Abstract
Objective
Sodium–glucose cotransporter 2 inhibitors (SGLT2i) target the reabsorption of sodium and glucose in the kidney proximal tubules to reduce blood sugar levels. However, clinical randomized controlled trials on SGLT2i have yielded inconsistent results, necessitating further research into their efficacy and safety for specific cardiac and renal diseases.
Methods
“Sodium in urine” was selected as a downstream biomarker of SGLT2i. Single nucleotide polymorphisms were extracted from genome-wide association study data as instrumental variables. Mendelian randomization analysis was then conducted for cardiac and renal diseases and potential adverse events. The causal effects of SGLT2i on these diseases were determined based on inverse variance weighted results, followed by sensitivity and pleiotropy tests.
Results
SGLT2i had a significant protective effect against nephrotic syndrome (odds ratio [OR] 0.0011, 95% confidence interval [CI] 0.000–0.237), chronic glomerulonephritis (OR 0.0002, 95% CI 0.000–0.21), and hypertensive nephropathy (OR 0.0003, 95% CI 0.000–0.785). No causal effects were observed between SGLT2i and cardiac diseases or potential adverse events.
Conclusions
SGLT2i can act as protective factors against nephrotic syndrome, chronic glomerulonephritis, and hypertensive nephropathy.
Keywords
Introduction
Sodium–glucose cotransporter 2 inhibitors (SGLT2i) reduce blood glucose by inhibiting glucose reabsorption in the proximal renal tubules. 1 Large-scale, multicenter, randomized controlled trials have demonstrated the effectiveness of SGLT2i in reducing proteinuria, preserving the glomerular filtration rate, and slowing the progression of chronic kidney diseases (CKD).2–4 Studies such as DAPA-CKD (Dapagliflozin in Patients with Chronic Kidney Disease) and EMPA-KIDNEY (Empagliflozin in Patients with Chronic Kidney Disease) have shown the significant protective effects of SGLT2i on CKD with heterogeneous etiologies including diabetic nephropathy, glomerulonephritis, and immunoglobulin A (IgA) nephropathy.5–7 However, the diverse spectrum of CKD etiologies warrants further investigation into the effects of SGLT2i. Cardiac outcomes studies such as DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure), the EMPEROR-Reduced (Empagliflozin in Patients With Heart Failure, Reduced Ejection Fraction, and Volume Overload) trial, EMPEROR-Preserved (Empagliflozin in Heart Failure with a Preserved Ejection Fraction), and EMPA-REG OUTCOME (Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes) have indicated that SGLT2i significantly reduce the risks of hospitalization and cardiac death in patients with heart failure.8–10 However, EMPA-KIDNEY and DECLARE-TIMI 58 (Dapagliflozin Effect on Cardiovascular Events–Thrombolysis in Myocardial Infarction 58) did not find a significant protective effect against major adverse cardiovascular events.3,11 Notably, these trials have focused more on heart failure and less on atrial fibrillation, stroke, myocardial infarction, and coronary artery disease, which are common comorbidities in patients with CKD and significant risk factors for morbidity, hospitalization, and mortality. 12 To date, large-scale clinical trials exploring the impact of SGLT2i on these cardiovascular conditions are lacking. In addition, the CANVAS (Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes) study found an increased incidence of adverse events such as genital infection, amputation, and fracture in patients receiving canagliflozin as compared with those in the placebo group. 13 The CREDENCE (Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy) study found a high occurrence of diabetic ketoacidosis.3,4 Although studies on other SGLT2i such as empagliflozin and dapagliflozin have reported similar rates for these adverse events, the potentially severe risks of acute kidney injury, hypoglycemia, diabetic ketoacidosis, fracture, and amputation remain a concern and limit the broad use of SGLT2i. Further evidence is needed to clarify the efficacy of SGLT2i in cardiac and renal diseases and to establish their safety profile.
Mendelian randomization (MR) leverages genetic variations in the analysis of causal effects between exposures and outcomes, offering new insights into disease etiology.14,15 This approach is less susceptible to confounding factors and reverse causation compared with traditional observational studies. 15 Under the assumptions of relevance, independence, and exclusivity, MR analyses remain a principled and reliable means to probe causal questions, regardless of whether the results are positive or negative.16,17 For example, MR studies found no causal effect of interleukin 6 signaling on the risk of pulmonary arterial hypertension, and no evidence that elevated urate levels reduce the risk of neurodegenerative outcomes, demonstrating the crucial value of MR in negative interaction between exposure and outcome.18,19 Recent proteomic MR analyses have focused on establishing positive relationships between proteins and diseases such as multiple sclerosis and diabetic nephropathy, aiming to identify drug targets and provide new insights for diagnosis and treatment.20,21
Drug-target MR uses single nucleotide polymorphisms (SNPs) within or near genes encoding drug targets as proxies for drug efficacy, simulating pharmacological effects to explore potential causal relationships between exposures and outcomes from a genetic perspective.22–25 To address inconsistencies in clinical observations and the lack of in-depth research on SGLT2i and common cardiac diseases, genome-wide association study (GWAS) data were used in two-sample drug-target MR analysis. In our study, we evaluated the causal relationships between SGLT2i and CKD with various etiologies, multiple cardiac conditions, and potential adverse reactions to enhance understanding of the efficacy and safety of SGLT2i (Figure 1).

Overview diagram of SGLT2i drug-target Mendelian randomization analysis. Note: The diagram was created with MedPeer (www.medpeer.cn). SGLT2i: Sodium–glucose cotransporter 2 inhibitors; OR, odds ratio; CI, confidence interval.
Methods
Data sources
In this MR study, we used exposure and outcome data derived from publicly available GWAS datasets, focusing on European populations. The data were located and downloaded after searching by GWAS ID in the Medical Research Council Integrative Epidemiology Unit (IEU) OpenGWAS project (https://gwas.mrcieu.ac.uk), as detailed in Table 1. All summary-level GWAS data used in the analyses are publicly available; therefore, this study was exempt from ethics review and the need for informed consent. Ethical approval for the GWASs can be found in the corresponding GWAS publications cited in the manuscript.
Outcome data information.

Study design
Sodium in urine (GWAS ID: ebi-a-GCST90013989) was selected as a downstream biomarker for MR analysis using the TwoSampleMR package (version 0.5.7) in R (version 4.2.3; www.r-project.org) to mimic the pharmacological effects of SGLT2i. The extract_instruments function was applied to retrieve GWAS data with P < 5 × 10−5. Linkage disequilibrium was addressed using the ld_clump function in the ieugwasr package (kb = 10,000, r2 = 0.3). We selected SNPs located within 1 Mb of the gene’s start site, specifically, on chromosome 16, positions 31,483,123–31,490,769 (GRCh37). Using this process, we identified four SNPs: rs11642632, rs72800849, rs8060136, and rs8063700 (Supplementary Table 1). All SNPs demonstrated F-statistics greater than 10 without weak instrument bias. PhenoScanner examination (www.phenoscanner.medschl.cam.ac.uk) revealed no association between these SNPs and known diseases. The identified SNPs satisfied the core assumptions of relevance, independence, and exclusivity required for MR.
With the four SNPs as exposures and potassium in urine (GWAS ID: ebi-a-GCST90013988) as a positive control, relevant outcome data were extracted using the extract_outcome_data function. Data were harmonized using the harmonise_data function. MR analysis was conducted according to five methods: inverse variance weighted (IVW), MR-Egger, weighted median, simple mode, and weighted mode. On the basis of the IVW results, we assessed the ability of the four SNPs to reflect the clinical effects of SGLT2i. The exposure of these SNPs in relation to kidney diseases, cardiac diseases, and certain potential adverse effects were then analyzed through MR analysis, including sensitivity and pleiotropy tests (Figure 2). The results were visualized using the forestploter package (version 1.1.1) in R.

Workflow of Mendelian randomization analysis for SGLT2i. The F-statistic was calculated as R2 = β2/(β2 + N × SE2) and F = (R2 × (N − 2))/(1 − R2), where β represents the effect size of the SNP, N is the total sample size, and SE is the standard error of the effect size. SLC5A2, solute carrier family 5 member 2 gene, encoding sodium–glucose cotransporter; SGLT2i, sodium–glucose cotransporter 2 inhibitor; SNP, single nucleotide polymorphism; IgA, immunoglobulin A; Chr, chromosome.
Results
In the MR analysis with urinary potassium as a positive control, the IVW method yielded a significant result, with P = 3.86 × 10−7 and odds ratio (OR) > 1 (Supplementary Table 2), consistent with the clinical observation that an increased urinary sodium level leads to increased potassium excretion. This finding demonstrated that the four SNPs identified through preliminary screening can reflect the pharmacological action of SGLT2i.
MR analysis suggested that SGLT2i can provide substantial protection against a variety of CKD (Figure 3, Supplementary Table 3). Significant protection effects were observed for nephrotic syndrome (OR 0.0011, 95% confidence interval [CI] 0.000–0.237, P = 0.013), chronic glomerulonephritis (OR 0.0002, 95% CI 0.000–0.210, P = 0.017), and hypertensive nephropathy (OR 0.0003, 95% CI 0.000–0.785, P = 0.043). For diabetic nephropathy (OR 0.143, 95% CI 0.008–2.690) and chronic interstitial nephritis (OR 0.432, 95% CI 0.001–315), a protective effect was indicated, although the results were not statistically significant. No causal relationship was observed for pyelonephritis (OR 1.577, 95% CI 0.251–9.901) or IgA nephropathy (OR 1.462, 95% CI 0.431–4.963).
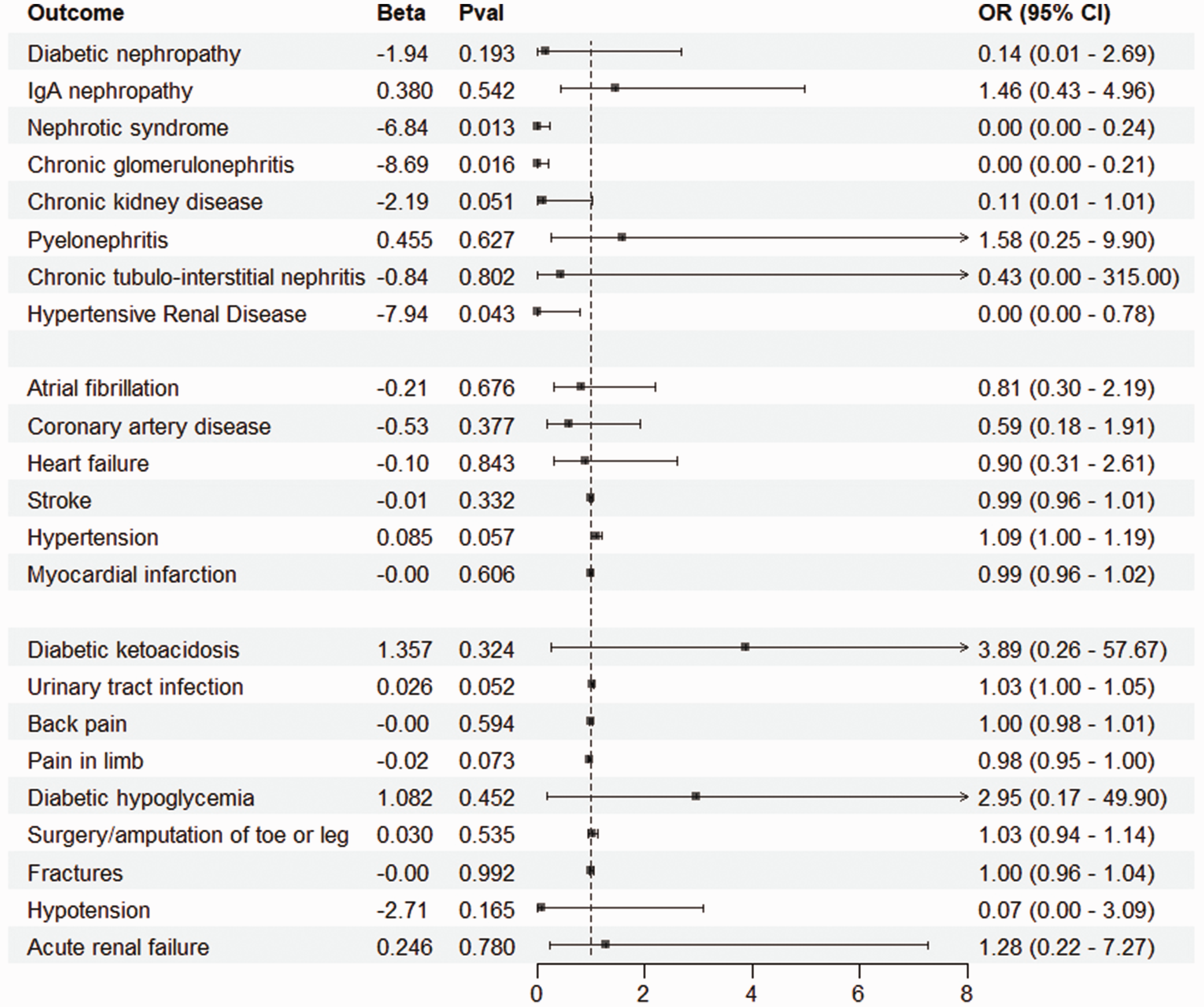
Results of Mendelian randomization analysis. Outcome: events included in the analysis; Beta: effect estimate of SGLT2 inhibitors on outcomes. IgA, immunoglobulin A; Pval, P-value. OR, odds ratio. CI, confidence interval.
In the MR analysis for cardiac diseases such as atrial fibrillation, coronary artery disease, stroke, heart failure, and myocardial infarction, SGLT2i were identified as potentially protective factors for most conditions (95% CI 0.30–2.19 for atrial fibrillation, 95% CI 0.18–1.91 for coronary artery disease, 95% CI 0.31–2.61 for heart failure, 95% CI 0.96–1.01 for stroke, and 95% CI 0.96–1.02 for myocardial infarction). However, the P-values did not show statistical significance (Figure 3, Supplementary Table 4).
MR analysis was conducted for adverse events commonly reported in clinical trials on SGLT2i. Conditions such as diabetic ketoacidosis, acute kidney injury, fracture, amputation, back and leg pain, and urinary tract infection did not show a definitive causal relationship with SGLT2i from the perspective of genetic variations (Figure 3, Supplementary Table 5).
All Mendelian analyses were subjected to heterogeneity and sensitivity tests, revealing no pleiotropy or heterogeneity. Scatter plots were used to illustrate the association strength between the SNPs and the exposure/outcome, with different colored lines representing fits from various models. Leave-one-out tests demonstrated stability in the MR outcomes between the exposure and outcome after the removal of any of the SNPs (Figure 4).

Scatter plots and sensitivity analysis. (a, b) Scatter plot and leave-one-out analysis for the Mendelian randomization (MR) of SGLT2 inhibitors and nephrotic syndrome. (c, d) Scatter plot and leave-one-out analysis for the Mendelian randomization of SGLT2 inhibitors and chronic glomerulonephritis and (e, f) Scatter plot and leave-one-out analysis for the Mendelian randomization of SGLT2 inhibitors and hypertensive nephropathy. SGLT2, sodium–glucose cotransporter 2; SNP, single nucleotide polymorphism.
Discussion
The MR results indicated that SGLT2i can delay the progression of CKD with most etiologies, particularly hypertensive nephropathy, chronic glomerulonephritis, and nephrotic syndrome. SGLT2i exhibit significant protective effects, corroborating the clinical trial finding that drugs such as dapagliflozin and empagliflozin significantly slow the progression of glomerulonephritis and hypertensive nephropathy.5,7 The renoprotective mechanisms of SGLT2i extend beyond direct glucose reduction and proteinuria reduction to lowering the glomerular filtration rate, improving oxidative stress, protecting renal microvasculature, and alleviating renal hypoxia.1,26,27 In mouse models, SGLT2i have been shown to modulate inflammation and metabolism, enhance mitochondrial function, and reduce inflammation and fibrosis, contributing to nephroprotective effects.28–30 These actions likely delay the onset and progression of hypertensive nephropathy and chronic glomerulonephritis. Furthermore, the analysis of SGLT2i in relation to nephrotic syndrome can provide new insights into the treatment of this disease. Nephrotic syndrome, characterized by substantial proteinuria, hypoalbuminemia, severe edema, and hyperlipidemia, can be inhibited or delayed with use of SGLT2i by significantly reducing proteinuria and exhibiting anti-inflammatory and antifibrotic effects.31,32 Although large-scale clinical trials specifically investigating the impact of SGLT2i on nephrotic syndrome are yet to be conducted, the MR analysis results and the clinical efficacy of SGLT2i in current studies suggest that these drugs may become a frontline medication for treating nephrotic syndrome in the future.
MR analysis did not establish a causal relationship between SGLT2i and cardiac events such as atrial fibrillation, coronary artery disease, heart failure, stroke, and myocardial infarction. These negative results warrant careful interpretation and consideration of several factors. The genetic variants used as instrumental variables in the analysis may not fully capture the complex pharmacological effects of SGLT2i. Studies including DAPA-HF, EMPEROR-Reduced, EMPEROR-Preserved, EMPA-REG OUTCOME, CREDENCE, and CANVAS have demonstrated that SGLT2i significantly reduce the risk of cardiac death and hospitalization in patients with heart failure, decrease the incidence of atrial fibrillation, and improve the prognosis of myocardial infarction.33–36 The discrepancy between our results and those of clinical studies could be attributed to differences in study populations, endpoints, or unmeasured confounding in observational studies, which MR aims to address. Negative MR findings do not negate these mechanisms but suggest that the overall causal impact on cardiovascular disease may be more complex than can be captured through genetic instruments alone. SGLT2i may exert direct effects against myocardial hypertrophy, fibrosis, and remodeling37,38 or indirectly protect the heart by slowing the deterioration of renal function, diuresis, glucose control, stimulation of erythropoiesis, and inhibition of the sympathetic nervous system.39–42 Further large-scale clinical studies are necessary to clarify the long-term influence of SGLT2i on cardiac diseases.
Our analyses did not identify a causal link between SGLT2i and potential adverse events such as amputation, fracture, dehydration, urinary tract infection, and acute kidney injury. It is essential to interpret these findings within the context of the existing literature. The findings from well-designed systematic reviews and meta-analyses of randomized clinical trials that have established a causal relationship between SGLT2i and certain adverse effects, particularly urinary and reproductive tract infections and diabetic ketoacidosis.43,44 Little or no evidence was found for the effect of SGLT2i on limb amputation, blindness, eye disease, neuropathic pain, or health-related quality of life. 45 These findings highlight the importance of closely monitoring and managing potential safety concerns associated with SGLT2i use in clinical practice.
In summary, we analyzed the causal relationship between SGLT2i and diseases from a genetic variation perspective through the innovative use of MR. Our results indicated that SGLT2i may have potential protective effects against chronic glomerulonephritis, hypertensive nephropathy, and nephrotic syndrome. However, these results need to be verified in further clinical trials. This study provides a theoretical basis for expanding the clinical application scope of SGLT2i.
This study has several limitations. First, MR analysis is not a true randomized controlled trial, and the clinical benefits of the medication cannot be directly estimated merely based on P-values and ORs. 22 Second, in selecting instrumental variables, the P-value threshold for SNPs was set at 5 × 10−5 instead of the conventional 5 × 10−8, increasing the risk of false positives. Finally, the exposure and outcome data were derived from GWAS studies of European populations, preventing the analysis of drug responses across different races and ethnicities as performed in multicenter clinical trials. This limitation may contribute to the negative MR results for diabetic nephropathy and IgA nephropathy.
Conclusions
SGLT2i may act as protective factors against nephrotic syndrome, chronic glomerulonephritis, and hypertensive nephropathy, as revealed in drug-target MR.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241272639 - Supplemental material for Mendelian randomization study of sodium–glucose cotransporter 2 inhibitors in cardiac and renal diseases
Supplemental material, sj-pdf-1-imr-10.1177_03000605241272639 for Mendelian randomization study of sodium–glucose cotransporter 2 inhibitors in cardiac and renal diseases by Lei Chen, Yongdi Zuo, Manrong He, Lijin Duo and Wanxin Tang in Journal of International Medical Research
Footnotes
Acknowledgements
This study extensively used GWAS data from the IEU database. The authors thank the participants and investigators from the contributing studies; this research would not be possible without them.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Lei Chen, Yongdi Zuo, Manrong He, and Lijin Duo. The first draft of the manuscript was written by Lei Chen and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data availability statement
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a Chengdu Science and Technology Bureau Grant (grant no. 2019-YF09-00090-SN) and the Science and Technology Department of Sichuan Province (grant no. 24ZDYF1187).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
