. IL-18 in combination with IFN-γ seems to be involved in the impairment of β-cell function by apoptotic cell death, at least in vitro. 28
In the sub-group analyses, we found higher levels of AT IL-18 expression in women compared to men. In hypertensive patients, lower levels of IL-18 and NLRP3 expression in leukocytes were observed compared to normotensive patients, which further supports the theory of leukocytes’ possible protective role in this setting. An association between HT and elevated levels of circulating and vascular levels of IL-18 and its regulators has previously been reported, but it is unknown whether elevated circulating IL-18 is a cause or a mere consequence of long-standing elevated blood pressure. 29
Patients with previous AMI and/or diabetic microvascular complications were defined as those with advanced vascular disease. When stratifying patients by these criteria in the main EXCADI study and excluding patients with advanced vascular disease, an improvement in HbA1c and VO2max was observed in the exercise group. 16 We could, however, not demonstrate any differences on the investigated markers in this sub-group in the present investigation.
Limitations
The limitations of this study is that it was initially designed for the purpose of measuring effects on HbA1c after exercise training; 16 hence, the initial power calculations have not considered effects on the investigated pro-inflammatory mediators. The subjects in our study were also heavily medicated with ACEI/A2 blockers, statins, platelet inhibitors and anti-diabetics. Some of these medications, like statins (93% users) and platelet inhibitors (94% users), are known to have anti-inflammatory properties, 30 which may have suppressed the values of the investigated inflammatory mediators, thereby masking potential effects of exercise training. As occurs from Table 1, there was significant higher number of aspirin users in the control group. In AT tissue, aspirin has been shown to reduce expression of the pro-inflammatory cytokines IL-1β and IL-6 in macrophages and further to increase IL-10 expression, which have anti-inflammatory properties. 31 The effects of aspirin specifically on IL-18, Caspase-1 and NLRP3 have, to the best of our knowledge, not previously been reported on.
Similarly, there was higher number of sulfonylurea users in the control group, the difference approaching statistical significance. Sulfonylurea seems to possess several anti-inflammatory attributes, such as suppressing the pro-inflammatory cytokines IL-8 and IL-1β production by leukocytes.32,33
Due to limited number of AT samples after intervention, effects on Caspase-1 and NLRP3 could not be investigated. The strength of this study was the randomization principle with long-term intervention with equal distribution of the study population and few differences between groups at baseline.
Conclusion
We could not demonstrate any significant effect of long-term exercise intervention on the pro-inflammatory mediator IL-18 and the upstream activators Caspase-1 and NLRP3 in our population of patients with a combination of CAD and T2DM.
We did observe inverse associations between leukocyte expression of IL-18, NLRP3 and Caspase-1 and glucometabolic variables, indicating their protective role. A close link between overweight, insulin resistance and expression of IL-18 in AT, probably representing an increased inflammatory state in AT, was confirmed.
This study may indicate that a population with less comorbidities and less medicated would benefit more from exercise training to reduce the inflammatory burden than the population in our study.
Footnotes
Acknowledgements
The authors thank Vibeke Bratseth and Beate Vestad for laboratory assistance. Trial registration: clinicaltrials.gov, NCT01232608.
Availability of data and materials
The data sets used and analysed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stein Erik Hagen Foundation for Clinical Heart Research and the Ada og Hagbart Waages Humanitære og Veldedige Stiftelse, Oslo, Norway.
