Abstract
Objective
The effect of renin–angiotensin system inhibitors (RASIs) in patients with heart failure (HF) and atrial fibrillation (AF) remains unclear. This study aimed to investigate associations between RASI use and all-cause mortality and cardiovascular outcomes in patients with AF and HF.
Methods
Using data from the China Atrial Fibrillation Registry study, we included 938 patients with AF and HF with a left ventricular ejection fraction <50%. Cox regression models for RASIs vs. non-RASIs with all-cause mortality as the primary outcome were fitted in a 1:1 propensity score-matched cohort. A sensitivity analysis was performed by using a multivariable time-dependent Cox regression model. As an internal control, we assessed the relation between β-blocker use and all-cause mortality.
Results
During a mean follow-up of 35 months, the risk of all-cause mortality was similar in RASI users compared with non-users (hazard ratio: 0.92; 95% confidence interval: 0.67–1.26). Similar results were obtained in the sensitivity analysis. In contrast, β-blocker use was associated with significantly lower all-cause mortality in the same population.
Conclusions
RASI use was not associated with better outcomes in patients with AF and HF in this prospective cohort, which raises questions about their value in this specific subset.
Trail Registration: ChiCTR-OCH-13003729.
Keywords
Introduction
Chronic heart failure (HF) and atrial fibrillation (AF) are two common illnesses associated with a substantial morbidity and risk of death. AF and HF frequently coexist, and up to 50% of patients with HF develop AF throughout their disease. 1 HF promotes ultrastructural changes and neuroendocrine processes, which eventually lead to atrial remodeling and increase the risk of developing AF.2–4 However, AF impairs cardiac function, leading to worsening symptoms of HF.5,6 Patients with both AF and HF have an even higher mortality and hospital admission rates compared with having only one of these conditions, irrespective of which illness arises first.7–9
Oral therapies for HF, including β-blockers, renin–angiotensin system inhibitors (RASIs), and mineralocorticoid receptor antagonists, markedly improve outcomes in heart failure and reduced ejection fraction (HFrEF).10–13 According to recent European and American guidelines, using these cardiovascular medications in patients with HFrEF has a class I recommendation.14–16 Nonetheless, the efficacy of using these medications in patients with HF who have a comorbidity of AF remains unclear. Previous analyses in this patient subset have lacked statistical power, but further large randomized studies are unlikely because most of these drugs are no longer patented. There is little incentive in the pharmaceutical industry to sponsor such trials. In the absence of such randomized evidence, evaluating the treatment effect from observational cohorts using specific methods to decrease attribution bias would be helpful. Post-hoc analysis of the Atrial Fibrillation and Congestive Heart Failure trial showed that β-blocker therapy was associated with a significantly lower mortality, but not hospitalization rate, in patients with HFrEF and AF. 17 These data enriched current evidence-based recommendations in patients with HF and added to the limited current evidence about treatment in patients with HF and AF.
With regard to RASIs, large randomized, controlled trials have established the benefits of angiotensin-converting enzyme inhibitors or angiotensin-II receptor blockers in reducing morbidity and mortality in HFrEF.11–13,18 Recently, HF with a left ventricular ejection fraction (LVEF) of 40% to 49% was defined as a distinct phenotype of HF, namely HF with a mid-range left ventricular ejection fraction (HFmrEF). 15 Observational studies have shown that RASI use is associated with a better prognosis in patients with HFmrEF.19,20 Therefore, this study aimed to investigate the association between routine RASI use and mortality and other cardiovascular outcomes in patients with HF and AF, specifically those with HFrEF and HFmrEF.
Materials and methods
Study population
The current analysis is based on the China Atrial Fibrillation Registry (CAFR) study, which was a prospective, multicenter, hospital-based, ongoing registry study of patients diagnosed with AF. Details of the cohort have been described previously. 21 This observational study aimed to understand the current clinical practice patterns, prognoses, and related factors in Chinese patients with AF and to compare different treatments in real-world practice. This study was approved by the Human Research Ethics Committee of Beijing Anzhen Hospital (approval number: D111107300000). The study is in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from all patients. The reporting of this study conforms to the STROBE statement. 22 Between August 2011 and December 2018, patients with AF were prospectively recruited from outpatient clinics and those in the hospital patients in 31 hospitals located in Beijing.
Patients with AF were included in the present analysis if they had an LVEF <50%, and they had symptomatic congestive heart failure and/or a medical history of HF.
RASI use was defined as the use of an angiotensin–converting enzyme inhibitor or angiotensin-II receptor blocker at baseline or during the follow-up. Patients who never used an RASI were classified as non-users at baseline and throughout the follow-up period (until death or 31 December 2018). Information on angiotensin receptor-neprilysin inhibitor (ARNI) therapy was not collected in this registry because the use of an ARNI was recommended for patients with HFrEF in 2014. 10 RASIs and data on other medications were collected at every follow-up.
We excluded patients aged younger than 18 years (n = 17), those with valvular AF (n = 626), and those who had <6 months’ follow-up (n = 3226). The following information on the patients’ baseline characteristics was collected: age, sex, smoking and drinking status, the medical history, including hypertension, diabetes mellitus (DM), myocardial infarction, stroke/transient ischemic attack (TIA), peripheral artery disease, the presence of a permanent pacemaker or implantable cardioverter-defibrillator (ICD), and catheter ablation, and the CHA2DS2-VASc score (HF, hypertension, age ≥75 years, DM, previous stroke/TIA, vascular disease, age 65–74 years, female sex). We also collected results of a physical examination, laboratory and imaging tests, and medication. All data were entered into a specific electronic data capture system. Data elements and definitions of each variable were in accordance with the American College of Cardiology/American Heart Association recommendations on AF clinical data standards.14,16,23,24
Follow-up and outcomes
Each enrolled patient was followed up at 3 months, 6 months, and every 6 months thereafter at an outpatient clinic or by a telephone interview by trained staff. Information on medical or interventional therapies, events of hospitalizations, and deaths was collected at each follow-up. The primary outcome was all-cause mortality. The secondary outcomes were cardiovascular mortality, cardiovascular hospitalization, and a composite endpoint of all-cause mortality or cardiovascular hospitalization.
Statistical analysis
Data are expressed as mean (standard deviation) for normally distributed continuous variables and number (%) for categorical variables. We compared patients’ characteristics between baseline RASI users and non-RASI users using the t test for continuous variables and the χ2 test for categorical variables.
For the main analysis, we carried out a propensity score-matched Cox proportional hazard analysis. Propensity score-matching (1:1) was performed using a greedy algorithm with a caliper of 0.2 to adjust for measured confounders between patients receiving and those not receiving an RASI at baseline before comparison. Propensity scores were calculated using a logistic regression model in which RASI use at baseline was treated as the dependent variable. Twenty-one baseline variables were included as covariates (Table 1). These covariates were selected on the basis of considering substantive knowledge and statistical associations. Improvements in balance across covariates were measured by absolute values of standardized differences in means or proportions of each covariate across exposure groups and expressed as a percentage of the pooled standard deviation. An absolute standardized difference <10%, as applied in our approach, was generally accepted as indicative of inconsequential residual bias.25,26 Drug use (i.e., use of RASIs, digoxin, β-blockers, oral anticoagulants, and statins) was treated as a time-dependent covariate.
Baseline characters according to RASI use before and after propensity score matching

Values are mean ± SD or n (%).
*Covariates included in the propensity score matching analysis.
RASI, renin–angiotensin system inhibitor; SD, standard deviation; TIA, transient ischemic attack; ICD, implantable cardioverter-defibrillator; eGFR, estimated glomerular filtration rate; BMI, body mass index; NYHA, New York Heart Association; LVEF, left ventricular ejection fraction; CHA2DS2-VASc, HF, hypertension, age ≥75 years, diabetes mellitus, previous stroke/TIA, vascular disease, age 65 to 74 years, female sex; OAC, oral anticoagulant; AF, atrial fibrillation.
Sensitivity analysis was performed with multivariate Cox regression models to estimate the hazard ratios (HRs) with 95% confidence intervals (CIs) for RASI use in clinical outcomes, in which drug use was also treated as a time-varying variable. The following variables that were included in the multivariable adjustment were as follows: age, sex, smoking, drinking, estimated glomerular filtration rate (eGFR), heart rate, systolic blood pressure, New York Heart Association (NYHA) class, LVEF, medical history (stroke/TIA, peripheral arterial thrombosis, hypertension, DM, MI, peripheral artery disease, permanent pacemaker or ICD, and CA), and drug use (i.e., RASIs, digoxin, β-blockers, oral anticoagulants, and statins).
With observational data, there needs to be evaluation of whether methodological limitations or decisions could bias findings. As an internal validity check of the present analyses, we assessed the association between β-blocker use and clinical outcomes in the same non-HF with preserved ejection fraction population using the same analytical methods as those described above.
To investigate whether any particular feature modulated the effects of an RASI on the primary outcome, subgroup analyses were performed in the main analysis. Patients were stratified by age (age ≥65 years or age <65 years), the eGFR (eGFR ≥60 mL/minute or eGFR <60 mL/minute), the LVEF (LVEF ≥40% or LVEF <40%), or the AF pattern (persistent AF or non-persistent AF). Heterogeneity across subgroups was tested by introducing interaction terms of RASI use and stratification variables into the Cox proportional hazard regression models.
All statistical tests were two-tailed with a significance level of 0.05. Data were analyzed using SAS software version 9.4 (SAS Institute, Cary, NC, USA).
Results
Baseline characteristics
A total of 25,512 patients were prospectively enrolled in the CAFR study. Of these, 938 patients with AF and HF with an LVEF <50% were included in the analysis, with a mean follow-up of 35 months. The baseline characteristics of the patients are shown in Table 1. Before matching, 568 (60.6%) patients were RASI users and 370 (39.4%) were non-RASI users. RASI users had a higher systolic blood pressure (p < 0.001), heart rate (p < 0.05), CHA2D2-VASc score (p < 0.001), and prevalence of hypertension (p < 0.001) compared with non-RASI users. Furthermore, RASI users were more likely to be receiving digoxin (p < 0.01) and to be on statins (p < 0.001) than non-RASI users.
After propensity matching, the population was restricted to 590 patients among whom 295 (50%) were RASI users and 295 (50%) were non-RASI users. A flow chart showing the patients’ selection is shown in Figure 1. After propensity matching, there were no significant differences in baseline characteristics between those who received an RASI and those who did not receive an RASI (Table 1). Post-matching standardized differences for all 21 baseline covariates were <10%. Matched patients had a mean age of 65.9±13.3 years, 70.0% were men, and the mean LVEF was 38.9% ± 6.9%.
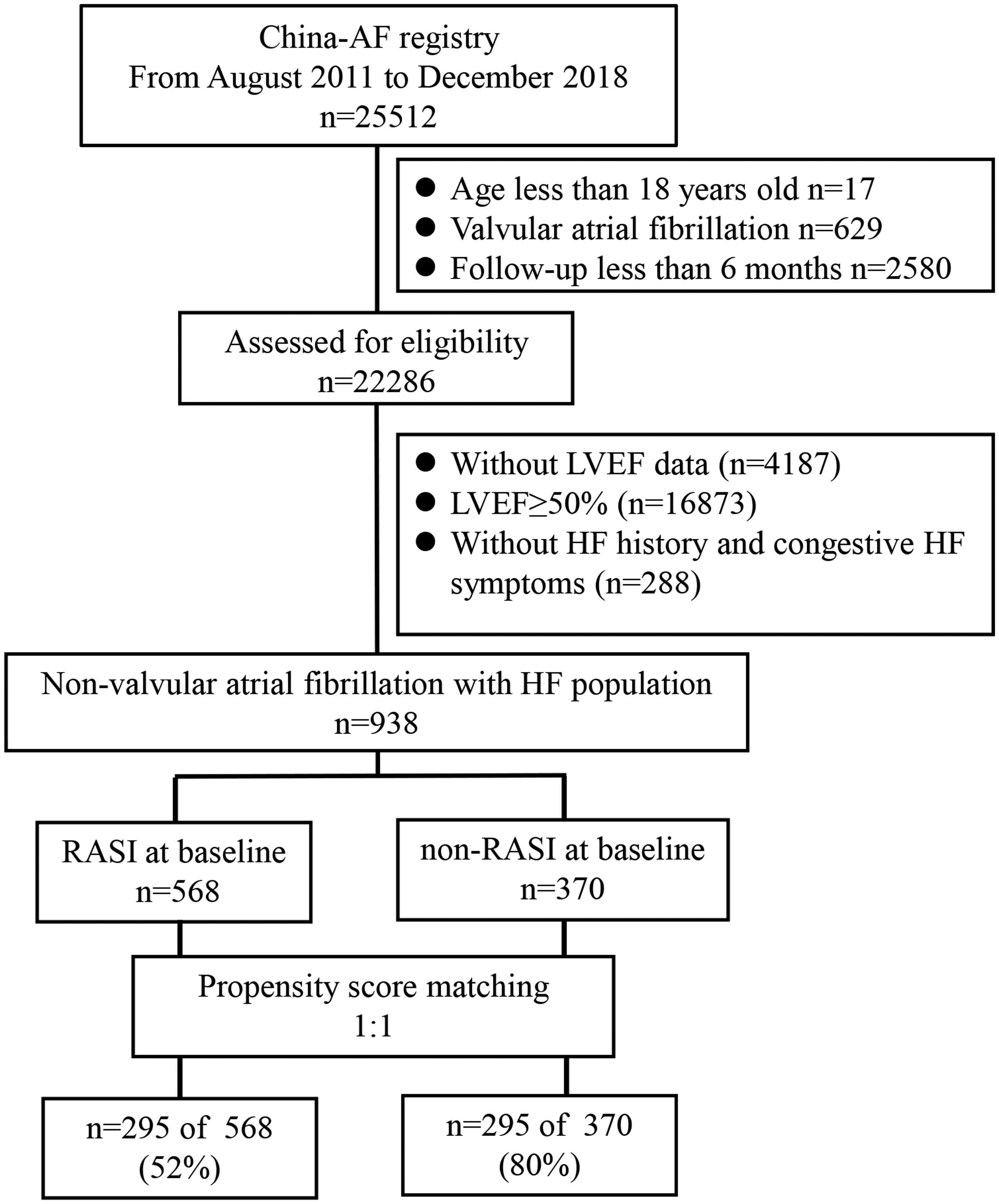
Overview of the study cohort
Primary and secondary outcomes
In the total population, 294 patients died during the follow-up. A total of 177 (9.06/100 patient-years) RASI users and 117 (9.64/100 patient-years) non-RASI users died. In the propensity score-matched population, 205 patients died during the follow-up, and these comprised 101 (9.98/100 patient-years) RASI users and 104 (11.37/100 patient-years) non-RASI users. The rate of the primary outcome (all-cause mortality) was similar in RASI users compared with non-RASI users (HR: 0.92; 95% CI: 0.67–1.26) (Table 2). Analyses of secondary outcomes yielded similar results. The HR was 1.02 (95% CI: 0.68–1.52) for cardiovascular mortality, 0.99 (95% CI: 0.72–1.36) for cardiovascular hospitalization, and 0.89 (95% CI: 0.69–1.14) for the composite endpoint of all-cause mortality or cardiovascular hospitalization.
Incidence rates and associations between RASI use and outcomes with propensity score matching analyses and sensitivity analyses

aData for RASI users and non-RASI users are shown as the incidence rate (number of events/100 patient-years).
bMultivariate time-varying Cox regression was performed in the 938 patients with non-heart failure with preserved ejection and adjusted for age, sex, smoking, drinking, the estimated glomerular filtration rate, heart rate, systolic blood pressure, New York Heart Association class, the left ventricular ejection fraction, the medical history (stroke/transient ischemic attack, peripheral arterial thrombosis, hypertension, diabetes mellitus, myocardial infarction, peripheral artery disease, permanent pacemaker or implantable cardioverter-defibrillator, and catheter ablation), and use of β-blockers, digoxin, oral anticoagulants, and statins.
RASI, renin–angiotensin system inhibitor; HR, hazard ratio; CI, confidence interval.
Similar results were obtained in sensitivity analysis with multivariate Cox regression models (HR: 0.93 for all-cause mortality, HR: 0.92 for cardiovascular mortality, HR: 1.07 for cardiovascular hospitalization, HR: 1.07 for cardiovascular hospitalization, and HR: 0.94 for the composite outcome).
Subgroup analyses
The main analyses by the propensity score-matching approach were repeated in key patient subgroups. There were no significant interactions between RASI use and AF patterns (i.e., persistent AF or non-persistent AF), age (i.e., age ≥65 years or <65 years), the eGFR (i.e., eGFR ≥60 mL/minute or <60 mL/minute), or the LVEF (i.e., LVEF ≥40% or <40%) regarding all-cause mortality (Figure 2).
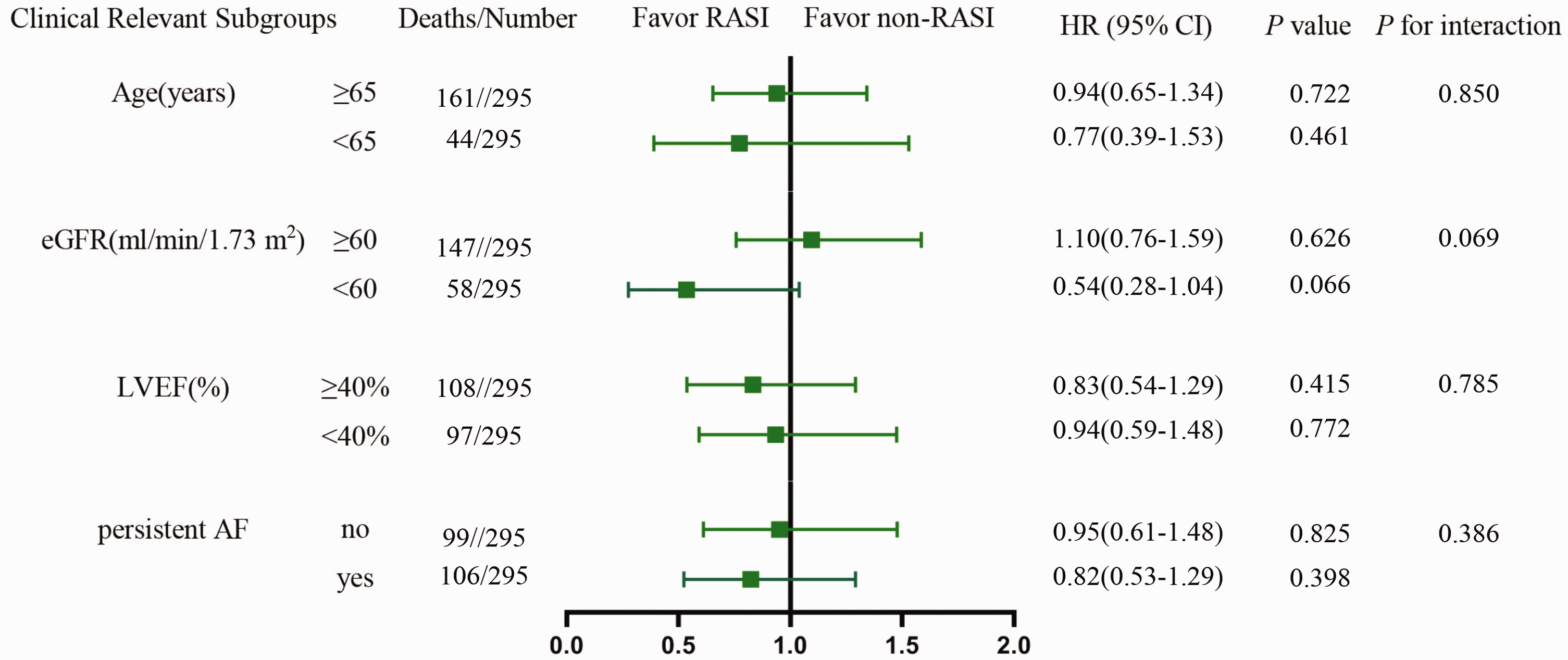
RASI use and outcomes in clinically relevant subgroups
Internal control
The associations of β-blocker use and all-cause mortality and cardiovascular outcomes were analyzed with the same population as that used for investigating RASI use. There were substantial differences in baseline characteristics between patients who received β-blockers and those who did not receive β-blockers before propensity matching (Table 3). After propensity matching, there were no significant differences in baseline characteristics between patients who received and those who did not receive β-blockers.
Baseline characters according to β-blocker use before and after propensity score matching

Values are mean ± SD or n (%).
*Covariates included in the propensity score-matching analysis.
SD, standard deviation; TIA, transient ischemic attack; ICD, implantable cardioverter-defibrillator; eGFR, estimated glomerular filtration rate; BMI, body mass index; NYHA, New York Heart Association; LVEF, left ventricular ejection fraction; CHA2DS2-VASc, HF, hypertension, age ≥75 years, diabetes mellitus, previous stroke/TIA, vascular disease, age 65 to 74 years, female sex; OAC, oral anticoagulant; RASI, renin–angiotensin system inhibitor; AF, atrial fibrillation.
β-blockers were associated with significantly lower all-cause mortality in the propensity-matched population (HR: 0.69; 95% CI: 0.51–0.93; p = 0.016) and in the total population after analysis with multivariate Cox regression (HR: 0.68; 95% CI: 0.50–0.92; p = 0.012) (Table 4). However, β-blockers were not associated with any secondary outcomes in different analytical methods.
Incidence rates and associations between β-blocker use and outcomes with multivariate time-varying Cox regression and propensity score matching analyses

aData for RASI use and non-RASI use are shown as the incidence rate (number of events/100 patient-years).
bMultivariate Cox regression was performed as described previously.
HR, hazard ratio; CI, confidence interval; RASI, renin–angiotensin system inhibitor.
Discussion
In this prospective cohort, we did not find any significant associations between the routine use of RASIs and better outcomes in patients with AF and HFrEF or HFmrEF. These findings were consistent across propensity score-matching analysis and sensitivity analysis with multivariate Cox regression models. Sub-group analyses showed similar results, regardless of the age, eGFR, type of AF, or LVEF. However, with similar methodological approaches, β-blocker use was significantly associated with a lower rate of all-cause mortality. This internal control reinforced the validity of the current analytical strategy. Therefore, a benefit of routine RASI use was not found in patients with AF and HFrEF and/or HFmrEF in this prospective, observational cohort.
In our study, 3.8% of the cohort of the patients had HF with HFrEF and HFmrEF. This rate is much lower than that in Western world cohorts. More than 50% of patients with HF will experience AF at some time. A meta-analysis on individual-level data on the efficacy of β blockers showed that 17% of patients with HF had AF at baseline. 17 However, we did not include HF with preserved ejection fraction in our study. More than two thirds of patients with HF with preserved ejection fraction have AF, and most AF occurs before a diagnosis of HF with preserved ejection. 27 Furthermore, the severity of patients in our study was much lower than that in patients who only have HF. An example of this difference is that only approximately 20% of patients had NYHA class III/IV cardiac function in our study, whereas 60% of patients had NYHA class III/IV cardiac function in the previous meta-analysis. 17 AF is more likely to develop throughout illness as the severity of HF increases. This finding could be the reason for the prevalence of AF being much higher in a HF cohort than in our study.
HF with AF is a specific subset population; when one of these shows an increase in its prevalence and incidence, the prognosis of the other worsens. Their frequent coexistence raises several challenges, especially regarding how best to treat them with routine medication. Our study showed that the prognostic benefit identified in patients with HFrEF and HFmrEF should not be extrapolated to HF subsets with AF rhythm. Therefore, the benefit of RASI use might be different between HFmrEF with AF and HFrEF with AF. To address this hypothesis, interactions were tested between RASI use and the LVEF (≥40% vs. <40%). However, this interaction was nonsignificant. More specifically, we did not find any benefit of RASI use in improving all-cause mortality when we compared patients with an LVEF<40% or ≥40% in subgroup analysis.
Notably, our results are in contrast to a secondary analysis of the Candesartan in Heart Failure-Assessment of Reduction in Mortality and morbidity (CHARM) trial. 28 This trial reported that candesartan led to a significant 30% risk reduction in all-cause mortality (HR: 0.70; 95% CI: 0.50–0.90) in patients with AF and HFrEF (i.e., LVEF <40%). Because of the nature of a secondary analysis, the conclusion of the CHARM trial may be not reliable owing to not following the randomized control principle and introducing bias. The primary analysis of this trial was based on the diagnosis of AF using a baseline electrocardiogram. Patients in sinus rhythm at baseline, but with a history of AF or new development of AF during follow-up, were categorized as no AF in the CHARM trial. Therefore, our study probably underestimated the true relationship between AF and mortality.
The reason for the lack of an association between RASI and better outcomes in patients with AF and HF might be due to several physiological and characteristic differences. First, unlike sinus rhythm, an irregular rhythm in AF is associated with a detrimental effect on cardiac function,4,29 which might offset the benefit of RASI on mortality. Second, the statistical results should be evaluated with caution by fully understanding the detailed information and underlying biases of a study. An example of this required understanding is that a meta-analysis published in 2014 showed no evidence that β-blockers improved the prognosis in patients with HF concomitant with AF. 30 However, later studies found the opposite conclusion.17,31 In this previous meta-analysis, more patients with NYHA III/IV cardiac function may have weakened the therapeutic benefit of β-blockers. Fewer patients with HF had NYHA III/IV cardiac function in our study compared with previous trials in patients with HFrEF and HFmrEF. 30 Therefore, β-blocker use was associated with significantly lower all-cause mortality in patients with AF and HF, whereas RASI use was not associated with lower all-cause mortality in the same population. We speculate that the benefits of RASI are limited in this population. Third, there are also structural and cellular consequences of AF that might affect the treatment efficacy. 32 These reasons cannot fully explain why RASI was not associated with a prognostic benefit in patients with AF and HF. Therefore, this issue requires further investigation.
Even though we did not find evidence of a benefit from RASI use in this analysis, several factors suggest that these results are valid. First, we used data from the CAFR study.22,33 These prospective data reduced the number of misclassifications, helped to avoid recall bias, and increased the accuracy of the potential confounder measurements. Second, we used different analytical approaches to control confounding. To balance potential confounders, we adopted the propensity score matching approach. Among observational studies, using the propensity score ensures the closest design to a clinical trial. 34 We also adjusted for an extensive array of clinical variables (21 clinical covariates) in a multivariable Cox model. Time-varying coefficients are capable of addressing the time-varying nature of treatment and reducing potential immortal time bias. 35 We modeled medication use as a time-dependent covariate in the propensity score matching approach and multivariable Cox model analysis to account for treatment changes during the follow-up. We confirmed our conclusion across various subgroups. Third, the contrast between the lack effects of RASI on all-cause mortality and the marked consistent benefit of β-blocker use in our analysis suggested that drugs, rather than analytical methods or study design, may have accounted for our results.
Our study has several limitations. First, although we adjusted for many significant factors and used sensitivity analysis to examine the “true” association, residual confounding effects may have been present. Second, this was an observational study in nature. A causal relationship between RASI use and all-cause mortality or cardiovascular outcomes should not be ruled out unless randomized, controlled trials can confirm our findings. Third, we lacked information about ARNI use in this cohort, which may have had a role in the prognostic outcome. However, this potential bias is likely to be minimal because ARNI therapy is comparatively low in China. 36 Despite these limitations, our study provides essential data on the relationship between RASI use and clinical outcomes in patients with AF and HF.
Conclusions
In this prospective CAFR study, RASI use was not associated with better outcomes in patients with AF and HF with an LVEF <50%. In contrast, in the same population, identical methods showed a lower rate of all-cause mortality in patients on β-blockers than in those without β-blockers. These results suggest that RASIs are not useful in this specific subset, but their effects need to be critically evaluated in randomized trials.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (2016YFC0900901, 2016YFC1301002).
