Abstract
The objective of the study was to investigate the usefulness of serum lipoprotein(a) level in predicting the severity of new on-set coronary artery disease in type 2 diabetics. A total of 1254 new on-set, consecutive coronary artery disease patients were classified into two groups: diabetes group (n = 380) and non-diabetes group (n = 874). The relationship between serum lipoprotein(a) levels and the severity of coronary artery disease assessed by Gensini score was analysed. Data showed that the diabetes group had higher serum triglyceride and high sensitivity C-reactive protein levels but lower high-density lipoprotein cholesterol levels (all p < 0.05). The multivariate logistic regression analysis suggested that lipoprotein(a) was an independent predictor for high Gensini score (odds ratio = 1.82, 95% confidence interval: 1.10–3.12, p = 0.029) after adjusting for traditional cardiovascular risk factors. Additionally, lipoprotein(a) levels were positively correlated with Gensini score (rho = 0.15, p = 0.014) and significantly elevated according to the tertiles of Gensini score (p = 0.008) in diabetics. However, no such results were observed in non-diabetics. Our data indicate that lipoprotein(a) is an independent predictor for the severity of new on-set coronary artery disease patients accompanied by type 2 diabetes, suggesting that these patients may benefit from lipoprotein(a) management in clinical assessment.
Introduction
Cardiovascular disease (CVD) is a well-known worldwide health threat and the major cause of death. Dyslipidemia is closely associated with the progression of coronary atherosclerosis. Lipoprotein(a) (Lp(a)), which is a genetically determined low-density lipoprotein (LDL) particle with a unique ‘apolipoprotein(a) (apo(a))’ molecule, may promote the progression of atherosclerosis. 1 It has been suggested that an apo(a) molecule contained in Lp(a) particle may interfere with fibrinolysis. 2 According to current knowledge, the serum concentration of Lp(a) among individuals is always stable. Meanwhile, it is minimally responsive to environmental and lifestyle-related factors and currently available lipid-lowering medications3,4 except for niacin 5 and hormone replacement therapy. 6 Furthermore, epidemiological evidence has shown that high Lp(a) levels can increase the risk of CVD independent of LDL cholesterol (LDL-C) levels. 7
However, although Lp(a), used as a unique cardiovascular risk factor, has been widely, intensively studied for many years, particularly in recent years, whether the level of Lp(a) is an independent risk factor for CVD remains controversial till now.8–13 Several studies indicated that Lp(a) might initiate atherosclerosis14,15 and possibly promote thrombosis at very high levels, 16 while other data did not support the notion that Lp(a) was an independent predictor for CVD risk. 17 At the same time, few studies have analysed the relationship between serum Lp(a) levels and the severity of coronary artery disease (CAD), in particular using the well-recognized quantitative coronary scores in a large sample size. More importantly, less data are currently available in diabetes, a unique diseased state with significant disorder of metabolism including dyslipidemia. This study, therefore, explored the usefulness of Lp(a) in predicting the severity of new on-set CAD assessed by Gensini score in patients with type 2 diabetics.
Methods
Study population
The study complied with the Declaration of Helsinki and was approved by the hospital ethical review board (FuWai Hospital and National Center for Cardiovascular Diseases, Beijing, China). Informed written consent was obtained from all patients included in this analysis.
In this study, there were 1254 new on-set CAD patients enrolled consecutively, and all patients underwent diagnostic coronary angiography in the Division of Dyslipidemia of FuWai Hospital from April 2011 to December 2013. All patients were classified into diabetes group (n = 380) and non-diabetes group (n = 874). Patients with previous percutaneous coronary intervention, previous coronary artery bypass grafting, post-myocardial infarction, abnormal baseline cardiac troponin I levels and/or valvular heart disease were excluded. Clinical characteristics, lipid parameters and laboratory test information of all enrolled patients were investigated.
Hypertension was diagnosed when repeated blood pressure was ⩾140/90 mmHg (at least two times in different environments) or when patients were taking antihypertensive drugs. Type 2 Diabetes was diagnosed with fasting serum glucose level ⩾126 mg/dL in multiple determinations or was under treatment with insulin or oral hypoglycaemic agents. Dyslipidemia was diagnosed with fasting total cholesterol (TC) ⩾200 mg/dL, triglyceride (TG) ⩾150 mg/dL and/or high-density lipoprotein cholesterol (HDL-C) <40 mg/dL (for male) or 50 mg/dL (for female).
Laboratory tests
Baseline laboratory data were obtained from venous blood samples taken after a 12-h overnight fast before coronary angiography. The lipid profiles were determined by automatic biochemistry analyser (Hitachi 7150, Tokyo, Japan). In detail, the LDL-C concentration was analysed by selective solubilization method (low-density lipid cholesterol test kit; Kyowa Medex, Tokyo). HDL-C concentration was determined by a homogeneous method (Determiner L HDL; Kyowa Medex, Tokyo). TC, TG, apolipoproteinB (apoB) and apolipoproteinA-1 (apoA-1) were measured by enzymatic methods. Lp(a) levels were measured with a latex turbidimetric method (LASAY Lp(a) auto; SHIMA laboratories, Tokyo). The determination of Lp(a) levels ranged from 0.5 to 100 mg/dL, and the normal reference value was less than 33 mg/dL. The interassay variation for samples in our laboratory was <10%. The levels of high-sensitive C-reactive protein (hs-CRP) were measured by immunoturbidometry (Beckmann Assay 360; Bera, CA, USA). All other including biomarkers were analysed by standard haematological and biochemical tests.
Angiographic examination
The selective coronary angiography was performed for all enrolled individuals, and the results were analysed by at least two interventional physicians. The CAD was defined by obstructive stenosis reaching or more than 50% of the vessel lumen diameter in any of the main coronary arteries including the left main coronary artery (LM), the left anterior descending artery (LAD), the left circumflex coronary artery (LCX), the right coronary artery (RCA) or the main branch of the vascular.
The severity of CAD was evaluated by Gensini score assessment system, which was defined according to stenosis severity as 1-point for <25% stenosis, 2-point for 26%–50% stenosis, 4-point for 51%–75% stenosis, 8-point for 76%–90% stenosis and 32-point for total occlusion. The score was then multiplied by a factor that represents the importance of the lesion’s position in the coronary arterial system. For example, 5 for the LM, 2.5 for the proximal LAD or proximal LCX, 1.5 for the mid-region and 1 for the distal LAD. 18
Statistical analysis
All analyses were performed using SPSS version19.0 software (Chicago, IL, USA). To test the distribution pattern, the Kolmogorov–Smirnov test was used. Continuous variables were presented as mean ± standard deviations (SDs) (normal distribution data) or median with 25th and 75th percentile (abnormal distribution data, including Gensini score, Lp(a), TG, apoA-1, hs-CRP, etc.); variables were compared by Student’s t-test (normal distribution data), Mann–Whitney U-test or Kruskal–Wallis test (abnormal distribution data). Categorical variables were summarized as frequencies with percentages and compared with chi-square test.
In order to identify the factors which were independently associated with high Gensini score in diabetes and non-diabetes group, binary univariate and multivariate logistic regression analyses were performed, respectively, by using forward stepwise selection process. Correlation between Lp(a) levels and Gensini score was examined using Spearman correlation analysis. A p value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics
The study population consisted of 895 men (71.4%) and 359 women (28.6%), aged 22–82 years (mean age 58.7 ± 9.4 years). All enrolled patients were classified into type 2 diabetes group (n = 380) and non-diabetes group (n = 874).
Characteristics of the patients in two groups were shown in Table 1. In brief, patients with type 2 diabetes tended to be older and had higher proportion of hypertension and dyslipidemia but lower proportion of recent smokers. Meanwhile, there were significantly higher TG levels but lower HDL-C levels in diabetes group compared with those in non-diabetes group (p = 0.013, p = 0.008, respectively). Characteristics of diseased vessels and Gensini score in the study population were shown in Table 2. Patients with diabetes had significantly higher Gensini score and percentage of three-vessel coronary disease compared with those in non-diabetes group (all p < 0.001).
Baseline characteristics of the study population.
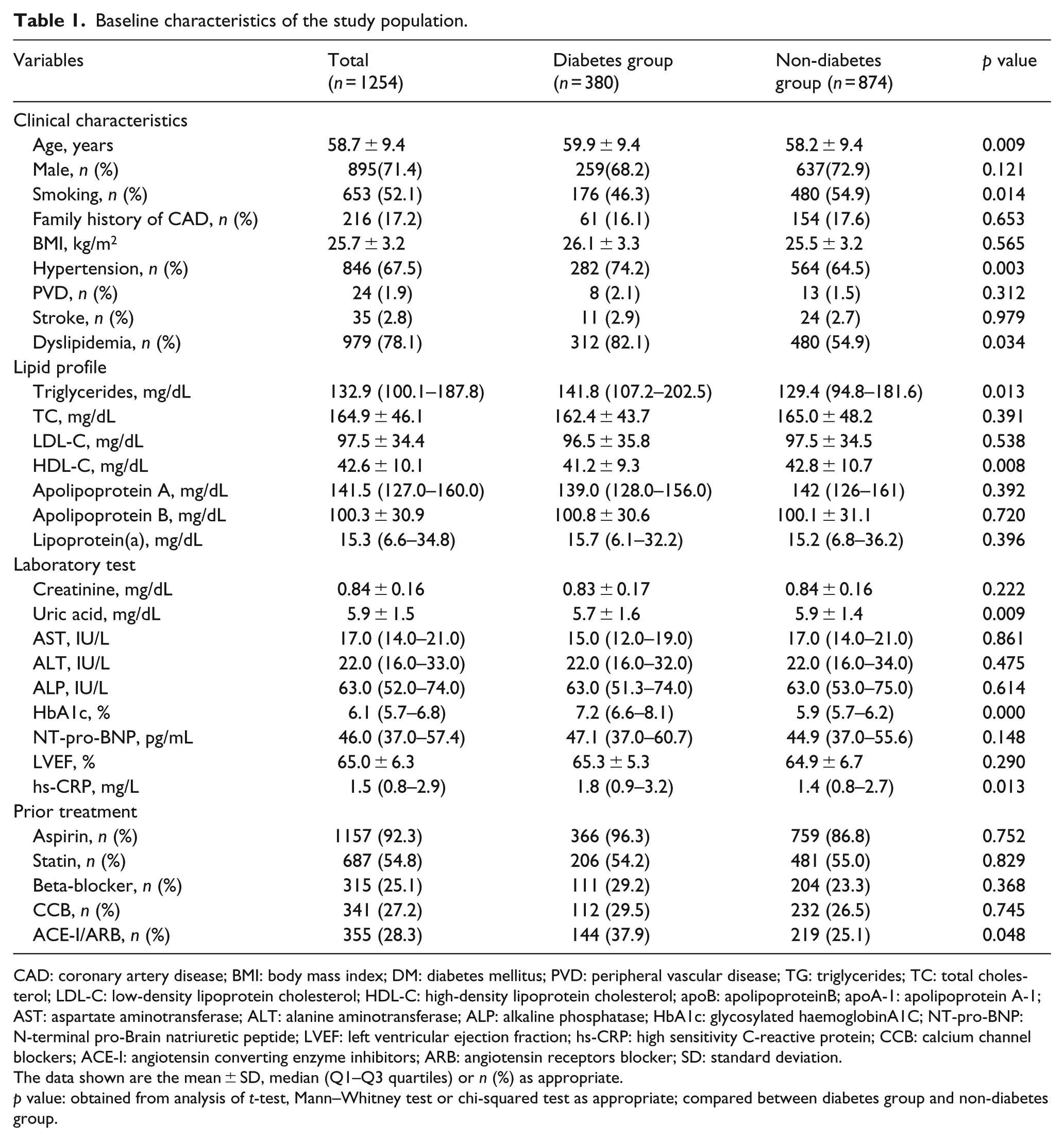
CAD: coronary artery disease; BMI: body mass index; DM: diabetes mellitus; PVD: peripheral vascular disease; TG: triglycerides; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; apoB: apolipoproteinB; apoA-1: apolipoprotein A-1; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; HbA1c: glycosylated haemoglobinA1C; NT-pro-BNP: N-terminal pro-Brain natriuretic peptide; LVEF: left ventricular ejection fraction; hs-CRP: high sensitivity C-reactive protein; CCB: calcium channel blockers; ACE-I: angiotensin converting enzyme inhibitors; ARB: angiotensin receptors blocker; SD: standard deviation.
The data shown are the mean ± SD, median (Q1–Q3 quartiles) or n (%) as appropriate.
p value: obtained from analysis of t-test, Mann–Whitney test or chi-squared test as appropriate; compared between diabetes group and non-diabetes group.
Characteristics of diseased vessels and Gensini score in the study population.
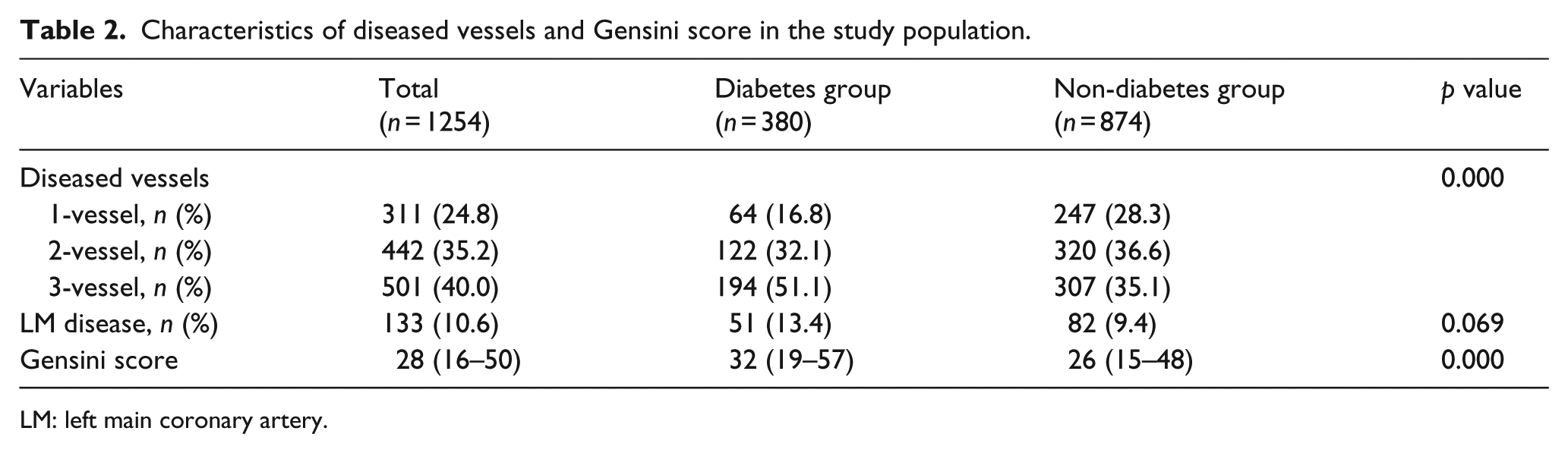
LM: left main coronary artery.
Logistic regression analysis
For detecting independent predictors for high Gensini score, univariate and multivariate logistic regression analyses were performed. As shown in Table 3, after adjusting for gender, age, current smoking, hypertension, dyslipidemia, family history of CAD, left ventricular ejection fraction (LVEF), glycosylated haemoglobinA1C (HbA1c) and hs-CRP, Lp(a) was demonstrated to be a significant and independent predictor for high Gensini score in diabetes patients (odds ratio (OR) = 1.82, 95% confidence interval (CI): 1.10–3.12, p = 0.029) but not in non-diabetes patients.
Logistic regression analysis for detecting independent predictors of high Gensini score in diabetes and non-diabetes groups.
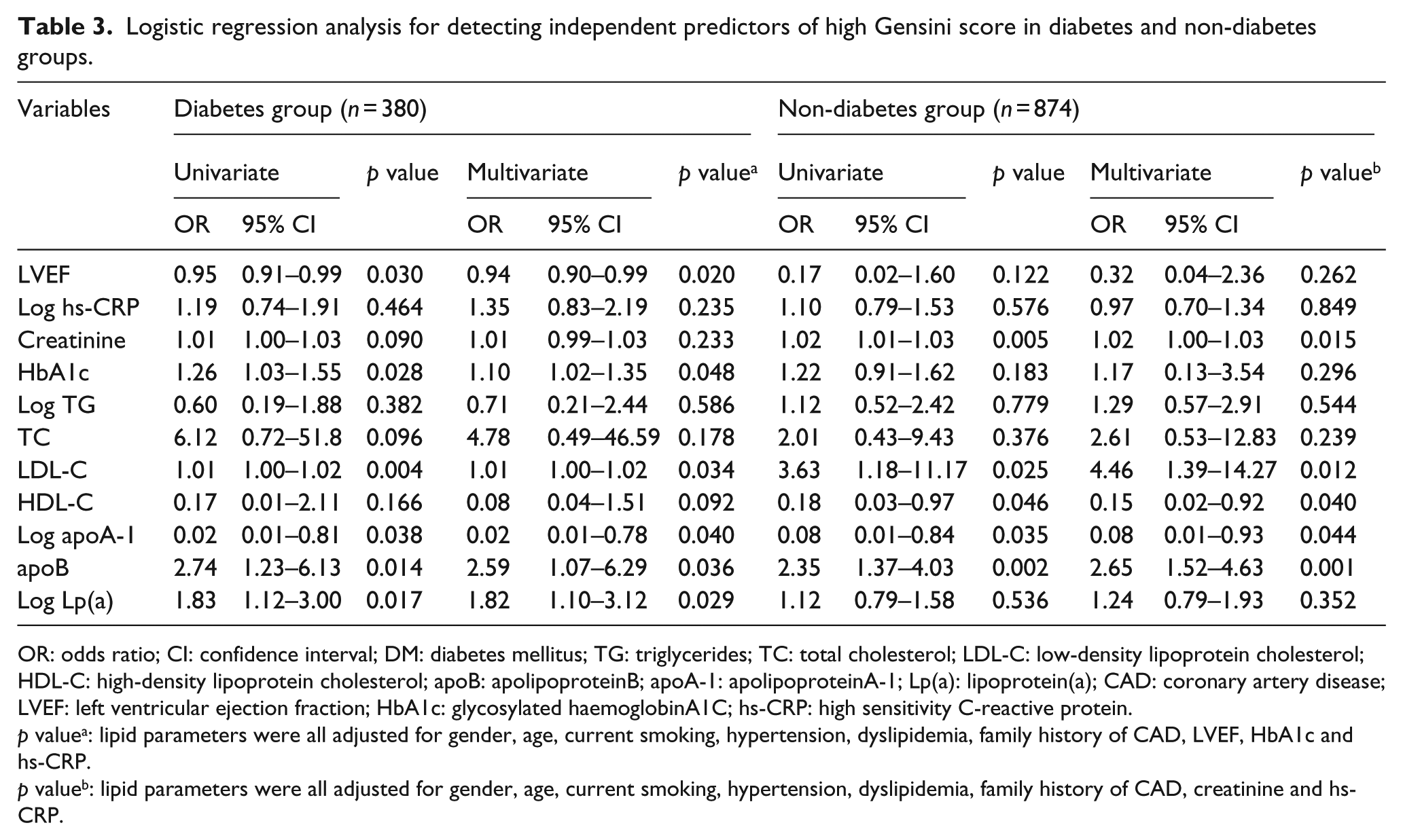
OR: odds ratio; CI: confidence interval; DM: diabetes mellitus; TG: triglycerides; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; apoB: apolipoproteinB; apoA-1: apolipoproteinA-1; Lp(a): lipoprotein(a); CAD: coronary artery disease; LVEF: left ventricular ejection fraction; HbA1c: glycosylated haemoglobinA1C; hs-CRP: high sensitivity C-reactive protein.
p valuea: lipid parameters were all adjusted for gender, age, current smoking, hypertension, dyslipidemia, family history of CAD, LVEF, HbA1c and hs-CRP.
p valueb: lipid parameters were all adjusted for gender, age, current smoking, hypertension, dyslipidemia, family history of CAD, creatinine and hs-CRP.
Correlation analysis of Lp(a) levels with Gensini score
Correlations between Lp(a) levels and Gensini score in diabetes and non-diabetes group were examined separately using Spearman correlation analysis. As shown in Figure 1, there was a significant and positive correlation of Lp(a) levels with Gensini score in type 2 diabetes group (rho = 0.15, p = 0.014), but not in non-diabetes group.
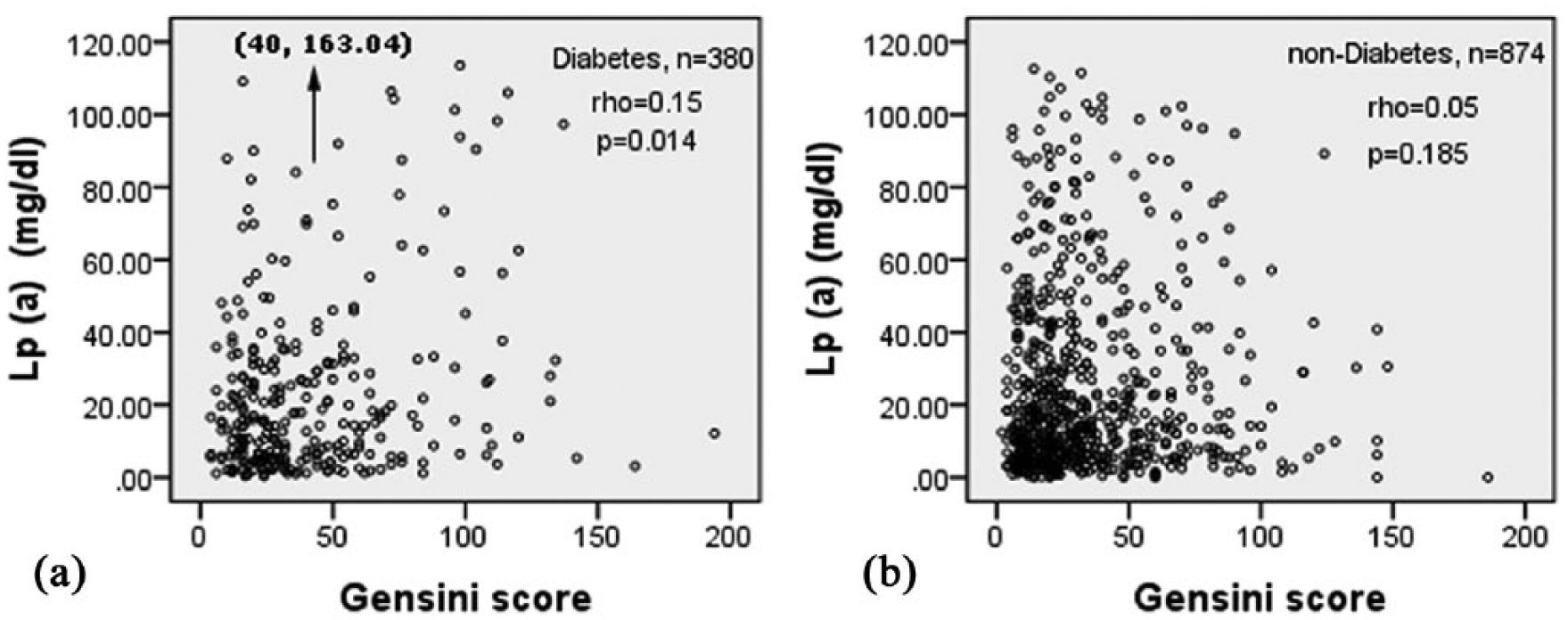
Correlation analysis of lipoprotein(a) (Lp(a)) levels with Gensini score in type 2 diabetes group and non-diabetes group.
Based on the tertiles of Gensini score, non-diabetes patients were divided into three groups: low Gensini score (<19, n = 290 (33.2%)), intermediate Gensini score (19–35, n = 282 (32.3%)) and high Gensini score (>35, n = 302 (34.6%)). Meanwhile, the patients with diabetes were also classified into three groups according to this cut-off value: low Gensini score (n = 93, 24.5%), intermediate Gensini score (n = 112, 29.5%) and high Gensini score (n = 175, 46.0%). Importantly, as indicated in Figure 2, the serum Lp(a) levels were highest in the upper Gensini score tertile, whereas similar between the first and second tertile (p for trend = 0.008) in diabetes group; however, no similar results were found in non-diabetes group (p for trend = 0.301). Besides that the distribution of patients among three groups of Gensini score was markedly unbalanced, and higher proportion of patients tended to have high Gensini score in diabetes group.
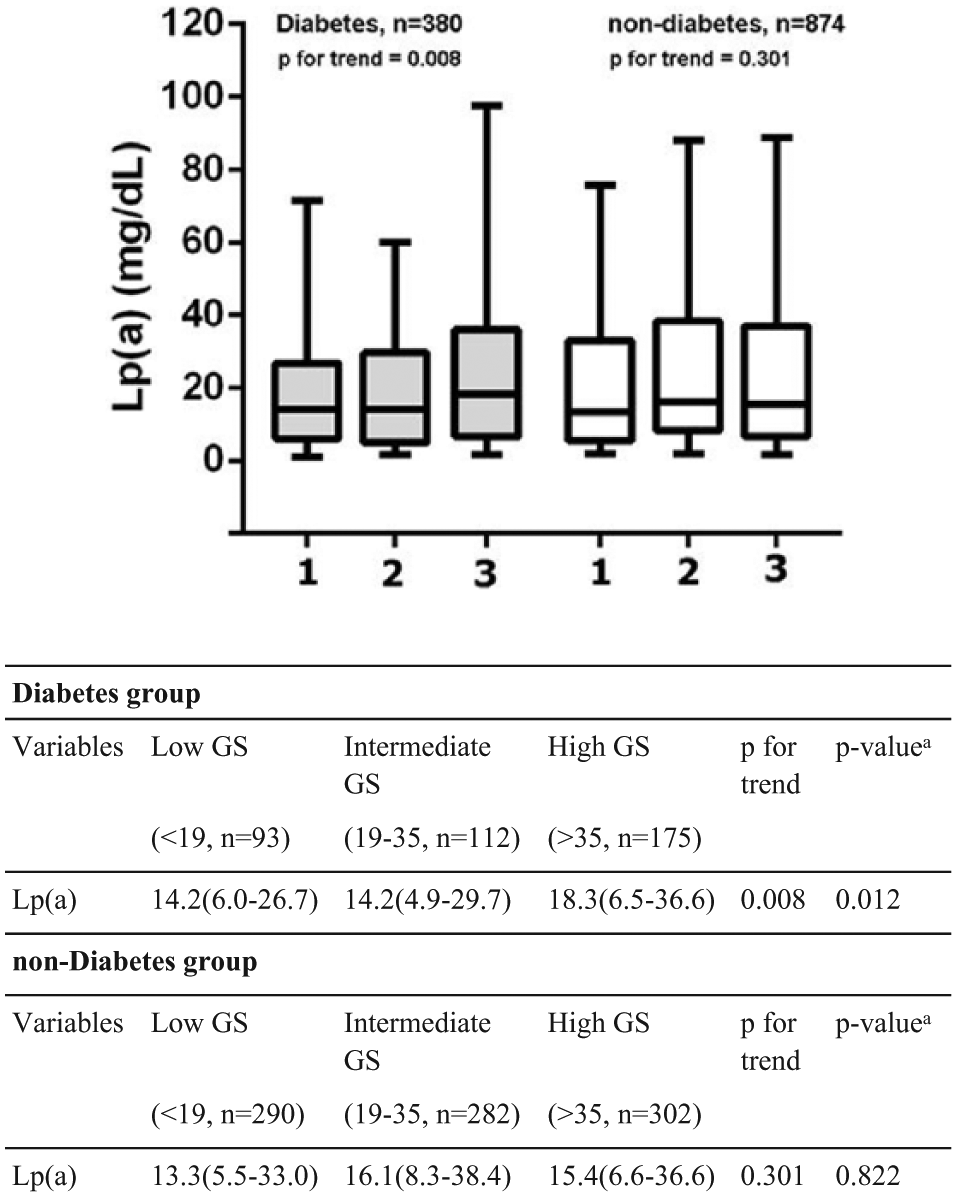
The lipoprotein(a) (Lp(a)) levels in type 2 diabetes group and non-diabetes group according to the tertiles of Gensini score (GS).
Discussion
The main findings of the study are that (1) the serum Lp(a) level was an independent predictor for the severity of CAD assessed by Gensini score in patients with diabetes (OR = 1.82, 95% CI: 1.10–3.12, p = 0.029) in our Chinese Han populations; (2) there was a significantly positive correlation of serum Lp(a) levels with Gensini score in CAD patients with diabetes (rho = 0.15, p = 0.014); (3) the serum Lp(a) levels were significantly increased according to the tertiles of Gensini score in CAD patients with diabetes (p = 0.008). However, no such results were observed in CAD patients without diabetes. Our present data suggest that more attention should be paid to Lp(a) in CAD patients with diabetes.
Although Lp(a) has long been an attractive subject in basic and clinical research, until recently, it has begun to be considered as a unique risk predictor of CAD and been broadly utilized. Evidence from genetic studies has recognized that Lp(a) is a causal risk factor for CVD rather than being merely a marker. 19 Mentionable, this causal relationship is continuous and independent of LDL-C and HDL-C levels. In spite of unknown mechanisms of Lp(a) in promoting atherosclerosis, proposed hypothesis including an increased Lp(a)-associated cholesterol entrapment in the arterial intima, inflammation mediated, carrying of proinflammatory oxidized phospholipids, impairing fibrinolysis and enhancing coagulation are strongly suggested. 20 Hence, the European Atherosclerosis Society (EAS) has recommended screening for high levels of Lp(a) in individuals at elevated risk of CVD. 7
Concerning the role of Lp(a) in the development of CAD, there were considerable studies that have been previously reported. Several studies have shown a strong relationship between serum levels of Lp(a) and the presence of CAD. Yazici et al. 21 investigated the importance of Lp(a) in pathogenesis of the high-risk unstable angina. Their results indicated that Lp(a) might be a risk factor for plaque destabilization and thrombosis rather than severity of CAD and related to myocardial injury in patients with unstable angina. 22 More recently, Langsted et al. 23 demonstrated that elevated Lp(a) levels could predict ischaemic heart disease and myocardial infarction in the general population, and the predictive ability was not affected by normal food intake but was minimally increased at high levels of CRP. Interestingly, Lp(a) levels vary across individuals, families and ethnic groups. 22 Associations between elevated Lp(a) levels and CAD in women are less robust than in men, possibly because of the cardioprotective and vasoprotective effects of endogenous oestrogen in premenopausal women 24 and the direct effects of oestrogens on Lp(a) levels. 25
On the other hand, a few studies have reported the correlation of Lp(a) with the severity of CAD. Besides, Momiyama et al. 26 studied 143 patients with coronary and aortic atherosclerosis and found that serum Lp(a) levels increased stepwise with the number of stenotic coronary vessels. Sposito et al. 27 also found that Lp(a) was an useful marker for the severity of CAD among 182 postmenopausal women. Insufficiently, these reports were limited by either the small sample size or lack of a strict assessment system of the severity of CAD. As we know, Gensini score assessment system is a simple, wieldy and widely used tool in evaluating the severity of coronary stenosis. 18 That is the reason why we chose Gensini score as an approach for evaluating the severity of the CAD in this Chinese cohort study. In 2006, Boroumand et al. performed a cross-sectional study including 826 patients who underwent angiography assessed by Gensini score. They found that the serum LP(a) concentration is an independent risk factor for coronary atherosclerosis in the Iranian population especially at the ages below 55 years. 11 In the study, they only used Gensini score = 6 as a cut-off point for coronary atherosclerosis, whereas we more accurately classified the subjects into three groups according to Gensini score tertiles. In addition, they mainly focused on the whole population underwent coronary angiography. However, considering the multiple metabolic disorders induced by diabetes, we specifically investigated whether diabetes modified this relationship in patients with CAD.
More importantly, according to our knowledge, although there were three studies concerning the role of Lp(a) in predicting the severity of CAD in patients with type 2 diabetes until now, the results were conflicting.12,13,17 Data from Watts et al. 12 group containing 36 diabetics with coronary angiography in 1995 and Gazzaruso et al. 13 group enrolling 227 CAD patients with diabetes in 2006 suggested that Lp(a) might be a reliable predictor of CAD severity in type 2 diabetic patients, whereas the study performed by Pedreno et al. 17 in 2000 suggested that there was lack of an association in CAD patients with diabetics (n = 355, 101 patients with diabetes and 254 patients without diabetes). In this study, we investigated the relationship between Lp(a) and the severity of new-onset CAD in patients with diabetes in a relatively large sample size and compared with those without diabetes. We found that serum Lp(a) level was an independent predictor for the severity of CAD assessed by Gensini score only in CAD patients with type 2 diabetes but not in non-diabetics. Accordingly, our data may confirm and extend the previous studies. Although the causes accounting for this interesting relationship are still obscure, we speculate that the following reasons may be involved. Primarily, the complicated metabolic disorders especially dyslipidemia induced by diabetes may be potential contributors. Additionally, the diabetic patients tend to have more severe coronary atherosclerosis than the non-diabetics. Finally, the Lp(a) levels are significantly increased by the severity of coronary atherosclerosis in diabetics but not in non-diabetics. All the preceding factors may partly explain this specific relationship. Beyond that, there were definite evidence that Lp(a) was increased in the very early stages of renal disease 28 and the apo(a) levels were higher in diabetic patients with albuminuria than in those without. 29 It has been well known that a size polymorphism at the apo(a) gene locus is the most important determinant of Lp(a) levels and exhibits marked interindividual variation. 28 Thus, whether renal disease or albuminuria would influence this relationship need to be tested in the future studies.
There were several limitations in this study. First, it is a cross-sectional study, so it is difficult to identify the causal relationship between Lp(a) and severity of CAD. Second, we did not explore the exact mechanisms of this relationship. Third, although there was statistical significance between Lp(a) and severity of CAD in patients with diabetes, the power was relatively weak, and further investigations are needed. Finally, we did not measure microalbuminuria, albuminuria and glomerular filtration rate in this study, which may be potential confounding factors.
In conclusion, this data provide a valuable evidence that higher Lp(a) level may be a useful marker in predicting the severity of coronary atherosclerosis in CAD patients with type 2 diabetes, suggesting that these patients may benefit from inclusion of Lp(a) measurement in clinical assessment.
Footnotes
Acknowledgements
Juan Chen, Yan Zhang and Jun Liu contribute equally to this study.
Declaration of conflicting interests
The authors have no relevant conflicts of interest to disclose.
Funding
This work was partly supported by National Natural Science Foundation (81070171, 81241121), Specialized Research Fund for the Doctoral Program of Higher Education of China (20111106110013), Capital Special Foundation of Clinical Application Research (Z121107001012015), Capital Health Development Fund (2011400302) and Beijing Natural Science Foundation (7131014) awarded to Dr Jian-Jun Li, MD, PhD.
