Abstract
Background:
Silent coronary artery disease is a frequent complication of type 2 diabetes (T2DM). Based on its multiple roles in inflammation, atherogenesis and glucose homeostasis, we hypothesised that activin A could be related to coronary atherosclerosis in T2DM.
Methods:
Activin A and follistatin were measured in 102 patients with T2DM and 20 age- and sex-matched healthy controls. Coronary angiography was performed in a sub-population of patients and associations with activin A were examined using multiple linear regression.
Results:
Serum activin A and the activin A/follistatin ratio were increased in patients with T2DM and coronary artery disease (CAD) compared with healthy volunteers and the elevated activin A was associated with the severity of coronary atherosclerotic burden as determined by the proportion of ≥2 vessel disease (p = 0.035) after multivariable-adjusted trend analysis. No significant association between presence of CAD or extent score and activin A was observed.
Conclusion:
In patients with T2DM, increased activin A may reflect chronic underlying pathophysiological processes involved in development of cardiovascular disease.
Introduction
Patients with type 2 diabetes mellitus (T2DM) have significantly increased risk of cardiovascular disease (CVD), including coronary artery disease (CAD), which is the leading cause of premature morbidity and mortality in these patients.1,2 The true prevalence of CAD in T2DM is often underestimated as classical symptoms of CAD may be absent.1,2 Thus, we and others have demonstrated a high prevalence of asymptomatic CAD in patients with moderate to long-standing T2DM.3–5
Low-grade inflammation plays an important role in the pathogenesis of T2DM and CAD, and may not only promote atherogenesis, but also contribute to insulin resistance and beta-cell impairment. 6 Activin A, a member of the transforming growth factor beta superfamily, may play an important role in glucose homeostasis by modulating insulin secretion. 7 Activin A may also modulate inflammatory responses and play a role in angiogenesis, vascular remodeling and development of atherosclerosis.8,9 In an experimental model, increased hepatic expression of activin A associated with high circulating levels, inhibited the formation of vascular smooth muscle cell (VSMC) hyperplasia in mice challenged by carotid artery ligation. 9
The activity of activin A is attenuated by its endogenous inhibitor follistatin, and the activin A/follistatin ratio may be of importance for activin A-mediated effects. 10 Based on the multiple roles of activin A in inflammation, atherogenesis and glucose homeostasis, we hypothesised that activin A and follistatin could be related to coronary atherosclerosis in T2DM. This was investigated by evaluating serum activin A and follistatin in patients with T2DM with and without CAD and assessing associations between these parameters and coronary angiographic scores in these patients.
Patients and methods
Study population
The present study is a sub-study of the Asker and Baerum Cardiovascular Diabetes (ABCD), a prospective, randomised, controlled study of intensive versus usual care in patients with T2DM. 4 Participating subjects were recruited through advertisement in the local newspaper and by letter to all general practitioners in the area (population 150 000) and from the hospital’s outpatient clinic. The present cross-sectional study consecutively enrolled 133 of these subjects aged 18–75 years for a cross-sectional investigation of the prevalence of CAD as assessed with non-invasive and invasive cardiological examinations. 4 Serum samples of activin A and follistatin were available for 102 patients who all had ≥ 1 CV risk factor for CVD in addition to diabetes (hypertension, past or ongoing smoking, premature CAD in first degree family, microalbuminuria or dyslipidaemia). Patients underwent diagnostic coronary angiography using standard clinical methods. 4 For comparison, serum levels of activin A and follistatin were also obtained from 20 sex- and age-matched healthy individuals (based on disease history, clinical examination and routine laboratory tests recruited from the hospital staff. Angiographic measurements were not performed in healthy controls. All participants gave written informed consent and the studies were conducted in accordance with the Declaration of Helsinki and approved by the Regional Ethical Committee.
Biochemistry and blood sampling
Peripheral venous blood was drawn into pyrogen-free tubes with or without EDTA as an anticoagulant. After coagulation, samples were centrifuged and serum samples were stored at −80°C. The samples were thawed only once prior to analysis of activin A and follistatin (samples were thawed in a water bath at 37°C). Plasma levels of glucose and C-peptide and urine levels of µ-albuminuria were analysed by routine laboratory methods at a central laboratory, while high-sensitivity C-reactive protein (hsCRP) was assayed on a MODULAR platform (Roche Diagnostics, Basel, Switzerland). Serum activin A was analysed by enzyme immunoassay (EIA) from Serotec (Oxford, UK). Follistatin and tumour necrosis factor (TNF)-α levels 4 were analysed by EIA (R&D Systems, Stillwater, MN, USA). All intra- and inter-assay coefficients of variation (CVs) were <10%.
Statistics
For statistical comparisons parametric and non-parametric tests were used as appropriate depending on distribution. Cross-tab statistics were analysed by chi-square. Univariable and forced multiple linear regression was used to evaluate associations between coronary measures and activin A (expressed in quartiles or log transformed) and adjust for covariates including age, T2DM duration, sex, hypertension, insulin use, smoking, microalbuminuri, hsCRP, TNF-α. Tests are two-tailed and considered to be significant when p < 0.05.
Results
Activin A and follistatin in type 2 diabetes mellitus in relation to coronary artery disease status
Patient characteristics according to CAD status are shown in Table 1. Compared with healthy controls, patients with T2DM had elevated median levels of serum activin A (Table 1). In contrast, serum follistatin levels were reduced while the ratio between activin A and follistatin was increased in T2DM patients as compared with controls (Table 1).
Patient characteristics according to CAD status in patients with T2DM.
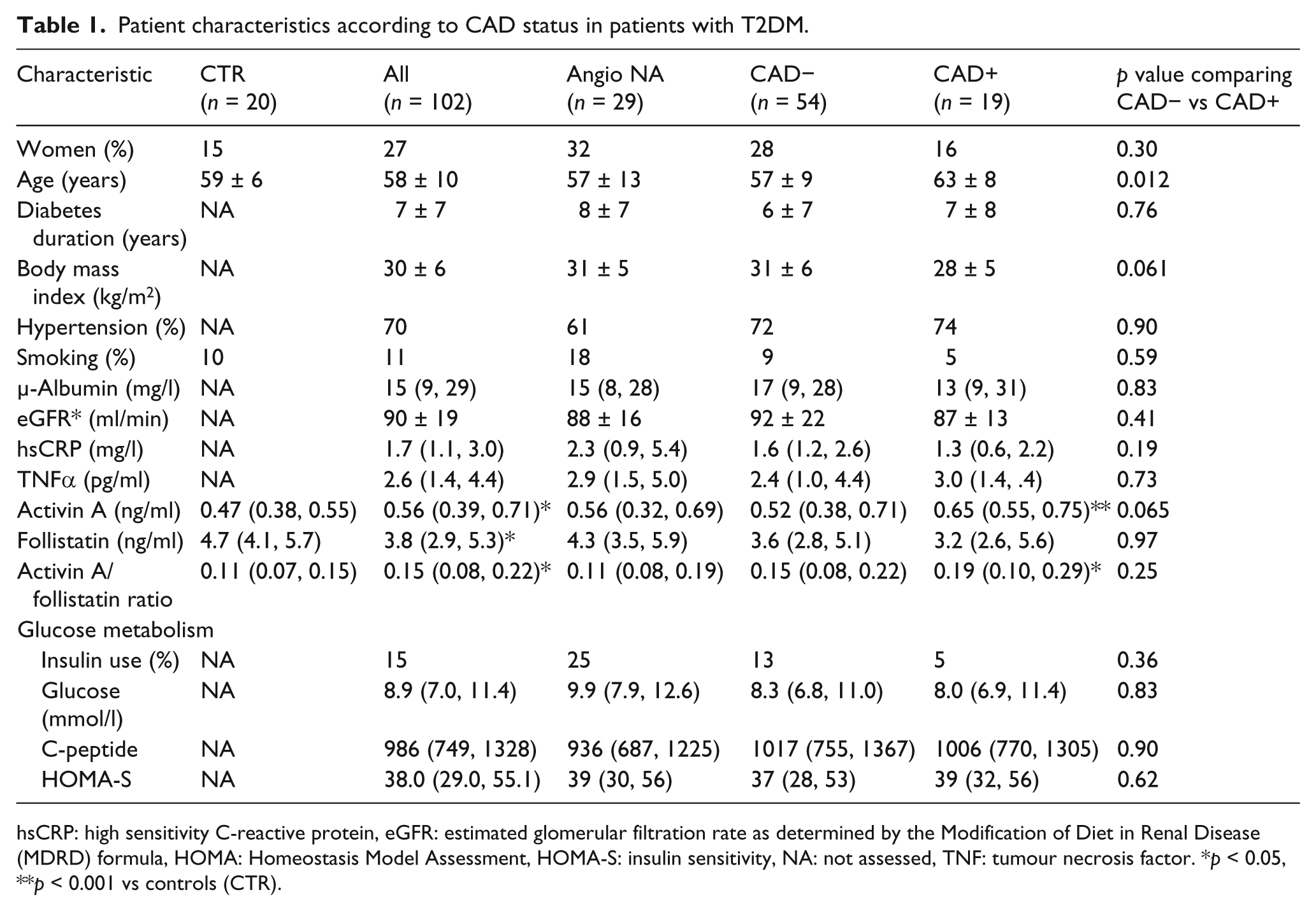
hsCRP: high sensitivity C-reactive protein, eGFR: estimated glomerular filtration rate as determined by the Modification of Diet in Renal Disease (MDRD) formula, HOMA: Homeostasis Model Assessment, HOMA-S: insulin sensitivity, NA: not assessed, TNF: tumour necrosis factor. *p < 0.05, **p < 0.001 vs controls (CTR).
Among 73 subjects with T2DM who voluntarily underwent coronary angiography, activin A levels tended to be higher (p = 0.065) in those with significant CAD versus those without and were markedly higher compared with controls (Table 1). While no differences were found for follistatin, the activin A/follistatin ratio was higher in patients with CAD compared with healthy controls (Table 1).
Associations between activin A, follistatin and coronary angiographic scores
Univariable analysis showed that when divided into quartiles, an increasing proportion of CAD was observed with increasing quartiles of activin A: 11%, 22%, 29% and 40% for quartile 1–4 respectively (p = 0.038). However, this trend was not significant after mutlivariable adjustment (Figure 1, Table 2). A similar trend was observed in relation to the proportion of affected vessels with 0%, 6%, 24% and 30% with ≥2 vessel disease with increasing activin A quartiles, respectively (p = 0.003). This association between quartiles of activin A and proportion of ≥2 vessel disease remained significant after multivariable adjustment (Figure 1, Table 2), although when activin A was entered as a continuous variable, the association was not significant. For extent score (number of segments exhibiting lesions ≥ grade 1, adjusted to 16 coronary segments) the mean ± SD for increasing quartiles of activin A (in ng/ml) were 0.11 ± 32, 0.28 ± 0.57, 0.53 ± 0.87 and 0.70 ± 0.92 (p = 0.022). The association with extent score was not significant after adjustment (Table 2). For follistatin and the activin A/follistatin ratio, no significant associations with angiographic measures were observed, except for an increasing activin A/follistatin ratio in patients with ≥2 vessel disease (univariable, p = 0.005).
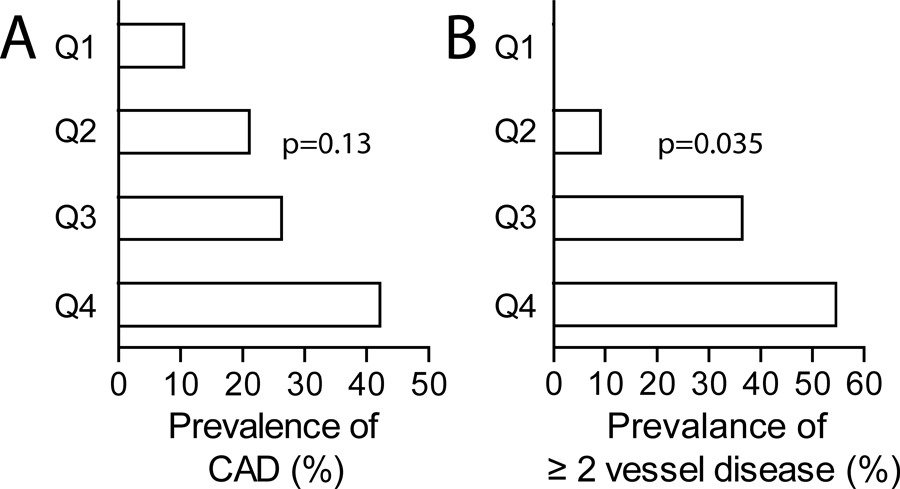
Prevalance of (A) CAD and (B) ≥2 vessel disease in relation to quartiles of activin A in type 2 diabetes mellitus. p values shows trend after adjustment in multiple regression (see statistics).
Multiple linear regression showing associations between activin A and coronary measures in patients with T2DM.
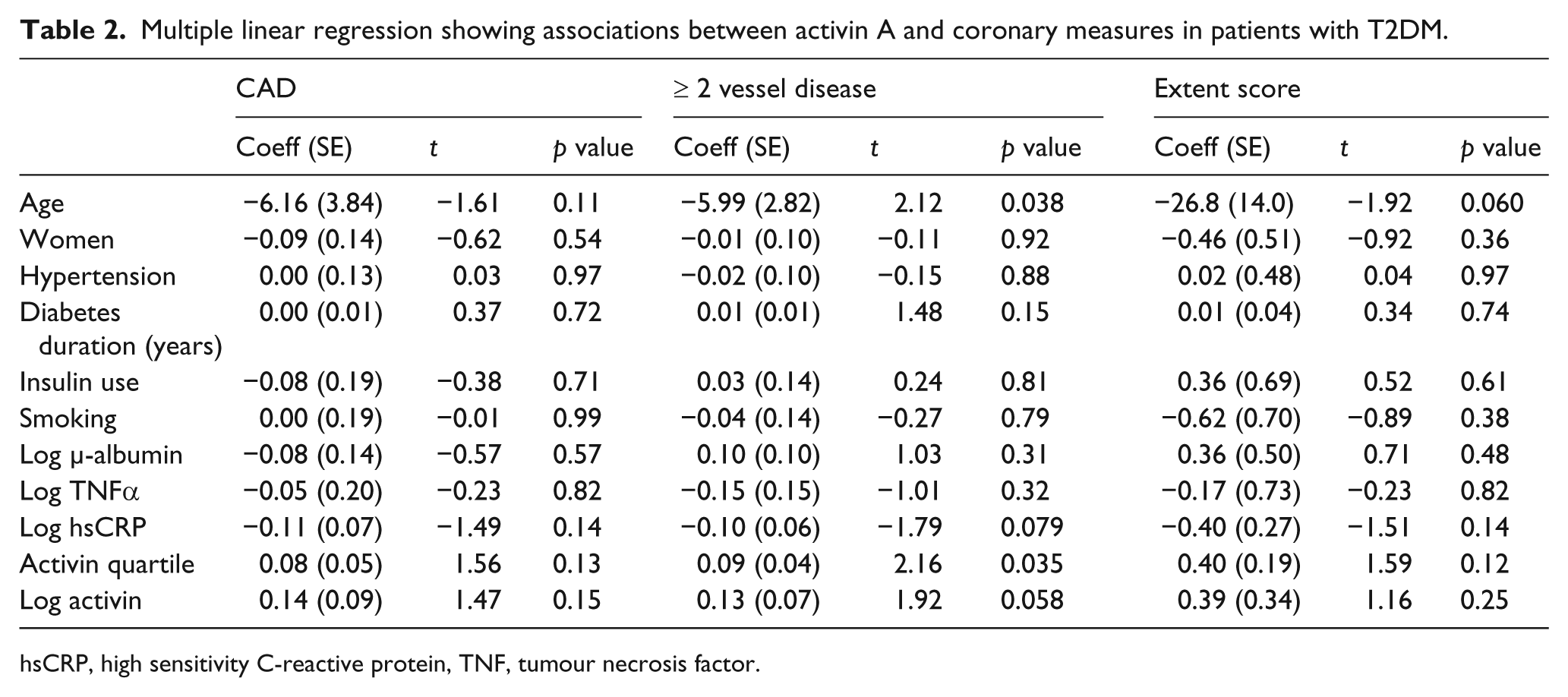
hsCRP, high sensitivity C-reactive protein, TNF, tumour necrosis factor.
Discussion
This study shows increased circulating activin A in patients with T2DM. Within the T2DM patients, elevated activin A was associated with the severity of coronary atherosclerotic burden indicating that increased activin A may reflect some chronic pathophysiological processes involved in development of coronary atherosclerosis.
Our findings indicate enhanced activin A activity in T2DM, and its associations with the extent of coronary angiographic scores (i.e. ≥2 vessel disease) support previous investigations of activin A in populations with established CV disease.8,10–12 The effects of activin A are complex and during systemic inflammation, both inflammatory and anti-inflammatory effects have been reported, depending on the degree of cellular pre-activation. 13 The rapid induction of activin A during systemic inflammation may prevent inappropriate inflammatory responses. 13 Furthermore, we and others have demonstrated various beneficial effects of activin A in relation to atherogenesis, such as inhibition of foam cell formation and VSMC activity potentially leading to plaque stabilisation.8,10 More recently we found that activin A attenuated the expression of the pro-atherogenic chemokine interleukin-8 and the anti-oxidative enzyme metallothionein from endothelial cells. 11 As the endothelium is an important target for vascular complications in T2DM and CAD, it is tempting to hypothesise that the increased levels of activin A in our patients with T2DM and CAD may represent a counteracting anti-inflammatory and anti-oxidative mechanism. However, caution is needed when interpreting these results, as the cross-sectional design of our study does not provide any insight into the regulation of activin A during progression of CAD. Thus future studies will have to determine if activin A is a marker or a mediator of CAD in T2DM.
Limitations to our study are that no angiographic examination was performed in controls to ensure that they were free of CAD. Also, a higher number of controls and more equal sex distribution between patients and controls would be preferred. Finally, the number of patients with CAD was low. To investigate if activin A may be a viable marker for CAD in patients with T2DM, a larger population would have to be studied.
Our study shows increased activin A in patients with T2DM with particularly high levels in patients with increased atherosclerotic burden, potentially reflecting a counteracting mechanism of activin A on inflammation in these patients. However, studies in larger cohorts would be needed to establish if activin A is a useful surrogate marker to define risk among these patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
There are no conflicts of interest.
