Abstract
Matrix metalloproteinases (MMPs) and their tissue inhibitors (TIMP) play a critical role during vascular remodelling, in both health and disease. Impaired MMP regulation is associated with many diabetes-related complications. This study examined whether exercise-induced regulation of MMPs is maintained in the skeletal muscle of patients with uncomplicated type 2 diabetes (T2DM). Subjects [12 T2DM, 9 healthy control subjects (CON)] underwent 8 weeks of physical training. Messenger RNA (mRNA) was measured at baseline, during and after 8 weeks of training. Protein was measured pre- and post-training. At baseline, there were no effects of diabetes on MMP or TIMP mRNA or protein. mRNA and protein response to training was similar in both groups, except active MMP-2 protein was elevated post training in T2DM only. Our results indicate that exercise-induced stimulation of MMPs is preserved in skeletal muscle of patients with T2DM. This early stage of diabetes may provide an opportunity for intervention and prevention of complications.
Keywords
Introduction
Cardiovascular complications remain a leading cause of morbidity and mortality for individuals with diabetes. 1 At the root of many diabetes-related complications lies a thickening of the capillary basement membrane (CBM), long regarded as a characteristic feature of microangiopathy.2,3 In the hyperglycaemic environment, interactions between aldose sugars, such as glucose, and free amino groups on peptides or lipids result in rearrangement of the molecular structure, oxidation and the irreversible formation of advanced glycosylation end products (AGE). 4 The CBM and surrounding extracellular matrix (ECM) are particularly susceptible to AGE-induced damage because these structures consist of long half-life proteins, such as collagens and laminins, which consequently have an increased exposure time to aldose sugars. 5 Not exclusive to poorly controlled diabetes, AGE modification of protein occurs even in modest hyperglycaemia. 6
AGE accumulation in the vasculature accelerates pro-inflammatory signalling 7 and enhanced deposition of CBM proteins. 8 Inhibiting the formation of AGE minimizes retinal CBM expansion in long-term diabetes. 9 The biochemical alteration of CBM proteins by AGE impairs proteolysis by matrix metalloproteinases (MMPs), thus compromising the physiological degradation of basement membrane and matrix proteins. Furthermore, decreased MMP-2 expression appears to result directly from glucose-induced modulation of integrin expression in the vascular ECM. The imbalance of degradation to deposition, in turn, contributes to the expansion of the CBM and ensuing vascular disease. 10 Alterations in the microvascular environment, such as these, are implicated in many of the deleterious complications associated with the diabetic state.
MMPs constitute a multi-gene family of secreted and cell surface proteolytic enzymes that process and degrade a variety of extracellular substances. Implicated in both pathological and physiological degradation of CBM proteins, regulation occurs via a tenuous balance between MMPs and their endogenous tissue inhibitors (TIMP). Induction of MMPs, either at gene transcription or at pro-enzyme activation, results in substrate-specific proteolysis, dependent on the particular action of the MMP subtype. 11 MMPs exist in either latent (pro) or active forms and may depend on other MMP subtypes for activation. For example, MT1-MMP (membrane type 1-MMP) cleaves the propeptide of MMP-2, thereby initiating an autoproteolytic activation process. 12 MMPs play a critical role in angiogenesis (wound healing, inflammation, pregnancy and tumour growth) through the degradation of CBM proteins, enabling endothelial cell migration into the interstitial matrix.13,14 While there is general agreement in the literature that diabetes affects normal MMP regulation in the vasculature, there is no consensus as to whether there is an enhanced 15 or diminished activity.16,17 The dysregulation may also result from an imbalance between MMPs and TIMPs. 18
Endurance exercise training has been shown to affect MMP messenger RNA (mRNA), protein and activity in human skeletal muscle. 19 Exercise exerts an effect on MMPs due to multiple potential stimulating factors, such as mechanical loading, 20 local ischaemia 21 and tensile forces in the muscle.22,23 Exercise-induced angiogenesis requires MMP degradation of the ECM in order to allow for endothelial sprouting. 24 Inhibition of MMP-2 and MT1-MMP in rat hindlimb muscles impairs the formation of new capillaries in response to chronic electrical stimulation. 25
Regular physical exercise is an efficient therapeutic strategy in the prevention of cardiovascular disease in the diabetic population. 26 One of the many benefits of training is the potential to decrease CBM width, 27 although the mechanisms explaining this observation have remained largely unexplored. Recent work in the aging rat has shown that exercise reduces fibrosis and MMP dysregulation in cardiac tissue; 28 therefore, it is reasonable to consider that exercise training may also reduce fibrosis and MMP dysregulation in the vasculature of patients with T2DM. To date, the only work published in this area has shown that regular exercise improved MMP and TIMP levels in the serum of patients with T2DM. 29 The authors concluded that these results offer insight into the cardioprotective mechanisms of exercise in T2DM. At this time, no studies that use muscle biopsies from exercising humans with T2DM or that investigate MMPs and TIMP on both mRNA and protein levels have been published.
The purpose of this study was to explore whether exercise training affects MMP levels (pro- and active-MMP-2, pro- and active-MT1-MMP) and/or levels of their tissue inhibitors (TIMP2) in the skeletal muscle of patients with T2DM. We chose to examine subjects with recently diagnosed diabetes and without overt cardiovascular disease, in order to gain insight into whether this is a population which can particularly benefit from regular exercise in order to preserve cardiovascular health, prior to the manifestation of disease. This work is a part of a larger study that investigated the effects of 8 weeks of moderate intensity exercise training on endothelial function in patients with T2DM.
Methods
Subjects
A total of 12 patients with type 2 diabetes (T2DM) and 10 healthy control subjects (CON) were originally recruited for this study, although 1 CON was excluded after initial assessment due to undiagnosed cardiovascular disease (exclusion factor). All results reported here are based on 12 T2DM and 9 CON.
Five patients with T2DM were treated with diet alone, two patients with metformin and five patients with metformin plus sulfonylurea. With the exception of medically controlled hypertension and hypercholesterolaemia in some T2DM patients, none of the subjects had any sign or history of cardiovascular disease. Two patients with T2DM were treated with a statin (hypercholesterolaemia), two patients with ace inhibitors (hypertension), one patient with beta blockers (hypertension) and two patients were treated with anti-diuretics (hypertension). Only one CON was on regular medications (allopurinol for gout).
No differences in blood pressure or cholesterol profile were detected between groups (see Table 1). HbA1C was significantly higher in subjects with T2DM as compared to controls (p < 0.05), thus confirming diabetes status. Average was 5.1 ± 1.1 years with diagnosed T2DM.
Subject characteristics.
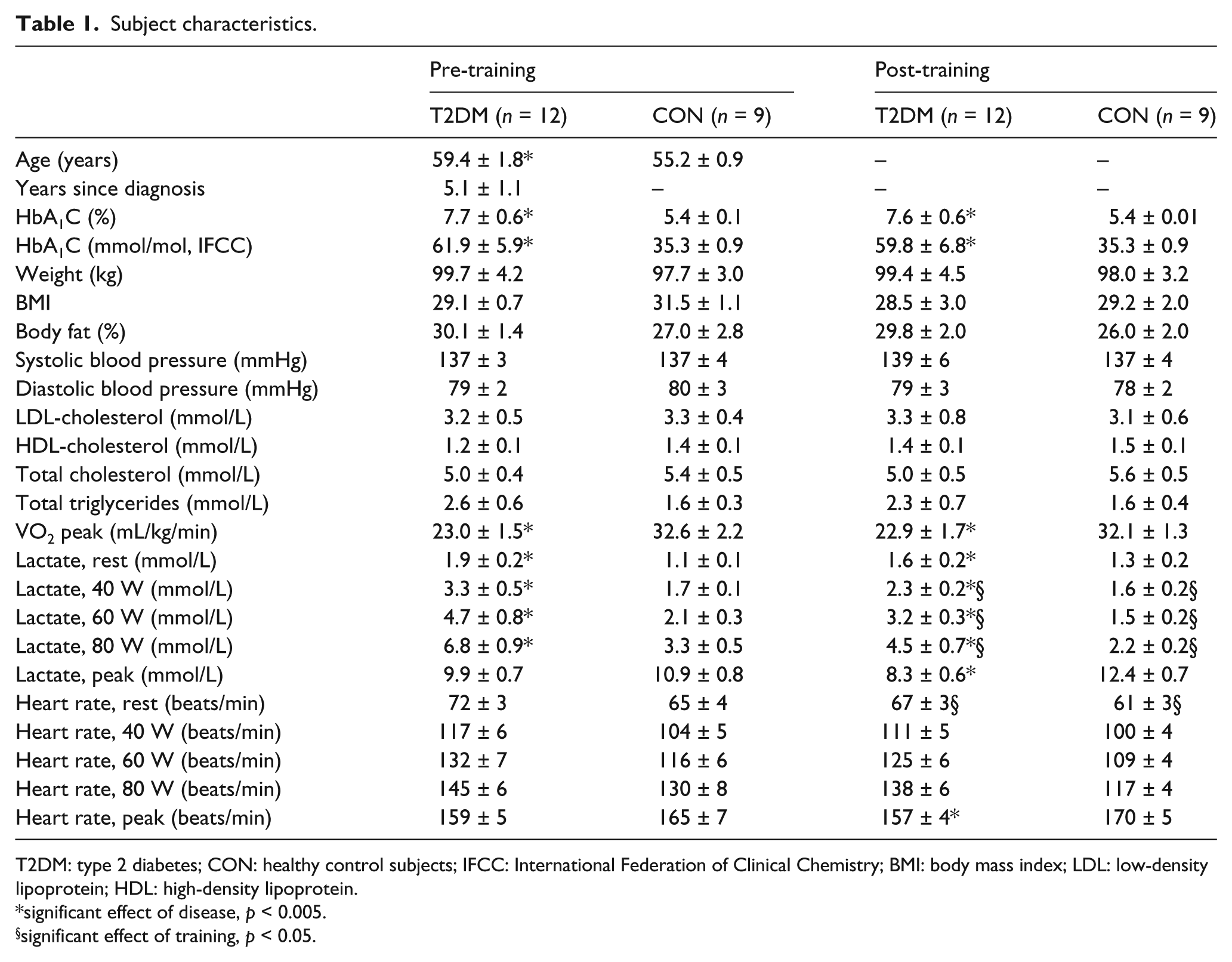
T2DM: type 2 diabetes; CON: healthy control subjects; IFCC: International Federation of Clinical Chemistry; BMI: body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
significant effect of disease, p < 0.005.
significant effect of training, p < 0.05.
There was one smoker in the group of patients with T2DM and two smokers among the CON (less than 15 cigarettes per day). None of the CON had a family history of T2DM. Only males were accepted into the experiment in order to control for possible effects of hormonal status. We attempted to match all subjects participating in this experiment for age, weight, body fat and previous sedentary behaviour (by self-reported habitual activity levels over past 6-month period), although the subjects with T2DM ended up being statistically older and less fit than CON (p < 0.05; see Table 1 for further details of subject characteristics). The study protocol was approved by the Ethical Committee of Copenhagen and Frederiksberg (01-069/02) and conducted in a manner that conforms to the principals outlined in the Declaration of Helsinki. All subjects gave informed written consent before participating in the experiment.
Pre-study evaluations
During an initial evaluation, subjects had body fat percent measured by Dual X-ray Absorption (DEXA) (Expert-XL; Lunar Corporation, Madison, WI, USA) and had a general medical examination that included routine hospital blood tests (Table 1) and resting electrocardiography (ECG) (abnormal would meet exclusion criteria). Normal glucose tolerance was confirmed in all CON through a standard 75 g oral glucose tolerance test.
All subjects underwent a submaximal exercise test on a rowing ergometer (Concept2; Modest Sport, Tikøb, Denmark) in order to be able to assess the modality-specific effects of our training programme. Additionally, a maximal exercise test was conducted on a cycle ergometer (Ergoline, Ergonometrics 800, Jaeger, Hoechberg, Germany; Siemens, Denmark) to provide data for exercise prescription for this study [i.e. maximal heart rate (HR), lactate, peak VO2: as presented in Table 1].
Exercise training
The exercise programme consisted of 8 weeks of home-based training on a rowing ergometer (Kettler Coach 2002; Kettler GmbH, Ense-Parsit, Germany). Subjects exercised 30 min every other day (approximately 28 sessions in total) and were instructed to maintain a HR, corresponding to a work intensity of 65%–70% of VO2 peak as indicated from the maximal exercise test. During the training sessions, all subjects maintained the prescribed intensity with a portable HR monitor (Polar, Kempele, Finland). Subjects recorded a training diary registering number of training bouts, duration, HR and any additional comments pertaining to each exercise session. Training adherence was monitored with regular follow-up phone calls, home visits, training diary verifications and the download of data from Polar monitors.
Experimental protocol
Subjects were instructed to refrain from exercising at least 24–36 h prior to their scheduled appointment to avoid the potential effects of acute exercise. They were required to come into the laboratory on three separate days for muscle biopsies: prior to the start of exercise training (for mRNA, protein and electron microscopy), after 2 weeks of training (approximately 5–6 sessions, for mRNA only) and upon the completion of 8 weeks (for mRNA and protein).
Muscle biopsies were obtained under local anaesthetic from comparable sites in the vastus lateralis muscle (Bergström technique). Tissue was immediately flash frozen in liquid nitrogen or preserved for electron microscopy (see below). Frozen samples were stored at −80°C until analysis.
RNA extraction
Total RNA was isolated from muscle biopsies with phenol extraction using a polytron (Ultra-Turrax T8; IKA Labortechnik, Staufen, Germany) and TRI Reagent (MRC, Cincinnati, OH, USA) according to the manufactures protocol. Total RNA concentrations and purity were determined by spectroscopy at 260, 280 and 240 nm. RNA integrity was ensured by gel electrophoresis.
Real-time reverse transcription–polymerase chain reaction (RT-PCR)
A measure of 500 ng total RNA was converted into complementary DNA (cDNA) in 20 µL using the Omniscript reverse transcriptase (Qiagen, Valencia, CA, USA) and 1 µM poly-dT (Invitrogen, Naerum, Denmark) according to the manufacture’s protocol (Qiagen). For each target mRNA, 0.25 µL cDNA was amplified in a 25 µL SYBR Green polymerase chain reaction (PCR) containing 1× QuantiTect SYBR Green Master Mix (Qiagen) and 100 nM of each primer (Table 2). The amplification was monitored real time using the MX3000P Real-time PCR machine (Stratagene, LaJolla, CA, USA). The threshold cycle (Ct) values were related to a standard curve made with known concentrations of cloned PCR products. The specificity of the PCR products was confirmed by melting curve analysis after amplification. The large ribosomal protein P0 (RPLP0) mRNA was chosen as internal control. To validate this use, another unrelated ‘constitutive’ mRNA, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA, was measured and normalized with RPLP0. As no differences were seen between the groups, both GAPDH and RPLP0 appear stable, and therefore, RPLP0 was considered suitable for normalization.
Primers for real-time RT-PCR.

RT-PCR: reverse transcription–polymerase chain reaction; RPLP0: large ribosomal protein P0; MT1: membrane type 1; MMP: matrix metalloproteinases; TIMP: tissue inhibitors of matrix metalloproteinases.
Immunoblotting
Muscle tissue was homogenized in lysis buffer containing 250 mM NaCl, 50 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) (pH 7.5), 10% glycerol, 1% triton X-100, 1.5 mM MgCl2, 1 mM ethylene glycol tetraacetic acid (EGTA), 10 mM Na4P2O7, 1 mM NaF, 800 µM Na3VO4, left on ice for 1 h and centrifuged at 12,000 × g for 15 min. The supernatants were collected and protein content was measured using Pierce bicinchoninic acid (BCA) Protein Assay Kit (Thermo Scientific, Boucherville, QC, Canada). A measure of 20 µg of total protein wasseparated by molecular weight using 10%–12.5% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred to nitrocellulose membranes (0.45 µm, 162-0115; Bio-Rad, St. Laurent, QC, Canada) using a 10 mM sodium tetraborate buffer. Following the transfer, the membranes were stained with 0.1% Ponceau S as a loading control [ImageJ software; NIH (http://rsbweb.nih.gov/ij/)]. The membranes were washed in distilled water and subsequently blocked in 5% bovine serum albumin (BSA) in Tris-buffered saline with Tween 20 (TBS-T) buffer (10 mM Tris-HCl, pH 7.5, 150 mM NaCl, 0.05% Tween 20) for 1 h at room temperature followed by overnight incubation at 4°C with the following primary antibodies: MMP-2 1:1000 (NB200-113, specific for both pro- and active-MMP-2; Novus Biologicals, Oakville, ON, Canada), MT1-MMP 1:4000 (AB19058, specific for both pro- and active-MT1-MMP; Chemicon (Millipore), Hellerup, Denmark) and TIMP2 1:1000 (ab1828; Abcam). The blots were washed with TBS-T and incubated with horseradish peroxidase-conjugated secondary antibodies (anti-mouse ab6728 andanti-rabbit ab6721; Abcam, Cambridge, UK). Bands were visualized with a chemiluminescence system (Immun-Star Chemiluminescent, 1705070; Bio-Rad) on clear blue X-ray film (CL-XPosure; Thermo Scientific) and analysed with ImageJ software. For quantification, the following bands were analysed: 72 kDa (pro-MMP-2), 62 kDa (active-MMP-2), 65 kDa (pro-MT1-MMP), 63 kDa (active-MT1-MMP) and 24 kDa (TIMP2).
Electron microscopy
At time of tissue extraction, samples were fixed with 2% v/v glutaraldehyde in 0.05 M sodium phosphate buffer (pH 7.2). Following isolation of suitable specimen blocks, the samples were rinsed three times in 0.15 M sodium cacodylate buffer (pH 7.2) and subsequently post-fixed in 1% w/v OsO4 in 0.12 M sodium cacodylate buffer (pH 7.2) for 2 h. The specimens were dehydrated in graded series of ethanol, transferred to propylene oxide and embedded in Epon according to standard procedures. Sections, approximately 80 nm thick, were cut with a Reichert-Jung Ultracut E microtome and collected on copper grids with Formvar supporting membranes, stained with uranyl acetate and lead citrate, and subsequently examined with a Philips CM 100 TEM (Philips, Eindhoven, The Netherlands), operated at an accelerating voltage of 80 kV and equipped with an OSIS Veleta digital slow scan 2k × 2k charge-coupled device (CCD) camera. Digital images were recorded with the ITEM software package.
Statistics
All subject characteristics, mRNA and immunoblotting were analysed by two-way analysis of variance (ANOVA) for repeated measures (time × disease). Pairwise multiple comparison procedures were performed by Holm–Sidak method. Subject characteristics are presented as mean ± standard error of the mean (SEM). All mRNA data were log transformed before statistical analyses and are presented as the geometric mean ± back transformed SEM. Immunoblotting data are presented as mean ± SEM. All analyses were performed on SigmaStat 4.0. For all data presented, p < 0.05 is considered significant.
Results
Subject characteristics and effects of disease and/or training
There were no effects of training or diabetic status on weight, body mass index (BMI), body fat, blood pressure, cholesterol [high-density lipoprotein (HDL), low-density lipoprotein (LDL), total], triglycerides or HR during submaximal testing (40, 60 and 80 W). However, there was an effect of disease on HbA1C, VO2 peak and blood lactate at rest and during submaximal exercise (40, 60 and 80 W). Subjects with T2DM also had a lower post-training peak HR and peak lactate level than CON. For both study groups, there was an effect of training that was detectable in blood lactate levels during submaximal testing (40, 60 and 80 W). Resting HR was lower post training in both groups. Data are presented in Table 1.
mRNA
Baseline comparisons
At baseline, there were no differences in MT1-MMP and MMP-2 mRNA levels between T2DM and CON. TIMP2 mRNA levels were also similar between the two groups. All data are presented in Figure 1.
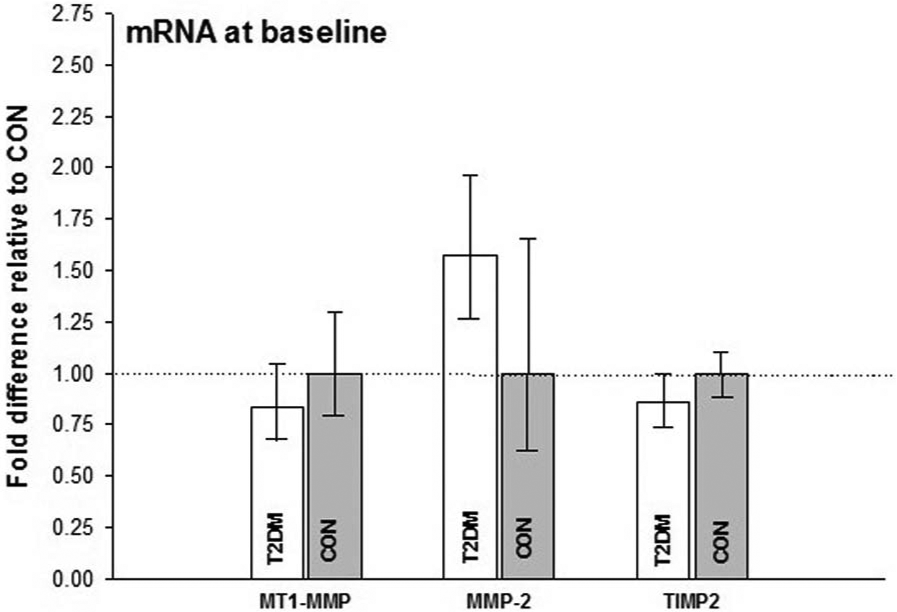
mRNA at baseline – mean values for CON set to 1.00, T2DM compared to CON. No effect of disease was detected on MT1-MMP, MMP-2 and TIMP2.
MT1-MMP, MMP-2 and TIMP2: effects of exercise or disease
Both groups had similar significant increases in MT1-MMP mRNA after 2 weeks of exercise training from baseline values. These elevations at baseline remained through to the end of the exercise training programme, with no change in MT1-MMP mRNA levels between 2 and 8 weeks of training. There was also no detectable difference due to diabetes.
Similarly, after 2 weeks of exercise training, both groups had similar increases in MMP-2 levels from baseline values. This increase persisted through to the end of 8 weeks of training. There were no differences in MMP-2 mRNA between 2 and 8 weeks of training, nor were there differences detected due to metabolic status.
Although there was an overall increase in TIMP2 mRNA due to training, it was not possible to detect whether the difference occurred at 2 or 8 weeks of training. No disease-related differences in mRNA expression were detected.
Data pertaining to the effects of disease or exercise on mRNA are presented in Figure 2.
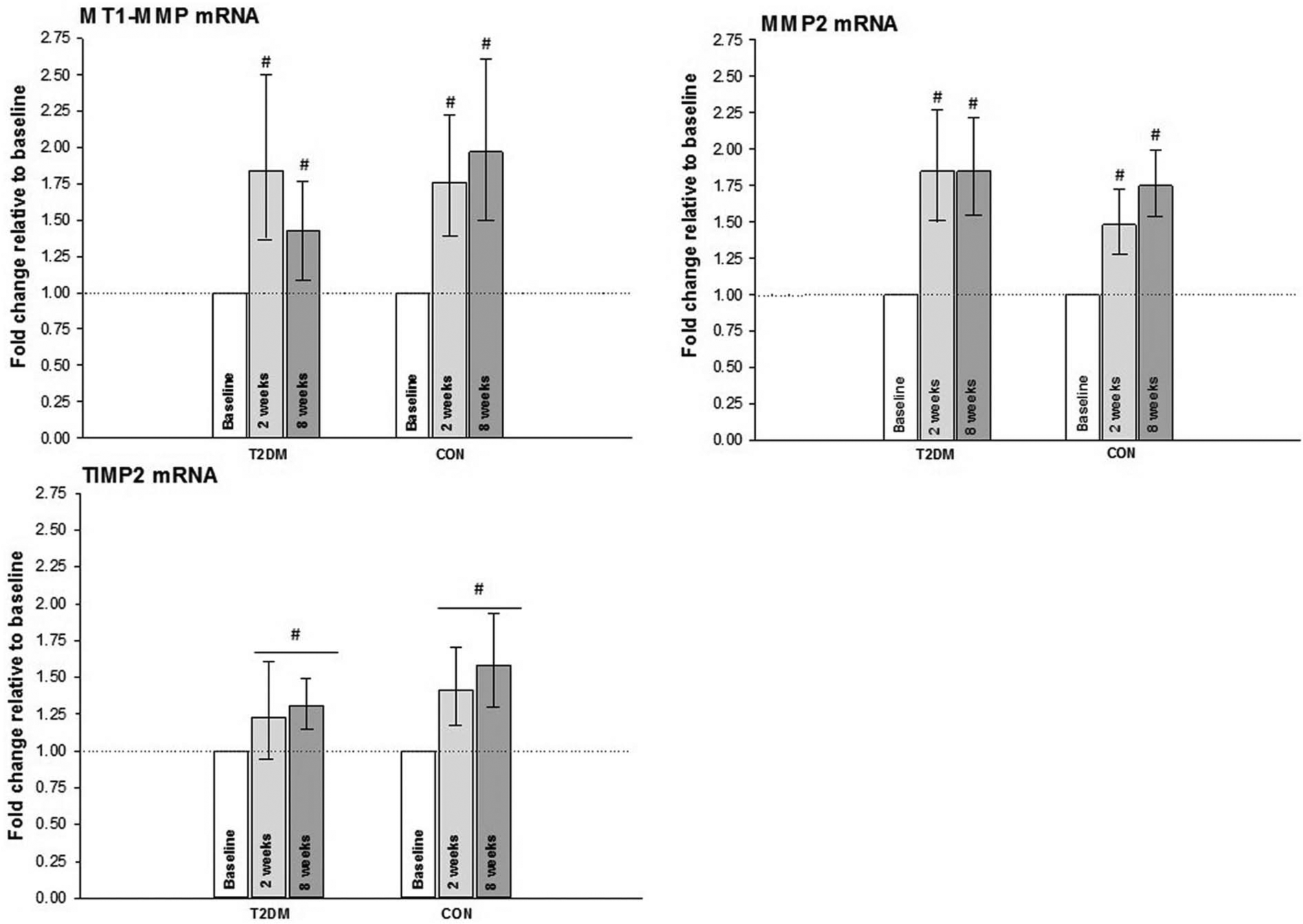
mRNA during exercise training – biopsies were taken at baseline, after 2 weeks and at conclusion of the study (8 weeks). Baseline measurements were set to 1.00 in each subject, with 2- and 8-week measurements compared to baseline. In the case of TIMP2, although a difference from baseline was detected, it was not possible to isolate it to either 2 or 8 weeks.
Immunoblotting
Representative gels are shown in Figure 3.
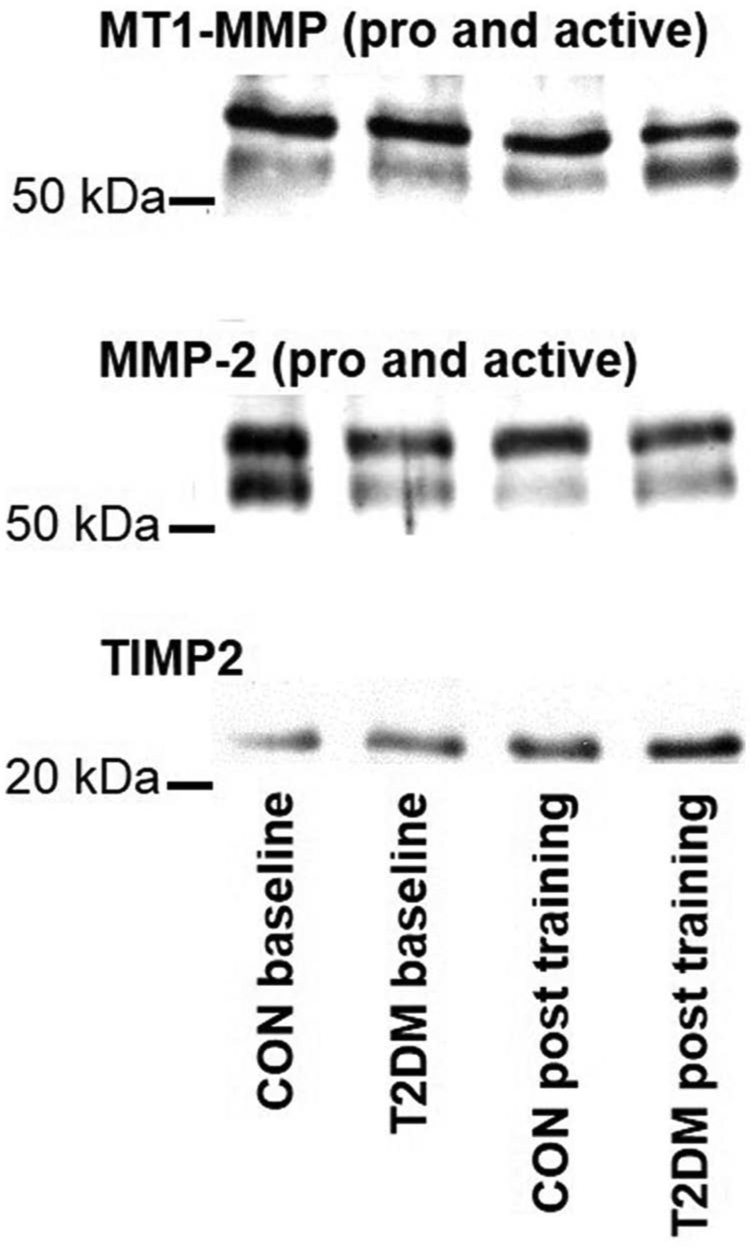
Representative gels for immunoblotting. For quantification, the following bands were analysed: 72 kDa (pro-MMP-2), 62 kDa (active-MMP-2), 65 kDa (pro-MT1-MMP), 63 kDa (active-MT1-MMP) and 24 kDa (TIMP2).
Baseline comparisons
At baseline, there were no differences in MT1-MMP (pro and active), MMP-2 (pro and active) and TIMP2 protein levels between T2DM and CON. These data are presented in Figure 4.
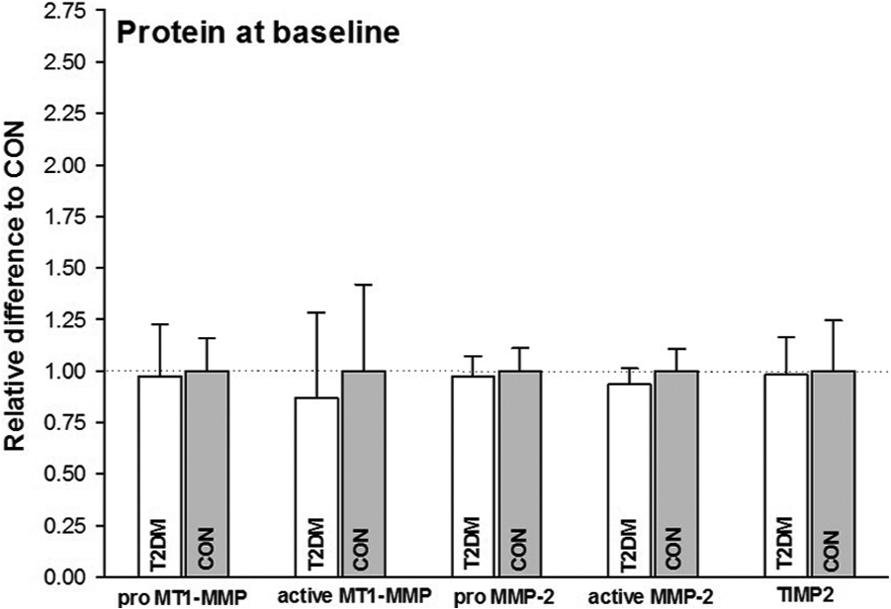
Protein measurements at baseline – mean values for CON set to 1.00, T2DM compared to CON. There was no effect of disease detected on MT1-MMP (pro and active), MMP-2 (pro and active) and TIMP2.
MT1-MMP (pro and active), MMP-2 (pro and active) and TIMP2 protein: effects of exercise or disease
No effect of training or disease was detectable for pro-MT1-MMP or pro-MMP-2 protein in either group. For active MT1-MMP, there was no effect of disease but there was a significant overall effect of training.
There was no effect of disease on active MMP-2 protein in either study group. The effects of training were evaluated, revealing a statistically significant increase in active MMP-2 protein in patients with T2DM only.
There were no detectable differences in TIMP2 protein, either due to training or disease. Data pertaining to the effects of disease or exercise on protein are presented in Figure 5.
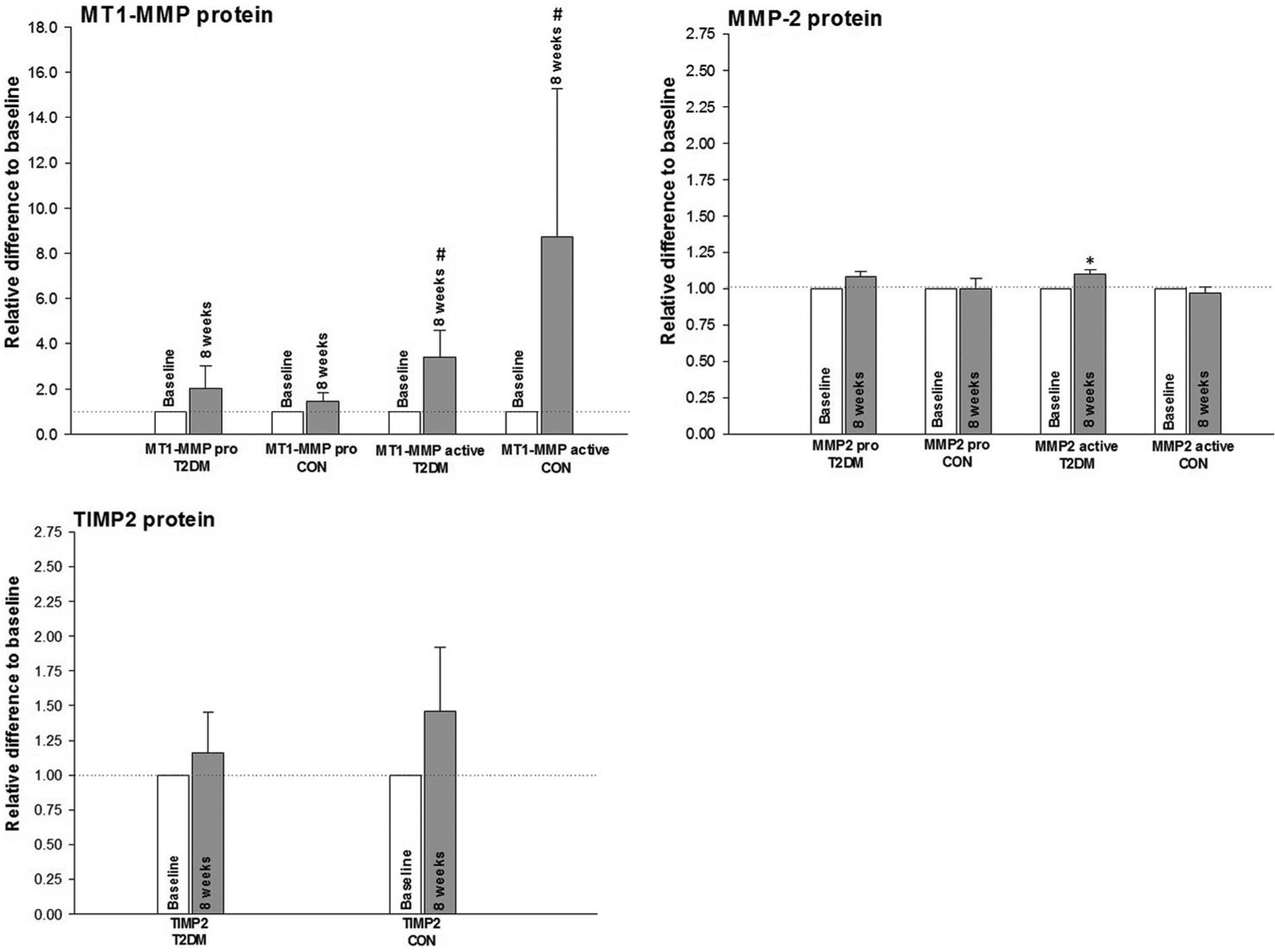
Protein during exercise training – biopsies for protein were taken at baseline and at conclusion of the study (8 weeks). Baseline measurements were set to 1.00 in each subject, with 8-week measurements compared to baseline.
Electron microscopy
Electron microscopic imaging of muscle biopsies was conducted on a subset of our subject group (T2DM: n = 7; CON: n = 5) that was included uniquely upon order of entry into the study. The images demonstrate a typical expansion of CBM when compared to CON, which is consistent with previous work done in this area. 2 Examples are presented in Figure 6.
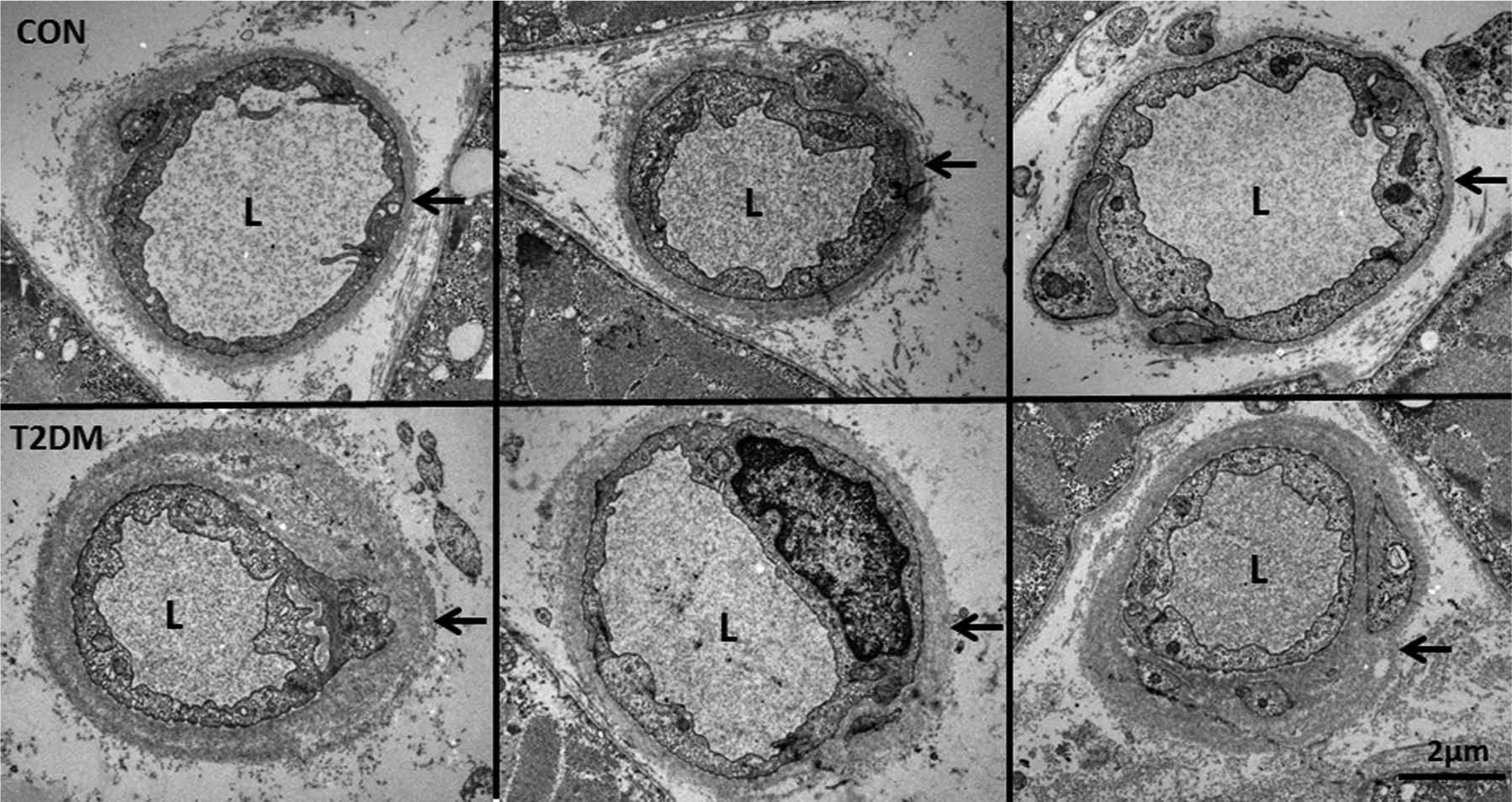
Capillary basement membrane and extracellular space: arrows indicate capillary basement membrane. L represents lumen of blood vessel. Each image represents a different subject.
Discussion
These results indicate that MT1-MMP and MMP-2 levels are preserved in the skeletal muscle of patients with uncomplicated T2DM. We also demonstrate that exercise training provides an effective stimulus for upregulating MMPs in this population, in a manner comparable to CON. There were no effects of training or disease on TIMP2 levels.
At baseline, there was no difference between subjects with T2DM and CON when evaluating MT1-MMP, MMP-2 or TIMP2 at either the mRNA or protein level. In both subject groups, MT1-MMP mRNA increased after 2 weeks of exercise and was maintained until the end of the training period. Active MT1-MMP protein was increased at the end of the 8-week training period. Similarly, MMP-2 mRNA levels also increased after 2 weeks of regular exercise and remained elevated at the end of the exercise programme; however, upregulation of mRNA did not translate into an increase of either pro or subsequent active protein in CON. Only the subjects with T2DM demonstrated an increase in active MMP-2 protein, and it can only be speculated whether this is a result of either (1) the diabetic state, (2) a discrepancy in the time course of protein formation between subjects groups, (3) a difference in fitness level or (4) a type of compensatory mechanism for the accumulation of extracellular proteins in the patients with T2DM. Exercise intensity appears to modulate the expression of active MMP-2; 30 therefore, it is possible that in our study, the exercise stimulus was physiologically perceived as more strenuous for the less fit subjects with T2DM. Also, it may be that MT1-MMP, being a membrane-bound MMP, serves roles in intracellular signalling, independent of MMP-2, that result from physical training. 31
Furthermore, there is no effect of T2DM on either TIMP2 mRNA or protein at baseline; therefore, at least in this population, there is no inhibitory or enhanced effect of TIMP2 on MMP activity due to disease. Exercise training induces an increase in TIMP2 mRNA over the 8 weeks; however, at which time point this occurs was not statistically detectable. Despite the rise in TIM-2 mRNA, there is no apparent exercise-induced increase in TIMP2 protein, at least within the 8 weeks of training included in this study.
We use electron microscopy to image the CBM in both subject groups, in order to confirm that our subjects with uncomplicated T2DM still demonstrated an expansion of the CBM as previously reported.2,3 A representation of these images, presented in Figure 6, clearly demonstrates that, despite the fairly recent diagnosis and lack of overt cardiovascular disease, these subjects with T2DM are already prone to an expansion of the CBM width, when compared to CON. As reductions in CBM width due to physical training have only been previously reported after 9 months of exercise, 27 we did not anticipate any measurable improvements after the 8 weeks of training included in our study and, therefore, did not image the CBM post training.
The important information presented here is that in skeletal muscle samples, uncomplicated T2DM does not have a major negative effect on MMPs at baseline nor is there an impaired MMP induction due to an 8-week moderate intensity exercise programme. Past studies have demonstrated a dysregulation of MMPs due to T2DM, which results in either an increase 32 or decrease 17 in these enzymes. Variations in MMP levels due to diabetes, however, may be due to the tissue sampled or the degree of cardiovascular disease present. Dichotomous angiogenic signalling is known to occur in T2DM, where complications arising from either an enhanced or diminished neovascularization may develop concurrently. For example, in a single patient, impaired wound healing may occur in the periphery due to lack of collateral flow while also demonstrating uncontrolled angiogenesis in the eye. This paradoxical signalling is tissue specific, 33 and given the role that MMPs play in angiogenesis, their own regulation may also affected in a similar manner. As for age, duration of diabetes, blood pressure and serum lipid concentrations, there is evidence that at least in plasma, these factors may not be significantly associated with MMP-2, MMP-9 and TIMP-1concentrations. 32 However, in other tissue, there appears to be a stronger relationship between age and MMP dysregulation.28,34
In cardiac tissue, exercise training seemingly protects against age-associated reductions in active MMP-1, MMP-2 and MT1-MMP in a rodent model. Regular physical activity attenuates elevations in TIMP-1 that were detected in aging versus young rats. 28 These authors conclude that exercise training plays a role in the reduction of fibrosis and MMP dysregulation in the heart. Based on our results, we offer similar conclusions: exercise training elevates MMP-2 and MT1-MMP mRNA and induce active MT1-MMP protein in both subjects with T2DM and CON, thus contributing to the maintenance of cardiovascular health in this population. There is also no associated elevation in TIMP2, which affects the ratio between active MMP and its inhibitor. Although there is previous evidence for MMP dysregulation due to diabetes, our subjects with T2DM, at least during this period of their disease trajectory, still appear to be able to regulate MMPs. In a study by Kadoglou et al., 16 weeks of exercise training had no effect on plasma MMP-2 and TIMP-1 in either subjects with T2DM or CON. Additionally, they report a training-induced decrease in plasma MMP-9 and concurrent increase in plasma TIMP2. 29 These results are inconsistent with our own as we detected an increase in active MMP-2 protein in subjects with T2DM but not in CON; however, as previously indicated, these discrepancies may result from the different tissues investigated.
The maintenance of physiological MMP regulation is important because the expansion of the CBM and ECM is linked to many diabetes-related complications.10,35–37 Impaired MMP proteolysis of matrix proteins is believed to negatively impact the critical interactions between pericytes and their ECM environment, resulting in their death and subsequent failure of normal capillary functions. 38 Furthermore, disease-induced remodelling of the ECM has been proposed as an early step in the progression of insulin resistance via altered mechano-signal transduction and its subsequent effect on mitochondrial gene expression signalling. 39 Thus, dysfunctional or impaired proteolysis contributes to the progression of both disease and its related complications. We intentionally recruited subjects with T2DM that were at an ‘early phase’ of disease (i.e. diagnosis within past 5–7 years, with modest hyperglycaemia), in order to investigate exercise-induced MMP regulation prior to the development of overt diabetes-related complications. The comparable response of the MMPs in our subjects with T2DM to CON suggests that intervention at this stage may be strategically important as it may prevent or minimize further progression of the disease trajectory.
Despite the established benefits of regular physical exercise for patients with T2DM,26,40–43 the majority of the molecular mechanisms implicated in health improvement remain largely speculative. Physiological adaptations to training include angiogenesis and the remodelling of skeletal muscle fibres and ECM. MMPs have been shown to be a critical part of this process; 25 therefore, MMPs, rather than being considered a part of the ‘problem’, 44 may also play an important therapeutic function in T2DM when induced under healthy conditions. 45
We conclude that exercise training remains an important regulator of MMPs in uncomplicated T2DM and this may be a means by which regular physical activity offers protection from diabetes-related vascular disease. At this early stage of the disease, these subjects still respond in a similar fashion as those with normal glucose tolerance; thus, intervention at this point of the disease trajectory may be a critical window of opportunity to promote and maintain vascular health and diminish risk in this population. Further work that addresses the direct link between reductions in CBM and ECM volume, MMPs, angiogenesis signalling and the preservation of cardiovascular health in patients with T2DM is clearly warranted. Strategic interventions that target the maintenance of physiological systems by exercise training may offer a cost-effective and efficient means of preventing associated disease in this population.
Footnotes
Acknowledgements
The authors wish to thank David Benée Olsen and Danny Reving for their role in conducting these experiments and acknowledge the technical expertise of Jeppe Bach, Thomas Beck and Regitze Kraunsøe. Invaluable administrative assistance was provided by Vibeke Hvass and Jacqueline van Hall.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
Funding for this study was obtained from the Novo Nordisk Foundation, the Danish Diabetes Association, the Danish Heart Foundation, Jacob Madsen and Olga Madsen’s Foundation, the Foundation of 1870, the Danish National Research Council, the Aase and Ejnar Danielsens Foundation and the Faculty of Health Sciences, University of Copenhagen (doctoral grant for C.S-B.).
