Abstract
Objective
Arterial elasticity is important for assessing the state of an artery. This cross-sectional study aimed to non-invasively examine stiffness parameters of the ascending aorta in patients with type 2 diabetes mellitus (T2DM).
Methods
We studied 58 patients, including 38 with T2DM and 20 controls. The stiffness of the aorta was evaluated during transthoracic echocardiography. Aortic parameters of stiffness, such as the stiffness index, elasticity index, and compliance index, were calculated using the aortic maximal diameter, aortic minimal diameter, and blood pressure.
Results
Pulse pressure values were significantly higher patients with T2DM than in controls. The β index was significantly higher in patients with T2DM lasting for >7 years compared with those with T2DM lasting for <7 years. Mean aortic compliance was significantly lower in patients with a longer duration of diabetes than in those with a shorter duration of diabetes. Aortic elasticity was significantly lower in patients with diabetes and arterial hypertension compared with patients without diabetes with concomitant arterial hypertension.
Conclusions
Patients with T2DM, especially when T2DM is long-term, have increased stiffness and decreased compliance of the ascending aorta. Pulse pressure, which is a cardiovascular risk factor, is also significantly increased in patients with T2DM.
Introduction
Arterial elasticity is important for assessing the state of the artery. During heart systole, stroke volume interacts with aortic characteristics to produce a pulsatile pressure wave (forward pressure). During diastole, this elastic force recoils the aorta and squeezes the blood forward into the peripheral tissues, thereby ensuring continuous flow. 1 Factors such as age, arterial hypertension, genetic predispositions, smoking, and excessive salt intake diminish arterial elasticity and increase cardiovascular risk. Aortic stiffening leads to increased pulse wave velocity (PWV) and faster return of the reflected wave to the ascending aorta. These phenomena cause an increase in systolic blood pressure, a decrease in diastolic blood pressure, and an increase in pulse pressure (PP). 2
Type 2 diabetes mellitus (T2DM) is an important factor that diminishes arterial elasticity because it leads to glycoside accumulation within vessel walls. 3 Loss of aortic elasticity increases systolic blood pressure and oxygen consumption, impairs diastolic function of the heart, and may lead to heart valve dysfunction. 4 Local elasticity of the aorta is assessed by echocardiographic parameters, such as aortic maximal diameter (Aomax), aortic minimal diameter (Aomin), and blood pressure measurement. Arterial stiffness is increased in the course of T2DM and this phenomenon is believed to constitute an independent risk factor of death and also specifically cardiovascular death. 5 Parameters of aortic stiffness are vital in assessing the state of the vessel wall. Therefore, measurement of these parameters may be useful in establishing vascular age. 6
This study aimed to non-invasively assess parameters of ascending aortic stiffness using transthoracic echocardiography in patients with T2DM. Patients in this study were divided into subgroups on the basis of the duration of diabetes, management of diabetes, and concomitant arterial hypertension.
Subjects and methods
All subjects signed written informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Bioethics Committee of the Medical University of Lublin (Lublin, decision no. KE-0254/272/2011, 24 November 2011).
This study was conducted in individuals who were hospitalized in the Internal Medicine Department. The study group consisted of patients who were diagnosed with T2DM in accordance with the World Health Organization criteria. 7 Patients without carbohydrate metabolism disorders were included in the control group. Exclusion criteria were as follows: heart arrhythmia, aortic valve disease, chronic heart failure with New York Hear Association class >II, malignancy, renal or liver failure, and acute or chronic inflammatory diseases. Obesity and arterial hypertension that were diagnosed on the basis of European Society of Cardiology criteria 8 were not among the exclusion criteria.
Examination of the patients included a thorough medical history, anthropometric measurements, and blood pressure value measurement of the brachial artery during the 3 following days in the patients’ rooms. Mean systolic blood pressure (SBP) and mean diastolic blood pressure (DBP) were calculated. PP was calculated according to the following formula below: PP [mm Hg] = SBP – DBP. Mean arterial pressure (MAP) was established using the following formula below: MAP [mm Hg] = DBP + PP/3.
Transthoracic echocardiography was performed in comparable conditions in every patient. Patients were examined at noon in the supine position after a few minutes of rest in silence at room temperature and in a slightly dim room. Echocardiography was performed with the Vivid 4 device (GE Healthcare, Chicago, IL, USA) and a 2.2-MHz convex transducer. Simultaneous electrocardiography was also performed. The following parameters of the ascending aorta at approximately 3 cm from the aortic valve were assessed in M mode echocardiography using the long axis parasternal view:9,10 Aomax when the aortic valve was open at the peak of the T wave in an electrocardiogram; and Aomin at the peak of the R wave in an electrocardiogram.
Aortic diameters were measured using mean value of results obtained from three following heart cycles. These results were used to calculate parameters of aortic stiffness. The aortic stiffness index (β) was calculated as the quotient of the natural logarithm of pressure alterations to the change in the vessel’s diameter according to the following formula:6,11,12
Epsilon (Ep), which is a pressure-related vessel elasticity index and shows the value of the change in blood pressure necessary to increase the vessel diameter by 100%, was calculated according to the following formula:6,11,12
The aortic compliance index (D), which is the reciprocal of Ep, was calculated according to the following formula:11,13
Aortic stiffness increases with an increase in the β index and Ep and a decrease in D.
Statistical analysis was performed using Statistica 10 PL software (StatSoft Polska, Krakow, Poland). Results are presented as mean values ± standard deviation for quantitative variables and as the percentage for qualitative variables. The studied parameters/variables’ convergence with a normal distribution was tested with the Shapiro–Wilk test. The Student’s t-test, analysis of variance, and the chi-square test were used for comparisons as appropriate. Statistical significance was set as p <0.05. We did not perform a sample size calculation. The limited number of samples may have affected the statistical significance of certain obtained results.
Results
Overall characteristics of the T2DM and control groups
Fifty-eight patients were studied, including 38 patients in the T2DM group and 20 in the control group. Age, as well as other parameters, were not significantly different between the T2DM and control groups (Table 1). The mean duration of diabetes was 10.3 ± 8.1 years. One third (34.2%) of the patients with diabetes were treated with oral agents and two thirds (65.8%) of patients with diabetes received insulin for treatment.
Characteristics of the study and control groups.

Values are mean ± standard deviation, n (%), or n.
T2DM, type 2 diabetes mellitus; BMI, body mass index.
Blood pressure
SBP, DBP, and MAP values were not significantly different between the groups. However, mean PP was significantly higher in the T2DM group compared with the control group (p = 0.028, Table 2).
Blood pressure values in the T2DM and control groups.
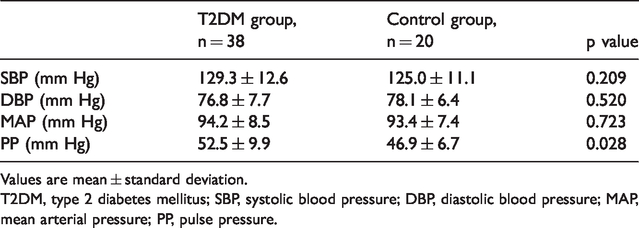
Values are mean ± standard deviation.
T2DM, type 2 diabetes mellitus; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; PP, pulse pressure.
Effect of the duration of diabetes on aortic stiffness parameters
Parameters of aortic stiffness in subgroups of patients were subjected to analysis (Figures 1–3). The β index was significantly higher in patients with T2DM lasting for >7 years compared with those with T2DM lasting for <7 years (p = 0.019, Figure 1). Furthermore, mean aortic compliance (D) was significantly lower in patients with a longer duration of diabetes than in those with a shorter duration of diabetes (p = 0.023, Figure 3).

Aortic stiffness index (β) in patients depending on the duration of diabetes, treatment type, and concomitant arterial hypertension and diabetes.
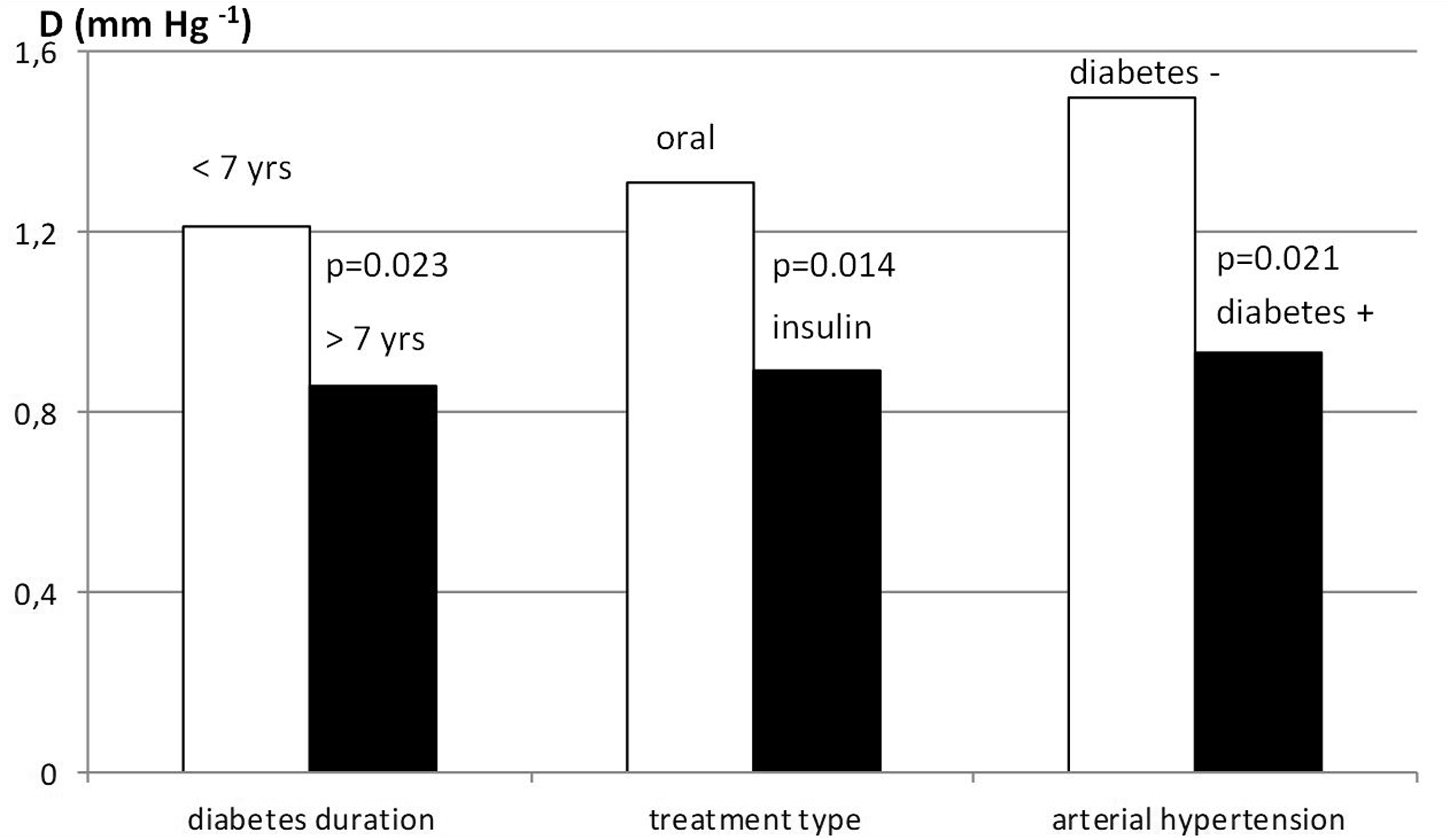
Aortic compliance (D) in patients depending on the duration of diabetes, treatment type, and concomitant arterial hypertension and diabetes.

Epsilon (Ep) in patients depending on the duration of diabetes, treatment type, and concomitant arterial hypertension and diabetes.
Effect of the diabetes treatment method on aortic stiffness parameters
Parameters of aortic stiffness in subgroups of patients who were treated with oral agents and insulin were subjected to analysis. Patients who were treated with oral agents had a lower Ep elasticity index compared with those who were treated with insulin (p = 0.034), which indicated higher aortic elasticity in patients treated with insulin (Figure 2). Aortic compliance (D) in patients who received insulin was significantly lower than that in patients who were treated with oral agents (p = 0.014, Figure 3).
Comparison of patients with coexisting arterial hypertension in the T2DM and control groups
We performed analysis of results obtained from patients with coexisting arterial hypertension in the two groups. Ep was significantly higher in patients with diabetes and arterial hypertension compared with those without diabetes with concomitant arterial hypertension (p = 0.032, Figure 2). Furthermore, aortic compliance (D) was significantly lower in the subgroup of patients with diabetes and coexisting arterial hypertension compared with those without diabetes with coexisting arterial hypertension (p = 0.021, Figure 3).
Parameters such as β, D, and Ep were not different between subjects in the T2DM and control groups in case of concomitant conditions, such as coronary artery disease, obesity, and nicotine addiction.
Discussion
Because of the natural history of atherosclerosis with silent progression over several decades, reliable methods for risk estimations in individuals are required. 14 In the current study, parameters of aortic stiffness were calculated using the ascending aortic diameter and blood pressure measurement taken in the brachial artery. However, the normal range of aortic stiffness parameters has not been established. Intriguingly, among older patients and individuals suffering from arterial hypertension or T2DM, arterial stiffness is higher in the aorta than in the common carotid artery. 15 This phenomenon is not observed in the healthy population. Henry et al. 16 found that T2DM and impaired glucose tolerance were related to increased arterial stiffness. Łoboz-Rudnicka et al. 6 analyzed relations between vascular age (established by intima–media thickness of the carotid artery), cardiovascular risk factors, and stiffness parameters of the common carotid artery in healthy individuals. These authors found that the β stiffness index and Ep elasticity index were positively correlated with vascular and chronological age.
Our study showed that patients with arterial hypertension and T2DM had higher parameters of aortic stiffness parameters compared with patients without T2DM with arterial hypertension. Zapolski et al. 17 showed that the aortic stiffness index β as measured with echocardiography in patients with diabetes was higher than that in healthy individuals. Avgeropoulou et al. 18 found a similar finding. Using the stiffness index β, Ep, and PWV, they observed increased stiffness of the common carotid artery in patients with diabetes, regardless of coexistence of arterial hypertension. Furthermore, Aslan et al. 19 showed that patients with pre-diabetes had higher arterial stiffness as assessed with echocardiography compared with healthy individuals. Sciatti et al. 20 suggested that people suffering from T2DM and hypertension have more impaired aortic elastic properties than those with hypertensive alone. Non-enzymatic glycosylation of proteins in the arterial wall in the course of chronic hyperglycemia is associated with functional and structural changes within the aorta. 3 Furthermore, arterial hypertension is related to faster progression of atherosclerosis and contributes to left ventricular hypertrophy. In our study, arterial hypertension was the most common concomitant disease among patients in the study and control groups. This finding may be due to a relatively low number of patients and the finding that patients included in the study were often burdened with other cardiovascular risk factors, such as arterial hypertension, obesity, and nicotine addiction.
The presence of T2DM itself, as well as a longer duration of diabetes, were associated with increased impairment of arterial elasticity in our study. These results are in accordance with Avgeropoulou et al.’s study, 18 who described an association between the duration of diabetes and the stiffness index β, as well as Ep. Moreover, Tentolouris et al. 21 showed that there was a correlation between the duration of diabetes and decreased aortic compliance. Other studies conducted on patients with diabetes showed that these patients had significantly higher arterial stiffness compared with healthy individuals and PWV was positively correlated with the duration of diabetes.22,23 Arterial stiffening is a natural aging process in vessel walls, which is accelerated in the course of diabetes. A longer duration of diabetes is associated with higher exposure of vessel walls to pathological fluctuations in glucose levels.
In the present study, patients who received insulin had significantly higher arterial stiffness, which may be the effect of long-lasting, poorly controlled diabetes, leading to macroangiopathic complications. Liu et al. 24 investigated patients with T2DM who were aged <30 years, and they found correlations between PWV and age, body mass index, and insulin treatment. These authors concluded that patients who received insulin had higher arterial stiffness. Moreover, studies have shown that 12-week treatment with metformin reduces arterial stiffness as assessed with PWV in patients with non-alcoholic fatty liver disease and polycystic ovary syndrome.25,26 However, patients with type 1 diabetes who received continuous subcutaneous insulin infusion had lower arterial stiffness than those treated with multiple insulin injections. 27 This finding suggests that normalization of glucose levels with continuous insulin infusion might have a protective effect on vessel walls and act against their stiffening. Notably, fluctuations in glucose levels, not mean glucose levels as assessed with glycated hemoglobin, participate in arterial stiffening. 27 Our study suggests that patients with T2DM who are treated with insulin have more advanced arterial stiffness compared with patients taking oral agents.
Increased PP is also believed to be a cardiovascular risk factor. PP increases with age and it is determined by the left ventricular ejection fraction and arterial stiffness. Increased PP in older patients indicates advancement of arterial stiffening, and as a result, internal organ dysfunction. 6 In our study, patients with T2DM had significantly higher PP values than controls. Nilson et al. 28 showed that PP was a strong independent cardiovascular risk factor among patients with diabetes. Taking into consideration the fact that individuals with chronic heart failure with New York Heart Association class >II were excluded from this study, we conclude that high PP values were caused by increased arterial stiffness.
This study has some limitations. One limitation may be due to echocardiographic assessment issues. However, there were no reports of problems with obtaining high-quality images. According to Stefanadis et al., 10 high-quality echocardiographic imaging allows aortic measurements with high accuracy to be obtained. Another limitation of this study is that it was conducted on a low number of patients and within one department. Accordingly, we were not able to use statistical tools, such as multivariate linear regression analysis, and could not verify independent associations between the studied parameters and outcomes. We hope to extend this research to larger groups of patients in the future. Additionally, parameters of arterial stiffness were measured within the wall of the ascending aorta, while blood pressure was measured in peripheral arteries. Pulse wave amplitude within peripheral arteries is significantly higher than that in central arteries in young individuals than in older adults. 29 This is consistent with our finding that SBP and PP, which were measured in peripheral arteries, were higher than SBP and PP in central arteries. This is due to peripheral arteries being much stiffer than the aorta in young healthy people than in older adults; these differences in elasticity tend to disappear with age. Patients who were included in this study belonged to the older population; therefore, blood pressure was measured in the brachial artery in our study.
Patients with T2DM, especially when T2DM is long-term and is treated with insulin, have increased stiffness and decreased compliance of the ascending aorta. These data suggest that this group of patients has a higher risk of development and progression of atherosclerotic lesions. Therefore, echocardiographic assessment of local arterial stiffness in these patients may be useful in the early stages of the atherosclerotic process because it allows identification of functional disorders before occurrence of structural changes. In the future, this assessment may be useful in establishing cardiovascular risk in patients with diabetes and implementing proper secondary prophylaxis. PP, which is also a cardiovascular risk factor, is significantly increased in patients with T2DM.
Footnotes
Acknowledgments
We are grateful to the patients of this study and to the staff of the Department of Internal Diseases, Medical University of Lublin, for assistance with the patients.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.



