Abstract
Background:
Sclerostin and Dickkopf-1 are extracellular inhibitors of the canonical Wnt/β-catenin signalling pathway, which is implicated in the development of arterial stiffness. However, the correlation between aortic stiffness and sclerostin or Dickkopf-1 levels in patients with type 2 diabetes mellitus is unknown.
Methods:
Fasting blood samples were collected from 125 patients with type 2 diabetes mellitus. Aortic stiffness was measured by carotid–femoral pulse wave velocity, and high aortic stiffness was defined by a carotid–femoral pulse wave velocity of >10 m/s. The serum sclerostin and Dickkopf-1 concentrations were determined using commercially available enzyme-linked immunosorbent assays.
Results:
In total, 46 patients with type 2 diabetes mellitus (36.8%) had high levels of aortic stiffness. Compared to the control group without aortic stiffness, this group was significantly older, had higher systolic and diastolic blood pressures, had higher blood urea nitrogen, creatinine, urinary albumin-to-creatinine ratio and serum sclerostin levels, and had significantly lower high-density lipoprotein cholesterol levels and estimated glomerular filtration rates. After adjusting for confounders, serum sclerostin [odds ratio = 1.005 (1.002–1.007), p = 0.002] levels remained an independent predictor of aortic stiffness. Multivariate analysis showed that the serum sclerostin level (β = 0.374, adjusted R2 change = 0.221, p < 0.001) was positively associated with carotid–femoral pulse wave velocity.
Conclusion:
Serum levels of sclerostin, but not Dickkopf-1, are positively correlated with carotid–femoral pulse wave velocity and independently predict aortic stiffness in patients with type 2 diabetes mellitus.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disease characterized by hyperglycaemia, relative insulin insufficiency and insulin resistance and is often associated with obesity and a sedentary lifestyle. 1 The rapid growths of urbanization and economic development, coupled with altered lifestyles in the general population, have led to its prevalence skyrocketing since the late 20th century. According to estimates by the International Diabetes Federation, 336 million people were diagnosed globally in 2011, and that figure is expected to rise to 552 million by 2030. 2 The increasing prevalence of T2DM is creating a huge burden on global health expenditure, with associated cardiovascular episodes contributing to a lifespan reduction of at least 6 years and a 1.3- to 2.3-fold higher risk of mortality from various causes. 3
Sclerostin and Dickkopf-1 (DKK1) are upregulated in diabetes and inflammation status.4,5 Sclerostin, a glycoprotein of the SOST gene located on chromosome 17q12–q21 in humans, and secreted by osteocytes, inhibits the Wnt/β-catenin pathway and prevents osteoblast differentiation and proliferation by binding to the dual complex receptor composed of LRP5 or LRP6 and the seven-transmembrane receptor Frizzled.6,7 DKK1 also binds to LRP5 or LRP6 and the cell surface co-receptor Kremen-1 and Kremen-2 to promote internalization of the receptor complex and is another secreted inhibitor of the Wnt/β-catenin pathway. 7 Interestingly, the Wnt/β-catenin pathway affects not only the skeleton but also glucose metabolism, vascular calcification, arterial stiffness and atherogenesis.8–12 Previous studies showed that the expressions of Wnt/β-catenin signalling inhibitors sclerostin and DKK1 may avoid further progression of ossification in calcifying vascular smooth muscle cells.13,14 A cross-sectional study by Daniele et al. 8 showed that sclerostin levels were positively correlated with insulin resistance in adipose tissue, skeletal muscle and the liver, and that increased sclerostin levels were observed in prediabetic states. Also, a study by Morena et al. 9 revealed that high serum levels of sclerostin, but not DKK1, had a positive association with coronary arterial calcification in 241 patients with chronic kidney disease (CKD) not receiving dialysis. In an earlier study, we also showed that the serum sclerostin level was independently associated with peripheral arterial stiffness in renal transplantation recipients. 10
Aortic stiffness strongly predicts cardiovascular episodes and is a major contributor to T2DM complications and mortality. 15 The carotid–femoral pulse wave velocity (cfPWV), a useful non-invasive method for evaluating aortic stiffness, has been associated with the risk of future cardiovascular disease and all-cause mortality. 16 Despite associations being shown between serum sclerostin or DKK1 and arterial stiffness in different patient groups, including renal transplant recipients, hemodialysis patients and postmenopausal women, there are no data about a relationship in patients with T2DM.10,17,18 Therefore, we examined whether there was a correlation between serum sclerostin or DKK1 levels and aortic stiffness, as measured by cfPWV, in patients with T2DM.
Methods
Study design and participants
This prospective cross-sectional study of patients with T2DM was conducted from November 2014 to March 2015 at a metabolism and endocrinology outpatient department in Hualien, Taiwan. Patients were excluded if they had an acute infection, acute myocardial infarction, heart failure or malignancy at the time of blood sampling. Informed written consent was obtained from all participants before study enrolment. The study was approved by the relevant institutional review board (IRB103-136-B).
Anthropometric and blood pressure measurements
All anthropometric measurements were performed by the same operator. Body weight and height were measured with the participants in light clothing, without shoes and recorded to the nearest 0.5 kg and 0.5 cm, respectively. Body mass index was calculated as the weight in kilogrammes divided by the height in metres squared.10,19,20 The blood pressure (BP) of the right arm was measured by trained staff with the patient at rest for at least 10 min in the morning, using standard mercury sphygmomanometers and appropriate cuff sizes. The systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured three times at 5-min intervals and were averaged for analysis.
Biochemical investigations
Overnight fasting (8–12 h) blood samples of approximately 5 mL were obtained from participants and immediately centrifuged at 3000×g for 10 min. Serum levels of blood urea nitrogen (BUN), creatinine, triglycerides, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), fasting glucose and glycated haemoglobin (HbA1c) were measured by an autoanalyser (Siemens ADVIA 1800; Siemens Healthcare GmbH, Henkestr, Germany).10,19,20 The urinary albumin-to-creatinine ratio (UACR) was measured by a random spot urine test. Serum sclerostin and DKK1 levels were quantified by commercially available enzyme-linked immunosorbent assays (Biomedica Immunoassays, Vienna, Austria). 8 The estimated glomerular filtration rate (eGFR) was calculated by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. 21
Aortic stiffness using cfPWV measurements
Arterial stiffness was assessed by pressure applanation tonometry (SphygmoCor System; AtCor Medical, Sydney, NSW, Australia), using the cfPWV measurements as previously described.19,20 Measurements were performed in a temperature-controlled room with participants in a supine position after a minimum of 10-min rest in the morning. Pulse waves were recorded consecutively at two superficial artery sites (in the carotid–femoral segment) and an R-timing reference was provided by recordings with an electrocardiography signal. The mean time difference between the R-wave and pulse wave on a beat-to-beat basis, over an average of 10 consecutive cardiac cycles, was calculated by the integral software and used to process each pulse wave and electrocardiography dataset. The cfPWV was then calculated as the ratio of the distance and the mean time difference between the two recorded points. Low and high aortic stiffnesses were defined as cfPWV values of ⩽10 m/s and >10 m/s, respectively.16,19,20,22
Statistical analysis
Analyses were performed using IBM SPSS version 19.0 (IBM Corp., Armonk, NY, USA). Variable distributions were tested using the Kolmogorov–Smirnov test. Normally distributed data were compared by Student’s independent t-tests (two-tailed) and expressed as means ± standard deviations. Non-normally distributed data were compared using the Mann–Whitney U test and expressed as medians and interquartile ranges. Data expressed as the number of patients were analysed using the chi-square test. Variables shown to correlate significantly with aortic stiffness were tested for independence by multivariate logistic regression analysis and reported as odds ratios (ORs) and 95% confidence intervals (CIs). Any data that were not normally distributed at this stage underwent base 10 logarithmic transformations (i.e. log transformations) to achieve normality. Correlations between the clinical variables and the cfPWV values among patients with T2DM were evaluated by univariate linear regression analysis, and variables that were significantly correlated with serum cfPWV were tested for independence by multivariate forward stepwise regression analysis. A p-value < 0.05 was considered statistically significant in all analyses.
Results
The demographic, clinical and laboratory characteristics of the 125 included patients with T2DM are shown in Table 1. Of these, 69 (55.2%) had hypertension and 46 (36.8%) had high aortic stiffness. Compared with the low aortic stiffness group, patients in the high aortic stiffness group were older (p = 0.001), had elevated serum sclerostin (p < 0.001), SBP (p < 0.001), DBP (p = 0.029), BUN (p = 0.029), creatinine (p < 0.001) and UACR (p < 0.001) values and lower HDL-C (p = 0.016) and eGFR (p < 0.001) levels. However, serum DKK1 levels were not significantly different between the two groups. There were also no significant differences between the two groups in terms of sex, comorbid conditions (e.g. hypertension) or medication use.
Clinical variables of patients with diabetes by severity of aortic stiffness.
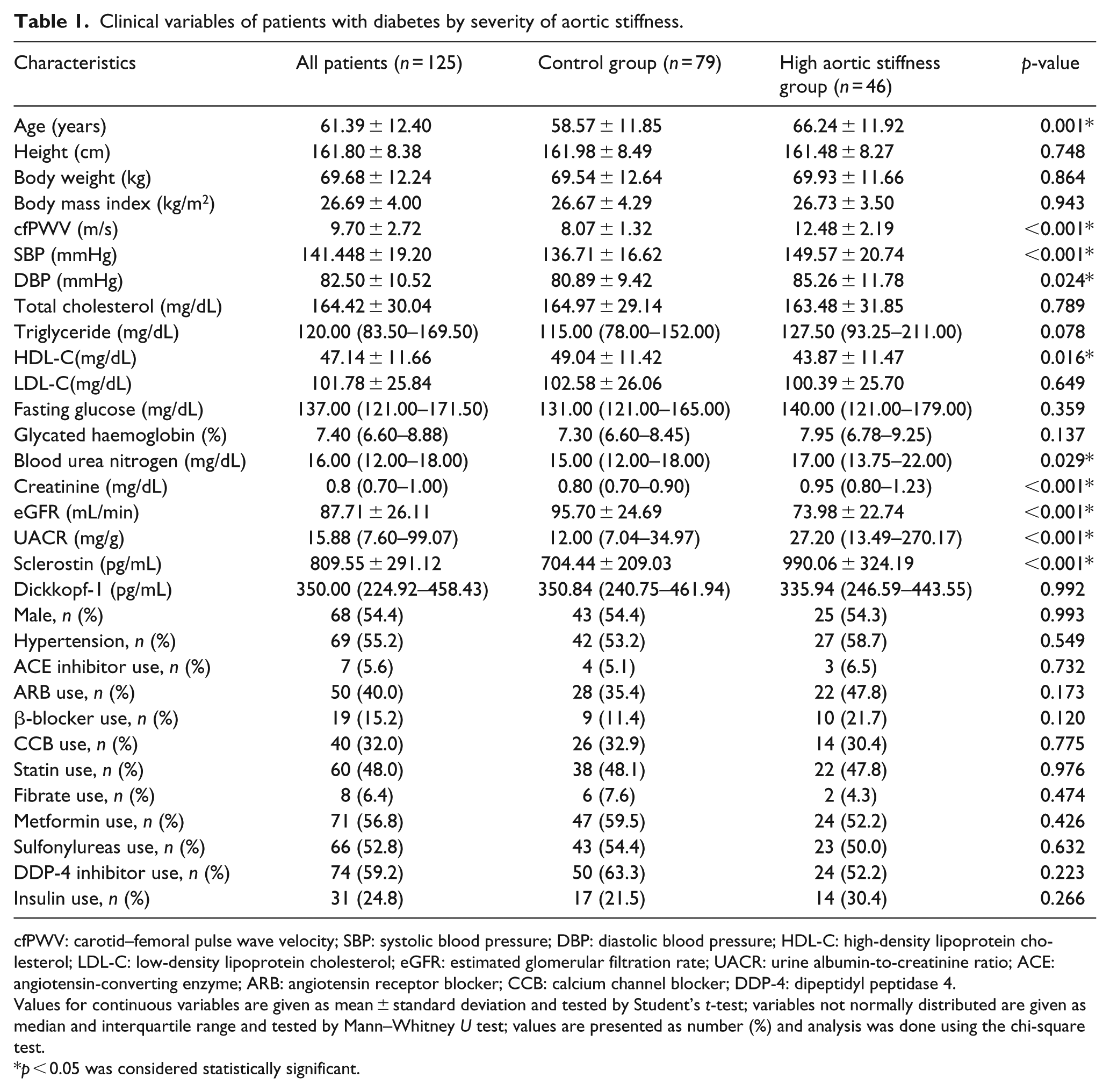
cfPWV: carotid–femoral pulse wave velocity; SBP: systolic blood pressure; DBP: diastolic blood pressure; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; eGFR: estimated glomerular filtration rate; UACR: urine albumin-to-creatinine ratio; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; CCB: calcium channel blocker; DDP-4: dipeptidyl peptidase 4.
Values for continuous variables are given as mean ± standard deviation and tested by Student’s t-test; variables not normally distributed are given as median and interquartile range and tested by Mann–Whitney U test; values are presented as number (%) and analysis was done using the chi-square test.
p < 0.05 was considered statistically significant.
As shown in Table 2, multivariate logistic regression analysis of factors that are significantly correlated with aortic stiffness revealed that serum sclerostin (OR: 1.005, 95% CI: 1.002–1.007, p = 0.002) and HDL-C (OR: 0.949, 95% CI: 0.902–1.000, p = 0.048) were correlated with, and were independent predictors of, aortic stiffness in patients with T2DM. Next, we assessed the correlation between cfPWV values and the clinical and biochemical parameters (Table 3). Univariate analysis revealed that sclerostin levels (r = 0.478, p < 0.001), age (r = 0.337, p < 0.001), SBP (r = 0.358, p < 0.001), DBP (r = 0.210, p = 0.019), logarithmically transformed triglyceride (log-triglyceride; r = 0.209, p = 0.019), log-creatinine (r = 0.304, p = 0.001) and log-UACR (r = 0.324, p < 0.001) were positively correlated, whereas eGFR (r = −0.341, p < 0.001) was negatively correlated with the cfPWV. By multivariable forward stepwise linear regression, sclerostin levels (β = 0.374, adjusted R2 change = 0.221, p < 0.001), age (β = 0.214, adjusted R 2 change = 0.035, p = 0.006), SBP (β = 0.191, adjusted R 2 change = 0.075, p = 0.018) and log-UACR (β = 0.186, adjusted R 2 change = 0.026, p = 0.020) remained positively correlated with cfPWV. Log-DKK1 levels showed no significant correlation with cfPWV.
Multivariate logistic regression analysis of the factors correlated to aortic stiffness in patients with diabetes.
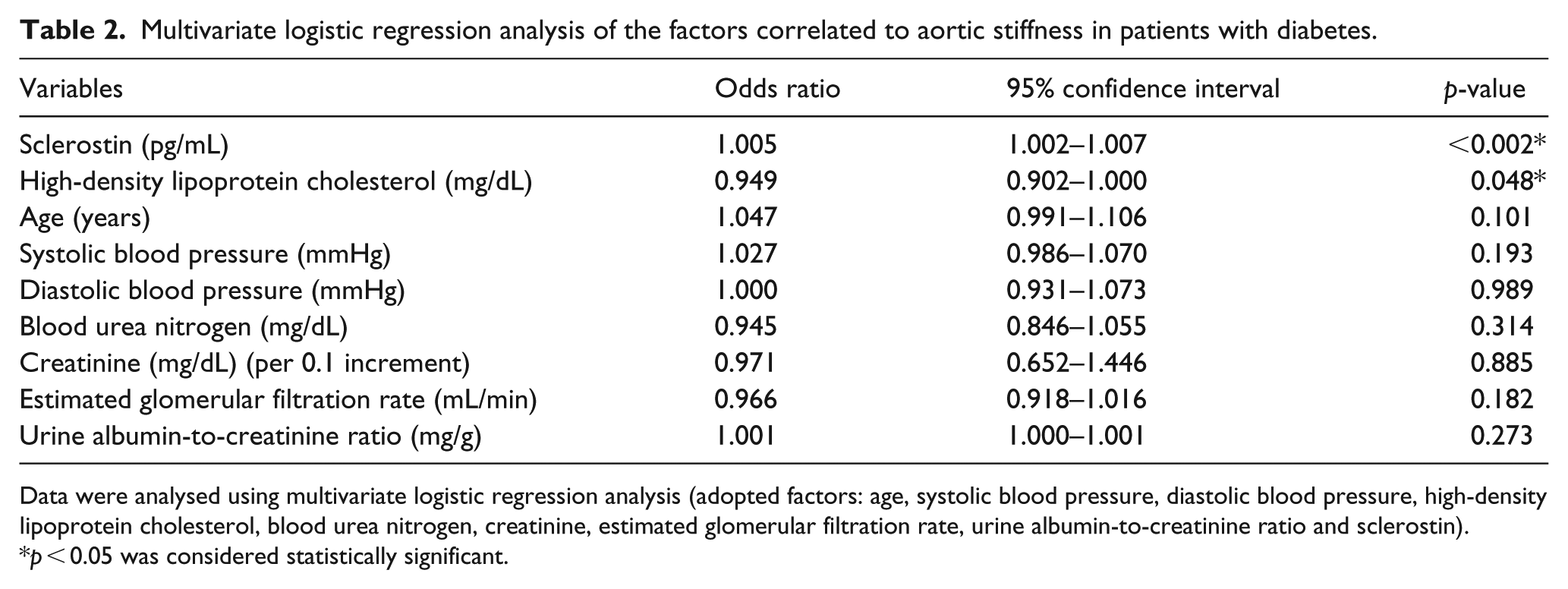
Data were analysed using multivariate logistic regression analysis (adopted factors: age, systolic blood pressure, diastolic blood pressure, high-density lipoprotein cholesterol, blood urea nitrogen, creatinine, estimated glomerular filtration rate, urine albumin-to-creatinine ratio and sclerostin).
p < 0.05 was considered statistically significant.
Correlation between carotid–femoral pulse wave velocity levels and clinical variables among patients with diabetes.
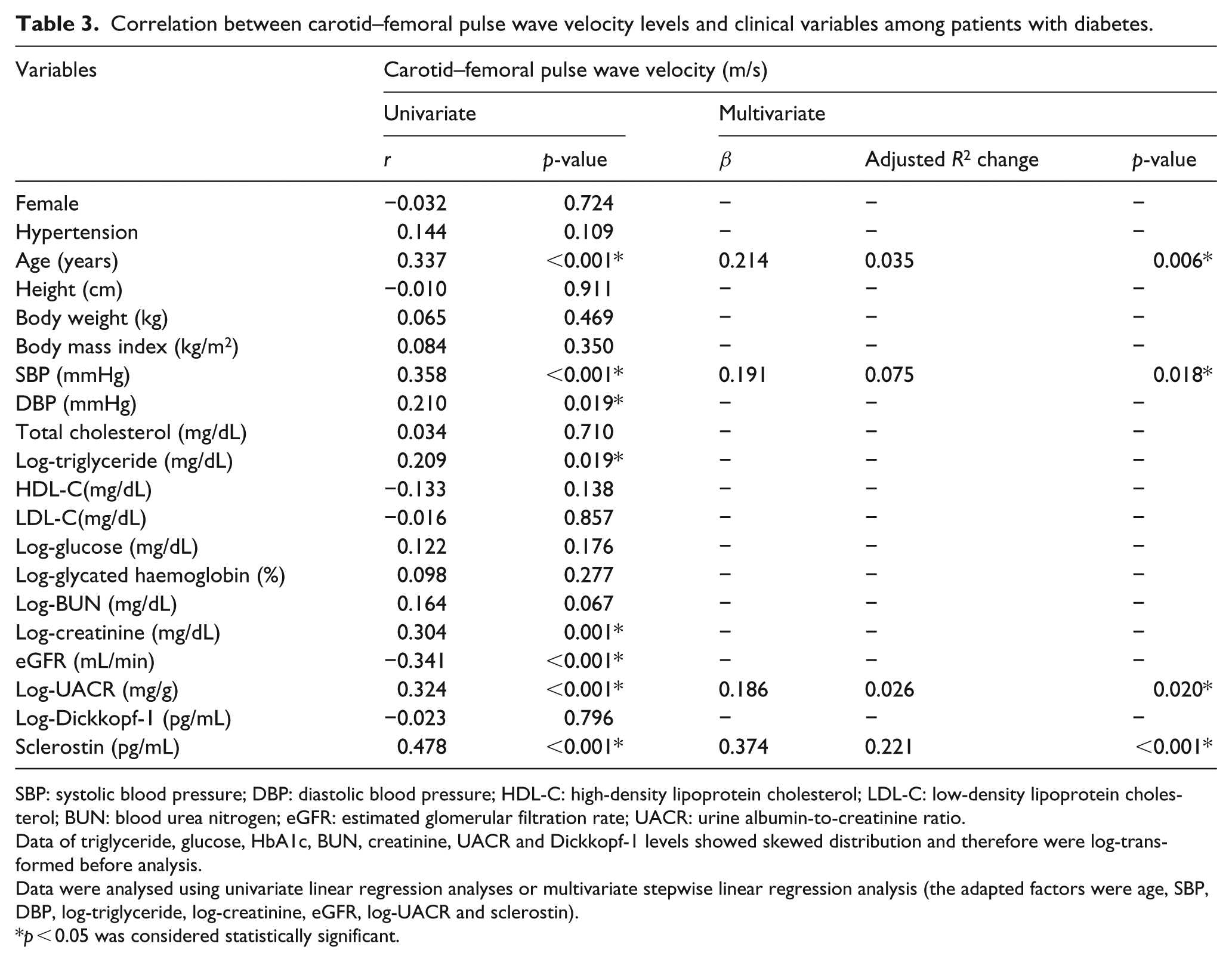
SBP: systolic blood pressure; DBP: diastolic blood pressure; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; BUN: blood urea nitrogen; eGFR: estimated glomerular filtration rate; UACR: urine albumin-to-creatinine ratio.
Data of triglyceride, glucose, HbA1c, BUN, creatinine, UACR and Dickkopf-1 levels showed skewed distribution and therefore were log-transformed before analysis.
Data were analysed using univariate linear regression analyses or multivariate stepwise linear regression analysis (the adapted factors were age, SBP, DBP, log-triglyceride, log-creatinine, eGFR, log-UACR and sclerostin).
p < 0.05 was considered statistically significant.
Discussion
In summary, we showed that serum sclerostin levels were higher and HDL-C levels were lower in patients with T2DM who had high aortic stiffness. Together with advanced age, high SBP and the log-UACR level, serum sclerostin levels were also an independent marker of cfPWV. However, serum DKK1 levels were not different among patients with T2DM by this measure of aortic stiffness.
As we age, degeneration of elastic fibres and increases in collagenous material, vascular smooth muscle cells and calcium deposition in the tunica media and adventitia contribute to increased arterial stiffness. 23 Our study found that patients with T2DM and high aortic arterial stiffness were older than those with low aortic stiffness. The high aortic stiffness group also had higher mean SBP and DBP levels. This is consistent with the evidence that BP both affects and is affected by stiffness, that is, increased BP-related distension of the arterial wall can increase stiffness, but stiffening can also increase SBP directly. 24 A study from Wang et al. 25 noted HDL-C is protective against cardiovascular disease. We found HDL-C levels to be lower in the group with high aortic stiffness in patients with T2DM. Moreover, it was also shown to be an independent predictor of aortic stiffness by multivariate logistic regression analysis. A similar result was obtained by Wang et al., 25 who reported that HDL-C was negatively correlated with the cfPWV values in 1447 participants in a community-based population in Beijing, China.
Significant differences were also noted in renal parameters in this study. We observed that patients with T2DM and high aortic stiffness had increased BUN and creatinine levels, but a decreased eGFR, consistent with previous studies showing a positive correlation between impaired renal function and arterial stiffness. 26 Patients with CKD have higher levels of uremic toxins that exert chemical stress on the arterial architecture, leading to chronic microinflammation, inadequate arterial repair and, ultimately, arterial stiffening. 25 In addition, the UACR was not only significantly elevated in patients with high aortic stiffness but also positively correlated with cfPWV values, again consistent with previous research.27,28 Kohara et al. 27 noted that microalbuminuria was associated with significantly higher cfPWV values in 136 community residents with no prior cardiovascular disease. Smith et al. 28 showed that microalbuminuria was a significant predictor of aortic pulse wave velocity among 134 patients with T2DM.
A recent study by Sciatti et al. 29 revealed that patients with hypertension and T2DM have a higher degree of aortic stiffness than patients with only hypertension. Although the mechanism underpinning this increased arterial stiffness in T2DM is multifaceted, hyperglycaemia-induced vascular inflammation appears to be central, involving monocyte macrophages, endothelial cells and vascular smooth muscle cells. 30 First, hyperglycaemia and advanced glycation end products (AGEs) stimulate the inflammatory activation of circulating monocytes, which then induce protein kinase C, nuclear factor κB and superoxide release, causing oxidative stress. Activation of nuclear factor κB increases the expressions of adhesion molecules to endothelial cells, which results in increased monocyte adhesion to vascular endothelial cells. 31 The AGE-induced decrease in endothelial nitric oxide production, coupled with hyperglycaemia, increases the expression of VCAM1 and impairs endothelial-dependent relaxation of vascular tone, another early marker of vascular injury. 32 The combination of increased monocyte activation and adhesion to endothelial cells causes vascular stiffening to progress.
Subsequently, the increased production of superoxide anions in the hyperglycaemic state increases the susceptibility of LDL-C to oxidation. 33 Oxidized LDL-C is more readily recognized by scavenger receptors on macrophages that have accumulated in arterial walls, and in patients with T2DM, the resulting lipid accumulation in arterial wall macrophages promotes atherogenesis and vascular stiffening. 34 Moreover, the presence of hyperglycaemia, AGEs and hyperinsulinaemia prompts the proliferation of vascular smooth muscle cells, resulting in an increased production of chondroitin sulphate and dermatan sulphate that bind oxidized LDL-C, leading to LDL-C retention in the arterial wall. Increased numbers of vascular smooth muscle cells also mean that there is increased collagen synthesis, further contributing to arterial stiffness. 35
Several inhibitors can regulate the Wnt/β-catenin pathway, but of these sclerostin and DKK1 have been most intensively studied. The expressions of sclerostin and DKK1 are not restricted to osteocytes. Sclerostin has been shown in cartilage, bone marrow, kidney, heart, aorta, pancreas and liver during development. However, expression of sclerostin is limited to osteocytes, chondrocytes and cementocytes postnatally. 36 Expression of DKK1 has been shown in skin, kidney, prostate, placenta and platelets, with lower levels of expression in endothelium. 37 Sclerostin, the main target of this study, functions in the canonical Wnt pathway and is stimulated by the microbiological responses to hyperglycaemia, including increased oxidative stress, LDL-C modification and vascular inflammation. 7 These responses upregulate tumour necrosis factor α and bone morphogenetic protein-2 (BMP-2) in vascular myofibroblasts and endothelial cells, which in turn upregulate multiple Wnt ligands.17,38 The enhanced Wnt pathway signalling then activates β-catenin, a coregulator of the expressions of RUNX2, SP7, MSX2 and alkaline phosphatases, to induce arterial calcification. 17 A study by Shao et al. 34 demonstrated that MSX2 promoted osteogenic differentiation and mineralization in primary aortic mesenchymal progenitors, contributing to arterial calcification. That study also found that MSX2 expression in the adventitia may alter the mineralizing potential of neighbouring mural calcifying vascular cells and even influence cells of the tunica media through increased paracrine Wnt signals. 38 In addition to altering bone formation and mineralization, sclerostin also affects serum concentrations of fibroblast growth factor 23 (FGF23) and calcitriol, hormones that regulate mineral accretion. 39 The European Uremic Toxin Work Group also found a positive correlation between serum sclerostin levels and arterial stiffness by multi-slice spiral computed tomography and pulse wave velocity measurements in patients with stage 2–5 CKD. 40 In our study, not only patients with T2DM and high aortic stiffness had significantly higher serum sclerostin levels, but multivariate logistic regression analysis also confirmed that serum sclerostin was an independent predictor of aortic stiffness. Univariate linear regression analysis and multivariate forward stepwise regression analysis further indicated that serum sclerostin levels were positively correlated with cfPWV values.
Another inhibitor of the Wnt/β-catenin pathway, DKK1, has not been clearly linked to the development of arterial stiffness to date. In 146 postmenopausal women, plasma DKK1 levels and pulse wave velocity failed to show an association. 18 Our previous study showed that the serum levels of sclerostin, but not DKK1, were independently associated with peripheral arterial stiffness in kidney transplant recipients. 10 In this study, we showed that there was no correlation between DKK1 and aortic stiffness in patients with T2DM. The discrepancy between sclerostin and DKK1 can be explained by the fact that, although both are inhibitors of the Wnt/β-catenin pathway, they are produced and distributed differently. Sclerostin is produced mainly by the skeleton, whereas DKK1 is also produced by platelets and vascular cells. Moreover, DKK1 may be sequestered in areas of calcified plaques, resulting in lower circulating levels. 18 Further studies are required to elucidate the true effect of DKK1 on arterial stiffening.
There are some limitations to this study. First, all participants were recruited from the same hospital, so the cohort may not truly represent the wider population. Second, the sample size might have been too small to render conclusive results. Third, some confounders (e.g. heart rate, smoking, alcohol consumption or serum intact parathyroid hormone) were not included and may have restricted the predictive power in our study. Fourth, even though the use of common antihypertensive, antihyperlipidaemic and antidiabetic drugs was not significantly different among patients in this study, we cannot exclude the potential for pharmacological effects on arterial stiffness exerted by other drugs. Larger studies are warranted to establish definitive conclusions.
Conclusion
Levels of the Wnt/β-catenin pathway inhibitor sclerostin, not DKK1, are positively correlated with aortic stiffness in patients with T2DM; moreover, sclerostin independently predicts aortic stiffness in this patient group. This study should contribute to ongoing research for sclerostin use to predict aortic stiffness and to assess cardiovascular risk in the wider population.
Footnotes
Acknowledgements
H.-Y.Y. and D.-A.W. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ministry of Science and Technology, Taiwan (MOST-106-2314-B-303-019-MY3).
Supplemental material
Supplemental material for this article is available online.
