Abstract
Degenerative aortic valve disease in combination with diabetes is an increasing burden worldwide. There is growing evidence that particularly small leucine-rich proteoglycans are involved in the development of degenerative aortic valve disease. Nevertheless, the role of these molecules in this disease in the course of diabetes has not been elucidated in detail and previous studies remain controversial. Therefore, the aim of this study is to broaden the knowledge about small leucine-rich proteoglycans in degenerative aortic valve disease and the influence of diabetes and hyperglycaemia on aortic valves and valvular interstitial cells is examined. Analyses were performed using reverse-transcription polymerase chain reaction, Western blot, enzyme-linked immunosorbent assay, (immuno)histology and colorimetric assays. We could show that biglycan, but not decorin and lumican, is upregulated in degenerated human aortic valve cusps. Subgroup analysis reveals that upregulation of biglycan is stage-dependent. In vivo, loss of biglycan leads to stage-dependent calcification and also to migratory effects on interstitial cells within the extracellular matrix. In late stages of degenerative aortic valve disease, diabetes increases the expression of biglycan in aortic valves. In vitro, the combinations of hyperglycaemic with pro-degenerative conditions lead to an upregulation of biglycan. In conclusion, biglycan represents a potential link between degenerative aortic valve disease and diabetes.
Introduction
Cardiovascular diseases collectively represent the leading cause for public health expenditures in industrialized countries. Within cardiovascular diseases, degenerative aortic valve disease (DAVD) is a serious element of increasing burden in terms of morbidity and mortality, as well as costs in the health care systems.1,2 Besides other risk factors, type 2 diabetes (T2D) is associated with an increased risk of DAVD3,4 and will be an incremental challenge in the future since its prevalence is estimated to double until 2030. 5
DAVD is characterized by morphological processes like connective tissue degeneration, lipid accumulation, inflammation and finally calcification.6,7 Recently, considerable evidence has accumulated to support the role of special extracellular matrix (ECM) components, that is, small leucine-rich proteoglycans (SLRPs), in the development of DAVD. SLRPs consist of a core protein with leucine-rich repeat motifs linked to dermatan sulphate (biglycan and decorin) or keratan sulphate (lumican) glycosaminoglycan chains. 8 Their potency to modulate lipid accumulation, their interaction with inflammatory molecules, the control of thrombin activity 9 and immune cells 10 turns proteoglycans into promising therapeutic targets.
In the healthy aortic heart valve (AV), SLRPs are abundant and mostly located in the inner part of the AV 11 where they ensure high viscoelasticity of the valve leaflet. 12 In degenerative processes affecting AV, localization and amount of these molecules are altered and especially biglycan and decorin have been described to be associated with DAVD.13–17 However, studies on biglycan using human AV samples comprise rather a small number of individual donors ranging from n = 5 to n = 87.18–20 Moreover, only sparse data exist on decorin and lumican with somewhat controversial results regarding the regulation of these SLRPs along DAVD.15,18,21
In connection with diabetic disorders, previous research on SLRP has so far focused on atherosclerosis,22,23 nephropathies, 24 obesity and fatty tissue, 25 as well as on monocytes. 26 Hyperglycaemia and its influence on SLRP turnover are assumed to be part of the clinically accelerated degeneration of the cardiovascular system in T2D.27–29 Nevertheless, current knowledge of the role of SLRP in diabetes-induced AV degeneration is limited.
Thus, the aims of this study are to analyse individual SLRP, for example, biglycan, decorin or lumican, in relation to the morphological stage of AV degeneration in human samples and their presence and abundance in valvular interstitial cells (VICs) prone to degeneration in vitro. The in vivo impact of biglycan on cell migration, proliferation, tissue remodelling and degeneration is analysed in biglycan loss-of-function mice. Moreover, influence of T2D on these key molecules in the context of degenerative processes of human AV and the interplay of hyperglycaemia and pro-degenerative conditions on the transformation of VICs in vitro is analysed.
Methods
Human valvular tissue
AVs were collected with an approval of the Institutional Ethics Committee (Medical Faculty, Heinrich Heine University, Düsseldorf) in accordance with the World Medical Association Declaration of Helsinki with patients’ informed written consent prior to elective isolated or concomitant AV replacement surgery (reference numbers: 3428, 4412R, 4567). Publication of the data does not compromise anonymity or confidentiality or breach local data protection laws. The data set does not contain any direct or indirect identifiers. No organs/tissues were procured from prisoners. The donors of the control AVs that were examined in this study underwent AV surgery for AV regurgitation (12%) or alternatively AV tissue of patients undergoing orthotopic heart transplantation (88%) was retrieved for analysis. Control AVs were only used unless presenting with structural changes upon macroscopic examination. For the fibrotic group, AV tissue with macroscopic appearance of cusp fibrosis and thickening but with absence of gross calcification was collected. Hence, patients from the AV fibrotic group had in 61% AV stenosis, in 26% AV regurgitation, in 3% aorta-related diseases and in 10% heart failure (and in consequence underwent heart transplantation) as the leading indication for surgery. AVs of the calcified group were derived solely from patients undergoing AV replacement due to stenosis. After excision, macroscopic images of individual AV cusps were taken, before separate snap-freezing of individual cusps. Demographic data of the donors comprising age and gender were documented. Moreover, cardiovascular risk factors, comorbidities, laboratory values and medication were documented. Patients with bicuspid AV were excluded from the study, since these valves are associated with altered hemodynamics predisposed for accelerated functional deterioration with early calcifications and stenosis as well as development of regurgitation. Bicuspid AVs were defined based on a combination of pre-op echography assessment if available, as well as intraoperative information by the surgeon. Patients with a medical history of rheumatic heart disease or active endocarditis were also excluded from analysis.
For classification of degree of degeneration, the cusps of each patient were categorized by macroscopic presence and extent of fibrotic lesions and calcific nodules. It is important to emphasize that this macroscopic classification of cusp tissue appearance does not reflect the functional performance of a given valve. Further analysis was performed on individual cusps without any signs of sclerosis (control group; n = 17), on cusps presenting fibrosis and structural thickening without signs of gross calcification (fibrotic group; n = 69) or cusps with severe calcification occupying major parts of the cusps (calcified group; n = 72). Cusps exposing partial or intermediate calcification were excluded from this study. One cusp per patient was analysed using the whole cusp without excision of any parts.
Animal experiments and surgical procedures
All animal care and experimental procedures were performed in accordance with the guidelines for the use of experimental animals as outlined in the Deutsches Tierschutzgesetz and were approved by the Local Ethics Committee for Animal Experimentation (Landesamt für Natur, Umwelt und Verbraucherschutz NRW, Germany; reference number: G404/11). Animal experiments were conducted in laboratories of ZETT (Zentrale Einrichtung für Tierforschung und wissenschaftliche Tierschutzaufgaben) of the Heinrich Heine University, Düsseldorf. For animal experiments, a biglycan loss-of-function mouse strain on a C57BL/6J background was used. Since we did not see gender-dependent differences in pilot experiments and in order to avoid unwanted interplay with female hormone system side effects, we chose male mice for the main experiments. Biglycan is located on the X chromosome, for which reason male mutant mice are Bgn−/0 and male wild-type mice are Bgn+/0. 30 For better visualization, Bgn+/0 are referred to as wild-type mice. Thus, all mouse experiments were performed with male Bgn−/0 and wild-type littermates in an ectopic tissue patch degeneration model. 31 Therefore, 5-mm2 biopsy punches of tissue material [raw bovine pericardium stored in proprietary ethylenediaminetetraacetic acid (EDTA) and isopropyl alcohol (Neovasc, Richmond, BC, Canada)] were prepared, washed in sterile phosphate-buffered saline (PBS) and subcutaneously implanted at the dorsal side of the anaesthetized 8-week-old mice. For anaesthesia, 5 mg/kg Rompun (Bayer, Leverkusen, Germany) and 100 mg/kg Ketanest (Pfizer, Freiburg, Germany) were injected intraperitoneally. To mitigate pain after surgery, mice received a subcutaneous injection of 5 mg per kg bodyweight Rimadyl (Pfizer) at the end of the surgical intervention. Mice received a vitamin D3 excess (76,500 IU) chow with high calcium (1.7%; cat. no. E15312-44) and water ad libitum and were kept in standard housing conditions. After 8 and 16 weeks, respectively, mice were sacrificed with CO2, the skin was opened and tissue patches were processed for subsequent analysis.
Isolation and culture of primary ovine VICs
Ovine AVs of six individual sheep (6- to 8-month old) were freshly obtained from a local abattoir. Leaflets were excised, washed in PBS and minced in small pieces. Pieces were incubated in Dulbecco’s modified Eagle’s medium (DMEM) with GlutaMAX™ supplement including 10% foetal calf serum (FCS), 1% penicillin/streptomycin and 1% non-essential amino acids either in control medium (DMEM containing 1 g/L glucose) or in DMEM containing 4.5 g/L glucose at 37°C and 5% CO2. Outgrown VICs were cultured for three to four passages prior to the experiments. For all in vitro experiments, cell culture medium containing 1 g/L (100 mg/dL; 5.5 mM) glucose referred to as ‘normoglycaemic conditions’ and cell culture medium containing 4.5 g/L (450 mg/dL; 25 mM) glucose was used referred to as ‘hyperglycaemic conditions’. In humans, a blood glucose level of 5.5 mM would be rated as normal, whereas 25 mM glucose levels would correspond to a critical high value, with an immediate need to intervention. 32 Beta glycerolphosphate/calcium chloride (10 mM/1.5 mM) was used for pro-degenerative treatment of the cells. Medium was changed every second day. All experiments with VIC were conducted after 5 days of treatment. Further extended treatment led to a strong degeneration and significant cell loss (data not shown).
Messenger RNA isolation and semi-quantitative real-time polymerase chain reaction analysis
Total RNA from human AV cusps was isolated using the TRIzol reagent according to the manufacturer’s instructions followed by a DNase digestion and RNA purification using the Qiagen RNeasy Mini Kit. Total RNAs of cultured ovine VICs were isolated by direct application of the RNeasy Mini Kt. Synthesis of complementary DNA (cDNA) was performed using the Qiagen QuantiTect Reverse Transcription Kit. Semi-quantitative real-time PCR was performed with an ABI 7300 reverse-transcription polymerase chain reaction (RT-PCR) system using the Platinum SYBR Green qPCR SuperMix-UDG/ROX kit (Invitrogen, Carlsbad, CA, USA). PCR was performed by starting with an initial step for 2 min at 50°C, followed by 2 min at 95°C. A total of 40 cycles were performed with 15 s at 95°C and 30 s at 60°C followed by single steps for 15 s at 95°C, 1 min at 60°C and 15 s at 95°C (primer sequences are listed in Table 1). Relative gene expression was determined by the comparative Ct method (2−ΔΔCt method).
Primer sequences.

Primer sequences used for semi-quantitative real-time polymerase chain reaction (PCR).
Biglycan core protein isolation and Western blot
Isolation of the core protein of biglycan was performed by anion-exchange chromatography using DEAE-Sephacel beads. Therefore, snap-frozen human AV cusps (n = 40 in total; n = 10 per subgroup) were pestled and solubilized in 5 mL DEAE buffer (8 M urea, 2 mM EDTA, 50 mM Tris and 0.5% Triton X-100, pH 7.5) and the samples were centrifuged to remove cell debris. Supernatant was used for measurement of the total protein concentration. For biglycan isolation, Sephacel beads were equilibrated with DEAE washing buffer containing 0.25 M NaCl. The solubilized samples were applied on the beads and washed five times with DEAE washing buffer. Elution was performed with 900 µL DEAE elution buffer containing 3 M NaCl. About 15 µL of chondroitin sulphate A (10 mg/mL) was added to the samples and an ethanol/potassium acetate precipitation was performed overnight. Precipitated samples were centrifuged. Pellets were solved in Aqua Dest and ethanol/potassium acetate precipitation was performed overnight. Samples were centrifuged and pellets were solved in Aqua Dest. Resolved pellets were digested with chondroitinase ABC in buffer [100 mM Tris, 15% bovine serum albumin (BSA) and 18 mM sodium acetate (pH 8)] containing a protease inhibitor mix [6-aminohexanoic acid, benzamidine and phenylmethylsulphonyl fluoride (PMSF)] for 3 h at 37°C. Proteins were separated on a 10% reducing polyacrylamide gel and blotted on nitrocellulose. Detection of biglycan signals was performed with an anti-biglycan (cat. no. sc-100857; clone 3E2; Santa Cruz Biotechnology, Dallas, TX, USA) and a secondary antibody (IRDye 800 CW; cat. no. 926-32210; LI-COR Biosciences, Lincoln, NE, USA) using an Odyssey scanner (LI-COR Biosciences). Intensity of protein bands was measured with the LI-COR Odyssey software and data were normalized to the total protein content of the input DEAE solution by a butterfly protein assay. Therefore, 10 µL of each sample was spotted in triplicates on Whatman paper, placed in 24-well plates and incubated for 10 min at room temperature, followed by Coomassie (2.5 g/L, 40% MeOH, 10% acetic acid) staining for 10 min. After destaining (3 × 5 min; 20% MeOH, 7% acetic acid), Whatman paper was incubated with 3% sodium dodecyl sulphate (SDS) solution at room temperature overnight. Extinction of samples was measured at 590 nm.
Tissue processing
Explanted tissue patches were processed for further analysis immediately after dissection. For (immuno-) histological analysis, patches were snap-frozen in cryo compound, and 5-µm-thin sections of frozen patch material were prepared and used for (immuno-) histological stainings. Patches for calcium assay were snap-frozen in liquid nitrogen and were shredded for analysis of calcium content.
Histochemical and immunohistochemical stainings
Tissue sections of tissue patches were stained according to a standard protocol of Movat’s pentachrome and host cell invasion into tissue patches was determined (wild type 8 weeks n = 6 and Bgn−/0 8 weeks n = 5; wild type 16 weeks n = 4 and Bgn−/0 16 weeks n = 7). For immunohistochemical stainings with monoclonal antibodies against biglycan (rabbit anti-biglycan; kindly provided by Larry W. Fisher, National Institute of Dental and Craniofacial Research, National Institutes of Health, Bethesda, MD, USA) and Mac2 (cat. no. CL8942AP; clone M3/38; Cedarlane, Burlington, ON, Canada), tissue sections were fixed 10 min with 4% formaldehyde and washed in PBS. After washing with PBS, sections stained for biglycan were incubated for 60 min with 2 U/100 µL chondroitinase at 37°C. Sections were blocked with 10% FCS/1% BSA in Tris-buffered saline for 60 min. Sections were washed with PBS and incubated with the primary antibody (biglycan 1:500 and Mac2 1:600 in 1% BSA/PBS) overnight. After washing steps in PBS, sections were treated with H2O2 (3%) for 10 min, again washed in PBS and incubated with a horseradish peroxidase (HRP)-coupled secondary antibody [goat anti-rabbit IgG-HRP polyclonal antibody (cat. no. sc-2004; Santa Cruz Biotechnology) or goat anti-rat IgG2A-HRP polyclonal antibody (cat. no. NB7126; Novus Biologicals, Littleton, CO, USA)] for 45 min. After washing steps in PBS, sections were treated with DAB Substrate Kit (Zytomed, Berlin, Germany) according to the manufacturer’s protocol and cell nuclei were stained with Meyer’s hematoxylin. Group sizes for immunohistochemical stainings were as follows: wild type and Bgn−/0 8 weeks, n = 6 each; wild type and Bgn−/0 16 weeks, n = 7 each. Immunohistochemical stainings of human calcified AV for biglycan were performed using the identical reagents and protocol as performed for the tissue patches with n = 5 AV of non-diabetics and n = 5 AV of diabetic patients. Quantification, that is, analysis of the integrated density of DAB stainings, was performed using the Fiji/ImageJ2 software.
Detection of calcium amount
For detection of calcium amounts of tissue patch material, a colorimetric calcium assay kit based on the cresolphthalein complexon method was used (cat. no. Cay-701220; Biomol, Hamburg, Germany). Shredded tissue patch material was pestled and lysed in 100 mM Tris-HCl buffer (pH 7) and centrifuged for 15 min at 4°C at 11,000 r/min. Afterwards, extinction of the supernatant was measured at 580 nm. Group sizes were as follows: wild-type and Bgn−/0 8 weeks n = 6, wild-type and Bgn−/0 16 weeks n = 7. Calcium amounts of cultured VICs were detected using a calcium assay kit (cat. no. KA1644; Abnova, Taipeh, Taiwan). Cells were washed with PBS and lysed in 100 mM Tris-HCl buffer (pH 7) containing 0.1% Triton-X-100 under rotation for 2 h at 4°C. Samples were centrifuged for 15 min at 4°C at 13,000 r/min. Extinction of supernatant was measured at 612 nm. Group sizes were n = 6 for each treatment. For measurements, a Tecan Reader Infinite M1000PRO was used.
Biglycan enzyme-linked immunosorbent assay
For determination of biglycan core protein in cell culture supernatant of VIC, a sandwich enzyme immunoassay for in vitro quantitative measurement of biglycan was used (cat. no. SEJ226; Cloud-Clone, Katy, TX, USA). Supernatants were collected after 5 days of treatment and centrifuged for 20 min at 1000g and stored at −80°C until usage. The enzyme-linked immunosorbent assay (ELISA) was performed according to the manufacturer’s instructions and extinction of the supernatants was measured at 450 nm. For measurements, a Tecan Reader Infinite M1000PRO was used. Group sizes were n = 4 for each treatment group. Data were normalized to the total protein amount of underlying cells using a DC™ Protein Assay (cat. no. 5000114; Bio-Rad, Hercules, CA, USA) according to the manufacturer’s instructions.
Statistical analyses
For statistical analyses, GraphPad Prism 6.0 was used. Reported data are represented as mean ± standard error of mean of all experiments performed. Since data were not parametric, unpaired two-tailed Mann–Whitney U tests were performed when comparing two groups with each other. If more than two groups were compared, a Kruskal–Wallis test with Dunn’s multiple comparison test was performed. For statistical analysis of patient data, chi-square test for categorical and Kruskal–Wallis test with Dunn’s post hoc test for continuous variables was used when comparing more than two groups. For comparison between two groups, Fisher’s exact test for categorical variables and an unpaired two-tailed Mann–Whitney U test for continuous variables were used. For all statistical analyses, p < 0.05 was considered as statistically significant. Power calculations were performed considering α = 0.05 (5%) with a specificity of 1 − α = 0.95 (95%) and β = 0.20 (20%) with a power of 1 − β = 0.80 (80%).
Results
SLRP in degenerated AV – only biglycan is significantly upregulated on mRNA level
Gene expression of biglycan, decorin and lumican was analysed comparing control AV cusps with AV cusps presented with morphological changes of fibrosis and with severe calcific degeneration (Figure 1; for patient characteristics, see Table 2). Here, significantly higher gene expression in degenerated AV compared to control AV was only observed for biglycan, whereas decorin and lumican expression in degenerated AV was unchanged. Moreover, gene expression of chondro-osteogenic markers like alkaline phosphatase and osteopontin showed a significant upregulation in degenerated valves as expected. Osteocalcin showed gradual increase in expression levels with increasing stages of degeneration, though not statistically significant. TGFβ was also significantly upregulated in degenerated AV (not shown).
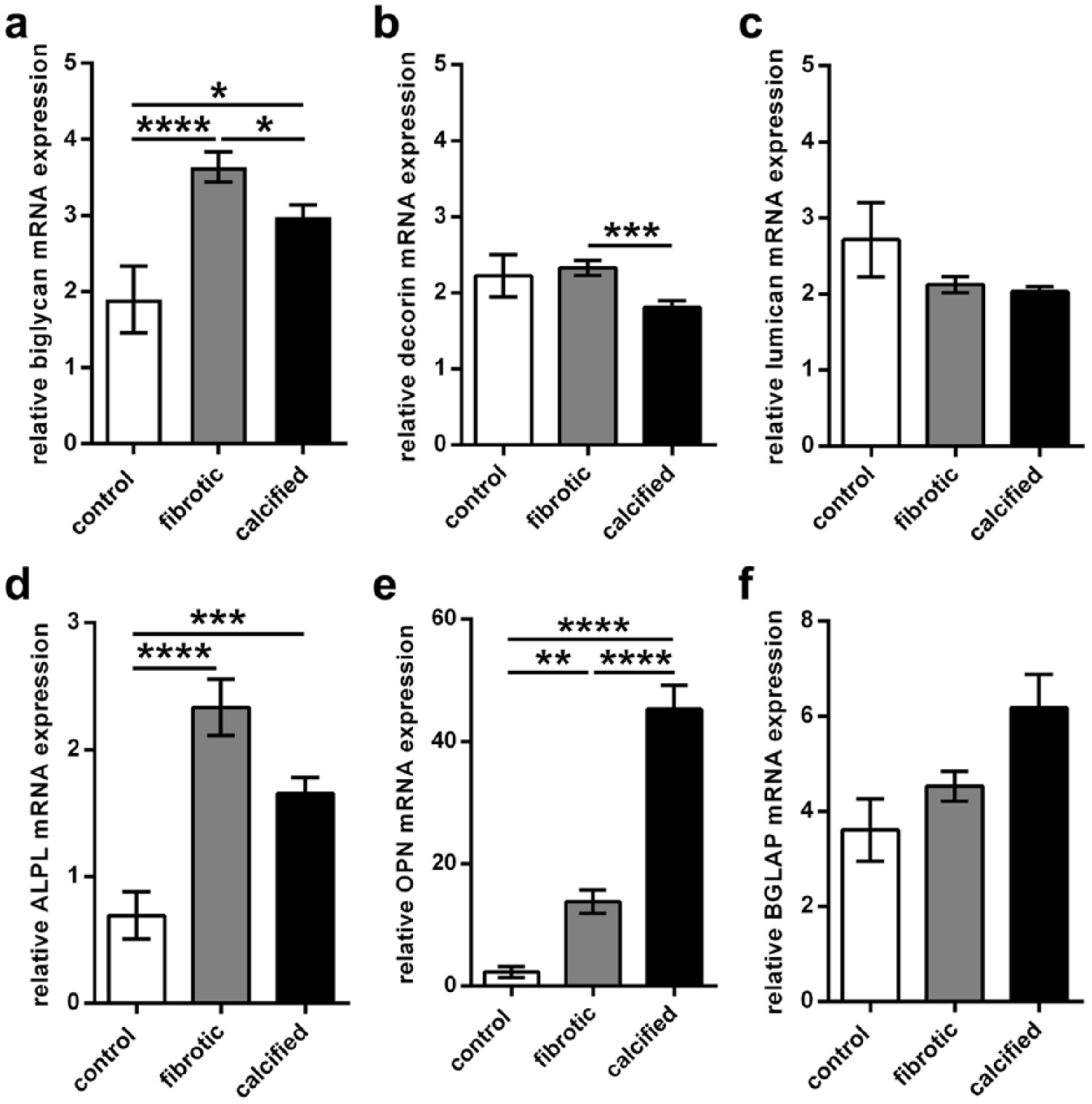
Biglycan, but not decorin or lumican, is significantly upregulated in degenerated AV. Biglycan and decorin are differentially expressed in various stages of degeneration. Gene expression analysis showed that (a) biglycan is significantly upregulated in fibrotic (n = 69) and calcified AV (n = 72) compared to control (n = 17), whereas (b) decorin and (c) lumican showed no difference to control. (a) Biglycan and (b) decorin are significantly upregulated in early stages of AV degeneration, that is, fibrotic AV compared to calcified AV, (c) whereas lumican remained unaltered. The chondro-osteogenic markers (d) alkaline phosphatase and (e) osteopontin showed significantly higher gene expression in diseased AV compared to control AV. (f) Osteocalcin gene expression showed an increase in degenerated AV. ALPL: alkaline phosphatase; OPN: osteopontin; BGLAP: osteocalcin; for statistical analysis, a Kruskal–Wallis with Dunn’s post hoc test was used. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Patient characteristics.
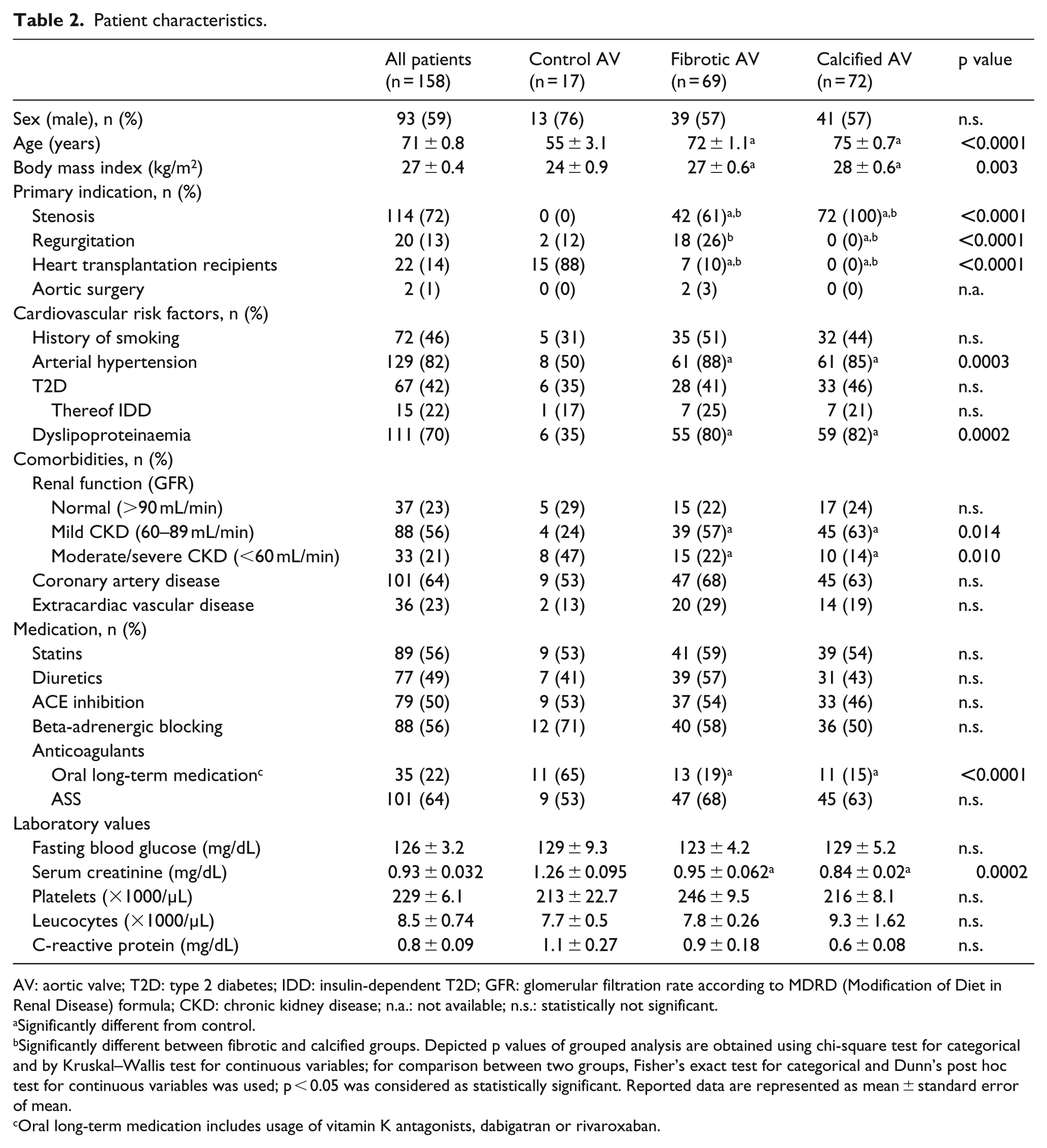
AV: aortic valve; T2D: type 2 diabetes; IDD: insulin-dependent T2D; GFR: glomerular filtration rate according to MDRD (Modification of Diet in Renal Disease) formula; CKD: chronic kidney disease; n.a.: not available; n.s.: statistically not significant.
Significantly different from control.
Significantly different between fibrotic and calcified groups. Depicted p values of grouped analysis are obtained using chi-square test for categorical and by Kruskal–Wallis test for continuous variables; for comparison between two groups, Fisher’s exact test for categorical and Dunn’s post hoc test for continuous variables was used; p < 0.05 was considered as statistically significant. Reported data are represented as mean ± standard error of mean.
Oral long-term medication includes usage of vitamin K antagonists, dabigatran or rivaroxaban.
Biglycan and decorin are upregulated in early stages of AV degeneration
Gene expression of biglycan and decorin was significantly higher in fibrotic AV compared to calcified AV (Figure 1), whereas lumican showed no differences. Analysis of chondro-osteogenic markers showed significant higher expression of osteopontin and by trend a higher expression of osteocalcin in late stages of degeneration. Alkaline phosphatase showed higher gene expression levels in fibrotic AV compared to calcified AV, though not statistically significant. Upregulation of biglycan on the messenger RNA (mRNA) level could also be shown on protein level in AV tissue of various stages of DAVD (Figure 2; for patient characteristics, see Table 3). Here, Western blot analysis showed that biglycan core protein was remarkably decreased in calcified AV when compared to fibrotic AV.
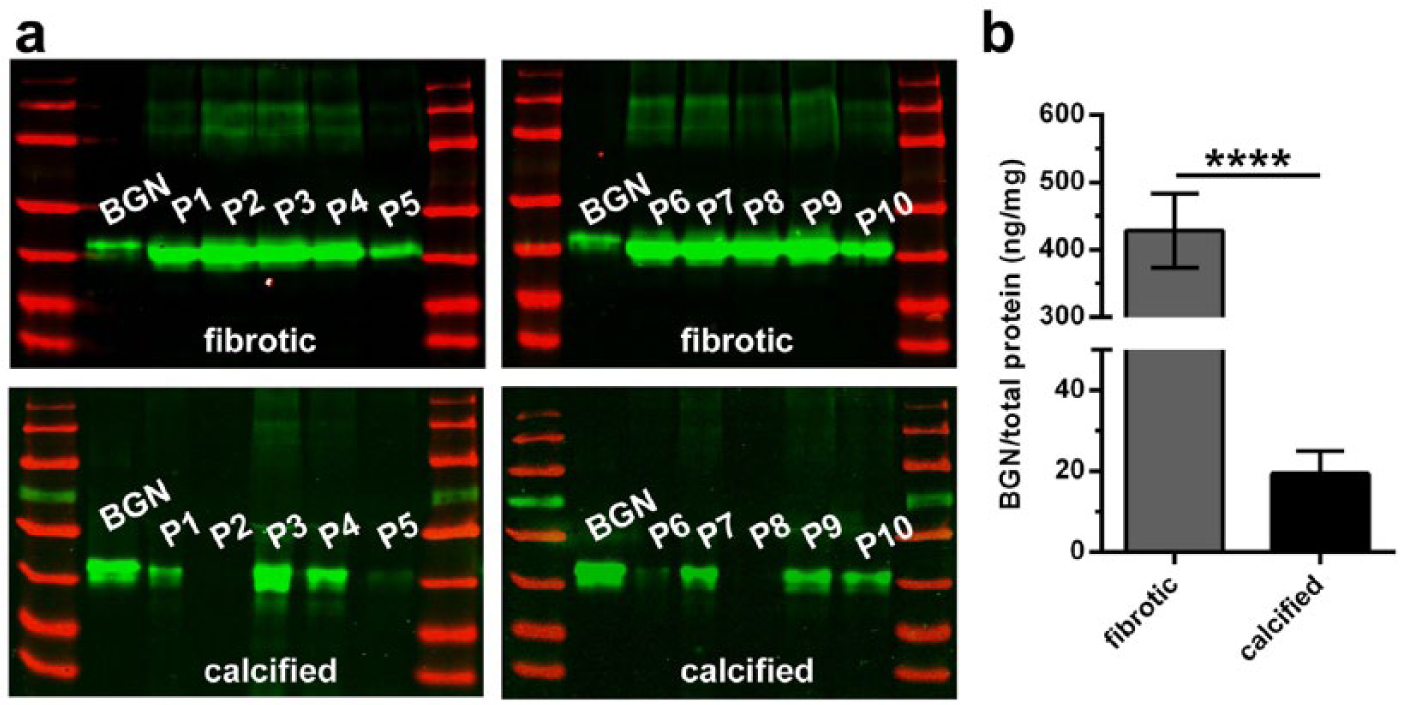
Biglycan core protein is elevated in early stages of AV degeneration. (a) Western blots of fibrotic and calcified AV (n = 10 each) were probed for biglycan content. Density analysis data of biglycan signals of AV were normalized to the signal of 50 ng recombinant biglycan (see first lane in each blot) and were normalized to total protein amount of input material. (b) Plotted data of Western blot analysis (all samples were derived at the same time and processed in parallel). Fibrotic AVs show a significantly higher amount of biglycan core protein compared to calcified AV. BGN: biglycan; for statistical analysis, an unpaired two-tailed Mann–Whitney U test was used. ****p < 0.0001.
Patient characteristics (subgroup for Western blot analysis presented in Figure 2).
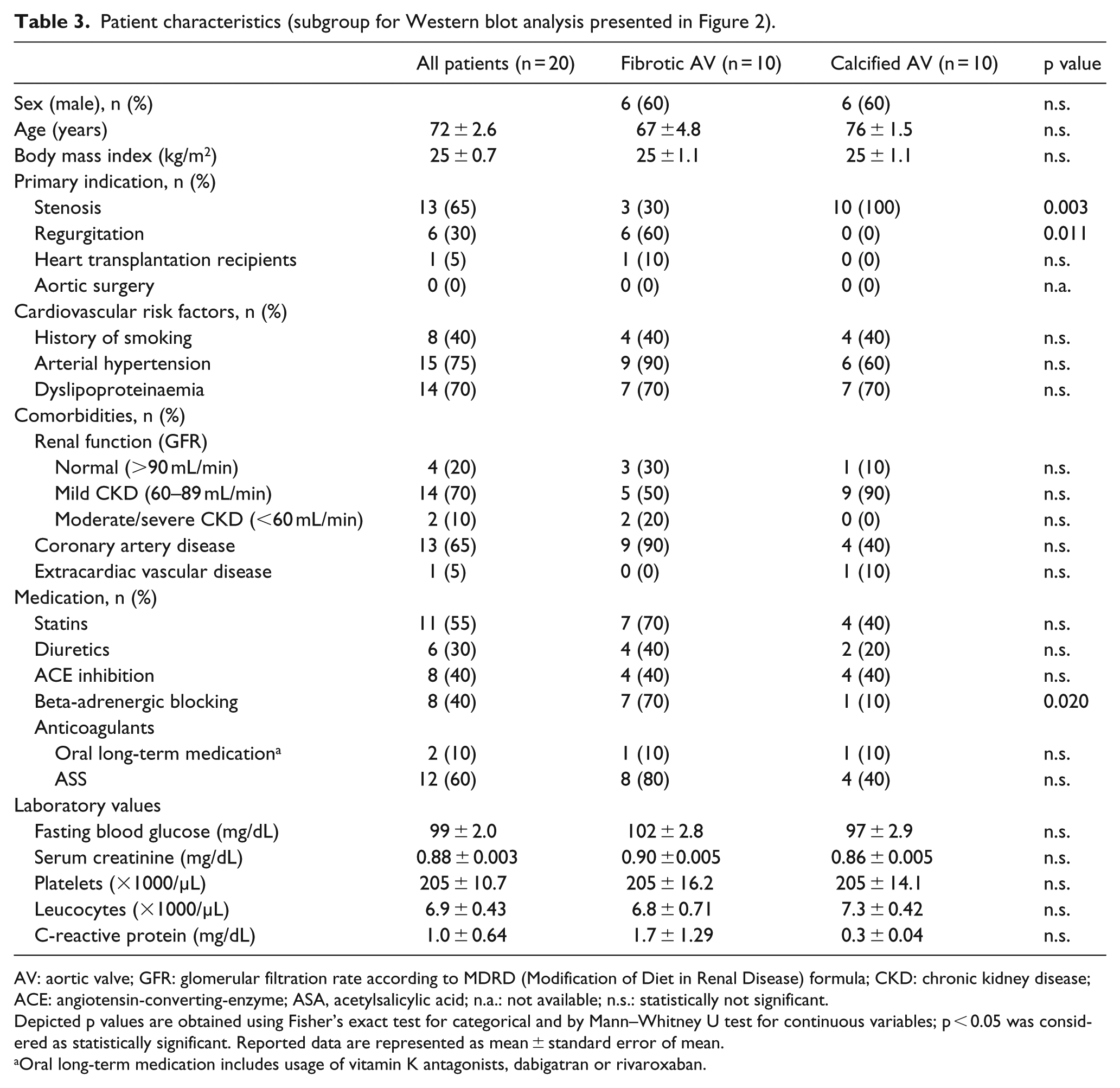
AV: aortic valve; GFR: glomerular filtration rate according to MDRD (Modification of Diet in Renal Disease) formula; CKD: chronic kidney disease; ACE: angiotensin-converting-enzyme; ASA, acetylsalicylic acid; n.a.: not available; n.s.: statistically not significant.
Depicted p values are obtained using Fisher’s exact test for categorical and by Mann–Whitney U test for continuous variables; p < 0.05 was considered as statistically significant. Reported data are represented as mean ± standard error of mean.
Oral long-term medication includes usage of vitamin K antagonists, dabigatran or rivaroxaban.
Loss of biglycan impairs degenerative and remodelling processes in vivo
The influence of a lack of biglycan on calcium accumulation, migration of invading interstitial cells as well as of invasion of macrophages was analysed in vivo (Figure 3). Therefore, an unfixed pericardial tissue patch was implanted into wild-type and biglycan loss-of-function (Bgn−/0) mice to study the biological role of biglycan in vivo on the aforementioned parameters.
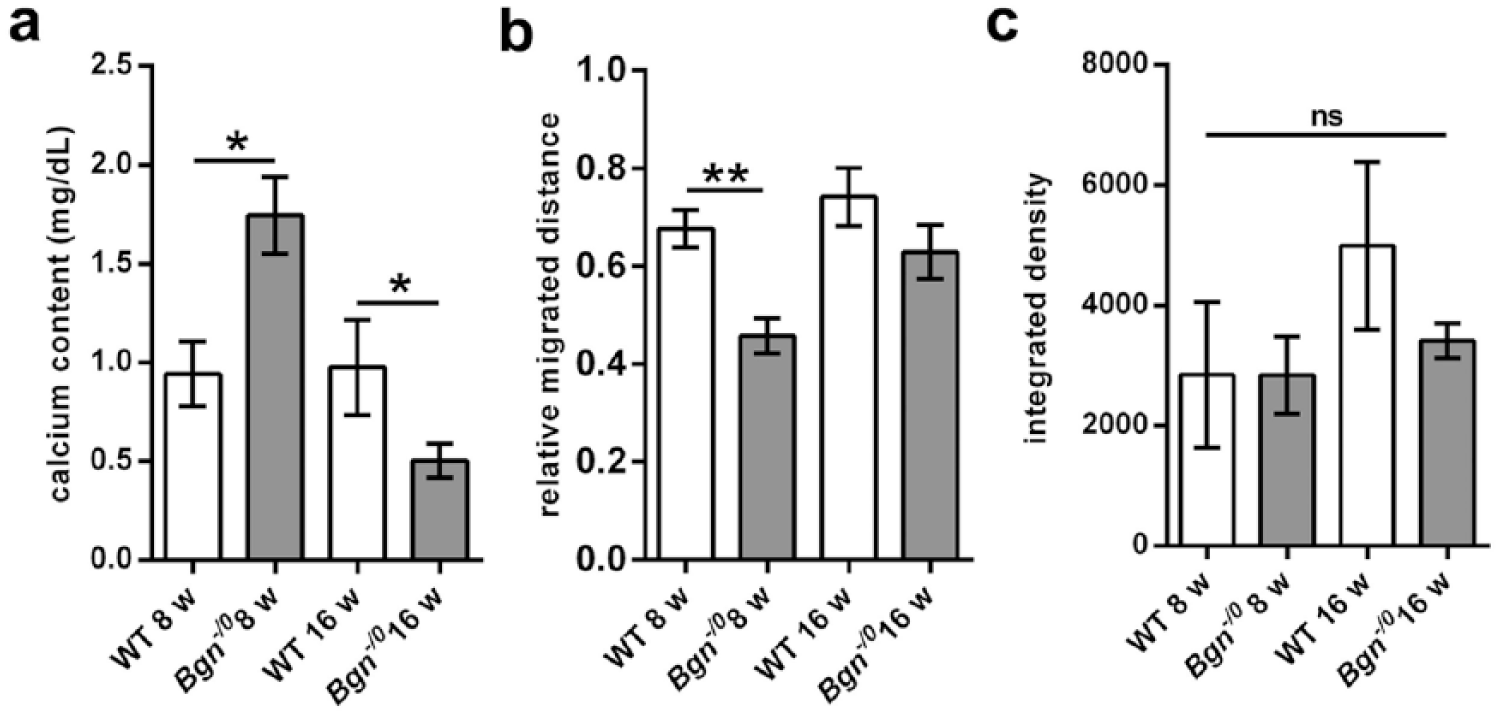
Loss of biglycan impairs degenerative and remodelling processes in vivo. (a) Loss of biglycan leads to a higher amount of calcium accumulation in tissue patches after 8 weeks in vivo (n = 6 each). Long-term conditions (16 weeks) lead to lower calcium accumulation in tissue patches of Bgn−/0 mice (n = 7 each). (b) Relative migrated distance of host cells into the patch is lower in Bgn−/0 mice (n = 5) compared to wild-type mice (WT; n = 6) at the early time point (WT 16 weeks n = 4, Bgn−/0 16 weeks n = 7). (c) Quantification data of immunohistochemical staining of macrophages in tissue patches show no differences between WT and Bgn−/0 or between 8- and 16-week time points. Bgn: biglycan; Bgn−/0: biglycan loss-of-function; WT: wild-type; for statistical analysis, a Kruskal–Wallis with Dunn’s post hoc test was used. *p < 0.05; **p < 0.01; ns: not significant.
Significantly greater calcium accumulation in the tissue patches was observed in Bgn−/0 compared to wild-type mice in the early time point, whereas in long-term experiments, the tissue patches of Bgn−/0 mice contained significantly lower amounts of calcium compared to wild-type mice (Figure 3(a)). A detailed analysis of the migration distance of invading host cells revealed an inhibitory effect of biglycan deletion on the overall migration capacity. Here, cells invaded the patch material to a significantly lesser degree when implanted in Bgn−/0 as compared to wild-type littermates. The differences observed in the long-term implantation groups did not reach statistical significance, although a trend was obvious (Figure 3(b)).
Since it has been shown that biglycan is involved in inflammatory responses by attracting macrophages, thus accelerating the progression of degenerative diseases like atherosclerosis,9,33,34 immunohistochemical staining with Mac2 was performed. The staining showed that macrophages were mainly present in areas in close proximity to surgical sutures (see Supplemental Figure) with very sparse invasion of cells into the patch tissue itself. Quantification of stainings showed that there were no differences in the amount of Mac2-positive cells between wild-type and Bgn−/0 mice or there were no differences between the two examined time points (Figure 3(c)). There was a trend towards higher density of Mac2 signals in tissue patches of WT of the late time point compared to that of the early time point WT group. Thus, a biglycan staining of the WT groups was performed to evaluate whether this might be due to enhanced biglycan accumulation in the late time point. However, quantification of immunohistochemical stainings showed that there were no differences in the amount of biglycan between the early and the late time points (see Supplemental Figure). A specific staining for proliferating cells revealed no remarkable level of cell proliferation in the patches (not shown). Taken together, the lack of biglycan led to no obvious differences in macrophage infiltration into the tissue patch and in proliferation of invading or surrounding cells, independent of the length of the in vivo approach. Nevertheless, lack of biglycan leads to a time-dependent altered calcium accumulation and has an anti-migratory effect in interstitial cells in vivo.
Biglycan expression in calcified AV is regulated by T2D
It is known that biglycan is able to influence the formation or assembly of other ECM molecules such as elastin or collagen in metabolic disorders.35–37 T2D as one of the risk factors leading to DAVD has also been shown to lead to fibrosis and matrix remodelling in metabolically induced cardiovascular diseases 38 substantially affecting ECM components like elastin and collagen.22,39 Thus, we analysed the influence of T2D on the expression of biglycan in our human DAVD samples. Therefore, in subgroup analysis, calcified AVs derived from non-diabetics were compared to respective samples derived from patients suffering from T2D. Here, biglycan expression was significantly higher in calcified AV tissue of patients with T2D (see Figure 4; for patient characteristics, see Table 4). In contrast, decorin and lumican showed no regulation associated with presence or absence of T2D, respectively. Western blot analysis revealed only a slight upward trend to higher amounts of biglycan core protein in calcified AV derived from patients with T2D compared to AV of non-diabetics (Figure 4(d) and (e)). Western blot data were then verified by immunohistochemical stainings of biglycan in calcified AV of diabetic and non-diabetic patients showing a statistically significant higher amount of biglycan in calcified AV of diabetic patients (Figure 4(f)–(h)).
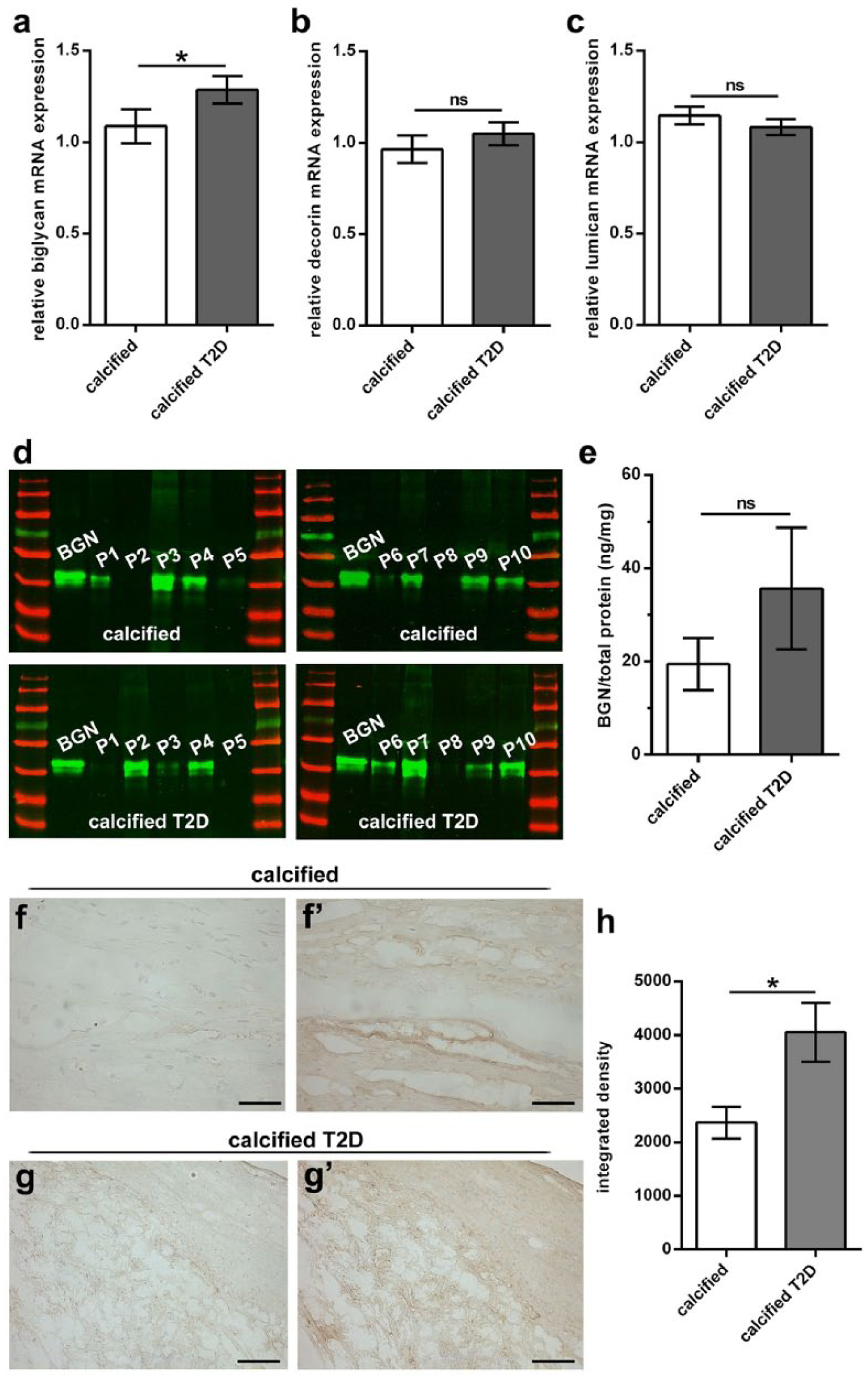
Type 2 diabetes increases the expression of biglycan in calcified AV. (a) Biglycan gene expression is upregulated in AV of diabetics (n = 33) compared to AV of non-diabetics (n = 39) in late stages of AV degeneration. (b) Decorin and (c) lumican gene expression in calcified AV is not influenced by the presence of T2D. (d) Western blots of calcified AV of non-diabetics and calcified AV of diabetics (n = 10 each) were probed for biglycan content. Density analysis data of biglycan signals of AV were normalized to the signal of 50 ng recombinant biglycan (see first lane in each blot) and were normalized to total protein amount of input material. (e) Plotted data of Western blot analysis (all samples were derived at the same time and processed in parallel). T2D leads to a slight upward trend in the amount of biglycan core protein in calcified AV. (f–g′) Exemplary immunohistochemical stainings of biglycan of calcified AV of non-diabetics (f′) and patients with T2D (g′; for specificity controls see (f) and (g)). (h) Quantification data of biglycan staining show a significantly higher biglycan protein expression in AV of diabetics (n = 5 patients each). For statistical analysis, an unpaired two-tailed Mann–Whitney U test was used. *p < 0.05; ns: not significant. BGN: biglycan; T2D: type 2 diabetes; scale bars/magnification: 50 μm/400× (f, f′) and 100 µm/200× (g, g′).
Patient characteristics of calcified AV subgroup.
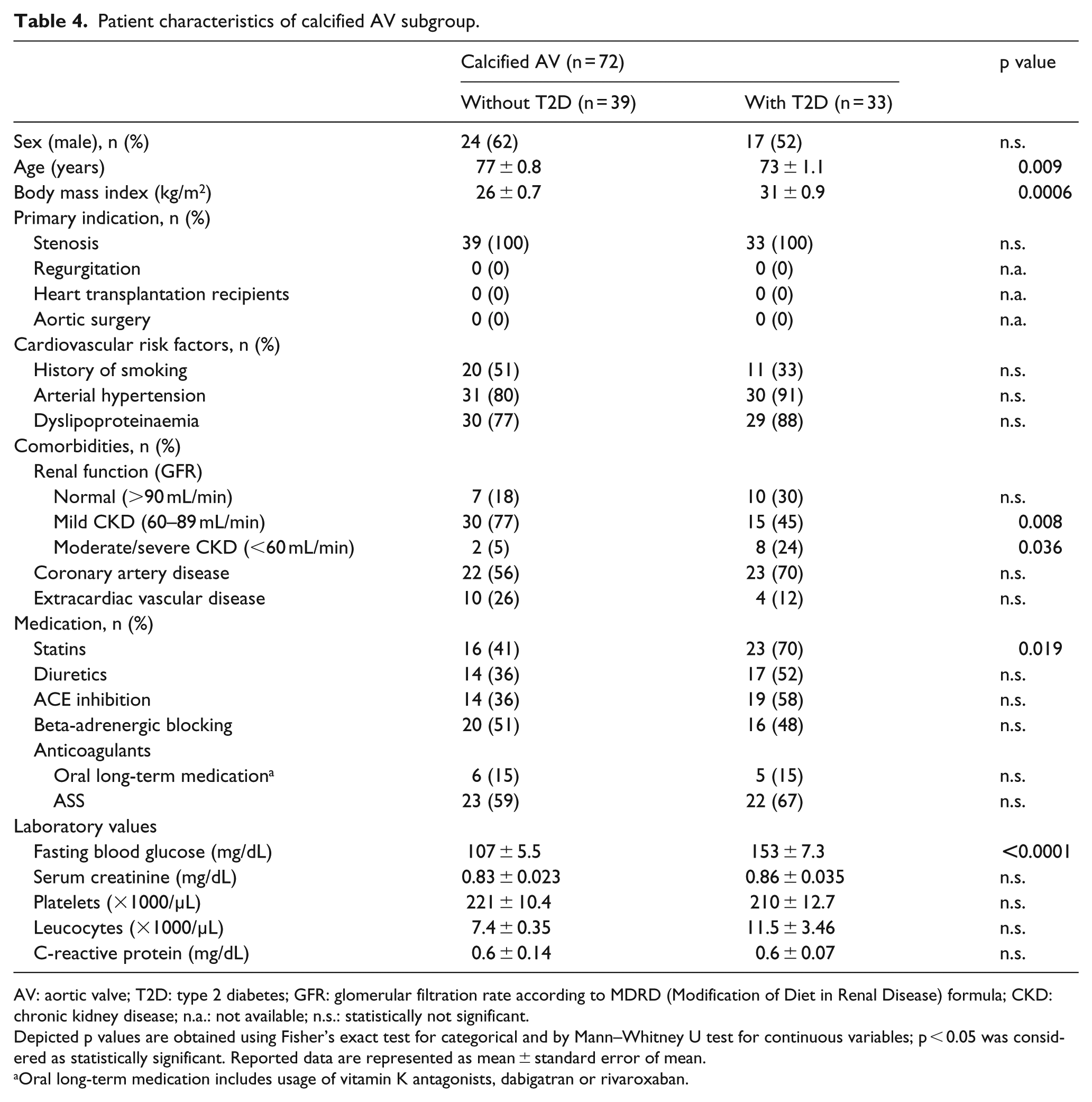
AV: aortic valve; T2D: type 2 diabetes; GFR: glomerular filtration rate according to MDRD (Modification of Diet in Renal Disease) formula; CKD: chronic kidney disease; n.a.: not available; n.s.: statistically not significant.
Depicted p values are obtained using Fisher’s exact test for categorical and by Mann–Whitney U test for continuous variables; p < 0.05 was considered as statistically significant. Reported data are represented as mean ± standard error of mean.
Oral long-term medication includes usage of vitamin K antagonists, dabigatran or rivaroxaban.
Hyperglycaemic conditions lead to changes in biglycan expression under βGP/CaCl2 stimulation and aggravate chondro-osteogenic differentiation of VIC
Isolated treatment with hyperglycaemic conditions or βGP/CaCl2, respectively, led to no changes in the gene expression of biglycan, decorin or lumican (Figure 5). However, the combination of hyperglycaemic conditions and βGP/CaCl2 led to a slight trend to higher, though not statistically significant upregulation of biglycan mRNA expression. Interestingly, using a biglycan ELISA assay, a statistically significant increase in biglycan protein expression by the combination of these factors could be shown. Nevertheless, decorin and lumican mRNA expression remained unchanged under this treatment. Notably, in this setting, hyperglycaemic conditions in combination with βGP/CaCl2 also induced an upregulation of osteogenic markers, further suggesting a detrimental effect of hyperglycaemia in the context of chondro-osteogenic transformation. Likewise, osteopontin expression was significantly enhanced when pro-degenerative treatment with βGP/CaCl2 was further supplemented with hyperglycaemic medium. TGFβ showed a trend towards higher mRNA expression under combined treatment compared to control. In addition, stimulation with βGP/CaCl2 alone and in combination with hyperglycaemia resulted in significantly higher calcium accumulation compared to control.
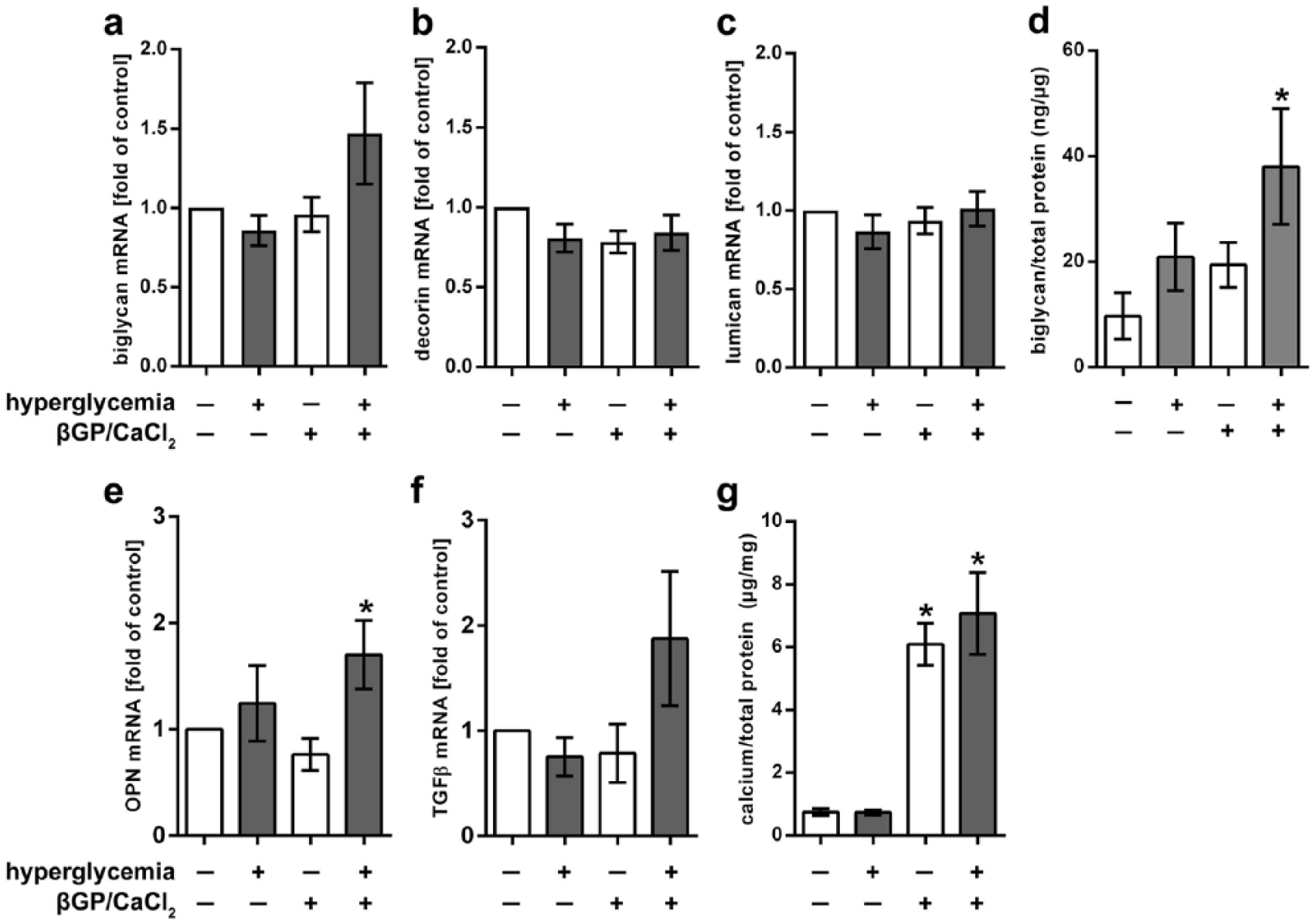
Hyperglycaemic conditions lead to changes in biglycan expression under βGP/CaCl2 stimulation and aggravate chondro-osteogenic differentiation. (a) Biglycan shows a slight trend to higher mRNA expression by combined treatment βGP/CaCl2 and hyperglycaemia. (b) Decorin and (c) lumican mRNA expression remains unchanged (n = 12). Biglycan ELISA shows significantly higher amounts of biglycan protein in supernatant of VIC grown under hyperglycaemic and pro-degenerative conditions (d; n = 4). Gene expression of osteopontin and TGFβ is also upregulated by the addition of hyperglycaemic conditions to βGP/CaCl2 stimulus (e, f; n = 6). Treatment with βGP/CaCl2 alone or in combination with hyperglycaemia leads to significantly higher calcium accumulation in VIC (g; n = 6). βGP/CaCl2: beta glycerolphosphate/calcium chloride; OPN: osteopontin; TGFβ: transforming growth factor beta; for statistical analysis, a Kruskal–Wallis with Dunn’s post hoc test was used. *p < 0.05.
Discussion
In this study, we focused on SLRP expression in various stages of DAVD analysed in human AV samples, and in parallel on the influence of a lack of biglycan on degenerative processes in an in vivo mouse model. Moreover, we sought to study the influence of T2D on SLRP in human AV degeneration and in an ovine in vitro model.
Our studies clearly demonstrate that biglycan, but not decorin or lumican, is upregulated in degenerated human AV tissue when compared to control AV (Figure 1), which has been controversially discussed in former studies often in inadequate numbers of cases.15,18,19,21 However, in our clinical cohort, TGFβ gene expression was also significantly higher in degenerated AV compared to control AV (not shown). TGFβ treatment has been shown to accelerate biglycan and decorin expression.40–42 This might be a cause for higher biglycan mRNA expression in affected AV compared to control AV, although we do not see the same effect on decorin expression. Regarding alterations in gene and protein expression between various stages of degeneration, that is, fibrotic and calcified AV, our data suggest that biglycan and decorin, but not lumican, are significantly upregulated in early stages of DAVD (Figures 1 and 2). Nevertheless, we did not see any differences in TGFβ expression between our subgroups of fibrotic and calcified AV (not shown) indicating that TGFβ might not be the exclusive trigger for SLRP accumulation in fibrotic AV. Fibrotic AV leaflets are characterized by leaflet thickening due to excessive ECM synthesis and accumulation, VIC proliferation 43 and myofibroblast differentiation possibly through interaction with TGFβ. 44 Since biglycan and decorin are involved in lateral assembly of collagen fibres 45 and even can protect collagen from proteolysis, 46 thickening of fibrotic AV might be indirectly linked to an upregulation of biglycan. In our in vitro experiments, we see an increased biglycan expression as a consequence of a degenerative stimulus in combination with hyperglycaemic conditions (Figure 5). Interestingly, we found that upregulation of TGFβ as a consequence of degenerative conditions was similarly dependent on glucose concentrations, a phenomenon that had been described in non-valvular settings in the literature before.47,48
Our in vitro data show that treatment with neither degenerative media alone did change SLRP gene expression nor did hyperglycaemic treatment alone. In contrast, the combination of both triggers led to a slightly higher gene expression and significantly higher protein expression of biglycan, an effect which could not be seen for decorin or lumican (Figure 5). Calcium accumulation yet was upregulated in a fashion that was independent of hyperglycaemia. Calcified AV of diabetics compared to AV of non-diabetics showed a significantly higher biglycan gene expression, whereas the other SLRP remained unchanged. However, protein data showed a slight upward trend in biglycan expression in Western blot analyses for AV of diabetics, but a significantly higher biglycan protein expression in immunohistochemical stainings (Figure 4).
The discrepant results in mRNA and protein expression between human calcified AV of diabetics and ovine VIC treated with pro-degenerative and hyperglycaemic conditions may be indicative of species-dependent effects. Moreover, the in vitro experiments using ovine VIC monolayer cultures are not entirely representative for in vivo conditions and the complex processes that take place in patients. The latter difference certainly involves a complex interaction of different comorbidities causing various effects beyond hyperglycaemia and deranged calcium and phosphate levels. Particularly, the time scales up to the point when morphological changes appear is largely different in vivo as compared to in vitro experiments.
T2D and its implications, such as hyperglycaemia and hyperlipidaemia, have been described to influence the expression of SLRP in vitro and in vivo.23,24 In the patient cohort analysed here, those patients donating calcified AV specimens were all in a pre-obese to obese state based on their body mass index (BMI; 49 see Table 2). Within the patients donating calcified AV (see Table 4), patients without T2D had a BMI of 26 ± 4 (classified as pre-adipose/overweight) and patients with T2D had a distinctly greater BMI of 31 ± 5 (classified as obese class I), which represents the real-world distribution of BMI among patients as observed in the clinical practice. Thus, higher biglycan expression in calcified AV of patients suffering from T2D may be a combined consequence of different effectors, including hyperglycaemia and hyperlipidaemia. Interestingly, an increased level of TGFβ due to diabetic conditions had been described to induce changes in SLRP leading to an enhanced binding of lipids. 50 Nevertheless, in our cohort, we did not see differences in TGFβ expression between AVs from diabetic compared to AVs from non-diabetic patients.
Besides their involvement in T2D, lipids might also be linked to the observed differences in biglycan expression when comparing control with degenerated AV (Figure 1). Here, patients of whom the degenerated AV specimens were derived from had a significantly higher BMI and also presented significantly more often with dyslipoproteinaemia as compared to patients donating control AV (Table 2). Increased biglycan expression in degenerated AV here may be linked to accelerated lipid accumulation, since it has been shown that proteoglycans co-localize with lipid-associated molecules in AV. 18 Moreover, in the initiation process of atherosclerosis, which is in large parts similar to processes taking place in DAVD,51,52 SLRPs are involved in lipid retention 53 and are assumed to worsen atherosclerotic processes. 54 However, in vivo studies in mice have shown that a lack of biglycan is not sufficient to induce an atheroprotective effect. 37
Since the age difference between the control and the DAVD groups is striking (see Table 2), it may be speculated that the difference in biglycan expression could be age-related. Previous reports on SLRP in AV along foetal and postnatal development have shown that composition of SLRP is strongly spatiotemporally organized, occasionally due to the interactions between collagen and SLRP in crucial steps of AV development.11,55 Nevertheless, initial high amounts of biglycan in foetal stages have been described to decrease perinatally with a further increase during progressive ageing. This process is often accompanied by coincidental similar changes in collagen. 56
In vivo analyses revealed that a loss of biglycan leads to an accelerated calcium accumulation in tissue patches in early time points and to a reversed effect in older mice (Figure 3). Higher amounts of calcium in the early time point here might be related to enhanced TGFβ signalling induced by the lack of biglycan,57,58 which in consequence may trigger remodelling and degenerative processes. Lower calcium accumulation at the later time point might be due to the fact that biglycan loss-of-function mice have an osteoporosis-like phenotype with diminished ability of calcium accumulation, which is increased and worsened in elderly mice.30,59 Thus, in vivo biglycan seems to act in a time-dependent manner. However, our in vivo data show that this is not due to increased macrophage infiltration. Decreased migratory ability of interstitial host cells due to a loss of biglycan was observed independently of the various stages of degeneration, though being more pronounced when mice were younger. In vitro experiments revealed that biglycan is able to increase migration of smooth muscle cells and fibroblasts.60,61 This might be indicative for a not migration-related role of biglycan action in the in vivo model of degenerative processes presented here.
Study limitations
Despite the fairly robust sample size composition, the findings presented here on patient-derived AV specimens warrant further validation in larger cohorts. Here, restricted access to human control AV tissue is a limitation. T2D is a complex disease with systemic and local effects in that their entire complexity is difficult to classify and hard to control for when cohorts of patients are analysed. With regard to the biological events in the course of DAVD as triggered by T2D, additional studies using further subgroups appear necessary. These studies may then allow examining single aspects associated with T2D, such as quality of blood glucose control, extent of insulin resistance or concomitant dyslipoproteinaemia. Moreover, cohort studies considering distinct ethnic groups than studied here are warranted. A limitation of the in vitro experiments of this study is the restricted comparability of animal-derived cell culture experiments to studies using human VIC. On the other hand, ovine VICs provide a reliable ‘healthy’ VIC phenotype and therefore may probably be less prone to side effects associated with typical patient-related comorbidities. Nevertheless, convincing data from several studies suggest that differentiation of VIC towards a pro-osteoblastic phenotype as induced by osteogenic medium may not be entirely comparable to the processes taking place on the level of VIC in vivo along initiation and progression of DAVD.62,63 Thus, direct comparisons of in vitro findings on VIC cultured under osteogenic stimulus with ex situ findings on degenerated AV must be drawn with caution.
Conclusion
In conclusion, our results represent a further piece of the puzzle extending our understanding of the detrimental interaction of T2D with proteoglycan regulation in AV tissue leading to further progression of DAVD. Our data further support the role of biglycan as a key factor in the course of DAVD in front of the background of T2D. However, further studies are necessary to prove whether biglycan upregulation in early stages of DAVD or in T2D may be considered as a protective mechanism or whether this special SLRP has a more pro-degenerative role as recent literature suggests. 19 Since biglycan fulfils both functions in vivo as a structural molecule and as a signalling molecule, these interrelations warrant further elucidations.
Supplemental Material
Supplemental_Figure – Supplemental material for Degenerative aortic valve disease and diabetes: Implications for a link between proteoglycans and diabetic disorders in the aortic valve
Supplemental material, Supplemental_Figure for Degenerative aortic valve disease and diabetes: Implications for a link between proteoglycans and diabetic disorders in the aortic valve by Mareike Barth, Jessica I Selig, Svenja Klose, Antje Schomakers, Lena S Kiene, Silja Raschke, Udo Boeken, Payam Akhyari, Jens W Fischer and Artur Lichtenberg in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank Annika Zimmermann for excellent technical assistance. The generous support of the S. Bunnenberg Foundation to the Cardiovascular Research Facilities at the Heinrich Heine University is greatly appreciated. Parts of the herein presented data have been demonstrated on the occasion of the annual meetings of the German Cardiac Society (DGK), the German Society for Surgery (DGCH) and the German Society of Thoracic, Heart and Vascular Surgery (DGTHG).
Author’s contribution
M.B. wrote the main manuscript text and prepared the figures. M.B., J.I.S., S.K., A.S. and L.S.K. carried out the experiments. S.R., U.B., P.A., J.W.F. and A.L. substantially contributed to the design of the project and evaluation of the data. All authors reviewed the manuscript and gave their final approval for publication. J.W.F. and A.L. contributed equally to this work.
Availability of data and material
Primary data and data sets used and/or analysed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The work was funded by the Research Committee of the Medical Faculty of the Heinrich Heine University (KS 9772546) and by the German Heart Foundation/German Foundation of Heart Research to M.B. (Dr. Rusche-Forschungsprojekt 2016). This work was supported by the Deutsche Forschungsgemeinschaft (SFB1116).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
