Abstract
We investigated the effects of 12-month exercise training on hypercoagulability in patients with combined type 2 diabetes mellitus and coronary artery disease. Associations with severity of disease were further explored. Patients (n = 131) were randomized to exercise training or a control group. Blood was collected at inclusion and after 12 months. Tissue factor, free and total tissue factor pathway inhibitor, prothrombin fragment 1 + 2 (F1 + 2) and D-dimer were determined by enzyme-linked immunosorbent assay and ex vivo thrombin generation by the calibrated automated thrombogram assay. Tissue factor and ex vivo thrombin generation increased from baseline to 12 months (p < 0.01, all), with no significant differences in changes between groups. At baseline, free and total tissue factor pathway inhibitor significantly correlated to fasting glucose (p < 0.01, both) and HbA1c (p < 0.05, both). In patients with albuminuria (n = 34), these correlations were strengthened, and elevated levels of D-dimer, free and total tissue factor pathway inhibitor (p < 0.01, all) and decreased ex vivo thrombin generation (p < 0.05, all) were observed. These results show no effects of exercise training on markers of hypercoagulability in our population with combined type 2 diabetes mellitus and coronary artery disease. The association between poor glycaemic control and tissue factor pathway inhibitor might indicate increased endothelial activation. More pronounced hypercoagulability and increased tissue factor pathway inhibitor were demonstrated in patients with albuminuria.
Keywords
Introduction
Patients with type 2 diabetes mellitus (T2DM) are at significant risk of developing cardiovascular disease (CVD) and having diabetes has been proposed as an equivalent to coronary artery disease (CAD) regarding risk for future cardiovascular events. 1 Endothelial cell activation, with change of phenotype to be proinflammatory and procoagulant, is thought to be the initial step in the pathogenesis of atherosclerosis and plaque development, the most important underlying condition of CAD.2,3 By plaque rupture or erosion, collagen and sub endothelial tissue factor (TF) are exposed to circulating blood, thus triggering the coagulation cascade with subsequent thrombin formation, which may lead to acute manifestations like acute coronary syndrome. 2
Patients with combined T2DM and CAD have more extensive atherosclerosis and thrombotic burden, 4 and hyperglycaemia is supposed to be one of the driving forces in this process. Also insulin resistance, typically present in T2DM, triggers atherothrombosis by inducing endothelial dysfunction, and hyperinsulinaemia has further been shown to increase TF procoagulant activity in T2DM patients, with subsequent accelerated thrombin generation.5–8 Albuminuria characterizes the development of kidney disease in diabetic patients and these patients are known to be hypercoagulable and often die of CVD complications.9,10
Regular physical exercise is well established in the management of both T2DM and CAD.11,12 In addition to improvement of conventional risk factors and glycaemic control, exercise training may have a favourable effect on the delicate balance of coagulation and fibrinolysis. 13
The calibrated automated thrombogram (CAT) is an overall function test of the haemostatic system that is sensitive to prothrombotic changes in plasma.14,15 The most used parameter, the endogenous thrombin potential (ETP), represents the total ex vivo thrombin activity and reflects the interplay between all relevant proteases and inhibitors in plasma. Furthermore, soluble tissue factor (sTF), free and total tissue factor pathway inhibitor (TFPI) and the coagulation activation markers prothrombin fragment 1 + 2 (F1 + 2) and D-dimer are useful tools to estimate the degree of in vivo hypercoagulability.
The aim of this study was to investigate the effects of 1 year of organized physical exercise on the degree of hypercoagulability, assessed by the above-mentioned parameters representative for both in vivo and ex vivo status, in patients with combined T2DM and CAD with the hypothesis that exercise has a favourable effect on the prothrombotic state in these high-risk patients. In addition, any associations between the haemostatic and glucometabolic variables and severity of disease were explored.
Methods
Study population
The study population (n = 137) consisted of patients with angiographically verified stable CAD and known T2DM. Subjects were enrolled at Department of Cardiology, Oslo University Hospital, Ullevål, Norway, between August 2010 and March 2012. The main study was a randomized clinical trial investigating the effects of 12 months physical exercise on progression of atherosclerosis and glucometabolic control.16,17 Exclusion criteria included severe diabetic complications, cancer, stroke or acute coronary syndrome within the last 3 months prior to inclusion, unstable angina, uncompensated heart failure, serious arrhythmia, serious valvular disease, serious rheumatological diseases, chronic obstructive pulmonary disease stadium GOLD IV, ongoing infections, thromboembolic disease, severe musculoskeletal disorders and other illnesses seriously limiting the ability for participating in physical activity. At inclusion, the subjects were randomized 1:1 to a combination of endurance and strength training or a control group with conventional follow-up by their general practitioner.
The regional Ethics Committee has approved the study, and all patients have given written informed consent to participate. The study was conducted in accordance with the Declaration of Helsinki and is registered at http://www.clinicaltrials.gov, NCT01232608.
Physical exercise intervention
The physical exercise programme was developed and conducted in collaboration with the Norwegian school of Sport Sciences. Details on the exercise programme have been previously described. 16 Briefly, the exercise group had two group-based sessions per week with qualified instructors, and a third home-based session, for a total volume of at least 150 min/week. About two-thirds were aerobic, and one-third resistance training and each of the supervised sessions included parts with high-intensity interval training.
Laboratory methods
Venous blood samples were drawn in fasting condition by standard venipuncture (21-gauge needle) between 08:00 and 10:00 AM, before intake of morning medication, at inclusion and after 1 year. Blood collected in Vacutainer tubes, containing 0.129 M trisodium citrate in dilution 1:10, was kept on ice until centrifugation at 3000g for 20 min at 4°C within 1 h. Plasma was stored frozen at −80°C until analysed. Routine blood samples, including HbA1c and insulin, were determined by conventional methods. To estimate insulin resistance, the updated homeostatic model assessment 2 of insulin resistance (HOMA2-IR) was applied.
Levels of the coagulation markers sTF, TFPI, F1 + 2 and D-dimer were determined by conventional enzyme immunoassays; Imubind TF kit recognizing TF-apolipoprotein, sTF and TF-VII complexes (American Diagnostic Inc., Greenwich, CT, USA), Asserachrom® free and total TFPI antigen, recognizing the full-length TFPI molecules, and the full-length and truncated TFPI molecules including TFPI bound to lipoproteins, respectively (Stago Diagnostica, Asniere, France), Enzygnost® F1 + 2 (monoclonal) (Siemens, Marburg, Germany) and Asserachrom® D-dimer (Stago Diagnostica), respectively. Inter-assay coefficients of variations (CVs) were 10.5%, 9.0%, 6.5%, 10.0% and 7.2%, respectively.
Ex vivo potential to generate thrombin was analysed by the CAT assay, conducted according to the manufacturer’s instruction (Thrombinoscope BV, Maastricht, The Netherlands). The method is described in detail elsewhere. 14 Briefly, citrated platelet poor plasma (PPP) was mixed with recombinant relipidated TF and phospholipids, with the final concentrations of 5 pM and 4 µM, respectively. Every experiment was performed with two separate readings, one for thrombin generation and a second, to which an inert thrombin calibrator with known concentration is added, to enable calculation of the results. A freshly made starting reagent containing CaCl2 (100 mM) and a thrombin-specific fluorogenic substrate (Z-Gly-Gly-Arg-AMC) (2.5 mM) in Hepes buffer were automatically added. The fluorescence intensity was recorded by the Fluoroskan Ascent® microplate fluorimeter (Thermo Fisher Scientific Oy, Vantaa, Finland). Reagents were provided by Thrombinoscope BV. The software (Thrombinoscope BV, version 3.0.0.29) enabled calculation of the thrombogram parameters, which include the lag time (LT, min), time to peak (ttPeak, min), peak height (PeakH, nM), the ETP (nM/min) and the slope of thrombin formation, named velocity index (VI). The respective inter assay CVs were 14.6%, 10.2%, 12.4%, 8.5% and 23.2%.
Statistical methods
Continuous data were mostly skewed and the data are presented as median values with 25th and 75th percentiles. Categorical data are presented as number and proportions. Spearman rank test was used for correlation analyses. Adjusted correlations were calculated by partial rank correlations. Mann–Whitney U test, independent samples T-test, Kruskal–Wallis test and chi-square test were applied for group comparisons, as appropriate. Wilcoxon’s test was used to explore within group changes, and for differences in changes between the randomized groups, Mann–Whitney U test was applied. p-values ≤0.05 were considered statistically significant. The statistical analyses were performed by IBM© SPSS© statistics for windows, v 21.0 (IBM Corp., New York, NY, USA).
Results
Of the total 137 patients included in the main study, 6 patients were on warfarin and have been excluded. Baseline characteristics of the population (n = 131) are presented in Table 1. Median age was 64 years, 83% were men, 57 patients (44%) presented with previous acute myocardial infarction (AMI) and 32 (25%) with albuminuria. A total of 14 patients did not complete the study and 9 participants were excluded due to low adherence to exercise intervention. These patients had an average exercise adherence of <40%. For the 108 patients who were analysed for the intervention effects, the average adherence was 67% (55%, 83%) and 21 patients had an average of >70%. 16 Clinical data did not differ between the randomized groups (Table 1).
Baseline characteristics of the total population (n = 131) and of the cohort completing the study according to the randomized groups (n = 108).
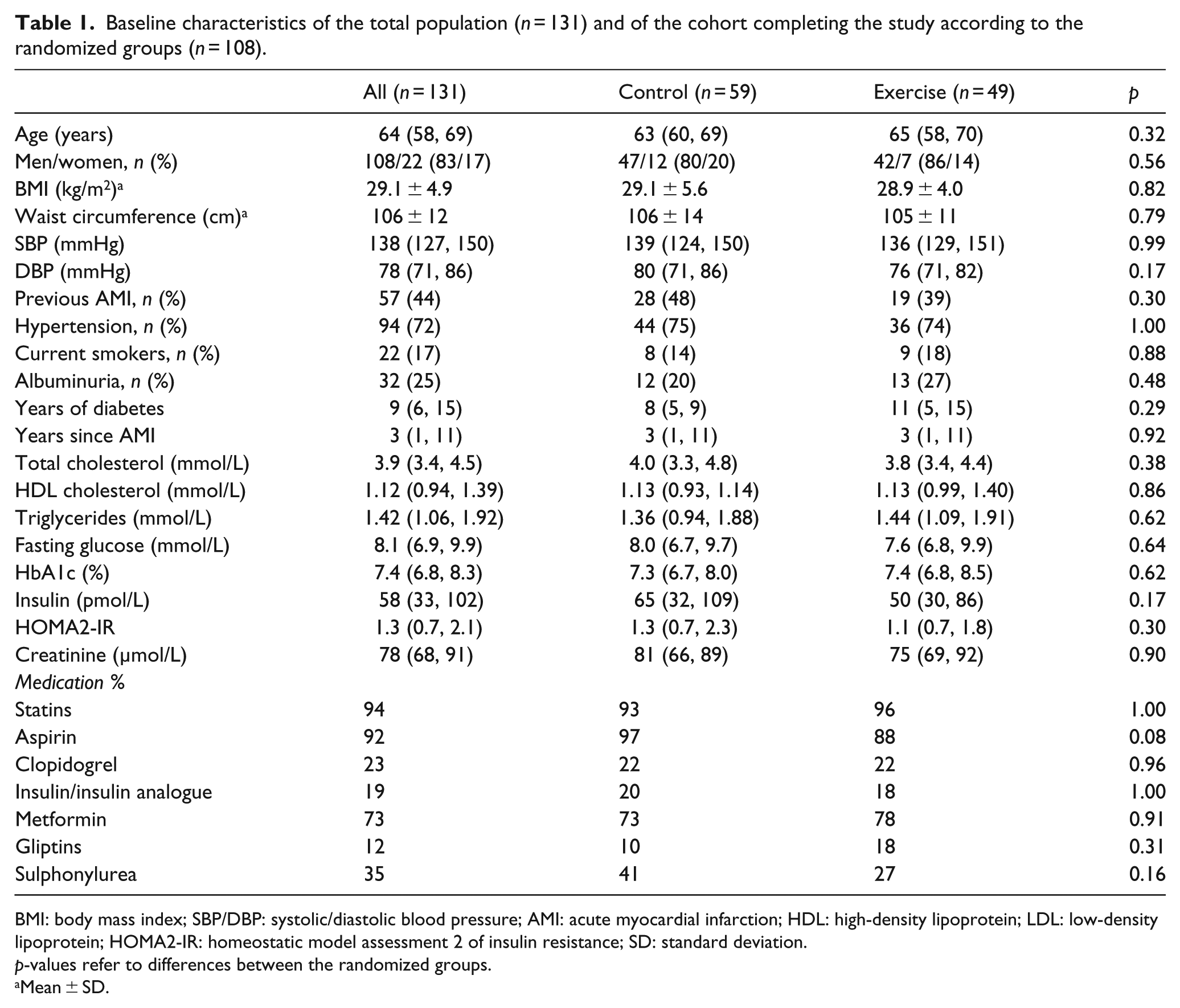
BMI: body mass index; SBP/DBP: systolic/diastolic blood pressure; AMI: acute myocardial infarction; HDL: high-density lipoprotein; LDL: low-density lipoprotein; HOMA2-IR: homeostatic model assessment 2 of insulin resistance; SD: standard deviation.
p-values refer to differences between the randomized groups.
Mean ± SD.
Effects of exercise training
After 12-month intervention, an increase in TF (p = 0.004) and ex vivo thrombin generation, the latter expressed by increased PeakH (p = 0.002) and VI (p = 0.001), and a reduction in ttPeak (p = 0.006) were observed in the exercise group. Increased ex vivo thrombin generation was also observed in the control group, shown by PeakH (p = 0.002), VI (p < 0.001) and a reduced ttPeak (p = 0.001). There were, however, no significant differences in changes between the groups (Table 2), also when baseline levels were taken into account for free and total TFPI which differed between the groups at baseline (p = 0.003 and p = 0.036, respectively).
Markers of procoagulant activity at baseline and after 12 months in the randomized groups.
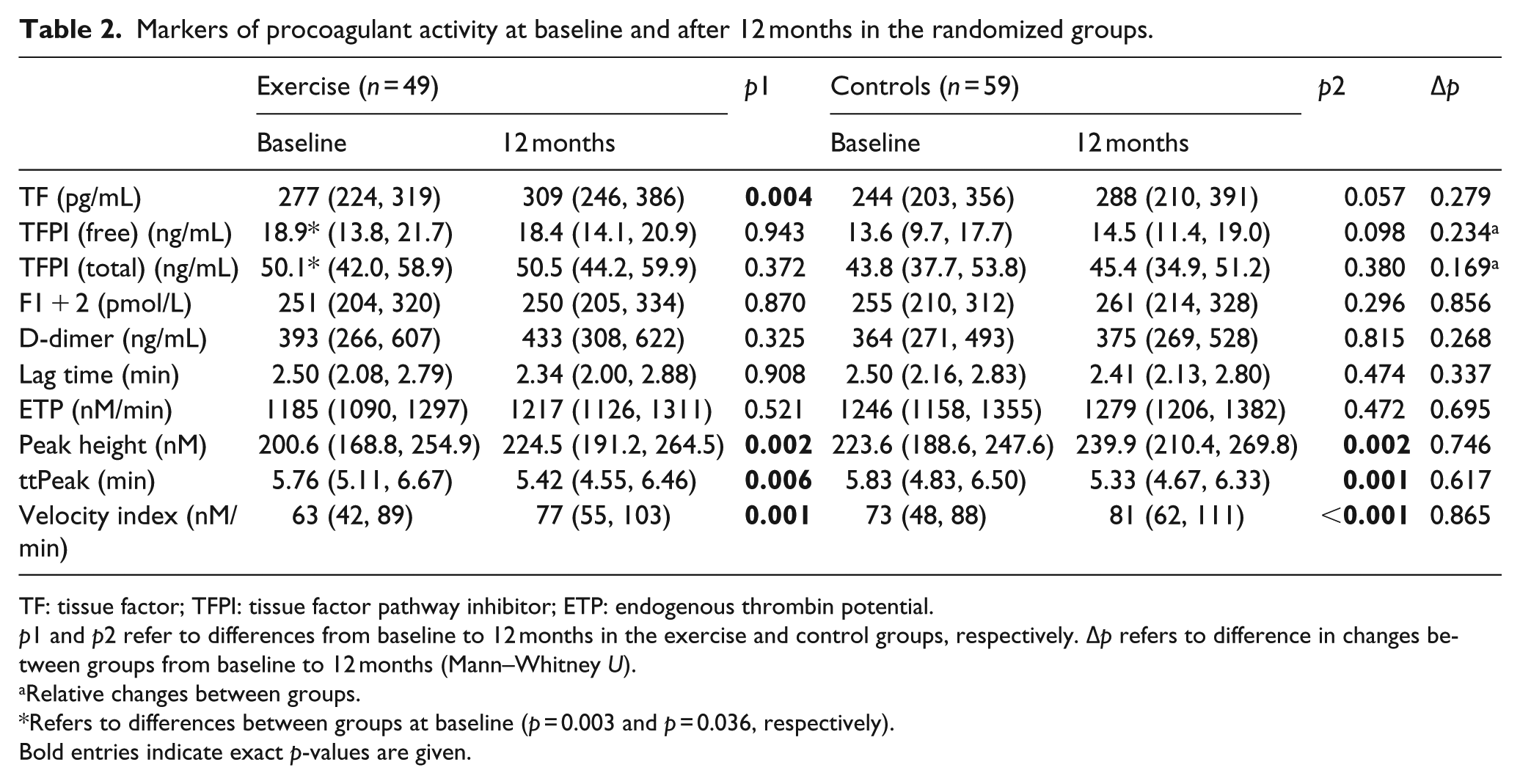
TF: tissue factor; TFPI: tissue factor pathway inhibitor; ETP: endogenous thrombin potential.
p1 and p2 refer to differences from baseline to 12 months in the exercise and control groups, respectively. Δp refers to difference in changes between groups from baseline to 12 months (Mann–Whitney U).
Relative changes between groups.
Refers to differences between groups at baseline (p = 0.003 and p = 0.036, respectively).
Bold entries indicate exact p-values are given.
Excluding patients with albuminuria (n = 25) did not have any impact on the results (data not shown). There were also no differences in changes between groups when analysed separately for patients with previous myocardial infarction (MI) (n = 47) or not (data not shown).
There was no difference in changes during the intervention period between the groups in HbA1c levels, although a numerical reduction was observed in the exercise group [7.3 (6.8, 8.0)% to 7.4 (6.5, 8.2)% in controls vs 7.4 (6.8, 8.4)% to 7.2 (6.6, 7.8)% in the exercise group] (p = 0.24, for difference in changes between groups). There were also no significant differences in changes between the randomized groups in weight, waist circumference, energy intake of main nutrients or diabetes medication during the study period (data not shown).
Baseline associations
In the total population (n = 131), free and total TFPI were significantly correlated to fasting glucose (p < 0.01, both) and to HbA1c (p < 0.05, both) (Table 3).
Baseline correlations (Spearman’s rho) between markers of procoagulant activity and glucometabolic variables in the total population (n = 131).
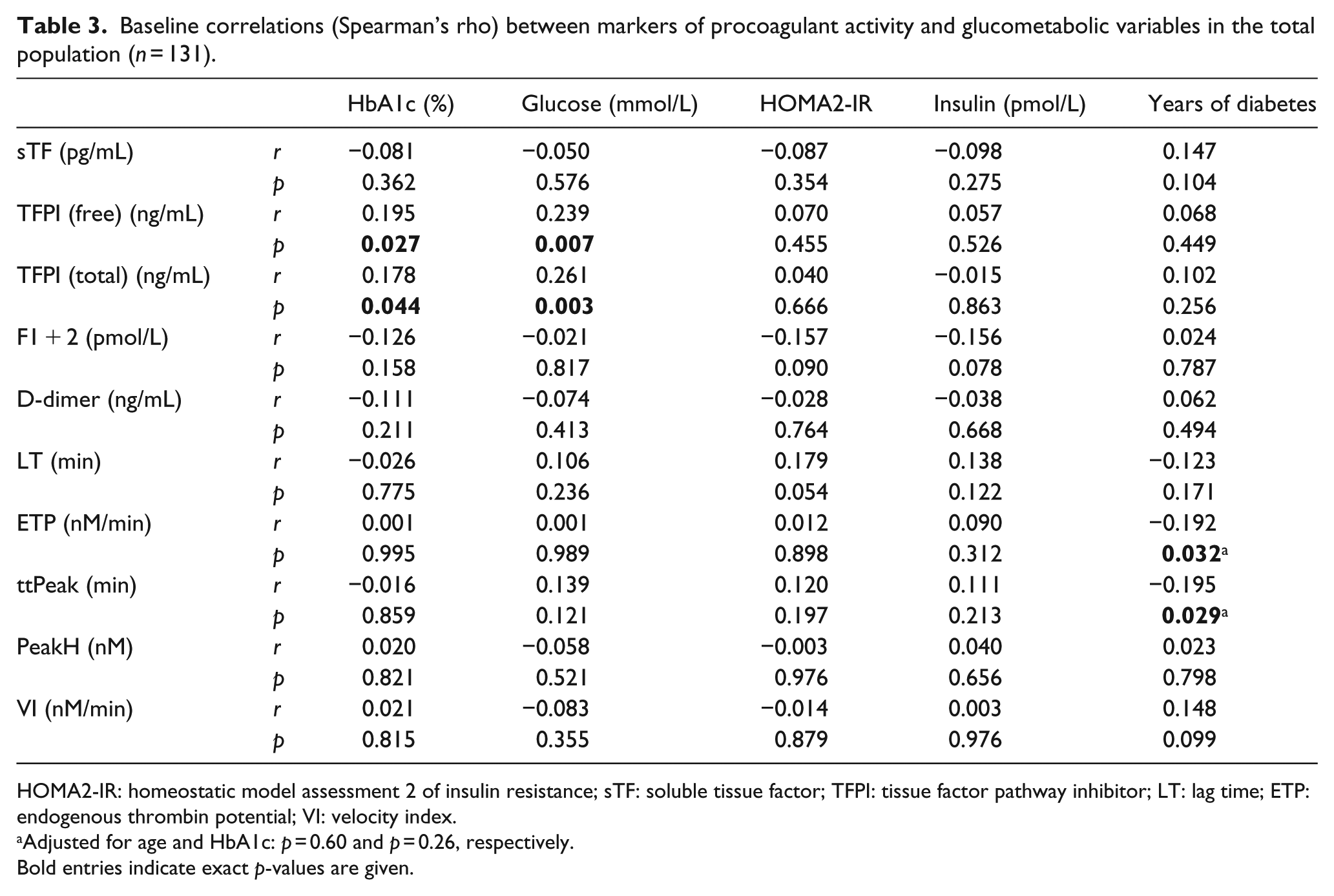
HOMA2-IR: homeostatic model assessment 2 of insulin resistance; sTF: soluble tissue factor; TFPI: tissue factor pathway inhibitor; LT: lag time; ETP: endogenous thrombin potential; VI: velocity index.
Adjusted for age and HbA1c: p = 0.60 and p = 0.26, respectively.
Bold entries indicate exact p-values are given.
Years of diabetes was inversely associated with ETP and ttPeak (p < 0.05, both) (Table 3); however, no longer significant after correction for age and HbA1c (p = 0.60 and p = 0.26, respectively). No other significant correlations between glucometabolic variables and the coagulation markers were observed. Furthermore, the levels of free and total TFPI were significantly inter-correlated to TF (r = 0.207 and r = 0.296, respectively, p < 0.05, both).
In the subgroup of patients having albuminuria (n = 32), the correlations between free and total TFPI and fasting glucose and HbA1c were strengthened (r = 0.476 and r = 0.503, respectively, p < 0.01, both) and (r = 0.352 and r = 0.444, respectively, p < 0.05, both).
These patients, as compared to those without albuminuria, had elevated levels of D-dimer, free and total TFPI (p < 0.05, all) (Figure 1, upper panel) in addition to delayed, slower and lower ex vivo thrombin generation, expressed by LT, ttPeak, VI and PeakH (p < 0.01, all) (Figure 1, lower panel). In patients with previous MI, no differences in any of the markers were observed (data not shown).
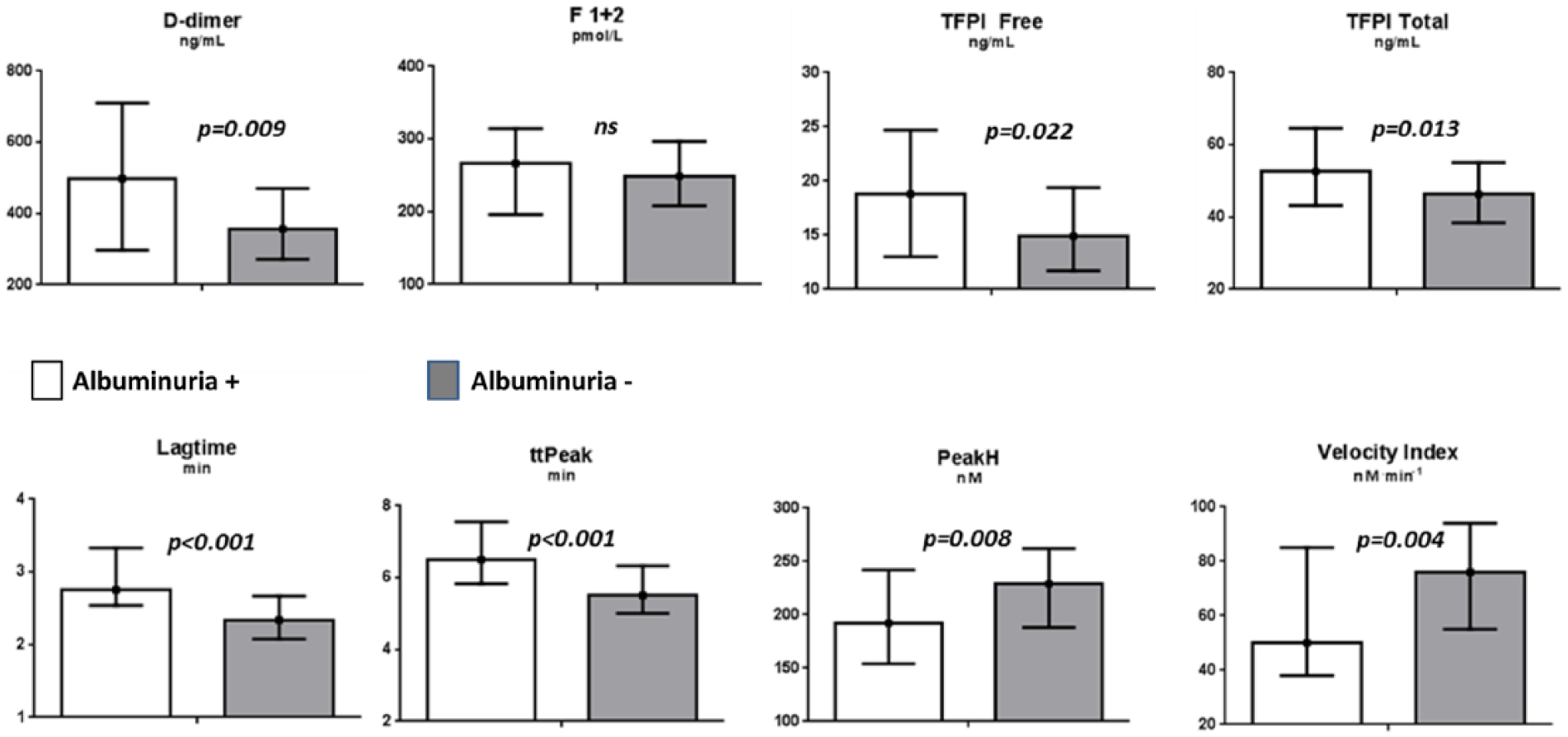
Levels of in vivo haemostatic variables (upper panel) and ex vivo thrombin generation (lower panel) according to patients with albuminuria (n = 32) (white column) and without (black column). Values refer to medians and 25th and 75th percentiles.
Discussion
In this randomized study, 1 year of organized physical exercise in patients with combined T2DM and CAD did not result in significant changes different from controls in the selected markers of procoagulant activity, also not when excluding patients with more severe disease states, that is, albuminuria or previous MI. In both groups, increased levels of TF and ex vivo thrombin generation were recorded at the end of the study.
At baseline, poor glycaemic control, assessed by HbA1c and fasting glucose, was significantly associated with levels of TFPI (free and total). These associations were strengthened when analysed in the subgroup of patients presenting with albuminuria, in which also the levels of TFPI and D-dimer were elevated and the ex vivo thrombin generation was decreased.
To the best of our knowledge, long-term effect of exercise training on markers of hypercoagulability in patients with combined CAD and T2DM has previously not been reported on. We observed, however, no differences in changes between the randomized groups in any of the measured variables, although our hypothesis was to achieve a shift towards a less prothrombotic state in the exercise group. This was also our hypothesis with regard to TFPI, which might be discussed to reflect endothelial activation. 18
Compliance to the study intervention for a period of 1 year is a challenge in our patient population. The mean adherence to the exercise intervention was 67% 16 which might be discussed not to be sufficient to achieve an effect on procoagulant activity. However, 21 of these patients obtained an adherence of >70%, assuming in total the compliance to be satisfactory.
In a study on patients with T2DM alone, randomized to only aerobic exercise or not, significantly reduced concentration of tissue plasminogen activator (tPA) antigen was shown, which could reflect reduced thrombotic potential as well as improved endothelial activation. 19 In a group of overweight men supposed to have prothrombotic traits similar to T2DM, a 3-month period with daily endurance exercise training resulted in significantly reduced levels of ETP in the exercise group compared to controls, also independent of exercise dose. 20 This is somewhat in contrast to our results; however, CAD patients were not included in that particular study. Also in post-MI patients, it has been shown that exercise training results in reduced procoagulant activity, assessed by activated prothrombin time (aPTT). 21
The combination of endurance and resistance exercise in our study was originally chosen in order to have good impact on glycaemic control. We did, however, obtain limited changes in HbA1c and also no weight change during the intervention, and it might be discussed if this combination is the best modality for dampening hypercoagulability. However, Ponjee et al. 22 who investigated prolonged effects of endurance exercise alone in sedentary subjects could also not demonstrate any changes in levels of von Willebrand factor, an endothelial marker, or in thrombin–antithrombin complex concentration.
The increase in levels of TF, the main activator of the extrinsic pathway of coagulation, shown in our study, might reflect a raise in prothrombotic potential, although in vivo coagulation per se might not be activated. These findings are strengthened by the increased and accelerated ex vivo thrombin generation. Why these changes occur also in controls is not easily explainable. Elevated levels of circulating TF have previously been demonstrated in both diabetes mellitus (DM) and CAD.23,24
Duration of diabetes was shown to be inversely related to ex vivo prothrombotic activity in univariate analyses; however, the correlation was no longer significant after adjusting for age and glycaemic control.
Diabetes patients who have developed albuminuria are supposed to have more severe and complicated disease state. We could, however, not find any effect on the measured markers also when those with albuminuria were excluded. We could, nevertheless, show that in this subgroup of patients, the coagulation system was activated, demonstrated by increased D-dimer levels. Furthermore, both free and total TFPI were elevated and the baseline correlations between the prothrombotic markers, especially TFPI, were strengthened, indicating that these patients have increased endothelial activation. Wakabayashi and Masuda 25 also showed D-dimer to be related to microalbuminuria, which is in accordance with our findings, suggesting a link between endothelial dysfunction, activation of the coagulation system and diabetic nephropathy. The reduced ex vivo potential for thrombin generation found in our study might be discussed to be due to ongoing in vivo coagulation, resulting in lower potential for ex vivo generation.
In contrast to our finding, enhanced ex vivo thrombin generation in T2DM patients with macrovascular disease, of which 46% of the total population (n = 160) had CVD, has been reported, and also significantly elevated ETP in patients with albuminuria, as compared to those without. 26 In this study, patients with previous MI did not present with higher procoagulant activity. When analysed separately, no benefit of exercise training was obtained in this subpopulation. Thus, it seems that coronary heart disease severity did not influence the results.
The significant correlations shown at baseline between levels of TFPI and HbA1c and fasting glucose, but not other glucometabolic variables, indicate that hyperglycaemia rather than insulin resistance causes the raise in TFPI. Our results are in line with previous reports showing a positive correlation between HbA1c and plasma activity levels of TFPI,27,28 and a decrease in TFPI levels after optimization of glycaemic control has also been demonstrated. 29 Several studies have demonstrated increased levels of TFPI in diabetic patients compared to healthy controls.30,31 The significant interrelation between TF and TFPI we observed might indicate a compensating mechanism in order to attenuate the TF-dependent pathway of coagulation. This might also be discussed along with the lack of associations between glucometabolic control and variables of thrombin generation. This is supported by the report on insulin-dependent DM patients, in which TFPI was found to dampen the hypercoagulable state, demonstrated by inhibition of factor Xa activity, however not sufficient to inhibit in vivo thrombin generation, shown by increased levels of F1 + 2. 30 In another study, accelerated thrombin generation was demonstrated in patients with diabetes as compared to healthy controls, with the most pronounced increase in the poorly controlled group. 31
Study limitations
The mean adherence to the exercise intervention in our study was 67% 16 which might have been too liberal in order to achieve an effect on procoagulant activity. In addition, study participants being engaged in physical exercise at the time of inclusion were not excluded, but their training programme should not exceed the one in the study. The main study was powered for between-group difference in HbA1c 14 and not for change in markers of hypercoagulability. The study participants were well controlled for their diabetes.
Conclusion
We could not demonstrate any effects of exercise training on markers of procoagulant activity in our population of patients with combined T2DM and CAD. However, both groups experienced increased coagulation potential after 12 months, which might be discussed along with progression of diseases. The significant association between poor glycaemic control and levels of TFPI observed might be indicative of increased endothelial activation. A more pronounced in vivo procoagulant state, as well as increased TFPI, was demonstrated in a subgroup of patients presenting with albuminuria.
Footnotes
Acknowledgements
The authors thank Sissel Åkra and Beate Vestad for valuable laboratory assistance to the study.
Authors’ contribution
H.A., S.S., R.B., I.S. and V.B. contributed to study conception and design; V.B., R.B, I.U.N. and I.S. contributed to execution of experiments and data analyses; V.B., H.A., S.S. and I.S. contributed to result interpretation; V.B. and I.S. contributed to drafting the manuscript; V.B., R.B., I.U.N., S.S., H.A. and I.S. contributed to manuscript revision and editing; and V.B. and I.S. contributed to manuscript approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Stein Erik Hagen Foundation for Clinical Heart Research and ‘Ada og Hagbart Waages Humanitære og veldedige stiftelse’, Oslo, Norway.
