Abstract
Aim:
To describe nationwide complication patterns in patients with atrial fibrillation and diabetes mellitus.
Methods:
All (n = 326,832) patients in Sweden with non-valvular atrial fibrillation during 2006–2012 were identified, and information on events, comorbidities and pharmacological therapy was extracted using nationwide mandatory registers. Patients were followed until 31 December 2013 and the mean follow-up time was 3.7 years (0.9–8 years).
Results:
Diabetes was present in 17.7%. The most frequent events in those with and without diabetes were mortality (48.8% vs 36.4%; p < 0.001), heart failure (21.4% vs 13.1%; p < 0.001), ischaemic stroke (8.2% vs 6.8%; p < 0.001), myocardial infarction (7.3% vs 4.3%; p < 0.001) and any bleeding (6.3% vs 5.2%; p < 0.001), respectively. Diabetes predicted mortality (hazard ratio = 1.28; 95% confidence interval = 1.25–1.31), combined event (first of mortality, heart failure, ischaemic stroke or myocardial infarction; hazard ratio = 1.22; 95% confidence interval = 1.20–1.25), single events and bleeding (hazard ratio = 1.12; 95% confidence interval = 1.06–1.19). The standardised mortality ratio for patients with atrial fibrillation and diabetes compared to the general population was 2.06 (95% confidence interval = 2.00–2.12) and for patients with atrial fibrillation without diabetes was 1.33 (95% confidence interval = 1.31–1.35).
Conclusion:
In this real-world setting, patients with atrial fibrillation and diabetes have a high cardiovascular risk, with mortality and heart failure rates exceeding those for stroke.
Introduction
Globally, cardiovascular disease is the main contributor to mortality, accounting for about 17.5 million deaths, or 46.2% of deaths from non-communicable diseases. 1 Atrial fibrillation (AF) is present in approximately 3% of the general adult population and the prevalence is expected to rise, mainly due to the ageing population. 2 With the estimated future rise in the number of persons with AF from 14 to 17 million in Europe by 2030, together with the associated five-fold increased risk of stroke and two-fold increased risk of mortality, AF will have a significant impact on future healthcare costs. 3
Diabetes mellitus is a well-established risk factor for AF, increasing the risk for AF by 40%. 4 Even in patients with increased fasting glucose 5 or metabolic syndrome, 6 the AF risk is increased by a similar magnitude. Retrospective data have shown a 70%–90% increase in the risk of stroke in those with coexisting diabetes and AF compared to those with AF alone. 7 In the CHA2DS2-VASc risk score system for calculating the risk for stroke in patients with AF, the presence of diabetes adds one point. 8 However, the risk of mortality and other adverse cardiovascular outcomes is not estimated using this score system. Updated information on the risk for complications associated with diabetes in AF is important for determining future preventive strategies and for planning of healthcare resources.
The aim of this study was to analyse the prevalence of diabetes and its impact on outcome in a contemporary population with AF from a nationwide perspective.
Methods
Study population and data sources
This study was based on all-inclusive national registers. Four registers were used: (1) the Swedish National Patient Register [hospitalisations and visits to hospital-affiliated outpatient clinics with diagnoses coded by International Classification of Diseases 10 codes (ICD codes)]; (2) the Swedish Dispensed Drug Register (prescriptions dispensed in Sweden since 1 July 2005 and coded by international standards into the Anatomical Therapeutic Chemical classification system code); (3) the Cause of Death register and (4) the LISA register (maintained by the Statistics Sweden governmental agency), which stores information about socioeconomic conditions. The ICD codes and a list of definitions used in our study can be found in the supplemental material (Supplemental Table S1).
We identified 345,123 patients with a diagnosis of AF between 1 January 2006 and 31 December 2012 who survived 30 days after the index event. All were followed for the incidence of mortality, heart failure, ischaemic stroke, myocardial infarction or any bleeding until 31 December 2013. Patients ⩽20 years (n = 409) and patients with valvular AF (n = 17,882) were excluded. The final study population included 326,832 patients. A flowchart with patient selection is depicted in the Supplemental Figure S1.
The study was approved by the Regional Ethical Review Board in Stockholm (reg. nos 2012/60-13/2 and 2016/1255-32) and complies with the Declaration of Helsinki.
Statistical analysis
Categorical variables are presented with absolute and relative (percentage) frequencies and are compared using Pearson’s chi-square. Continuous variables are presented as the mean ± standard deviation and are compared using the non-parametric Kruskal–Wallis H test. Time-to-event data by diabetes status, age and glucose-lowering therapy (GLT) are displayed using Kaplan–Meier curves and were compared with the log-rank test. Event data are presented in absolute numbers and percentage frequencies. Incidence rates are presented as events per 100 patient-years at risk. Cox’s proportional hazard regression analysis was used to calculate hazard ratios (HR) and their 95% confidence intervals (CIs) for the different events in patients stratified by diabetes status, GLT, age groups and duration of AF. A stepwise multivariate regression analysis was performed with adjustments for comorbidities and socioeconomic factors in the first step. In the second step, adjustments for medication at baseline were added. A list of comorbidities and medication we adjusted for is presented in the Supplemental Table S2. To calculate the standardised mortality ratios (SMR), we used Swedish population data on mortality in different age groups for the year 2012, freely available from Statistics Sweden, for reference purposes. A two-sided probability of p < 0.05 was considered statistically significant in all tests. All analyses were performed with STATA version 14 (STATA Corp., College Station, TX, USA).
Results
Among 326,832 individuals with non-valvular AF, 17.7% (n = 57,953) had diabetes. Of those with diabetes, 28.9% had no GLT, 35.4% were treated with oral antiglycaemic drugs (OAD) alone and 35.7% were treated with insulin (with or without OAD). Baseline characteristics stratified by diabetes status and treatment are presented in Table 1. Patients with diabetes were slightly older, were more often male, and scored higher on the stroke risk scheme CHA2DS2-VASc and on the bleeding risk scheme HAS-BLED. Diabetes individuals more frequently had comorbidities such as hypertension, ischaemic heart disease, stroke, heart failure, previous coronary revascularisation, peripheral artery disease or chronic kidney disease. Accordingly, cardiovascular drugs were used more frequently in those with diabetes. Patients with coexisting AF and diabetes had a lower degree of education, more frequently lived alone and had a lower annual income. The same risk factor burden and treatment patterns were seen at ages below 65 years, apart from the fact that male gender was more prevalent and alcohol consumption was more common in the diabetes group (Supplemental Table S3). Anticoagulants (warfarin or newer oral anticoagulants) were more commonly used in those with diabetes (47.2% vs 43.2%, p < 0.001). The proportion of diabetes patients with a CHA2DS2-VASc score of ⩾2 was 99% (n = 57,278). Among these, 53% were not treated with anticoagulants and 13% had neither anticoagulants nor antiplatelet agents. In general, diabetes individuals treated with insulin had a higher cardiovascular risk burden, and higher CHA2DS2-VASc and HAS-BLED scores.
Baseline characteristics stratified by diabetes status and treatment (percentage figures in parentheses).
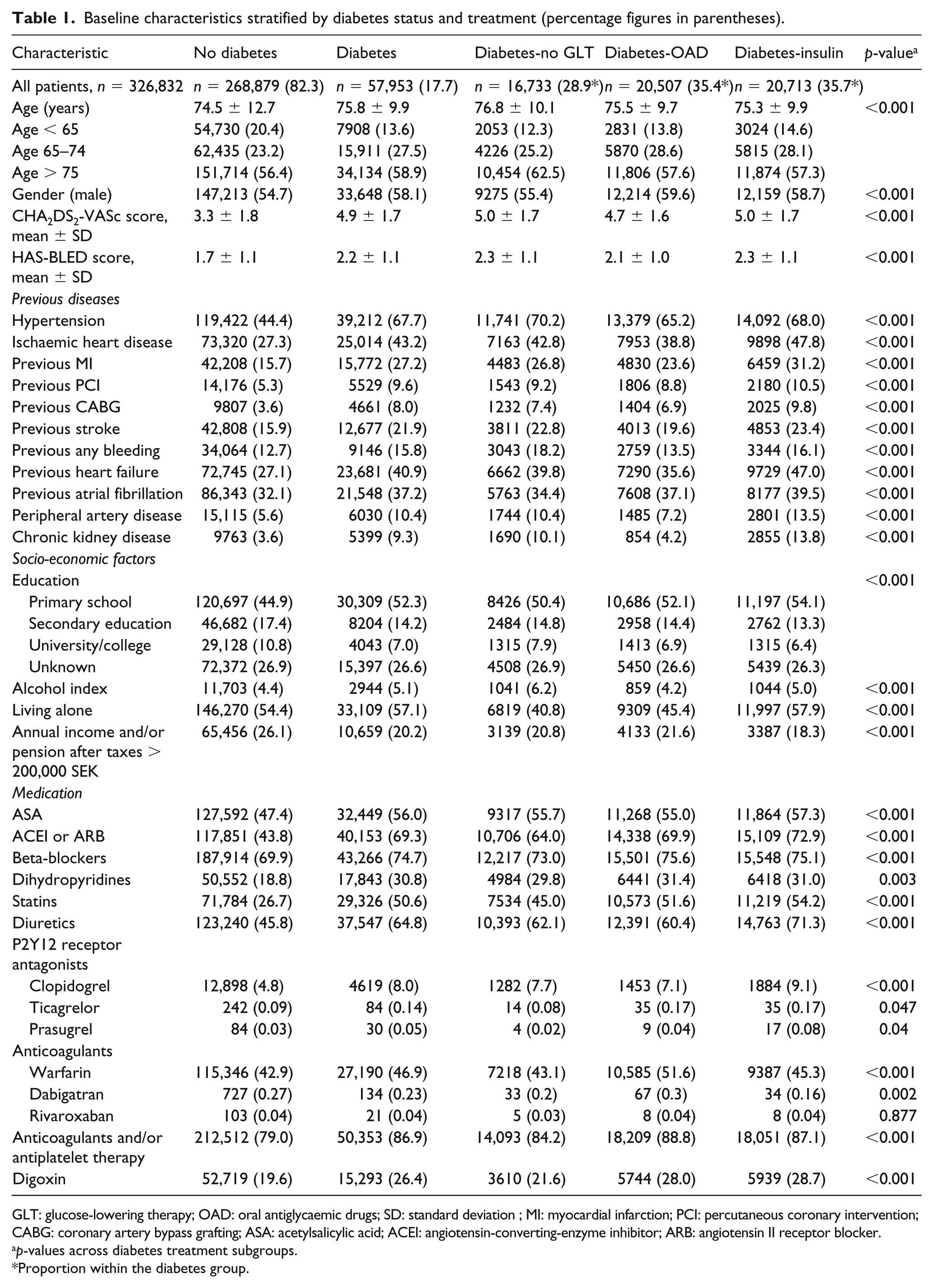
GLT: glucose-lowering therapy; OAD: oral antiglycaemic drugs; SD: standard deviation ; MI: myocardial infarction; PCI: percutaneous coronary intervention; CABG: coronary artery bypass grafting; ASA: acetylsalicylic acid; ACEI: angiotensin-converting-enzyme inhibitor; ARB: angiotensin II receptor blocker.
p-values across diabetes treatment subgroups.
Proportion within the diabetes group.
Long-term event rate
The mean follow-up time was 3.7 years (range = 0.9–8 years). Number of events and incidence rate stratified by diabetes status and treatment are shown in Table 2. The most frequent event was mortality (48.8% vs 36.4%; p < 0.001), followed by heart failure (21.4% vs 13.1%; p < 0.001), ischaemic stroke (8.2% vs 6.8%; p < 0.001), myocardial infarction (7.3% vs 4.3%; p < 0.001) and any bleeding (6.3% vs 5.2%; p < 0.001) in those with and without diabetes, respectively (Supplemental Figure S2). Within the diabetes group, all events except bleeding occurred more frequently in those treated with insulin. The same pattern was also seen at ages below 65 years (Supplemental Table S4) and in patients without previous AF at baseline, that is, with a first occurrence of AF (Supplemental Table S5). Kaplan–Meier curves for time to events are depicted in Figure 1. Forest plots of the HR (95% CI) for different events, adjusted for comorbidities and medication, stratified by diabetes status and treatment are illustrated in Figure 2. After adjustment for comorbidities, socioeconomic factors and medication, diabetes was independently associated with an increased mortality risk (HR = 1.28; 95% CI = 1.25–1.31) which was even more pronounced at ages below 65 years (HR = 1.59; 95% CI = 1.43–1.77) (Supplemental Table S6). The corresponding figures for the combined event were 1.22; 1.20–1.25 and at ages below 65 years 1.41; 1.31–1.53. In patients with a first occurrence of AF, diabetes was associated with a similar increase in risk (Supplemental Table S7).
Number of events and events per 100 person-years stratified by diabetes status and treatment.
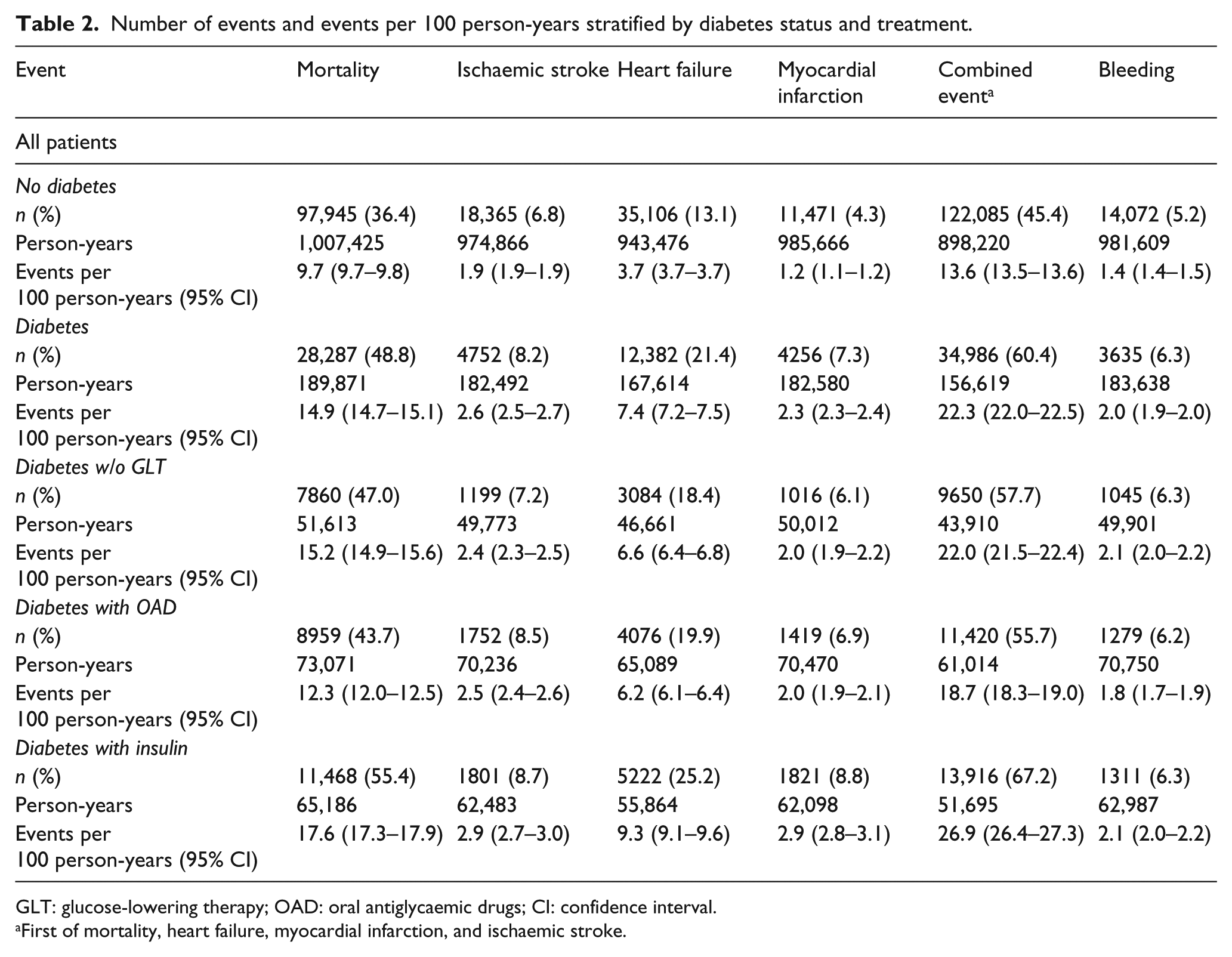
GLT: glucose-lowering therapy; OAD: oral antiglycaemic drugs; CI: confidence interval.
First of mortality, heart failure, myocardial infarction, and ischaemic stroke.
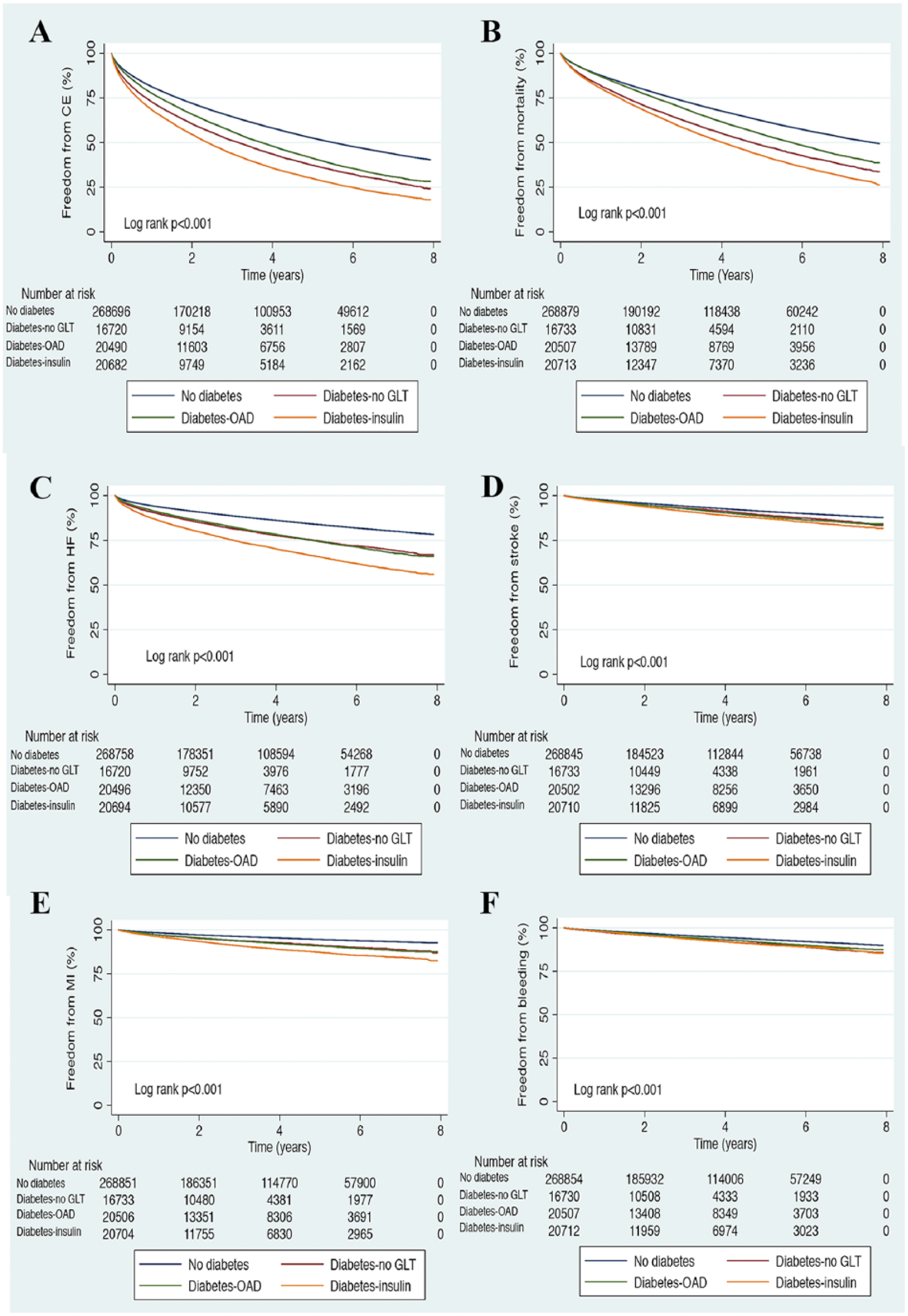
Kaplan–Meier curves showing time to freedom from A. Combined event (CE; first of mortality, heart failure, ischaemic stroke and myocardial infarction), B. Mortality, C. Heart failure, D. Ischaemic stroke, E. Myocardial infarction and F. Any bleeding.
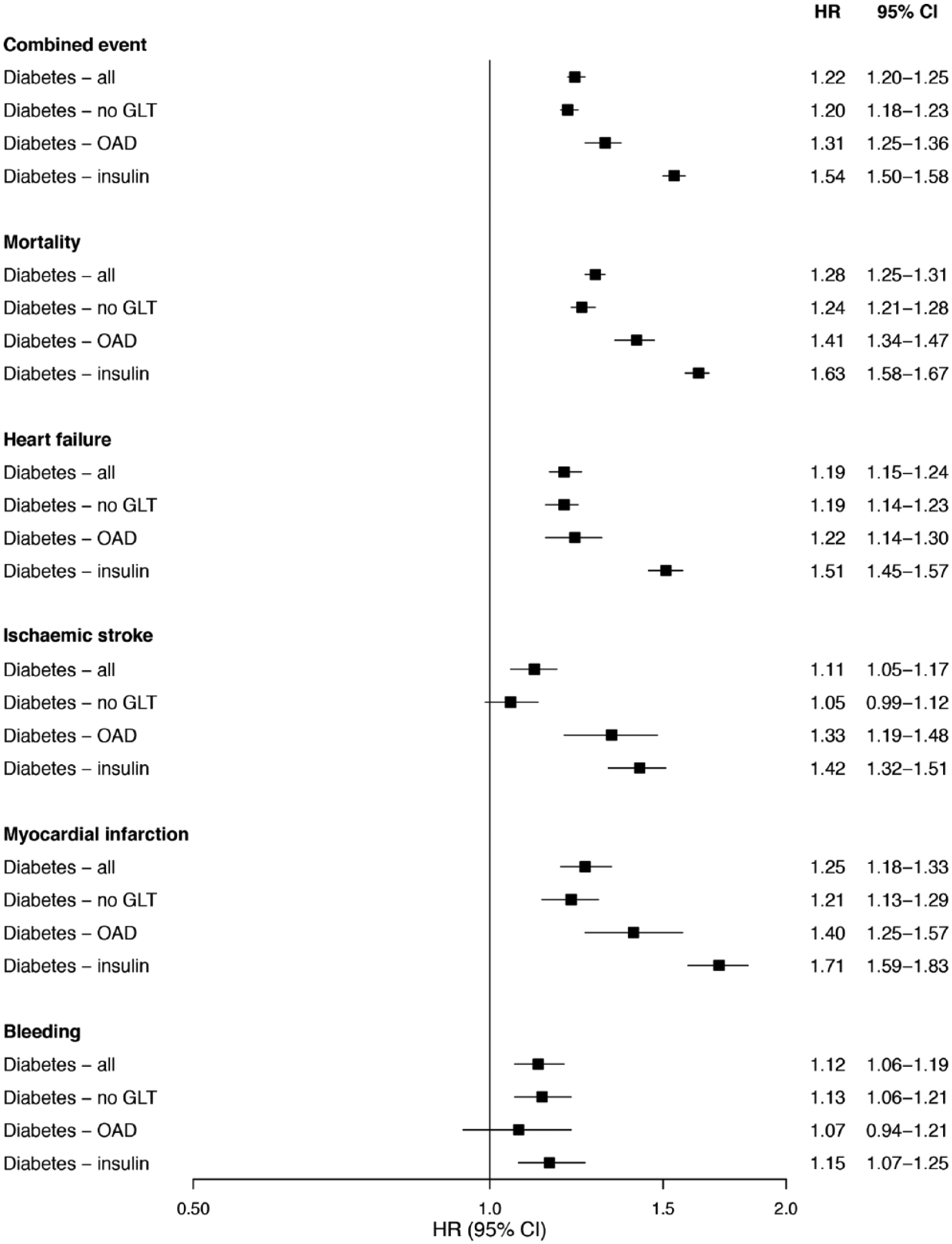
Forest plots with HR (95% CI) for combined event (first of mortality, heart failure, ischaemic stroke, and myocardial infarction), single events and any bleeding, adjusted for comorbidities and medication, stratified by diabetes status and treatment as compared to patients without diabetes (HR = 1.00).
The SMR for year 2012 for all patients with AF and diabetes was 2.06 (95% CI = 2.00–2.12) and for those with AF alone 1.33 (95% CI = 1.31–1.35). The SMR in those with AF and diabetes at the age of 50–54 years was 12.60 (95% CI = 7.05–20.78), at 55–59 years 5.94 (95% CI = 3.88–8.70) and at 60–64 years 5.96 (95% CI = 4.83–7.28). The SMR in patients with AF alone at the age of 50–54 years was 2.63 (95% CI = 1.83–3.65), at 55–59 years 2.58 (95% CI = 2.07–3.17) and at 60–64 years 2.17 (95% CI = 1.89–2.47) (Supplemental Figure S3).
Mortality causes are listed in the Supplemental Table S8. The most common causes of mortality in patients with and without diabetes, respectively, were cardiovascular disease (53.0% vs 55.4%) and cancer (14.9% vs 18.1%). Death from endocrine/metabolic disease was more common in patients with diabetes (10.2% vs 0.7%). Death from cerebrovascular disease was less common in patients with AF and diabetes (8.7% vs 9.5%).
Discussion
This nationwide study based on real-life data in an unselected population shows that patients with coexisting AF and diabetes have more cardiovascular events and higher mortality rates compared to those without diabetes. A similar pattern is observed in younger patients and in those with a first occurrence of AF. The combination of diabetes and AF increased the mortality rate two-fold compared to the general population and over five times at ages below 65 years. In contrast, the mortality rate in those with AF but without diabetes was only slightly increased (1.3 times). All cardiovascular events occurred more frequently in the diabetes population and somewhat surprisingly, heart failure was the second most common event after mortality, while stroke occurred at a lower rate. This stresses the importance of addressing not only the stroke risk but also the overall cardiovascular risk in patients with AF and coexisting diabetes.
The recently updated AF guidelines from the European Society of Cardiology (ESC) state that three (i.e. I: acute rate and rhythm control, II: management of precipitation factors and III: assess stroke risk) of the five recommended treatment domains in AF patients will have an impact on outcome and further highlight the fact that deaths and hospitalisations are less related to stroke and more to deteriorating heart failure and sudden cardiac death. 2 Our data further strengthen that this also applies to those with coexisting diabetes, where heart failure and mortality rates exceeded those for stroke. Lifestyle modification is a new and highlighted treatment domain in the guidelines. In patients with diabetes, several lifestyle factors such as obesity, physical inactivity and as shown in our study, alcohol overconsumption in the younger age groups might contribute to AF and the subsequent associated worse prognosis and multifactorial intervention is indeed important in prevention of diabetes complications. 9 In the small randomised ARREST-AF study, in which 17% of patients with AF had known diabetes, multifactorial intervention reduced the risk of recurrent AF. 10 In the recent LEGACY study, in which around 30% had diabetes and 10% impaired glucose tolerance, sustained weight loss reduced the burden of recurrent AF. 11 Interestingly, a contemporary improvement in echocardiographic abnormalities with a reduction in left atrial volume and left ventricular septal thickness was found. 11 A similar effect as a result of multifactorial intervention in AF was reported in a recent single-centre trial from Australia. 12 Those studies were too small to evaluate the effect in the subgroups with diabetes and AF, but since the clustering of cardiovascular risk factors is even more pronounced in those with diabetes, there are reasons to believe that such multifactorial intervention could be even more beneficial for these patients.
We found the highest event rates among those treated with insulin. The most likely explanation is that insulin treatment is a proxy for longer diabetes duration, which is also supported from a recent nationwide study from Denmark that reported higher mortality and thromboembolism risk with longer diabetes duration in AF patients. 13 Due to the observational character of our study, it cannot be concluded that insulin per se is responsible for the adverse outcome, rather that insulin use signifies a high-risk individual.
There are several possible explanations for the increased risk of incident AF and subsequent cardiovascular events in patients with diabetes. Risk factors associated with the metabolic syndrome and diabetes, such as hypertension, ischaemic heart disease, obesity and arterial stiffness, are all individually associated with an increased AF risk and also with the development of cardiovascular complications. 2 At the myocardial level, several mechanistic explanations have been suggested as a consequence of diabetes, including structural, metabolic, electrical and electromechanical atrial remodelling changes. 14 Moreover, diabetes might lead to the development of diabetic cardiomyopathy and heart failure with either a preserved or a reduced ejection fraction, increasing the risk of AF, as well as worsening prognosis. 15
Until recently, clinical trials studying glucose-lowering effects on cardiovascular outcomes have been disappointing. However, a paradigm shift was introduced when trials on novel diabetes medications, such as sodium-glucose co-transporter 2 (SGLT2) inhibitors and glucagon-like peptide-1 (GLP-1) analogues, showed a reduction in total and cardiovascular mortality in patients with high cardiovascular risk.16,17 Considering the high event rate in diabetes individuals with AF, this mortality reduction, together with findings including reduced hospitalisations for heart failure with the SGLT2 inhibitor empagliflozin, 16 and weight reduction seen with both empagliflozin and the GLP-1R-analogue liraglutide are of particular importance.16,17 Therefore, future studies exploring the effects of these therapies in a population with coexisting diabetes and AF, where heart failure and overweight are often overrepresented, would be of interest.
Although we found lower rates of stroke compared to mortality and heart failure in the diabetes cohort, stroke is a severe disabling complication of AF and diabetes. 7 In this real-life study, we found that even though 99% of diabetes patients had a CHA2DS2-VASc score of ⩾2, less than 50% were treated with oral anticoagulants and the use of novel oral anticoagulants (NOAC) was very low (only 0.4%). In a meta-analysis of the four Phase-III trials comparing the efficacy of warfarin versus NOACs in patients with AF and diabetes, a favourable trend, but no significant risk reduction, was found in the occurrence of ischaemic stroke with the use of NOACs as compared to warfarin, though significance was reached in favour of NOACs in the composite endpoint of stroke/systemic embolism.18,19 A possible better outcome with NOACs in patients with diabetes mellitus cannot be ruled out and future studies in this population would be of interest. Bleeding complications were only marginally increased after adjustment for comorbidities and other medications, with no major difference between diabetes treatment groups. It could be possible that the rate of bleeding complications could be improved with the increased use of NOACs in the future, especially the risk of intracranial bleeding. 18 Interestingly, cardiovascular disease-related mortality in our study did not substantially differ in patients with and without diabetes, estimated to 53% and 55%, respectively, although occurring at a higher rate and at earlier occasion in those with diabetes.
Strengths and limitations
One major strength of our study is the inclusion of all AF patients in Sweden, apart from those diagnosed only in the primary care setting and not hospitalised or seen in hospital-based, outpatient clinics. The main limitation is the lack of information on body mass index (BMI), smoking, left ventricular function, staging of chronic kidney disease and diabetes variables. Another limitation is the extreme low NOAC utilisation in our population, which does not reflect the current treatment trends. Regarding mortality causes, there is uncertainty to a certain extent in death certificates with a possible misclassification of mortality cause in some cases. Finally, the Drug Register only provides information on filled prescriptions and not medication adherence.
Conclusion
In conclusion, this nationwide study shows that patients with AF and diabetes have a high overall cardiovascular risk, with rates of mortality and heart failure exceeding those for stroke. This underlines that other preventive treatment strategies, beyond preventing stroke with anticoagulants, need to be implemented and further explored.
Footnotes
Declaration of conflicting interests
Dr Norhammar has received honoraria for expert group participation and speaker’s fees from Astra-Zeneca, MSD, Boehringer-Ingelheim, Lilly and Novo Nordisk; Dr Friberg is a consultant to Sanofi, Bayer, Pfizer and BMS; Dr Karayiannides has received speaker’s fees from Novo Nordisk, Astra-Zeneca, Boehringer-Ingelheim and Lilly, and honorarium for expert group participation from Astra-Zeneca; and Dr Lundman has received speaker’s fees from Astra-Zeneca and honoraria for expert group participation from MSD (Sweden) and Sanofi.
Funding
This work was supported by the Swedish Heart-Lung Foundation and research funds from the Department of Internal Medicine at Danderyd University Hospital, Karolinska Institutet and Stockholm County Council.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
